My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Nutrición Hospitalaria
On-line version ISSN 1699-5198Print version ISSN 0212-1611
Nutr. Hosp. vol.34 n.5 Madrid Sep./Oct. 2017
https://dx.doi.org/10.20960/nh.1051
TRABAJO ORIGINAL / Paciente crítico
Preoperative metabolic status is associated with different evolution of resting energy expenditure after liver transplant in adults
Influencia del estado metabólico preoperatorio sobre los cambios en el gasto energético en reposo después del trasplante hepático en adultos
Ana Brito-Costa1, Luís Pereira-da-Silva2,3, Ana Luísa Papoila2,3, Marta Alves2, Élia Mateus4, Fernando Nolasco3,4 and Eduardo Barroso3,4
1Nutrition and Dietetics Unit. Hospital Curry Cabral. Centro Hospitalar de Lisboa Central. Lisbon, Portugal.
2Research Unit. Centro Hospitalar de Lisboa Central. Lisbon, Portugal.
3NOVA Medical School. Universidade NOVA de Lisboa. Lisbon, Portugal.
4Transplant Center. Hospital Curry Cabral. Centro Hospitalar de Lisboa Central. Lisbon, Portugal
ABSTRACT
Introduction: The resting energy expenditure (REE) evolution after liver transplantation is not fully understood.
Objective: To assess longitudinally the REE evolution in adults undergoing liver transplantation, in association with other nutritional measurements that characterize the metabolic profile.
Methods: A single-center cohort of consecutive end-stage liver disease patients with indication for liver transplantation was recruited. REE, subjective global assessment (SGA), handgrip strength and body composition measurements were assessed before transplantation (T0) and at median nine (T1) and 36 (T2) days after transplantation. Mixed effects regression models were used for analysis.
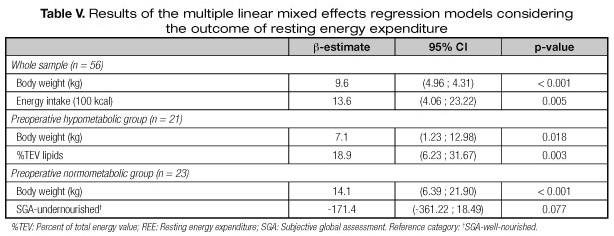
Results: Fifty-six patients with a mean age of 53.7 (8.5) years were included; 87.5% were males. In T0, 41% of patients were normometabolic, 37.5% were hypometabolic and 21.4% were hypermetabolic. After transplantation, the mean REE decreased progressively in patients initially hypermetabolic and the opposite occurred in those initially hypometabolic. The REE evolution was positively associated with body weight (β = 9.6, p < 0.001) and energy intake (β = 13.6, p = 0.005) in the whole sample; it was positively associated with body weight (β = 7.1, p = 0.018) and percentage of energy intake from lipids (β = 18.9, p = 0.003) in initially hypometabolic patients, and positively associated with body weight (β = 14.1, p < 0.001) and negatively associated with SGA-undernourishment (β = -171, p = 0.007) in initially normometabolic patients.
Conclusion: Different REE evolutions after liver transplantation are associated with the preoperative metabolic status. In patients initially hypometabolic, the REE evolution is positively associated with body weight and percentage of energy intake from lipids, and in those initially normometabolic, it is positively associated with body weight and negatively associated with SGA-undernourishment.
Key words: Body composition. Liver transplantation. Metabolic status. Nutritional assessment. Resting energy expenditure.
RESUMEN
Introducción: el gasto energético en reposo (GER) después del trasplante hepático no está totalmente esclarecido.
Objetivo: evaluar longitudinalmente la evolución del GER y de otros parámetros nutricionales que caracterizan el perfil metabólico en adultos que recibieron un trasplante hepático.
Métodos: cohorte consecutiva de pacientes con enfermedad hepática terminal con indicación para trasplante, reclutada en un centro. Las siguientes variables fueron medidas antes del trasplante (T0) y en las medianas de nueve (T1) y 36 (T2) días de postoperatorio: GER, valoración global subjetiva (VGS), dinamometría manual y composición corporal. Se utilizaron modelos de regresión de efectos mixtos.
Resultados: se incluyeron 56 pacientes con una edad media de 53,7 (8,5) años; el 87,5% de ellos eran varones. En T0, el 41% de los pacientes eran normometabólicos; el 37,5%, hipometabólicos, y el 21%, hipermetabólicos. Después del trasplante, el GER disminuyó progresivamente en los pacientes inicialmente hipermetabólicos y lo contrario ocurrió en los inicialmente hipometabólicos. La evolución del GER se asoció positivamente con el peso (β = 9,6, p < 0,001) y la ingesta energética (β = 13,6, p = 0,005) en toda la muestra. Asimismo, se asoció positivamente con el peso (β = 7,1, p = 0,018) y el porcentaje de ingesta energética de grasas (β = 18,9, p = 0,003) en los inicialmente hipometabólicos, mientras que en los inicialmente normometabólicos se relacionó positivamente con el peso (β = 14,1, p < 0,001) y negativamente con la subnutrición por VGS (β = -171, p = 0,007).
