INTRODUCTION
The diagnosis of metabolic syndrome (MetS) is understood as an important clinical resource to identify the risk of onset and development of cardiometabolic outcomes 1,2,3. MetS carriers are, respectively, two and five times more likely to present cardiovascular diseases and type II diabetes mellitus 1. MetS is identified when at least three of the following components are altered: abdominal fat, blood pressure, triglycerides, fasting plasma glucose, and HDL-cholesterol 1,2,3.
Although the cut-off points that define alterations in each component of MetS were initially proposed for use in adults, adaptations were proposed allowing the diagnosis to be made in the young population, and thus enabling intervention strategies delineated to minimize the possibility of occurrence of diabetes mellitus and cardiovascular diseases as early as possible 4,5,6.
In young people, the prevalence of MetS is naturally lower than in adults. Nevertheless, studies have shown proportions close to 5%, which in some cases may exceed 10%, depending on the demographic factors and diagnostic criteria adopted 7. In addition, nutritional status is capable of strongly impacting the prevalence of MetS. In a survey that gathered approximately 37 thousand Brazilian adolescents, the overall prevalence of MetS was equivalent to 2.6%; however, it reached 21.3% in obese youngsters 8.
In this perspective, investigating the factors associated with MetS in adolescents becomes relevant to knowledge in the area. Lifestyle-related determinants potentially associated with a higher risk of MetS are becoming increasingly frequent in adolescents, such as insufficient physical activity 9, a high time spent in sedentary activities 10 and inadequate dietary intake 11.
A meta-analysis study showed that adolescents with insufficient physical activity were 35% more likely to be diagnosed with MetS, while sedentary behavior, represented by recreational screen time, did not present a significant association. It should be considered that in both analyses limitations were identified, such as the practice of physical activity dichotomized in only two strata (low and high), when in order to present sufficient discriminatory capacity a minimum of three strata should be considered. In the case of sedentary behavior, only six studies were considered in the analysis, which does not allow for safe statements 12.
With regard to food consumption, the few studies that sought an association with MetS in adolescents found contradictory results 13,14,15,16. For example, the intake of sugary beverages increased the chances of presenting MetS by ten times 13, while consumption of soft drinks or fast food did not present a significant association 14. In addition, no associations were found between diet quality and MetS 15, whereas when investigating the consumption of different types of foods, an inverse association was identified only between fruit consumption and MetS 16).
Therefore, considering the importance of monitoring MetS from the earliest ages and the possibility of different factors influencing its diagnosis, the objective of the present study was to verify the association between lifestyle determinants (physical activity, sedentary behavior, and food consumption) and MetS, through control of demographic indicators and anthropometric nutritional status, in a sample of adolescents from southern Brazil.
METHODS
A cross-sectional survey was carried out in the city of Jacarezinho, Paraná, Brazil. The data collection extended from August to November 2014. The intervention protocols were approved by the Research Ethics Committee of the University of Paraná - UNOPAR (Opinion 1,302,963). All participants and their representatives signed a free and informed consent form.
SAMPLE AND SELECTION OF SUBJECTS
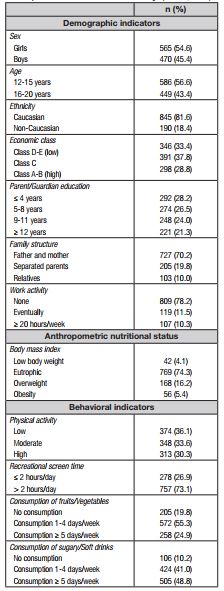
The reference population included adolescents of both sexes, between 12 and 20 years of age, enrolled in public and private elementary schools (6th to 9th grade) and high school (1st to 3rd year). The sample size was established assuming a 95% confidence interval, a sampling error of three percentage points, an increase of 10% to cover possible cases of losses and, as the sample planning involved conglomerates (school structure, sex, shift, and year of study), a delineation effect (deff) equivalent to 1.5 was added, initially providing a minimum sample of 1,000 participants. However, the final sample used in the data analysis was composed of 1,035 adolescents (565 girls and 470 boys).
The criteria adopted to exclude some adolescents drawn for the study were: a) refusal to participate in the study; b) non-confirmation by signing the free and informed consent form; c) any health problem that temporarily or permanently prevented participation in the study; d) the use of any type of medication that could induce changes in the study variables; e) being subjected to any type of specific diet; f) pregnancy; and g) non attendance at school on the day scheduled to begin data collection. In these cases, a new draw was carried out to recover any sample losses.
DATA COLLECTION
Anthropometric measures (weight, height, and waist circumference) and other components used to identify MetS were collected, as well as a questionnaire consisting of items distributed in four sections: demographic aspects, food consumption, physical activity, and sedentary behavior. The questionnaire was applied at a single moment, individually for each adolescent in the place and time of the classes.
