My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Nutrición Hospitalaria
On-line version ISSN 1699-5198Print version ISSN 0212-1611
Nutr. Hosp. vol.34 n.5 Madrid Sep./Oct. 2017
https://dx.doi.org/10.20960/nh.747
Inflammation and insulin resistance according to body composition in European adolescents: the HELENA study
Inflamación y resistencia a la insulina según composición corporal en adolescentes europeos: el estudio HELENA
Esther María González-Gil1,2,3,4, Luis Gracia-Marco5, Javier Santabárbara6, Denes Molnar7, Francisco José Amaro-Gahete8, Frederic Gottrand9, Aline Arouca10, Antonios Kafatos11, Kurt Widhalm12, Yannis Manios13, Alfonso Sian14, Marcela González-Gross4,15, Sonia Gómez-Martínez16, Ligia Esperanza Díaz16, Catherine Leclerq17, Jean Dallongeville18, Ascensión Marcos16 and Luis A. Moreno Aznar1,2,3,4; on behalf of the HELENA Study Group
1Growth, Exercise, Nutrition and Development (GENUD) Research Group. Faculty of Health Sciences. Universidad de Zaragoza. Zaragoza, Spain.
2Instituto Agroalimentario de Aragón (IA2). Zaragoza, Spain.
3Instituto de Investigación Sanitaria Aragón (IIS Aragón). Zaragoza, Spain.
4Centro de Investigación Biomédica en Red de Fisiopatología de la Obesidad y Nutrición (CIBERObn). Madrid, Spain.
5Children's Health and Exercise Research Centre. Sport and Health Sciences. University of Exeter. Exeter, United Kingdom.
6Department of Preventive Medicine and Public Health. Universidad de Zaragoza. Zaragoza, Spain.
7Department of Pediatrics. University of Pecs. Pecs, Hungary.
8Department of Medical Physiology. School of Medicine. Universidad de Granada. Granada, Spain.
9Univ Lille 2. INSERM U995. CHU-Lille, France.
10Department of Public Health. Ghent University. Ghent, Belgium.
11Preventive Medicine and Nutrition Unit. School of Medicine. University of Crete. Crete, Greece.
12Department of Pediatrics. Division of Clinical Nutrition. Medical University of Vienna. Vienna, Austria.
13Department of Nutrition and Dietetics. Harokopio University. Athens, Greece.
14Unit of Epidemiology and Population Genetics. Institute of Food Sciences. National Research Council. Avellino, Italy.
15ImFine Research Group. Facultad de Ciencias de la Actividad Física y del Deporte-INEF. Universidad Politécnica de Madrid. Madrid, Spain.
16Immunonutrition Group. Institute of Food Science, Technology and Nutrition. (ICTAN). Spanish National Research Council (CSIC). Madrid, Spain.
17CREA (Council for Agricultural Research and Economics). Research Center for Food and Nutrition. Rome, Italy.
18INSERM U1167. Institut Pasteur de Lille. Lille, France
The HELENA Study was supported by the European Community Sixth RTD Framework Programme (Contract FOOD-CT-2005-007034). This analysis was also supported by the Spanish Ministry of Science and Innovation (JCI-2010-07055) and the European Regional Development Fund (FEDER).
ABSTRACT
Introduction: Inflammation is related to insulin resistance in adults, especially on those individuals with high levels of body composition.
Objectives: The aim of this study is to assess the relationship between a set of inflammatory biomarkers and insulin resistance by levels of body composition in a sample of European adolescents.
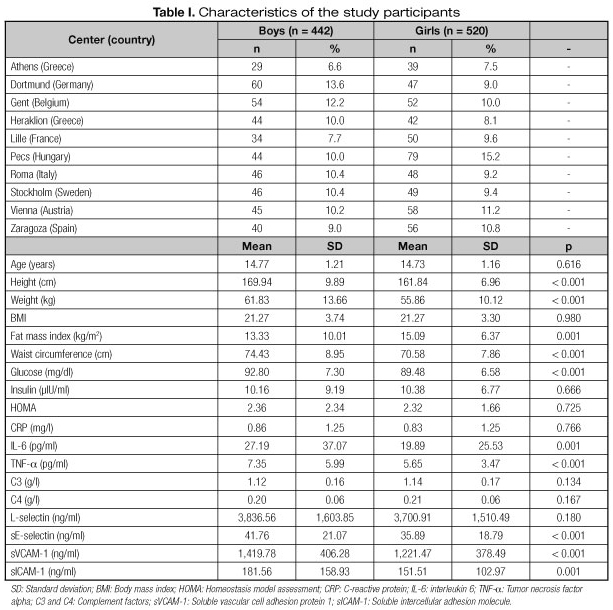
Material and methods: Nine hundred and sixty-two adolescents (442 boys and 520 girls) from nine European countries met the inclusion criteria of having measurements for the homeostasis model assessment (HOMA) and a set of inflammation-related biomarkers: C-reactive protein (CRP), tumor necrosis factor alpha (TNF-α), interleukin (IL-6), complement factors C3 and C4 and selected cell adhesion molecules. Body mass index (BMI), fat mass index (FMI) and waist circumference (WC) were categorized using tertiles. To assess the associations stratifying by body composition indexes, ANOVA and linear regression models were performed.
Results: Mean biomarkers' concentrations differed across BMI, FMI and WC tertiles (p < 0.05) by sex. In both sexes, insulin, HOMA, CRP, C3 and C4 were significantly different between categories (p < 0.001), always showing the highest mean concentration in the upper category of BMI, FMI and WC. The most consistent finding was an association between insulin resistance and C3 concentrations (p < 0.05), in the adolescents in the highest tertile of BMI, FMI and WC, except in the case of FMI in girls.
Conclusion: Inflammatory and glucose metabolism markers differed by tertiles of body composition, being usually higher in the highest tertile. C3 complement factor was associated with insulin resistance in adolescents, especially those with high total and abdominal adiposity.
