INTRODUCTION
Sleep, like food, is essential for life. Sleep has a restorative function and the brain signals the need for it, similarly to how it regulates thirst and hunger (1). Sleep hours are part of the sleep-wake cycle, which usually consists of an alternation of approximately 8 hours of night sleep and 16 hours of wakefulness (2,3). An alteration of sleep patterns has immediate effects, such as the presence of excessive daytime sleepiness, general fatigue, impaired concentration with increased sensitivity to distractors, reduced reactivity to stimuli, irritability, and emotional instability. In the long-term, it increases the risk of death from cardiac, respiratory, and metabolic disorders, and it increases the prevalence of obesity in healthy populations (4,5). For adults aged 18-64 years, it is recommended to sleep 7 to 9 hours to improve health. The appropriate amount of sleep is 6-10 hours, and it is not recommended to have less than 6 hours or more than 10 (6). Individuals who sleep more or less than indicated may have health problems that compromise their wellbeing (6). Sleep deprivation can alter dietary choices and this, in turn, can reduce the duration of sleep. Some experiments have found that subjects who sleep shorter tend to consume excessive food the following day (7,8). This suggests that sleep restriction increases dietary energy intake (DEI) (9) but not necessarily energy expenditure. A better understanding of this response can guide nutritional management in the population exposed to this condition.
In the last few years, some studies have shown a relationship between substances such as ghrelin (10 11 12-13), growth hormone, cortisol (12,14), cholecystokinin (CCK), or peptide tyrosine (PYY) and sleep-wake behavior. Ghrelin has been known to stimulate appetite and is important for energy balance. It is expressed primarily in the stomach and acts to stimulate growth hormone secretion and food intake to regulate energy homeostasis (12,14). As well, sleep debt has been associated with lower leptin levels, increasing hunger, and as a consequence higher food intake (15). This is probably one of the reasons why there is a continuously increasing amount of evidence linking nocturnal sleep to different characteristics of diet, appetite, or body composition (16).
Some reviews on the subject have been already published (5,17,18). Capers et al. (5), in a systematic review (SR) of clinical trials, found no significant association between serum leptin and ghrelin levels following sleep restriction. However, due to insufficient information, they were unable to perform an analysis for energy intake. Subsequently, in 2017, Khatib et al. (17) authored an SR that included randomized and non-randomized studies; they found an association between sleep restriction and an increase in energy intake, as well as in total macronutrients, without specifying when the highest energy intake occurred. Zhu et al. (18), in another SR, investigated the relationship between sleep restriction and different metabolic outcomes, and they found an inverse and significant relationship between the hours of nighttime sleep and appetite, as well as increased “energy intake from one meal of the day”.
Some studies have investigated the relationship between sleep and appetite, as measured subjectively. The relationship between sleep restriction and increased intake appears to be compelling, but it is unclear whether the study design has influenced the results. This is a reason why, in this review, we sought to delimit only randomized studies, hoping to obtain a better level of evidence.
In addition, it is necessary to know if the increase in energy intake is related to a specific meal timing. Breakfast irregularity, for example, has been associated with inflammation and change in body weight (19). Nutritional strategies can be implemented focused on the meal of greater intake and, thus, prevent weight gain in a population exposed to a short night's sleep. Therefore, the purpose of this review is to discuss and analyze the evidence on the effect of nocturnal sleep restriction versus habitual sleep on dietary energy intake including specific meals, as well as the relationship between ghrelin and leptin levels in healthy populations.
MATERIALS AND METHODS
This SR and meta-analysis was carried out under the guidelines of the Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0 (20), with additional support from other bibliographic resources (21).
TYPES OF PARTICIPANTS
Inclusion criteria
We included healthy adults, aged 18-64 years. This age range was chosen because the recommended hours of sleep within it are all the same (6).
Exclusion criteria
We did not include studies of patients with cancer, obstructive sleep apnea, or metabolic disorders (such as fatty liver, polycystic ovary, and autoimmune diseases).
Studies without randomization, information on diet and outcomes of interest, or control group, and those in which a long-term nutritional intervention was performed were also excluded.
TYPES OF INTERVENTION
We included all relevant experimental studies in English and Spanish, published until 2019, that included sleep restriction as an intervention (< 6 hours) compared with habitual sleep.
TYPES OF OUTCOMES MEASURES
Primary outcome: dietary energy intake, including meal timing and information about macronutrients—carbohydrates, proteins and fat, according to dietary energy intake.
Secondary outcome: changes in leptin and ghrelin levels after short sleep or habitual sleep.