Regarding the demographic aspects, in addition to sex and age, information related to ethnicity, economic class, parental/guardian education, family structure, and eventual work activity were collected. The family economic class was identified according to the guidelines proposed by the National Association of Research Companies 17. Information on food consumption was obtained using food items from the Youth Risk Behavior Survey (YRBS), translated, adapted, and validated for use in the Brazilian young population 18. In this case, the adolescents answered how often they consumed fruits/vegetables and sugary/soft drink products, taking as a reference the week prior to data collection. From the frequency of consumption reported, the following indicators were considered: no consumption, consumption 1-4 days/week and consumption ≥ 5 days/week.
The Physical Activity Questionnaire for Adolescents (PAQ-A) was used to measure the practice of physical activity. This questionnaire was translated and validated for use in young Brazilians 19. The PAQ-A consists of eight questions structured and directed to different aspects of the practice of physical activity in the previous seven days. The response options are coded using an increasing scale of 1 to 5 points, and the physical activity practice score is computed through the arithmetic mean of the scores assigned to each question. Subsequently, specific cut-off points for sex and age were used based on the distribution of tertiles. Thus, adolescents with PAQ-A ≤ 1st tertile scores were classified as having low physical activity, those with scores between the 1st and 2nd tertile as moderately active, and adolescents with scores ≥ 2nd tertile as having high physical activity.
Sedentary behavior was considered through recreational exposure to high screen time, with structured questions about watching TV and using a computer, video game, tablet, and smartphone in a typical week. For the responses, a predefined questionnaire was made available, in which the adolescent indicated their option from six categories, with options between "none" to "≥ 5 hours/day". The questions considered screen time equivalent to watching TV and using a computer, video game, tablet, and smartphone separately on weekdays and on weekends (Saturday and Sunday). Weighted means of the data on weekdays and weekends were used to identify the screen time per day reported by adolescents. Excessive screen time was defined by the combined use of TV and other screen devices for > 2 hours/day 20).
Regarding the anthropometric variables, measures of height, body weight, and waist circumference were performed according to a methodology described by the World Health Organization (WHO) 21. The body mass index (BMI) was calculated by the ratio between measurements of body mass in kilograms and height, expressed in square meters (kg/m2). The nutritional status of adolescents was determined according to sex and age cut-off points proposed by the International Obesity Task Force (IOTF): low body weight, eutrophic, overweight, and obesity 22.
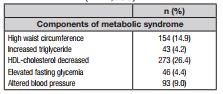
MetS was identified from the blood analysis of plasma lipids (triglycerides and HDL-C) and fasting blood glucose, resting blood pressure (systolic and diastolic), and abdominal fat accumulation (waist circumference), according to the criteria proposed by the International Diabetes Federation (IDF) 6. In this case, MetS is defined by the presence of a high waist circumference (< 16 years: both sexes ≥ percentile 90; ≥ 16 years: boys ≥ 90 cm and girls ≥ 80 cm) and at least two other compromised components: increased triglycerides (≥ 150 mg/dl), low HDL-C (< 16 years: both sexes < 40 mg/dl; ≥ 16 years: boys < 40 mg/dl and girls < 50 mg/dl), and altered blood pressure (systolic ≥ 130 mmHg or diastolic ≥ 85 mmHg).
STATISTICAL ANALYSIS
Prevalences observed in the outcome of interest (MetS) according to demographic indicators, nutritional status and lifestyle determinants (physical activity, sedentary behavior, and food consumption) are presented together with the respective 95% confidence intervals (95% CI). To analyze the linearity of the associations between MetS and the set of independent variables, prevalence ratio calculations were used. Statistical differences among the strata under investigation were treated by the Chi-square test (χ2). Subsequently, variables that indicated at least marginally significant associations (p ≤ 0.20) in the bivariate analysis were selected to be included in the hierarchical multiple regression procedures. In this case, the variables were treated in blocks, and the demographic aspects (level one) were the first to be entered in the model, followed by the anthropometric nutritional indicators (level two) and, finally, the lifestyle determinants (level three). The retrograde procedure was adopted (backward), in which all the variables that presented statistical significance (p < 0.05) remained in the multivariate model. Data were processed in the Statistical Package for Social Science (SPSS), version 22.
RESULTS
Descriptive data characterizing the study sample are provided in Table I. Girls represented 54.6% of the sample and the highest concentration of adolescents was between the ages of 12 and 15 years (56.6%). The majority of adolescents in the study were Caucasian (81.6%), living with their parents (70.2%), and reported not working (78.2%). Regarding the economic class and schooling of the parents/guardians, the proportions of adolescents were distributed proportionally in the strata considered. Furthermore, 21.6% of the adolescents demonstrated high body weight (overweight + obesity), approximately ⅓ were classified as low physically active (36.1%), and in each group of ten adolescents, seven reported staying > 2 hours in front of screen devices. Daily consumption of fruits and vegetables was reported by 24.9% of adolescents and 89.8% consumed sugary/soft drinks at least once a week. Regarding the individual components of MetS, decreased HDL-cholesterol was predominant (26.4%), while high fasting glycemia (4.4%) and high triglycerides (4.2%) were the least prevalent.