Conclusión: la evolución del GER después del trasplante hepático se asocia con el estado metabólico preoperatorio, de forma positiva con el peso y el porcentaje de ingesta energética de grasas en los inicialmente hipometabólicos, mientras que en los pacientes inicialmente normometabólicos se relaciona positivamente con el peso y negativamente con la subnutrición valorada por VGS.
Palabras clave: Composición corporal. Estado metabólico. Evaluación nutricional. Gasto energético en reposo. Trasplante hepático.
Introduction
Orthotopic liver transplantation (LTx) is currently the treatment of choice for end-stage liver disease (1,2). The success rate for this procedure has increased progressively during the past decades as a result of continued advances in surgical technique, anesthesia, immunosuppressive therapy and nutritional support (1,3).
After LTx, it is expected that many metabolic derangements improve with the recovery of allograft function, because the liver is the organ that encompasses the overall regulation of the energy metabolism process (4,5). However, some postoperative metabolic complications related to nutritional status, hepatic denervation, and prolonged immunosuppression may compromise the long-term clinical outcome (6).
The resting energy expenditure (REE) measurements have major interest in patients with end-stage liver disease on the waiting list for LTx. Hypermetabolism, mainly associated with extrahepatic factors, has been described in these patients as a negative prognostic factor in transplant-free survival patients with cirrhosis, independently of the model for end-stage liver disease (MELD) score (7).
When the energy metabolism, body composition, and dietary intake are assessed together, information regarding the relationship between the metabolic and the nutritional status is more accurate and complete (2). Very few studies have assessed the combined sequential changes in dietary intake, REE and body composition in patients with end-stage liver disease undergoing LTx (8-10), and longitudinal assessments that include the pre- and postoperative periods are even scarce (2,3).
This study aimed to longitudinally assess the REE before and shortly after orthotopic LTx in adults, as the primary outcome. To better characterize the metabolic profile, dietary intake, nutritional status, body composition, and grip strength were assessed together with REE, as secondary outcomes. We hypothesize that after LTx, the REE progressively normalizes, and different evolutions of REE are associated with the preoperative metabolic status.
Material and methods
STUDY DESIGN
This observational cohort study assessed the longitudinal changes in the REE of consecutive patients who underwent LTx within a period of two years (January 2013 to December 2014), at the Transplant Center of Hospital Curry Cabral, Centro Hospitalar de Lisboa Central. All prescriptions were provided by the treating physicians in compliance with the Transplant Center protocol described below. This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving patients were approved by the hospital ethical committee. Written informed consent was obtained from all patients and data confidentiality was ensured. Registration of race and ethnicity was not permitted under Portuguese law. The study protocol is described elsewhere in a secondary analysis of this study (11). The criterion for inclusion was elective orthotopic LTx due to chronic liver disease. The criteria for exclusion were inborn errors of metabolism, familial amyloidotic polyneuropathy, sequential transplantation, multi-organ transplantation, re-transplantation, and acute liver failure. Baseline characteristics potentially affecting REE were recorded, including age, sex, etiology of liver disease, co-morbidities, MELD score, and drug addiction. Other recorded pre- and postoperative variables with potential influence on the REE included postsurgical complications, immunosuppressive therapy, steroids and steroid dosages, beta-blockers, and blood biochemistries.
NUTRITIONAL SUPPORT PROTOCOL
In compliance with the Transplant Center protocol, the patients were scheduled to start enteral nutrition within the first 48 hours after LTx (via nasogastric tube or oral feeding), once gastrointestinal motility was re-established. The daily nutritional goals were 35-40 kcal/kg body weight and 1.2-1.5 g protein/kg body weight (12).
IMMUNOSUPPRESSIVE THERAPY PROTOCOL
As per the Transplant Centre protocol, patients were scheduled to start treatment with a combined therapy including a calcineurin inhibitor (cyclosporine or tacrolimus) and corticosteroids. The cyclosporine-based regimen was preferred for patients with hepatitis secondary to hepatitis C virus, human immunodeficiency virus and diabetes mellitus. According to the specific conditions, the immunosuppressive regimen was combined with mycophenolate mofetil.
ASSESSMENT SCHEDULE
Participants with stable conditions were scheduled for three follow-up assessments, as follows: at the last visit before LTx (T0), after surgery as soon as respiratory and functional autonomy were established (T1), and approximately one month after surgery, inpatient or outpatient (T2). For each follow-up visit, all assessments were performed on the same day by the same nutritionist trained in the equipment used in the study (AB-C), and included dietary assessment, subjective global assessment (SGA), REE, anthropometry, bioelectrical impedance analysis (BIA), and handgrip strength.