Key words: Inflammation. Insulin resistance. Body composition. European adolescents.
RESUMEN
Introducción: la inflamación está relacionada con la resistencia a la insulina en adultos, especialmente en individuos con altos valores de composición corporal.
Objetivos: valorar la relación entre diferentes marcadores inflamatorios y la resistencia a la insulina según valores de composición corporal en adolescentes europeos.
Material y métodos: novecientos sesenta y dos adolescentes (442 chicos y 520 chicas) de nueve países europeos cumplían el criterio de inclusión de tener medidos la evaluación del modelo de homeostasis (HOMA) y diferentes marcadores inflamatorios: proteína C-reactiva (PCR), factor de necrosis tumoral alfa (TNF-α), interleukina (IL-6), factores de complemento C3 y C4 y moléculas de adhesión. El índice de masa corporal (IMC), el índice de masa grasa (IMG) y la circunferencia de cintura (CC) se categorizaron en tertiles. Para valorar las asociaciones por índices de composición corporal se realizó ANOVA y regresión.
Resultados: las concentraciones de los marcadores diferían entre los tertiles de IMC, IMG y CC (p < 0,05), por sexo. En ambos sexos, insulina, HOMA, PCR, C3 y C4 fueron significativamente diferentes entre categorías (p < 0,001), presentando la mayor concentración en la categoría superior de IMC, IMG y CC. El resultado más consistente para los adolescentes del tertil superior de IMC, IMG y CC fue la asociación entre resistencia a la insulina y concentraciones de C3 (p < 0,05), excepto para IMG en chicas.
Conclusión: los marcadores del metabolismo inflamatorio y de la glucosa diferían según tertiles de composición corporal, siendo mayores en el tertil superior. El C3 se asoció con resistencia a la insulina en adolescentes, especialmente en aquellos con adiposidad total y abdominal.
Palabras clave: Inflamación. Resistencia a la insulina. Composición corporal. Adolescentes europeos.
Introduction
Obesity is a condition in which the adipose tissue mass is increased due to a high adipocytes number and size (1). In obese individuals, the endocrine function of the adipose tissue is impaired and the adipocytes, as well as the pre-adipocytes, macrophages, and adipose stem cells, contribute to the production of pro-inflammatory cytokines (2). Both the hypertrophied adipocytes and other adipose tissue immune cells could lead to chronic inflammation through innate and adaptive immune responses (3). Additionally, the adipose tissue dysfunction is related to the development of co-morbidities such as insulin resistance, type 2 diabetes and cardiovascular diseases (4). The inflammatory state triggered by the impaired function of the adipose tissue also seems to be related with these comorbidities (5,6).
Among a large number of inflammatory-related biomarkers, C-reactive protein (CRP) has been the one most widely used. Hepatic synthesis of CRP, complement factors C3 and C4, depends on pro-inflammatory cytokines released by the adipose tissue such as tumor necrosis factor alpha (TNF-α) or interleukin 6 (IL-6) (7). Moreover, cell adhesion molecules are elevated during inflammatory conditions and have been suggested as markers for atherosclerosis (8).
Inflammation seems to be an important step in the pathogenesis of insulin resistance (IR) (9). In obese subjects, the inflammatory response can lead to altered insulin mediated signaling pathway by directly inhibiting insulin receptors (10). The relation between traditional inflammatory cells, cytokines, and chemokines and insulin resistance has been studied in adult populations (10). In children, obesity-related adipose tissue dysfunction develops early in childhood and is related to IR (11). In a previous study in adolescents, an association between IR and some inflammatory biomarkers was observed, and these relationships were stronger in obese subjects (12). However, another study performed in adolescents and young adults suggested that low grade inflammation did not appear to play a role in the development of IR (13). Due to these controversial findings, there is a need of further research in early stages of life, as it is a critical period for the development of future co-morbidities. Body composition, especially the body fat, could determine the associations between inflammatory markers and insulin resistance. Thus, the aim of this study is to assess the relationship between inflammatory markers and insulin resistance by body composition in a sample of European adolescents.
Material and methods
STUDY DESIGN
The HELENA study is a cross-sectional multi-center study (n = 3,528) conducted between 2006 and 2007 in ten European cities: Athens and Heraklion, in Greece; Dortmund, in Germany; Ghent, in Belgium; Lille, in France; Pecs, in Hungary; Rome, in Italy; Stockholm, in Sweden; Vienna, in Austria; and Zaragoza, in Spain. General procedures and methodology of the HELENA study have been previously described (14).
The study was performed according to the ethical guidelines of the Edinburgh revision of the 1964 Declaration of Helsinki (2000), the International Conferences on Harmonization for Good Clinical Practice and the legislation on clinical research from each of the participating countries. The local Ethics Committees of each center approved the protocol. Written informed consent was obtained from the adolescents and their parents.
STUDY SAMPLE
Blood collection was randomly performed in approximately a third of the total sample (n = 1,089, 31%). Nine hundred and sixty-two participants (442 boys and 520 girls) met the inclusion criteria of having measured the homeostasis model assessment (HOMA) and the set of biomarkers related with inflammation: TNF-α, IL-6, CRP, complement factors C3 and C4 and cell adhesion molecules: vascular cell adhesion molecule-1 (sVCAM-1), intercellular adhesion molecule-1 (sICAM-1), sE-selectin and L-selectin.
PHYSICAL MEASUREMENTS
Weight and height were measured in underwear and barefoot with an electronic scale (SECA 861, Seca Ltd., Birmingham, UK) and a stadiometer (SECA 225, Seca Ltd.). In addition, body mass index (BMI) was calculated as body weight in kilograms divided by the square of height in meters. From skinfolds, percentage of fat was calculated using the Slaughter formula and then the fat mass index was also calculated (kg/m2) (15). Waist circumference was measured with an un-elastic tape. All anthropometric measures were taken following a standardized protocol. Inter-observer reliability for skinfold thicknesses and circumferences measurements was always greater than 90% (16).