SEARCH STRATEGY
A search of electronic literature was made in October 2016 and February 2019 in the PubMed, Scopus, Lilacs, and Embase databases. To identify relevant studies, the following combinations were used: “sleep” AND (“feeding behavior” OR “dietary energy intake” OR “energy intake” OR “meal” OR “appetite” OR “ghrelin” OR “leptin”) AND (random) NOT (“cohort” OR “observational” OR “review”); we used Medical Subjects Headings (MESH) terms and also free search terms with restrictions in human studies. Two authors (AGO and FLB) completed the title and abstract screening and full-text screening, and manually searched the reference lists of eligible studies and reviews to identify other potentially relevant studies. An additional effort was made to acquire unpublished data by contacting two authors. We used consensus to resolve disagreements.
DATA EXTRACTION AND MANAGEMENT
The extraction of data from all included studies was carried out by two reviewers working independently from each other (AGO and FLB). Collected data were transferred to a previously arranged and standardized database. When additional information that was not published in the articles was required, an e-mail was sent to the authors to request it. Mean ± SD values were extracted, as well as the number of participants in each study.
EVALUATION OF THE QUALITY OF EVIDENCE
To ensure that the studies met the inclusion criteria, they were independently reviewed by two authors (AGO and FLB). To evaluate the quality of publications, the Jaddad scale (22) was used. The two blinded authors extracted the relevant information using previously designed forms. Any disagreement was solved by discussion and analysis of the information.
The quality of peer-reviewed articles was evaluated using checklists for quantitative studies and the experience of the authors.
EVALUATION OF THE BIAS RISK IN INCLUDED STUDIES
Bias risk was assessed independently by the reviewers (AGO and FLB) based on the following aspects: generation of the allocation sequence, method used to conceal the allocation sequence, and blind evaluation of resulting data (23). Due to the heterogeneity of data, it was not possible to perform a meta-analysis of the secondary outcome. The risk for bias was assessed using the tool included in the Cochrane Handbook (23).
STATISTICAL ANALYSIS
Demographic data were extracted from the included studies. The bias risk of each study was analyzed and compared with the bias risk of the other studies. The data were reported as means or standardized (Std) mean difference and SD for kilocalories according to total energy intake and kcal per meals. A sensitivity analysis was performed where macronutrients were compared between the available studies. In order to carry these comparisons, a percentage conversion was made to kcal and then from kcal to grams according to the total per group. Statistical heterogeneity was assessed using the I2 test; when the test showed a value greater than 50 %, heterogeneity was considered moderate or high. A random-effects model was used. The Review Manager Software, Version 5.1® (RevMan) was used to analyze the data and create the figures.