- Dietary assessment. Food intake was assessed via 24-hour dietary recall (2) by the aforementioned experienced nutritionist. To achieve a more accurate estimation, complementary visual images of food portions were used. Food Processor Plus® version 6.0 (ESHA Research, Salem, Oregon, USA) was used to convert the consumption of food items into energy and macronutrients. The daily energy intake is expressed as the total energy intake (in kcal) and as the percentages of the total energy values (%TEV) generated per macronutrient.
- Subjective global assessment. This method is based on the nutritional histories and clinical examinations of the patients. The patients were classified as well nourished, moderately undernourished and severely undernourished (13).
- Resting energy expenditure. The REE was measured by indirect calorimetry using Fitmate® (COSMED, Rome, Italy), which has been validated for REE measurements in adults (14). The patients fasted for 12 hours. The device was automatically calibrated before each measurement. The volume of inspired oxygen was collected using a comfortable face mask with integrated bacterial filter. The exhaled gas was collected over 15 minutes, and the gas collected over the first five minutes was discarded (14). The mean volume of inspired oxygen per minute was used to calculate the REE according to the Weir (14) formula. The REE was predicted according to the Harris and Benedict equation (15), using the measured body weight or the adjusted body weight in case of ascites (16). Hypermetabolism was defined as a measured REE more than 120% of the predicted value; normometabolism, as a measured REE within 80-120% of the predicted value, and hypometabolism, as a measured REE less than 80% of the predicted value (17).
- Anthropometry. Anthropometric measurements included body weight (Tanita® SC-330 Tanita Arlington Heights, IL, USA, with a limit 270 kg and a precision of 0.1 kg), height (Seca® digital model 242, precision 0.1 cm), triceps, biceps and subscapular skinfolds thickness (Jamar® caliper, Sammons Preston, Bolingbrook, Illinois, USA, accuracy 1 mm), and mid-arm, waist and hip circumferences, using the recommended methods (18). The averages of three consecutive measurements were recorded for the analyses. Body mass index was calculated (18).
- Bioelectrical impedance analysis. Body composition was assessed with a validated tetrapolar multifrequency BIA (Body Composition Monitor, Fresenius Medical Care®) (19) using an alternating current of 500 μa, and frequencies of 5-1000 kHz (50 frequencies), which provided estimates of total body water, intracellular water, extracellular water, body cell mass, lean mass and fat mass. Automatic calculations were performed for the percentage of fat mass, lean mass index, and fat mass index (18).
- Handgrip strength. Handgrip strength was assessed using the Jamar® Handgrip Dynamometer (Sammons Preston, Bolingbrook, Illinois, USA). All measurements were collected with a precision of 1 kg with the patient in standardized positions (20), and the best value obtained from three measurements of the right and left hands was considered as recommended (20).
The dietary assessment and SGA were not assessed whenever the state of consciousness of the patient did not guarantee acquisition of reliable information (unless family members or caregivers provided reliable data).
STATISTICAL ANALYSIS
Exploratory analyses were performed for all variables. There were no missing values for any of the analyzed variables. The categorical data are presented as frequencies and percentages, and the continuous variables are presented as means or medians with the standard deviations (SD) or interquartile ranges (25th percentile - 75th percentile), respectively, as appropriate. Shapiro Wilk tests and Q-Q plots were used to test for the normality of the distributions of the variables.
Generalized linear mixed effects models for a continuous response variable were used to take into account the correlation structure between measures in time, to explore the association between the REE and other clinical, nutritional, metabolic and demographic data considered as covariates: age, body weight, lean mass, body cell mass, lean mass index, immunosuppressive therapy (calcineurin inhibitors and steroids), β-blockers, length of stay in intensive care and total hospital stay, MELD score, SGA-assessed nutritional status, and dietary intakes (energy and %TEV generated per macronutrient).
The level of significance α = 0.05 was used. Nevertheless, p-values between 0.05 and 0.10 were still considered, indicating evidence, although weak, of an association with the outcome. The statistical analyses were performed using the Stata (StataCorp. 2013. Stata Statistical Software: Release 13. College Station, TX: StataCorp LP) and R project (R Core Team 2013) (R: a language and environment for statistical computing; R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, http://www.R-project.org/).
Results
SAMPLE CHARACTERISTICS
A total of 64 patients were recruited but eight were excluded due to death in five, re-transplantations in two, and failure to achieve respiratory and functional autonomy in one. From the 56 included, 87.5% were men, and at enrollment they had a mean (SD) age of 53.7 (8.5) years, and a mean MELD score adjusted for hepatocellular carcinoma (HCC) of 18.5 (4.3). The etiologies of the liver disease included 13 (23.2%) cases of alcoholic chronic liver disease (CLD), ten (17.9%) cases of alcoholic CLD associated with HCC, eight (14.3%) cases of alcoholic and viral associations, seven (12.5%) cases of viral CLD, six (10.7%) cases of alcoholic and viral CLD associated with HCC, five (8.9%) cases of viral CLD associated with HCC, and seven (12.5%) cases with other diseases (three cases of primary sclerosing cholangitis, two cases of cryptogenic CLD, one case of autoimmune disease, and one case of primary biliary cirrhosis). The viral CLD cases were caused by infection with hepatitis B or C viruses. Sixteen (28.6%) patients had a history of drug addiction.