BLOOD ANALYSIS
Blood withdrawal was performed after 12 hours overnight fast. C-reactive protein (CRP) levels were quantified by immunoturbidimetry (AU 2700, Olimpus, Rungis, France). Adolescents with higher CRP concentrations than 10 mg/dl were excluded. Serum C3 and C4 complement factors were analyzed by nephelometry (Behring Diagnostics, CA, USA). The coefficient of variation (inter-assay precision) was 1.9% for CRP, 1.4% for C3, and 1.2% for C4. Serum cytokines IL-6 and TNF-α were determined using the High Sensitivity Human Cytokine MILLIPLEX™ MAP kit (Millipore Corp., Billerica, MA, USA) and collected by flow cytometry (Luminex-100 v.2.3, Luminex Corporation, Austin, TX, USA). The intra- and inter-assay precision CVs were: 3.5% and 4.5%, respectively, for IL-6; and 3.5% and 3.8%, respectively, for TNF-α. Detection limits (sensitivity) for all the analyses were 0.007 mg/l for CRP, 0.01 g/l for C3, 0.002 g/l for C4, 0.1 pg/ml for IL-6, and 0.05 pg/ml for TNF-α. Undetectable values were recorded as the specific detection limit. Children with values of 0.12 pg/ml for TNF-α and IL-6 were excluded as it was an assigned value for children with concentration values under the detection curve. The serum adhesion molecule sL-selectin was analyzed through commercial ELISA kits (Diaclone, France); the sensitivity of this kit was less than 1 ng/mL for L-selectin. The analyzed by Universal Microplate Spectophotometer (Power WaveTM XS, Biotek® Instruments, INC USA).
The multiplex assay kit was used to detect for the simultaneous quantification of the molecules sE-Selectin, sVCAM-1, sICAM-1, in serum. The samples were analyzed by citometry (Luminex® 100). The sensitivities of these assays were: Min DC 0.079 ng/ml for sE-Selectin, 0.016 ng/ml for sVCAM-1 and 0.009 ng/ml for sICAM-1. The intra-assay CVs were 11.2% for sE-Selectin, 4.5% for sVCAM-1 and 7.9% for sICAM-1.
STATISTICAL ANALYSIS
Analyses were performed separately for boys and girls. Normality of distributions was assessed with the Kolmogorov-Smirnov test. Glucose, insulin, HOMA, CRP, IL6, TNF-α, L-selectin, sE-selectin and sICAM were normalized by natural logarithm transformation. t-tests were used for comparisons of continuous variables by sex.
For BMI, FMI and WC, the standardized regression residuals by age were calculated and then categorized in tertiles.
Analysis of variance (ANOVA) with Bonferroni post-hoc correction was applied to compare mean differences of each biomarker between the categories of each indicator of body composition.
Finally, multiple linear regressions were performed to assess the association between HOMA-IR (dependent) and each marker of inflammation (independent) adjusted by center within body composition tertiles (BMI, FMI or WC). The effect of the city of residence was controlled in all regressions by using dummy variables. The dependent variable, HOMA-IR, was log-transformed. For markers of inflammation that were also log-transformed, results were expressed as percentage of change of the geometrical mean of the HOMA-IR for 10% increases of the corresponding biomarker. If the independent variable was normally distributed, results were expressed as percentage of change of the geometrical mean of HOMA per unit increase of the corresponding biomarker.