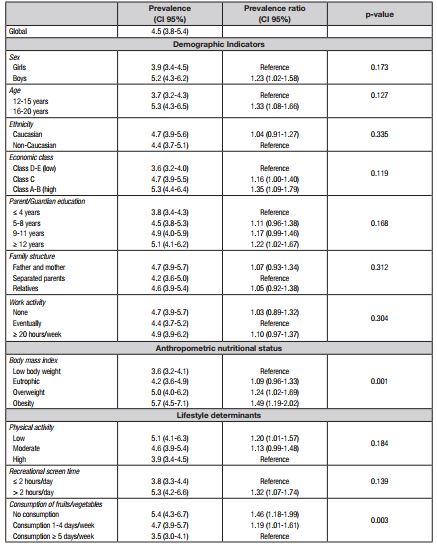
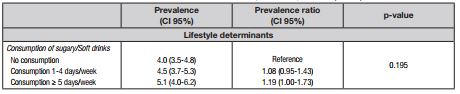
MetS prevalences with stratification for demographic indicators, anthropometric nutritional status, and lifestyle determinants are presented in Table II. The overall prevalence was equivalent to 4.5% (95% CI: 3.8-5.4). The bivariate analysis showed that, from the list of variables considered, differences in MetS prevalence stratified by ethnicity, family structure and labor activity were not considered as marginally significant (p ≤ 0.20).
Table II. Prevalence and prevalence ratio of metabolic syndrome with stratification for demographic indicators, nutritional status and determinants of the lifestyle of adolescents from Jacarezinho, Paraná, Brazil (2014)
Results of the hierarchical multiple regression are available in Table III. In the case of demographic indicators, the final model confirmed significant associations between MetS, age and economic class, indicating greater exposure among older adolescents and those belonging to the higher economic class. Likewise, anthropometric nutritional status remained significantly associated with MetS, in which obese adolescents presented a 62% greater chance of the outcome (OR = 1.62 [95% CI: 1.28-2.47]). Among the determinants of lifestyle, recreational screen time and fruit/vegetable consumption remained associated with MetS.
Table III. Hierarchical multiple logistic regression for demographic indicators (level 1), nutritional status (level 2) and determinants of lifestyle (level 3) of metabolic syndrome in adolescents from Jacarezinho, Paraná, Brazil (2014)

1Odds ratio not adjusted.
2Odds ratio adjusted by the other variables included in the model.
DISCUSSION
The objective of the study was to provide information on the association between lifestyle determinants (physical activity, sedentary behavior and food consumption) and MetS, through the control of demographic indicators and anthropometric nutritional status, in a sample of adolescents from southern Brazil. The main findings revealed an overall MetS prevalence of 4.5%. In addition, an independent association with MetS was found among: older adolescents, of higher economic class, who reported excessive screen time, with lower intake of fruits/vegetables, and who were obese.
Using the same diagnostic criteria (IDF), the prevalence of MetS observed in the current study was higher than that found in the young Brazilian population (4.5% vs 2.6%). However, when comparing the data of adolescents specifically from the interior of the southern region of Brazil, prevalences became similar (4.5% vs 4.1%) 8. When compared with international data, the prevalence of MetS identified in the present study is higher than that described in North American and European adolescents; however, it is lower than that found in adolescents from Asian countries 7. In this context, it is interesting to note that, in comparison to the diagnostic criteria proposed by the IDF, studies that use other criteria to diagnose MetS invariably present higher prevalences 7. This is due to the fact that the IDF diagnostic criteria have more rigorous cut-off points, in addition to considering the greater accumulation of abdominal fat as a compulsory risk component 6. However, the advantage of using the IDF diagnostic criteria is the possibility of minimizing possible false-positive cases in the diagnosis of MetS 23.
In line with previous findings 7,24,25,26), age and economic class were significantly associated with the presence of MetS in the adolescents of this study. A systematic review has shown that the prevalence of MetS increases in significant proportions in the young population with advancing age, presenting values ranging from 3% to 6% between the beginning and end of adolescence, respectively 7.
Initially, the expected increase in blood pressure, triglycerides, fasting glycemia, and visceral fat deposits in more advanced stages of biological maturation 27 may explain the higher proportion of MetS among older adolescents.