MEASURED OUTCOMES
The assessments were performed on three occasions: at a median (interquartile range) of 90.5 (P25: 44.2; P75: 134.5) days before LTx (T0), at a median of nine (P25: 7; P75: 12) days after LTx (T1), and at a median of 36 (P25: 31; P75: 43) days after LTx (T2). At T2, all patients were stable, and 94.7% were outpatients.
On SGA assessment, 21 (37.5%) patients were undernourished before LTx. At T1, the number of undernourished patients increased to 34 (60.7%), and a subsequent decrease to nine (16.1%) patients was observed (p = 0.012) (Table I). Noteworthy, only one patient was severely undernourished at T0 and T1, and none at T2.
The energy intake significantly decreased from T0 to T1 and subsequently increased (Table II). Additionally, a progressive increase in the %TEV from lipids was observed up to T2 (only significant between T0 and T2), without significant changes in the %TEV from proteins. The %TEV from carbohydrates significantly decreased from T1 to T2.
Regarding the preoperative metabolic status, 21 (37.5%) patients were hypometabolic, 23 (41.1%) were normometabolic, and 12 (21.4%) were hypermetabolic (Table III). From T0 to T1, the prevalence of normometabolic patients increased (41.1% to 71.4%, p = 0.015), and that of hypometabolic and hypermetabolic patients decreased (37.5% to 17.9% and 21.4% to 10.7%, respectively; p < 0.001). From T0 to T2 the prevalence of normometabolic patients also increased (41.1% to 57.1%, p < 0.001) (Table III). In the preoperative normometabolic group, the prevalence of hypermetabolic patients increased (4.3% to 43.5%, p < 0.001) from T1 to T2 (Table III).
There were no significant changes between the mean REE at the three assessment times (Table II), but different REE profiles were observed when stratified into the preoperative metabolic status categories. In the preoperative hypometabolic patients, significant progressive increases in mean REE (kcal) from T0 to T1 (1,030.6 to 1,436.1, p = 0.001) and from T1 to T2 (1,436.1 to 1,659.2, p < 0.001) were observed (Fig. 1). In the preoperative hypermetabolic patients, significant progressive decreases in mean REE (kcal) from T0 to T1 (2,097.1 to 1,662.5, p = 0.024) and from T1 to T2 (1,662.5 to 1,493.0, p < 0.001) were observed (Fig. 1). In the preoperative normometabolic patients no significant postoperative changes were found in the mean REE. Notably, at T2, the mean measured REE was similar to the predicted value in the three preoperative metabolic groups.
Regarding anthropometry (Table IV), body weight, body mass index, waist circumference, hip circumference, mid-arm circumference, and skinfolds thickness significantly decreased from T0 to T1. At T2, these values remained significantly decreased compared to T0, except for the skinfolds thickness catching-up to similar preoperative values.
Regarding body composition (Table IV), total body water, intracellular water, extracellular water, lean mass, body cell mass and lean mass index significantly decreased from T0 to T1, with subsequent catch-up to similar preoperative values, except for the extracellular water that remained decreased. The percentage of fat mass significantly increased from T0 to T1, with subsequent decrease to similar preoperative values and the fat mass index did not change from T0 to T1 and decreased subsequently (Table IV).
The mean handgrip strength (kg) significantly decreased from T0 to T1 (64.4 to 56.2, p < 0.001), with subsequent catch-up to a similar preoperative value (56.2 to 63.2, p < 0.001).
For the whole sample, the significant predictors of REE were body weight and energy intake (Table V). Specifically, each additional kg of body weight was associated with a 9.6 kcal mean increase in REE (p < 0.001), and each increment of 100 kcal energy intake was associated with a 13.6 kcal mean increase in REE (p = 0.005).
In the preoperative hypometabolic patients, the significant predictors of REE were body weight and %TEV from lipids (Table V). Specifically, each additional kg of body weight was associated with a 7.1 kcal mean increase in REE (p = 0.018), and each 1% increment in %TEV from lipid intake was associated with an 18.9 kcal mean increase in REE (p = 0.003).
In the preoperative normometabolic patients, the significant predictors of REE were body weight and SGA-assessed nutritional status (Table V). Specifically, each kg increment of body weight was associated with a 14.1 kcal mean increase in REE (p < 0.001); compared with the well-nourished patients according to the SGA, the SGA-undernourishment was associated with a 171 kcal mean decrease in REE (p = 0.007).
In the preoperative hypermetabolic patients, no multivariable model was found for REE.
Discussion
Early screening in LTx may help to further elucidate the mechanisms associated with post-transplant metabolic and nutritional disturbances, and contribute to the optimization of clinical strategies (21). Accordingly, the earlier impact of perioperative factors on the nutritional status was assessed as soon as it was possible after LTx, that is, at T1 when the patients had sufficient autonomy to collaborate with the measurements, and one month thereafter, as outpatients.