Data were managed and analyzed with the IBM SPSS Statistics v.21 (IBM Corp., New York, NY, USA, 2012).
Results
Descriptive characteristics are presented in table I. Boys were significantly taller and heavier than girls and had significantly higher values of waist circumference, whereas girls had higher levels of FMI. Regarding the biomarkers measured, boys had significantly higher concentrations of glucose, HOMA, IL-6, TNF-α, L-selectin, sE-selectin, sVCAM-1 and sICAM-1 than girls, while girls had significantly higher levels of insulin and C3 than boys.
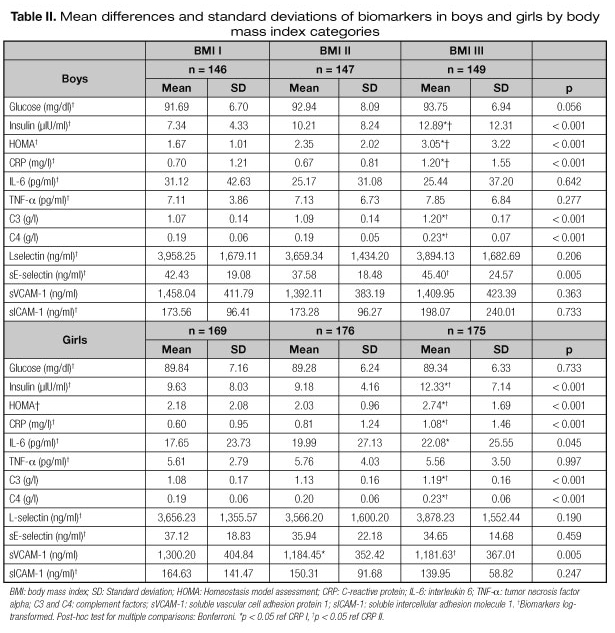
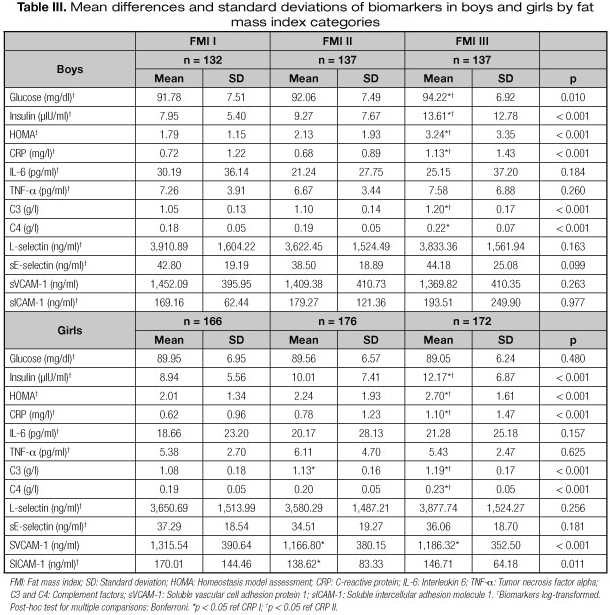
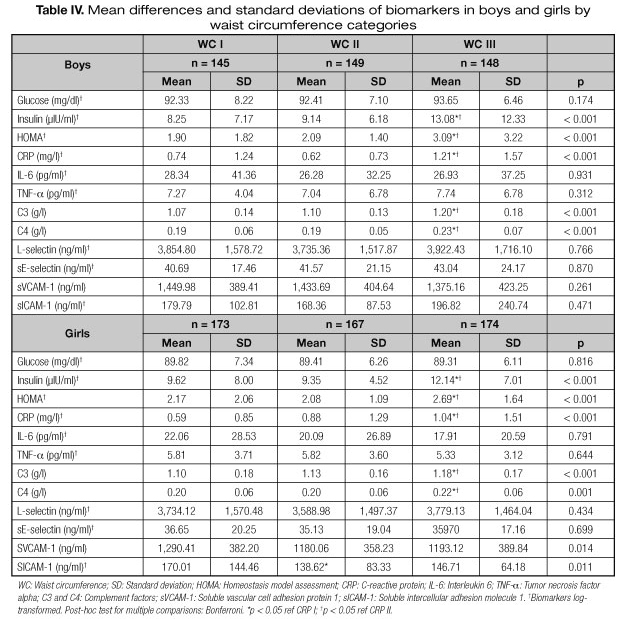
Results for the ANOVA are presented in tables II, III and IV. Significant differences were found in mean values of the measured biomarkers across categories of each marker of body composition, by sex. When BMI increased, the mean concentrations of insulin and HOMA in boys, CRP in girls and C3 and C4 in both sexes significantly increased (p < 0.001). In addition, CRP and sE-selectin in boys, and insulin, HOMA and sVCAM-1 in girls presented also significantly different mean values across categories. Also, when FMI increased, the mean levels of glucose in boys, CRP in girls and insulin, HOMA, C3 and C4 in both sexes, significantly increased (p < 0.001). Additionally, CRP in boys and sVCAM-1 presented significant differences (p < 0.001) across categories. Finally, when WC increased, the mean level of insulin in boys and CRP in girls increased (p < 0.001); HOMA, CRP, C3 and C4 significantly increased (p < 0.001) in both sexes. Mean values of sVCAM-1 presented as well significant differences (p = 0.024) between waist circumference categories, but only in girls.
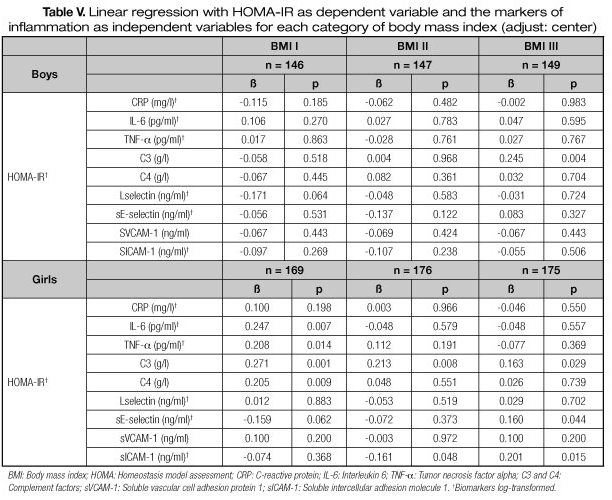
Tables V, VI and VII show results of the linear regression between HOMA-IR and the markers of inflammation for each body composition index. Table V shows the results of the linear regression by tertiles of BMI. In the lowest tertile, in girls, HOMA-IR increased in a 31.1% and 22.7% per each additional g/l of C3 and C4, respectively, and in 2.0% and in 2.4% when TNF-α and IL-6 increases by 10%, respectively. Also, in the second tertile of BMI, we expect about 23.7% increase in HOMA-IR per each additional g/l of C3 and about 14.8% decrease in HOMA-IR per each additional ng/ml of sICAM-1. Moreover, in the highest tertile of BMI, in girls, HOMA-IR increased by 22.3% and 17.7% per each additional ng/ml of sICAM-1 and 1.5% when sE-selectin increases by 10%. Significant associations between HOMA-IR and C3 in the highest tertile of BMI in both sexes were found: HOMA-IR increased in a 17.7% per each additional g/l of C3 in girls and 27.7% per each additional g/l of C3 in boys.
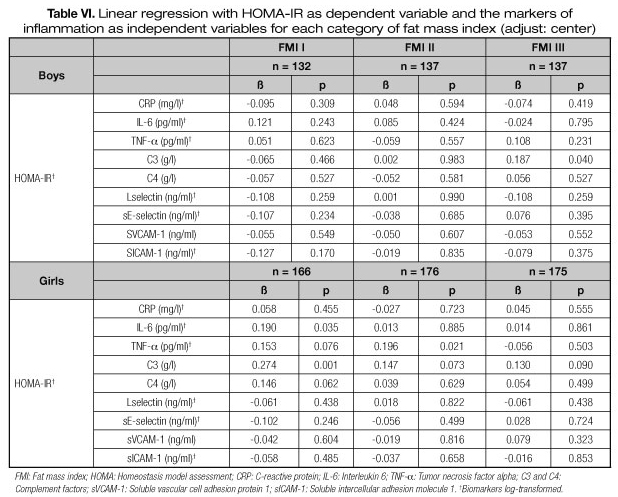
Table VI presents the results of the linear regression between HOMA-IR and the markers of inflammation by tertiles of FMI. In the lowest tertile, HOMA-IR increased in 1.8% when IL-6 increased 10% and in 31.5% per each additional g/l of C3 in girls. Furthermore, in the second tertile of FMI, we expect about 1.9% increase in HOMA-IR when TNF-α increases by 10% in girls. Moreover, in the highest tertile of BMI, HOMA-IR increased by 20.5% per each additional g/l of C3 in boys.