With regard to economic class, the prevalence of MetS was positively associated with the highest stratum. Another study involving Brazilian youths identified a significant association in the same direction (24); however, data collected in other countries indicate an inverse relationship between economic class and the presence of MetS 25,26. The different criteria used to classify the economic strata and the interactions between family income and attributes of the socio-cultural context in which it is inserted can contribute to understanding these differences. For example, unlike young people living in developed countries/regions, high-income adolescents from developing countries, such as Brazil, tend to have a higher prevalence of obesity and, in turn, a higher cardiometabolic risk, in comparison with those of low economic classes 28.
The findings of the present study demonstrated a significant and independent association between obesity and MetS. Similar findings are found in the literature 14,16,29, which reinforces the hypothesis that anthropometric nutritional status is strongly associated with the risk of MetS. In this sense, studies have detected important associations between obesity and MetS from very young ages. Using a longitudinal design, among a set of biological and behavioral variables, it was observed that childhood obesity is the strongest predictor of MetS and other predisposing risk factors for cardiovascular diseases in early adulthood 30.
However, even assuming that there may be consensus regarding the impact of overweight and obesity on the increased risk for MetS, lifestyle determinants invariably present significant associations with cardiometabolic risk parameters in adolescents, regardless of excess body weight 31,32. In the present study, through the control of demographic indicators and anthropometric nutritional status, no significant association between physical activity and MetS was identified. It is possible this occurred as the variable physical activity was evaluated by self-report, which weakens the reliability of the data, since adolescents may present some difficulty in recalling levels of intensity, volume and frequency of everyday activities 33. This limitation was evidenced in a meta-analysis study which, when taking into account only the accelerometry technique for the measurement of physical activity, found three times higher chances of less physically active adolescents presenting MetS; however, in the analysis of subgroups, when considering only the use of self-report, the association disappeared 12.
Regarding sedentary behavior, the findings demonstrated that excessive recreational screen time was independently associated with MetS. Nevertheless, a fact that must be considered is the cut-off point used to define excessive screen time, typically recommended by current international guidelines and used in the present study; in particular, the cut-off point for excessive recreational screen time > 2 hours/day, proposed as the lowest risk for cardiovascular health 20, has shown a significant association with MetS in adults 34. Additionally, a study conducted in a representative sample of Brazilian adolescents also identified an independent association between recreational screen time ≤ 2 hours/day and lower cardiometabolic risk scores 32. However, it is worth highlighting a meta-analysis study, in which primary and sensitivity analyses based on this cut-off point did not reveal significant associations between recreational screen time and MetS in adolescents. In this case, the individual studies included in the meta-analysis that indicated significant associations assumed a cut-off point close to four hours/day 12.
Another finding from the present study was the independent association detected between fruit/vegetable consumption and MetS. It is important to highlight that this food habit remained significantly associated even after adjustments for potential confounding variables. The protection attributed to higher consumption of fruits/vegetables is consistent with evidence presented by other studies involving different experimental designs and statistical treatment 16,31. The food consumption pattern exerts an important influence on MetS through specific effects on the plasma lipid-lipoprotein profile, blood pressure, and body fat. Unlike diets in which fat-rich foods predominate, diets with higher fruit/vegetable consumption tend to have lower intake of simple carbohydrates and saturated fat, and a higher intake of complex carbohydrates and fibers, which are inversely related to altered glycemia and triglycerides, greater accumulation of abdominal fat, and high blood pressure, and positively with more favorable HDL-C, all known MetS components 35. On the other hand, in the case of consumption of sugary/soft drinks, in agreement with findings of some studies 14,16, but divergent from others 13, the significant association with MetS identified through bivariate analysis was not confirmed after adjustment for demographic indicators and anthropometric nutritional status.
One of the limitations of the study is that the research method employed to identify the determinants of lifestyle involved self-report, thus allowing possible memory bias. In addition, the transverse nature of the data does not allow inferences of causality in the association between MetS and the other variables. Finally, residual confounding caused by unidentified and unmeasured factors may potentiate eventual inaccuracies of the findings.
On the other hand, one of the strengths of the study is the simultaneous investigation of demographic indicators, anthropometric nutritional status and lifestyle determinants, which enabled adjustments in the final data analysis model, pointing to variables that were independently associated with MetS, and making it possible to plan and direct, personalized and more effective intervention actions. An additional strength of the study is the participation of a random and representative sample of adolescents aged 12 to 20 years who, together with the robustness of the procedures used in the data analysis, offer indications that the findings can be considered and generalized to other groups of young people with similar characteristics.
In conclusion, the present study identified that, due to the significant and independent association with MetS, intervention strategies should be designed to reduce recreational screen time and encourage daily consumption of fruits/vegetables in this population, with special attention to older adolescents, with a high economic class and excess body weight.












 Curriculum ScienTI
Curriculum ScienTI