The same skilled investigator performed all the measurements, guaranteeing more methodological homogeneity of the data collection. As blinding the previous measurements was not feasible, the more subjective methods potentially influenced by the observer, such as the 24-hour dietary recall and the SGA, were performed before those essentially dependent on automatic measurements, such as indirect calorimetry (14) and tetrapolar multifrequency BIA (19).
In the studied cohort some common changes were observed in the postoperative period.
A progressive increase in percentage of energy intake from lipids was observed, as reported by others (2). This effect may result from liver denervation with loss of fat detection mediated through cholecystokinin, enterostatin, and hepatic fuel oxidation, resulting in hyperphagia and development of fat appetite (22).
The number of SGA-assessed undernourished patients increased from baseline to T1, and subsequently decreased significantly. Worsening of the nutritional status and implicated mechanisms has been reported elsewhere at 30-45 days (23) and 3-5 months (24) after LTx. In relation to these previous reports, our data better discriminates the nutritional status along the first median 36 days after LTx. A long-term improvement in the SGA-assessed nutritional status that resulted in 81.8% well-nourished patients at five years after LTx has been described (25).
In this study, most of the body compartments were observed to decrease during the first days after transplantation, with subsequent catch-up to similar preoperative values. Contrarily, a continuing decrease in BIA adiposity indicators was observed, in spite of a postoperative increase in skinfolds thickness. An adiposity decrease shortly after LTx was previously reported (2,3). The handgrip strength transitory decreased with subsequent return to baseline values. The initial postoperative decrease in handgrip strength was reported to be more associated with psychological and metabolic factors and central fatigue (3,26) than with reduced skeletal muscle and protein deposition (3).
The REE was assessed by indirect calorimetry, which is considered as a gold standard method for REE assessment in hospitalized patients (27). The mean REE did not change significantly in the postoperative study period, similarly to described (5). In several studies on patients who underwent LTx, the energy metabolism has been analyzed through the REE normalized either for body weight (28), or for fat-free mass (17) and body cell mass (17), assuming that the later compartments are principal contributors to energy requirements and used as surrogates for metabolically active tissue (29). However, this may be a misconception to certain extent. Both the fat-free mass and body cell mass pool together numerous organs and tissues that differ significantly in metabolic rate. In healthy adults, despite the combined weight of brain, liver, heart, and kidneys is less than 6% of the total body weight, or 7% of fat-free mass, together they account for approximately 60% of the REE (29). As alternative, the regression analysis of REE avoids conceptual problems associated with the use of the aforementioned ratios (29). This approach was used in a recent assessment of nutritional metabolism after LTx (5), as we did in the present study.
As mostly used (2,8,17), the REE was predicted according to the Harris and Benedict equation (15), and the metabolic status was defined based on the percentage of measured REE in relation to the predicted value (17). We found that before LTx, 37.5% of patients were hypometabolic and 21.4% were hypermetabolic. In patients with end-stage liver disease, preoperative heterogeneity of metabolic status (3,17) has been described, with predominance of hypermetabolism (3,17), which was associated with postoperative reduced survival rate (7,8). Although the pathophysiology behind an increased REE is unclear, high extra-hepatic metabolism related to catecholamine hyper-regulation and/or systemic inflammation, associated to liver failure and drugs, have been implicated (7,17,30). On the other hand, the preoperative true hypometabolic state, that is, independent of the lean mass reduction, has been associated with the severity of liver disease and considered as potentially catastrophic (17,31). Other authors have interpreted the preoperative hypometabolism as a metabolic efficiency in response of low energy intakes (31) and/or an adaptation mechanism to a previous hypermetabolism (32).
Normalization of the REE was observed in the whole sample after LTx. A progressive REE increase in the initially hypometabolic patients and a progressive REE decrease in the initially hypermetabolic patients were observed. A significant proportion of initially normometabolic patients became hypermetabolic at the end of follow-up, similarly to what had been previously reported (2); however, no significant postoperative changes in the mean REE were found in the preoperative normometabolic group. Similarly to our results, it was found that improvement of nutritional metabolism may require approximately four weeks after LTx (5). This recovery is not entirely explained by adequate food intake and may involve improvements of endogenous metabolism (5). Despite the associated immunosuppressive therapy, the LTx may normalize the glucose utilization in cirrhosis by increasing non-oxidative glucose disposal (33). In comparison to the diseased liver, in which the hepatic blood flow-dependent oxygen supply may become limiting for its metabolic capacity (4), the splanchnic and whole-body oxygen uptake normalizes in clinically stable long-term course after LTx (4).