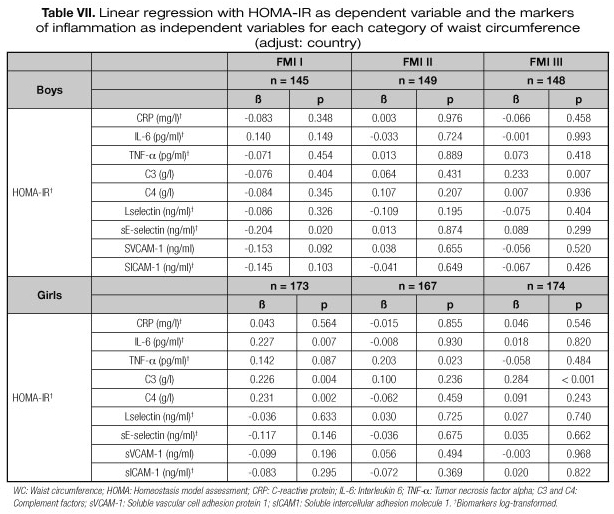
Finally, table VII shows the results of the linear regression by tertiles category of WC. In the lowest tertile of WC, HOMA-IR decreased 1.9% when sE-selectin increased 10% for boys; while in girls, HOMA-IR increased 2.2% when IL-6 increased 10% and 25.3% and 25.9% per each additional g/l of C3 and C4, respectively. In the second tertile of waist circumference, HOMA-IR increases 1.9% when TNF-α increases 10% in girls. In the highest tertile of waist circumference, significant associations between C3 were observed in both sexes: HOMA-IR increased 24.9% and 32.8% per each additional g/l of C3 in boys and girls, respectively.
Discussion
The main finding of this study is the consistent significant association between C3 complement factor and insulin resistance, irrespective of total and abdominal fat deposition. To our knowledge, this is the first study assessing the relationship between different inflammatory markers and insulin resistance in a relatively large sample of European adolescents from different cities.
In our sample, mean concentrations of glucose, insulin, HOMA and some inflammatory markers such as CRP or C3 and C4 were significantly higher in the highest tertile of each marker of body composition. Even in children, obesity has been related to low-grade inflammation (17). Adipocyte hypertrophy has been associated with HOMA- insulin resistance and inflammation in obese children (11). Results from our study also support the hypothesis that, even in adolescence, there is a link between adiposity, glucose metabolism and inflammation as some of these biomarkers were increased in the highest levels of total and abdominal adiposity.
Furthermore, in the present study there were linear associations between some inflammatory markers and HOMA as dependent variable, by categories of body composition indices. Previous studies suggest that inflammatory markers can interfere with insulin action by directly inhibiting insulin receptors (18). However, there are some discrepancies between studies regarding the relationship between inflammation and insulin resistance in adolescents (12,13). A recent study in obese adolescents failed to show a significant relation between obesity and IR mediated by low-grade inflammation using traditional inflammatory markers (19).
Although previous studies have associated some traditional inflammatory biomarkers with the development of diabetes or insulin resistance (20-22), we did not find any relationship between the traditional inflammatory markers and the HOMA for adolescents with the highest levels of BMI, FMI and WC. Out of all the inflammatory markers measured in the present study, only C3 was consistently related with insulin resistance, measured by HOMA, especially in the highest tertiles of total and abdominal adiposity, except FMI in girls. Our results are in line with those of some previous studies. Serum C3 was the strongest inflammatory marker related to insulin resistance in a study in an elderly population (23). This complement factor is an emerging cardio metabolic risk factor related to some comorbities such as type 2 diabetes (24). In a sample of Spanish adolescents, serum C3 levels were associated with body fat, especially with abdominal obesity (25), and were higher in adult subjects with insulin resistance (26). A previous study performed in adults showed that low-grade inflammation and insulin resistance might represent two independent pathways by which body fat leads to elevated C3 (27). However, it seems that changes in C3 levels over a 7-year follow-up period were associated with changes in several measures of insulin resistance and that baseline C3 was associated with the 7-year incidence of type 2 diabetes (28). Although the main production of the C3 is in the liver, C3 is also synthesized by activated macrophages (29) and adipocytes (30) as an inflammatory cytokine or an adipokine. Its hepatic production is induced by cytokines, such as interleukin-6 and TNF-α (31), which may interfere with insulin receptor functioning and cause insulin resistance (32). In addition, the complement system is a regulator of both the innate and adaptive system and, as a part of the inflammatory response, could also contribute to insulin resistance. We also found associations between insulin resistance and C4 complement factor in girls. Production of C4 depends, as production of C3, on proinflammatory cytokines released by the adipose tissue such as tumor necrosis factor alpha (TNF-α) or interleukin 6 (IL-6) (7). However, literature on the relationship between C4 and insulin resistance is scarce.
In our study, associations between TNF-α and insulin resistance were found in girls, in the lowest tertiles of body fat composition but not in the highest tertiles. TNF-α interferes negatively with the insulin signaling pathway, but also induces insulin resistance indirectly by altering adipocyte differentiation and adipocyte lipid metabolism (10).
This study has strengths as well as some limitations. First, its cross-sectional design, which does not allow drawing conclusions on causality; however, in adults it was observed that C3 was associated with the development of insulin resistance in a longitudinal study (28). Furthermore, the study is limited by the fact that blood samples only reflect inflammation, glucose and insulin concentrations at a given specific time point. On the other hand, the strengths of the study are: the use of traditional and non-traditional inflammatory markers that could be also involved in the pathogenesis of insulin resistance, and the use of standardized and harmonized information on body composition of adolescents from nine European countries.