In the multivariable analysis, different mixed effects regression models for REE profile were applied in our study. In the whole sample, the REE profile was positively associated with body weight and energy intake. When stratified into the preoperative metabolic status categories, the REE profile was positively associated with body weight and percentage of energy intake from lipids in those initially hypometabolic, and with body weight and SGA-well-nourishment status in those initially normometabolic. It is difficult to explain the positive association observed between the REE and body weight, rather than with metabolically active body compartments. It may be speculated that the association of the REE with the body mass in spite of with the mass of metabolically active compartments is attributable to the confounding effect of surgical stress on the REE, mediated by a combination of hormones, such as catecholamines, glucagons, and cytokines (17).
In the whole sample, the positive association observed between the REE profile and energy intake may be attributable to the thermic effect of food. This effect is proportional to the total food intake, and in a typical mixed diet the thermic effect makes up 8% to 10% of total energy expenditure (34).
In the preoperative hypometabolic patients, the positive association found between the REE profile and percentage of energy intake from lipids is consistent with the reported higher REE in high fat consumers (35). It may be speculated that the decrease in adiposity in the initially hypometabolic patients observed at the end of the follow-up period (data not shown), may involve leptin. Leptin was reported to be associated with metabolic rate increase and reverse of excessive adiposity (36). Moreover, leptin may be elevated in hypometabolic patients before LTx (36) and positively correlated with the percentage of energy intake from lipids (35).
In the preoperative normometabolic patients, the SGA-undernourishment was associated with REE decrease. The negative association between the REE profile and the nutritional status may be attributable to a better metabolic efficiency in response to low energy intakes (24). In fact, the hypermetabolism may contribute to the development of undernutrition in liver disease, but when this state is reached, it appears that the REE is adapted to the new condition (32).
In our study, factors potentially affecting REE, such as age (10), sex (37), etiology of liver disease (38), MELD score (17), immunosuppressive therapy (2,37), steroids (37), and beta-blockers (7) were not associated with the REE profiles. It would be interesting to explore differences in the measured energy expenditures between the different types of liver disease; however, the great heterogeneity of etiologies of liver disease unable this analysis.
To the best of our knowledge, only two studies have longitudinally assessed the combined changes in metabolism, body composition and muscle function after LTx (2,3). In both of these studies, relatively small samples were followed-up for 12 months after surgery. Plank et al. (3) examined 14 patients and reported hypermetabolism before surgery; after surgery, the REE increased and peaked approximately on the 10th day; subsequently, the hypermetabolism persisted at six months, and at 12 months the measured REE was close to predicted values (3). Ferreira et al. (2) examined 17 patients and found increased REE at one month after surgery, without cases with hypometabolism; subsequently, REE decreased until the end of the study period.
A strong point of our study is the use of mixed effects regression models for REE profile, considering covariates potentially affecting the outcome (REE changes) shortly after LTx, and the potential confounders' effects; moreover, this analysis also took the autocorrelation structure over time between measurements into account.
A limitation of this study might be the use of a non-validated method to assess body composition in patients with chronic liver disease. Nonetheless, multi-frequency BIA is validated in patients with hydro-electrolytic derangements (19) and it has been suggested that this method is superior in cirrhotic patients (39). Moreover, the multi-frequency BIA is particularly accurate in the measurement of extracellular water, which may be increased in liver disease prior to LTx due to overhydration of the fat-free mass (40).
To summarize, longitudinal changes in REE were assessed shortly after orthotopic LTx, and analyzed through a comprehensive multivariable analysis including main variables potentially affecting the metabolic status. The REE was assessed together with additional nutritional measurements, enabling a better characterization of the metabolic profiles. Some of our hypotheses were confirmed. After LTx, a significant normalization of REE was observed in the whole sample, reflected by a REE progressive increase in the initially hypometabolic patients, and a REE progressive decrease in the initially hypermetabolic patients.
An important finding of this study is the well characterized association between the preoperative metabolic status and different postoperative REE evolutions. In spite of positive associations of REE evolution with body weight and energy intake in the whole sample, in initially hypometabolic patients it was positively associated with body weight and energy intake from lipids, and in initially normometabolic patients, positively associated with body weight and negatively associated with SGA-undernourishment.
Acknowledgment
This study is part of the PhD thesis in Life Sciences - Clinical Medicine of one of the authors (Ana Brito-Costa), supervised and co-supervised by two authors (Luís Pereira-da-Silva and Fernando Nolasco, respectively) from the NOVA Medical School, Universidade NOVA de Lisboa, Lisbon, Portugal.
Authorship
Ana Brito-Costa: Study design, measurements, data collection, analysis and interpretation of results, and manuscript drafting.
Luís Pereira-da-Silva: Conception and study design, interpretation of results, and manuscript drafting.
Ana Luísa Papoila: Statistical analysis, interpretation of results, and critical revision for important intellectual content.
Marta Alves: Statistical analysis, and interpretation of results.
Élia Mateus: Interpretation of results, and critical revision for important intellectual content.
Fernando Nolasco: Critical revision for important intellectual content.
Eduardo Barroso: Critical revision for important intellectual content.
All authors gave approval for the submitted version.