Conclusions
In conclusion, results from the current study show that there is an association between C3 and HOMA in a multicenter sample of adolescents, especially in those with high levels of total and abdominal adiposity. To avoid chronic insulin resistance, efforts should be made to reduce deposition of total and abdominal fat in obese children and adolescents. This may impact on the reduction of serum C3 concentrations and prevent future insulin-related diseases such as diabetes. Longitudinal studies assessing this relationship between C3 and insulin resistance are needed to confirm these results.
Acknowledgements
Thanks to Anke Carstensen, Rosa María Torres and Ulrike Albers for the laboratory work.
Authors contributions
FG, MGG, AK, KW, JD, YS, DM, AM and LAM designed and directed the study; SGM, LED and AM conducted and directed the laboratory analysis; EMGG performed the statistical analysis and wrote the manuscript. EMGG, LGM, JS, DM, FA, FG, AA, AK, KW, YM, AS, MGG, SGM, LED, CL, JD, AM and LAM critically reviewed the manuscript.
HELENA Study Group
Co-ordinator: Luis A. Moreno.
Core Group members: Luis A. Moreno, Fréderic Gottrand, Stefaan De Henauw, Marcela González-Gross, Chantal Gilbert.
Steering Committee: Anthony Kafatos (President), Luis A. Moreno, Christian Libersa, Stefaan De Henauw, Sara Castelló, Fréderic Gottrand, Mathilde Kersting, Michael Sjöstrom, Dénes Molnár, Marcela González-Gross, Jean Dallongeville, Chantal Gilbert, Gunnar Hall, Lea Maes, Luca Scalfi.
Project Manager: Pilar Meléndez.
1. Universidad de Zaragoza (Spain)
Luis A. Moreno, Jesús Fleta, José A. Casajús, Gerardo Rodríguez, Concepción Tomás, María I. Mesana, Germán Vicente-Rodríguez, Adoración Villarroya, Carlos M. Gil, Ignacio Ara, Juan Fernández Alvira, Gloria Bueno, Aurora Lázaro, Olga Bueno, Juan F. León, Jesús Ma Garagorri, Manuel Bueno, Idoia Labayen, Iris Iglesia, Silvia Bel Serrat, Luis A. Gracia Marco, Theodora Mouratidou, Alba Santaliestra-Pasías, Iris Iglesia, Esther M. González-Gil, Pilar De Miguel-Etayo, Cristina Julián Almárcegui, Mary Miguel-Berges, Isabel Iguacel.
2. Consejo Superior de Investigaciones Científicas (Spain)
Ascensión Marcos, Julia Wärnberg, Esther Nova, Sonia Gómez, Ligia Esperanza Díaz, Javier Romeo, Ana Veses, Belén Zapatera, Tamara Pozo, David Martínez.
3. Université de Lille 2 (France)
Laurent Beghin, Christian Libersa, Frédéric Gottrand, Catalina Iliescu, Juliana Von Berlepsch.
4. Research Institute of Child Nutrition Dortmund, Rheinische Friedrich-Wilhelms-Universität Bonn
Mathilde Kersting, Wolfgang Sichert-Hellert, Ellen Koeppen.
5. Pécsi Tudományegyetem (University of Pécs) (Hungary)
Dénes Molnar, Eva Erhardt, Katalin Csernus, Katalin Török, Szilvia Bokor, Mrs. Angster, Enikö Nagy, Orsolya Kovács, Judit Répasi.
6. University of Crete School of Medicine (Greece)
Anthony Kafatos, Caroline Codrington, María Plada, Angeliki Papadaki, Katerina Sarri, Anna Viskadourou, Christos Hatzis, Michael Kiriakakis, George Tsibinos, Constantine Vardavas, Manolis Sbokos, Eva Protoyeraki, Maria Fasoulaki.
7. Institut für Ernährungs- und Lebensmittelwissenschaften -Ernährungphysiologie. Rheinische Friedrich Wilhelms Universität
Peter Stehle, Klaus Pietrzik, Marcela González-Gross, Christina Breidenassel, Andre Spinneker, Jasmin Al-Tahan, Miriam Segoviano, Anke Berchtold, Christine Bierschbach, Erika Blatzheim, Adelheid Schuch, Petra Pickert.
8. University of Granada (Spain)
Manuel J. Castillo, Ángel Gutiérrez, Francisco B Ortega, Jonatan R Ruiz, Enrique G Artero, Vanesa España, David Jiménez-Pavón,Palma Chillón, Cristóbal Sánchez-Muñoz, Magdalena Cuenca
9. Istituto Nazionalen di Ricerca per gli Alimenti e la Nutrizione (Italy)
Davide Arcella, Elena Azzini, Emma Barrison, Noemi Bevilacqua, Pasquale Buonocore, Giovina Catasta, Laura Censi, Donatella Ciarapica, Paola D'Acapito, Marika Ferrari, Myriam Galfo, Cinzia Le Donne, Catherine Leclercq, Giuseppe Maiani, Beatrice Mauro, Lorenza Mistura, Antonella Pasquali, Raffaela Piccinelli, Angela Polito, Romana Roccaldo, Raffaella Spada, Stefania Sette, Maria Zaccaria.
10. University of Napoli "Federico II" Dept of Food Science (Italy)
Luca Scalfi, Paola Vitaglione, Concetta Montagnese.
11. Ghent University (Belgium)
Ilse De Bourdeaudhuij, Stefaan De Henauw, Tineke De Vriendt, Lea Maes, Christophe Matthys, Carine Vereecken, Mieke de Maeyer, Charlene Ottevaere, Inge Huybrechts.
12. Medical University of Vienna (Austria)
Kurt Widhalm, Katharina Phillipp, Sabine Dietrich.
13. Harokopio University (Greece)
Yannis Manios, Eva Grammatikaki, Zoi Bouloubasi, Tina Louisa Cook, Sofia Eleutheriou, Orsalia Consta, George Moschonis, Ioanna Katsaroli, George Kraniou, Stalo Papoutsou, Despoina Keke, Ioanna Petraki, Elena Bellou, Sofia Tanagra, Kostalenia Kallianoti, Dionysia Argyropoulou, Stamatoula Tsikrika, Christos Karaiskos.
14. Institut Pasteur de Lille (France)
Jean Dallongeville, Aline Meirhaeghe.
15. Karolinska Institutet (Sweden)
Michael Sjöstrom, Jonatan R Ruiz, Francisco B. Ortega, María Hagströmer, Anita Hurtig Wennlöf, Lena Hallström, Emma Patterson, Lydia Kwak, Julia Wärnberg, Nico Rizzo.
16. Asociación de Investigación de la Industria Agroalimentaria (Spain)
Jackie Sánchez-Molero, Sara Castelló, Elena Picó, Maite Navarro, Blanca Viadel, José Enrique Carreres, Gema Merino, Rosa Sanjuán, María Lorente, María José Sánchez.
17. Campden BRI (United Kingdom)
Chantal Gilbert, Sarah Thomas, Elaine Allchurch, Peter Burgess.
18. SIK - Institutet foer Livsmedel och Bioteknik (Sweden)
Gunnar Hall, Annika Astrom, Anna Sverkén, Agneta Broberg.
19. Meurice Recherche & Development asbl (Belgium)
Annick Masson, Claire Lehoux, Pascal Brabant, Philippe Pate, Laurence Fontaine.
20. Campden & Chorleywood Food Development Institute (Hungary)
Andras Sebok, Tunde Kuti, Adrienn Hegyi.
21. Productos Aditivos SA (Spain)
Cristina Maldonado, Ana Llorente.
22. Cárnicas Serrano SL (Spain)
Emilio García.
23. Cederroth International AB (Sweden)
Holger von Fircks, Marianne Lilja Hallberg, Maria Messerer.
24. Lantmännen Food R&D (Sweden)
Mats Larsson, Helena Fredriksson, Viola Adamsson, Ingmar Börjesson.
25. European Food Information Council (Belgium)
Laura Fernández, Laura Smillie, Josephine Wills.
26. Universidad Politécnica de Madrid (Spain)
Marcela González-Gross, Raquel Pedrero-Chamizo, Agustín Meléndez, Jara Valtueña, David Jiménez-Pavón, Ulrike Albers, Pedro J. Benito, Juan José Gómez Lorente, David Cañada, Alejandro Urzanqui, Rosa María Torres, Paloma Navarro.
References
1. Greenberg AS, Obin MS. Obesity and the role of adipose tissue in inflammation and metabolism. Am J Clin Nutr 2006;83(2):461S-5S. DOI: 83/2/461S. [ Links ]
2. Murdolo G, Smith U. The dysregulated adipose tissue: A connecting link between insulin resistance, type 2 diabetes mellitus and atherosclerosis. Nutr Metab Cardiovasc Dis 2006;16(Suppl 1):S35-8. DOI: S0939-4753(05)00246-2(pii)10.1016/j.numecd.2005.10.016. [ Links ]
3. Ghigliotti G, Barisione C, Garibaldi S, Fabbi P, Brunelli C, Spallarossa P, et al. Adipose tissue immune response: Novel triggers and consequences for chronic inflammatory conditions. Inflammation 2014;37(4):1337-53. DOI: 10.1007/s10753-014-9914-1. [ Links ]
4. Hajer GR, Van Haeften TW, Visseren FL. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur Heart J 2008;29(24):2959-71. DOI: 10.1093/eurheartj/ehn387ehn387. [ Links ]
5. Hotamisligil GS. Inflammation and metabolic disorders. Nature 2006;444(7121):860-7. DOI: nature05485(pii)10.1038/nature05485. [ Links ]
6. Libby P, Ridker PM, Maseri A. Inflammation and atherosclerosis. Circulation 2002;105(9):1135-43. [ Links ]
7. Trayhurn P, Wood IS. Adipokines: Inflammation and the pleiotropic role of white adipose tissue. Br J Nutr 2004;92(3):347-55. DOI: S0007114504001795. [ Links ]
8. Hwang SJ, Ballantyne CM, Sharrett AR, Smith LC, Davis CE, Gotto AM Jr, et al. Circulating adhesion molecules VCAM-1, ICAM-1, and E-selectin in carotid atherosclerosis and incident coronary heart disease cases: The Atherosclerosis Risk In Communities (ARIC) study. Circulation 1997;96(12):4219-25. [ Links ]
9. Shoelson SE, Lee J, Goldfine AB. Inflammation and insulin resistance. J Clin Invest 2006;116(7):1793-801. DOI: 10.1172/JCI29069. [ Links ]
10. Makki K, Froguel P, Wolowczuk I. Adipose tissue in obesity-related inflammation and insulin resistance: Cells, cytokines, and chemokines. ISRN Inflamm 2013;2013:139239. DOI: 10.1155/2013/139239. [ Links ]
11. Landgraf K, Rockstroh D, Wagner IV, Weise S, Tauscher R, Schwartze JT, et al. Evidence of early alterations in adipose tissue biology and function and its association with obesity-related inflammation and insulin resistance in children. Diabetes 2015;64(4):1249-61. DOI: 10.2337/db14-0744db14-0744. [ Links ]
12. Aguilar MJ, González-Jiménez E, Antelo A, Perona JS. Insulin resistance and inflammation markers: Correlations in obese adolescents. J Clin Nurs 2013;22(13-14):2002-10. DOI: 10.1111/jocn.12034. [ Links ]
13. Adabimohazab R, Garfinkel A, Milam EC, Frosch O, Mangone A, Convit A. Does inflammation mediate the association between obesity and insulin resistance? Inflammation 2016;39(3):994-1003. DOI: 10.1007/s10753-016-0329-z10.1007/s10753-016-0329-z. [ Links ]