References
1. Adam R, Karam V, Delvart V, O'Grady J, Mirza D, Klempnauer J, et al. Evolution of indications and results of liver transplantation in Europe. A report from the European Liver Transplant Registry (ELTR). J Hepatol 2012;57(3):675-88. [ Links ]
2. Ferreira LG, Santos LF, Anastácio LR, Lima AS, Correia MI. Resting energy expenditure, body composition, and dietary intake: A longitudinal study before and after liver transplantation. Transplantation 2013;96(6):579-85. [ Links ]
3. Plank LD, Metzger DJ, McCall JL, Barclay KL, Gane EJ, Streat SJ, et al. Sequential changes in the metabolic response to orthotopic liver transplantation during the first year after surgery. Ann Surg 2001;234(2):245-55. [ Links ]
4. Tietge UJ, Bahr MJ, Manns MP, Böker KH. Decreased splanchnic oxygen uptake and increased systemic oxygen uptake in cirrhosis are normalized after liver transplantation. Liver Transpl 2001;7(12):1015-22. [ Links ]
5. Sugihara K, Yamanaka-Okumura H, Teramoto A, Urano E, Katayama T, Morine Y, et al. Recovery of nutritional metabolism after liver transplantation. Nutrition 2015;31(1):105-10. [ Links ]
6. Anastácio LR, Diniz KG, Ribeiro HS, Ferreira LG, Lima AS, Correia MI, et al. Prospective evaluation of metabolic syndrome and its components among long-term liver recipients. Liver Int 2014;34(7):1094-101. [ Links ]
7. Mathur S, Peng S, Gane EJ, McCall JL, Plank LD. Hypermetabolism predicts reduced transplant-free survival independent of MELD and Child-Pugh scores in liver cirrhosis. Nutrition 2007;23(5):398-403. [ Links ]
8. Selberg O, Böttcher J, Tusch G, Pichlmayr R, Henkel E, Müller MJ. Identification of high- and low-risk patients before liver transplantation: A prospective cohort study of nutritional and metabolic parameters in 150 patients. Hepatology 1997;25(3):652-7. [ Links ]
9. Merli M, Giusto M, Gentili F, Novelli G, Ferretti G, Riggio O, et al. Nutritional status: Its influence on the outcome of patients undergoing liver transplantation. Liver Int 2010;30(2):208-14. [ Links ]
10. Ribeiro HS, Anastácio LR, Ferreira LG, Lima AS, Correia MI. Energy expenditure and balance among long term liver recipients. Clin Nutr 2014;33(6):1147-52. [ Links ]
11. Brito-Costa A, Pereira-da-Silva L, Papoila AL, Alves M, Mateus É, Nolasco F, et al. Factors associated with changes in body composition shortly after orthotopic liver transplantation: The potential influence of immunosuppressive agents. Transplantation 2016;100(8):1714-22. [ Links ]
12. Plauth M, Cabré E, Riggio O, Assis-Camilo M, Pirlich M, Kondrup J, et al. ESPEN Guidelines on Enteral Nutrition: Liver disease. Clin Nutr 2006;25(2):285-94. [ Links ]
13. Detsky AS, McLaughlin JR, Baker JP, Johnston N, Whittaker S, Mendelson RA, et al. What is subjective global assessment of nutritional status? J Parenter Enteral Nutr 1987;11(1):8-13. [ Links ]
14. Nieman DC, Austin MD, Benezra L, Pearce S, McInnis T, Unick J, et al. Validation of Cosmed's FitMate in measuring oxygen consumption and estimating resting metabolic rate. Res Sports Med 2006;14(2):89-96. [ Links ]
15. Harris J, Benedict F. A biometric study of human basal metabolism. Proc Natl Acad Sci USA 1918;4:370-3. [ Links ]
16. Krenitsky J. Nutrition update in hepatic failure. Practical Gastroenterol 2014(128):47-55. [ Links ]
17. Ferreira LG, Santos LF, Silva TR, Anastácio LR, Lima AS, Correia MI. Hyper- and hypometabolism are not related to nutritional status of patients on the waiting list for liver transplantation. Clin Nutr 2014;33(5):754-60. [ Links ]
18. Lohman T, Roche A, Martorell R. Anthropometric reference manual. Champaign, USA: Human Kinetics Books; 1991. [ Links ]
19. Moissl UM, Wabel P, Chamney PW, Bosaeus I, Levin NW, Bosy-Westphal A, et al. Body fluid volume determination via body composition spectroscopy in health and disease. Physiol Meas 2006;27(9):921-33. [ Links ]
20. Arena R. Muscular strength and muscular endurance. In: Pescatello L, Arena R, Riebe D, Thompson P, eds. ACSM's Guidelines for Exercise Testing and Prescription. 9th ed. Indianapolis: American College of Sports Medicine; 2014. pp. 94-104. [ Links ]