14. Moreno LA, De Henauw S, González-Gross M, Kersting M, Molnar D, Gottrand F, et al. Design and implementation of the Healthy Lifestyle in Europe by Nutrition in Adolescence Cross-Sectional Study. Int J Obes (Lond) 2008;32(Suppl 5):S4-11. DOI: 10.1038/ijo.2008.177ijo2008177. [ Links ]
15. Slaughter MH, Lohman TG, Boileau RA, Horswill CA, Stillman RJ, Van Loan MD, et al. Skinfold equations for estimation of body fatness in children and youth. Hum Biol 1988;60(5):709-23. [ Links ]
16. Nagy E, Vicente-Rodríguez G, Manios Y, Beghin L, Iliescu C, Censi L, et al. Harmonization process and reliability assessment of anthropometric measurements in a multicenter study in adolescents. Int J Obes (Lond) 2008;32(Suppl 5):S58-65. DOI: 10.1038/ijo.2008.184ijo2008184. [ Links ]
17. Tam CS, Clement K, Baur LA, Tordjman J. Obesity and low-grade inflammation: A paediatric perspective. Obes Rev 2010;11(2):118-26. DOI: 10.1111/j.1467-789X.2009.00674.xOBR674. [ Links ]
18. Wellen KE, Hotamisligil GS. Inflammation, stress, and diabetes. J Clin Invest 2005;115(5):1111-9. DOI: 10.1172/JCI25102. [ Links ]
19. Cohen JI, Maayan L, Convit A. Preliminary evidence for obesity-associated insulin resistance in adolescents without elevations of inflammatory cytokines. Diabetol Metab Syndr 2012;4(1):26. DOI: 10.1186/1758-5996-4-261758-5996-4-26. [ Links ]
20. Bertoni AG, Burke GL, Owusu JA, Carnethon MR, Vaidya D, Barr RG, et al. Inflammation and the incidence of type 2 diabetes: The Multi-Ethnic Study of Atherosclerosis (MESA). Diabetes Care 2010;33(4):804-10. DOI: 10.2337/dc09-1679dc09-1679. [ Links ]
21. Glowinska B, Urban M, Peczynska J, Florys B. Soluble adhesion molecules (sICAM-1, sVCAM-1) and selectins (sE selectin, sP selectin, sL selectin) levels in children and adolescents with obesity, hypertension, and diabetes. Metabolism 2005;54(8):1020-6. DOI: S0026049505001277(pii)10.1016/j.metabol.2005.03.004. [ Links ]
22. Pradhan AD, Manson JE, Rifai N, Buring JE, Ridker PM. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA 2001;286(3):327-34. DOI: joc10096. [ Links ]
23. Muscari A, Antonelli S, Bianchi G, Cavrini G, Dapporto S, Ligabue A, et al. Serum C3 is a stronger inflammatory marker of insulin resistance than C-reactive protein, leukocyte count, and erythrocyte sedimentation rate: Comparison study in an elderly population. Diabetes Care 2007;30(9):2362-8. DOI: dc07-0637(pii)10.2337/dc07-0637. [ Links ]
24. Engstrom G, Hedblad B, Eriksson KF, Janzon L, Lindgarde F. Complement C3 is a risk factor for the development of diabetes: A population-based cohort study. Diabetes 2005;54(2):570-5. DOI: 54/2/570. [ Links ]
25. Warnberg J, Nova E, Moreno LA, Romeo J, Mesana MI, Ruiz JR, et al. Inflammatory proteins are related to total and abdominal adiposity in a healthy adolescent population: The AVENA Study. Am J Clin Nutr 2006;84(3):505-12. DOI: 84/3/505. [ Links ]
26. Weyer C, Tataranni PA, Pratley RE. Insulin action and insulinemia are closely related to the fasting complement C3, but not acylation stimulating protein concentration. Diabetes Care 2000;23(6):779-85. [ Links ]
27. Wlazlo N, Van Greevenbroek MM, Ferreira I, Jansen EJ, Feskens EJ, Van der Kallen CJ, et al. Low-grade inflammation and insulin resistance independently explain substantial parts of the association between body fat and serum C3: The CODAM study. Metabolism 2012;61(12):1787-96. DOI: 10.1016/j.metabol.2012.05.015S0026-0495(12)00212-0. [ Links ]
28. Wlazlo N, Van Greevenbroek MM, Ferreira I, Feskens EJ, Van der Kallen CJ, Schalkwijk CG, et al. Complement factor 3 is associated with insulin resistance and with incident type 2 diabetes over a 7-year follow-up period: The CODAM Study. Diabetes Care 2014;37(7):1900-9. DOI: 10.2337/dc13-2804dc13-2804. [ Links ]
29. Zimmer B, Hartung HP, Scharfenberger G, Bitter-Suermann D, Hadding U. Quantitative studies of the secretion of complement component C3 by resident, elicited and activated macrophages. Comparison with C2, C4 and lysosomal enzyme release. Eur J Immunol 1982;12(5):426-30. DOI: 10.1002/eji.1830120513. [ Links ]
30. Choy LN, Rosen BS, Spiegelman BM. Adipsin and an endogenous pathway of complement from adipose cells. J Biol Chem 1992;267(18):12736-41. [ Links ]
31. Baumann H, Gauldie J. The acute phase response. Immunol Today 1994;15(2):74-80. DOI: 0167-5699(94)90137-6. [ Links ]
32. Marette A. Mediators of cytokine-induced insulin resistance in obesity and other inflammatory settings. Curr Opin Clin Nutr Metab Care 2002;5(4):377-83. [ Links ]
![]() Correspondence:
Correspondence:
Esther María González Gil.
Growth, Exercise, Nutrition and Development (GENUD) Research Group.
Facultad de Ciencias de la Salud.
Universidad de Zaragoza.
C/ Pedro Cerbuna, 12.
50009 Zaragoza, Spain.
e-mail: esthergg@unizar.es
Received: 16/11/2016
Accepted: 19/01/2017