21. Briet F, Aqel BA, Jeejeebhoy KN, Scolapio JS. A low pretransplant peripheral blood mononuclear cell complex I activity predicts metabolic disturbances and inability to regain fat free mass in cirrhotic patients undergoing liver transplantation. Nutr Res 2009;29(1):26-34. [ Links ]
22. Mela D, Rogers P. Control of apetite and energy balance and imbalance. In: Hall Ca, ed. Food eating and obesity. London; 1998. [ Links ]
23. Hussaini SH, Oldroyd B, Stewart SP, Soo S, Roman F, Smith MA, et al. Effects of orthotopic liver transplantation on body composition. Liver 1998;18(3):173-9. [ Links ]
24. Merli M, Giusto M, Riggio O, Gentili F, Molinaro A, Attili AF, et al. Improvement of nutritional status in malnourished cirrhotic patients one year after liver transplantation. e-SPEN 2011;6(3):e142-e7. [ Links ]
25. Wagner D, Adunka C, Kniepeiss D, Jakoby E, Schaffellner S, Kandlbauer M, et al. Serum albumin, subjective global assessment, body mass index and the bioimpedance analysis in the assessment of malnutrition in patients up to 15 years after liver transplantation. Clin Transplant 2011;25(4):E396-400. [ Links ]
26. Schroeder D, Hill GL. Postoperative fatigue: A prospective physiological study of patients undergoing major abdominal surgery. Aust N Z J Surg 1991;61(10):774-9. [ Links ]
27. Boullata J, Williams J, Cottrell F, Hudson L, Compher C. Accurate determination of energy needs in hospitalized patients. J Am Diet Assoc 2007; 107(3):393-401. [ Links ]
28. Weinsier RL, Schutz Y, Bracco D. Reexamination of the relationship of resting metabolic rate to fat-free mass and to the metabolically active components of fat-free mass in humans. Am J Clin Nutr 1992;55(4):790-4. [ Links ]
29. Javed F, He Q, Davidson LE, Thornton JC, Albu J, Boxt L, et al. Brain and high metabolic rate organ mass: Contributions to resting energy expenditure beyond fat-free mass. Am J Clin Nutr 2010;91(4):907-12. [ Links ]
30. Tajika M, Kato M, Mohri H, Miwa Y, Kato T, Ohnishi H, et al. Prognostic value of energy metabolism in patients with viral liver cirrhosis. Nutrition 2002;18(3):229-34. [ Links ]
31. Campillo B, Bories PN, Pornin B, Devanlay M. Influence of liver failure, ascites, and energy expenditure on the response to oral nutrition in alcoholic liver cirrhosis. Nutrition 1997;13(7-8):613-21. [ Links ]
32. Kondrup J. Nutrition in end stage liver disease. Best Pract Res Clin Gastroenterol 2006;20(3):547-60. [ Links ]
33. Merli M, Leonetti F, Riggio O, Valeriano V, Ribaudo MC, Strati F, et al. Glucose intolerance and insulin resistance in cirrhosis are normalized after liver transplantation. Hepatology 1999;30(3):649-54. [ Links ]
34. Hill JO, Wyatt HR, Peters JC. Energy balance and obesity. Circulation 2012;126(1):126-32. [ Links ]
35. Cooling J, Blundell J. Differences in energy expenditure and substrate oxidation between habitual high fat and low fat consumers (phenotypes). Int J Obes Relat Metab Disord 1998;22(7):612-8. [ Links ]
36. Kalaitzakis E, Bosaeus I, Ohman L, Björnsson E. Altered postprandial glucose, insulin, leptin, and ghrelin in liver cirrhosis: Correlations with energy intake and resting energy expenditure. Am J Clin Nutr 2007;85(3):808-15. [ Links ]
37. Giusto M, Lattanzi B, Di Gregorio V, Giannelli V, Lucidi C, Merli M. Changes in nutritional status after liver transplantation. World J Gastroenterol 2014;20(31):10682-90. [ Links ]
38. Guglielmi F, Panella C, Buda A, Budillon G, Caregaro L, Clerici C, et al. Nutritional state and energy balance in cirrhotic patients with or without hypermetabolism multicentre prospective study by the "Nutritional Problems in Gastroenterology" section of the Italian Society of Gastroenterology (SIGE). Dig Liver Dis 2005;37(9):681-8. [ Links ]
39. Foster KR, Lukaski HC. Whole-body impedance - What does it measure? Am J Clin Nutr 1996;64(3 Suppl):388S-96S. [ Links ]
40. Lehnert ME, Clarke DD, Gibbons JG, Ward LC, Golding SM, Shepherd RW, et al. Estimation of body water compartments in cirrhosis by multiple-frequency bioelectrical-impedance analysis. Nutrition 2001;17(1):31-4. [ Links ]
![]() Correspondence:
Correspondence:
Ana Brito-Costa.
Nutrition and Dietetics Unit.
Hospital Curry Cabral.
Rua da Beneficência, 8.
1069-166 Lisbon, Portugal.
e-mail: anabritocosta@gmail.com
Received: 19/02/2017
Accepted: 05/04/2017