RESULTS
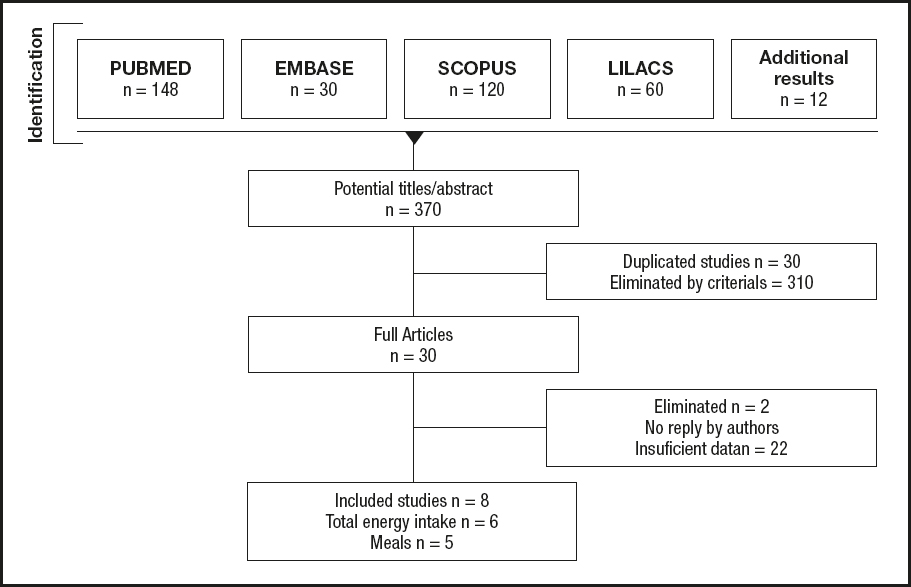
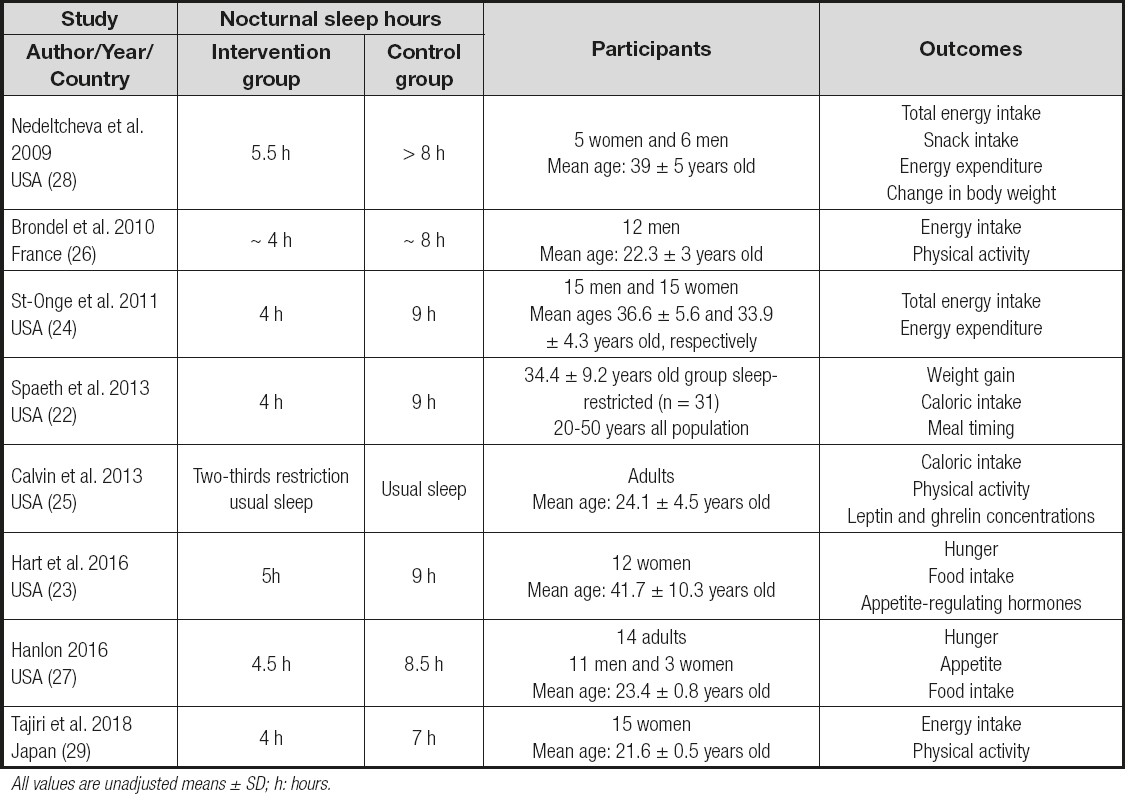
Among the 384 potential studies identified, a total of eight studies met the selection criteria and included 129 participants within the group with short sleep, and 126 participants in the group with habitual sleep (Fig. 1). The characteristics of the eight clinical trials included are shown in table I.
Table I. General characteristics of the studies included in the systematic review
All values are unadjusted means ± SD; h: hours.
Regarding the results, six of the studies reported total energy intake (24-29), the remaining two reported energy intake per mealtime (30,31). Five studies were conducted in the USA, one in France, and one in Japan. The mean age of the subjects was 31.6 ± 5.3 years, with a mean BMI of 24.1 ± 2.08 kg/m2 (Table I).
All the studies compared sleep restriction versus habitual sleep time to evaluate the effects of sleep deprivation.
The most widely used method for evaluating diet was weighted food in 6 studies, and only two of them carried out questionnaires (27,29); in all the studies these measurements were performed and carefully supervised by trained staff. Sleep was evaluated using actigraphy, polysomnography (PSG), and an accelerometer (Table I). Several evaluations were made in a lab in a controlled environment, and only one of them was at home with sleep measurements using a wristwatch-type accelerometer (29); others in both conditions controlled environment and free living (24) (Table I). To evaluate hunger, the instrument most commonly used was the Hunger Questionnaire (24,30). The analysis of ghrelin and leptin levels was not possible due to the great heterogeneity of the data, as well as the type of method used for the quantification of hormones and the time at which they were measured.
BIAS RISK OF THE INCLUDED STUDIES
Figure 2 shows the bias risk of the studies included. A high bias risk was found in approximately 70 % of included studies, mainly because the nature of the intervention made it difficult to conceal randomization and blinding (of participants, researchers, and measured results). Generation of the allocation sequence, selective reporting, and management of incomplete data showed no clear bias risk.
TOTAL ENERGY INTAKE
Figure 3 shows the estimation and description of the effect of partial sleep deprivation versus habitual sleep. Of a total of eight articles accepted in the review, six contained information available for the analysis of total energy intake. For this analysis Spaeth and cols. (28) showed results according to each day of sleep restriction, 1-5 days; following the comparison in the other studies, the mean of all days (1 2 3 4-5) was used in this analysis. The overall result shows a significant difference in energy intake between study groups (149.86 (95 % CI: 10.09-289.63); p = 0.04). The results showed low heterogeneity, with I2 = 0 % (Fig. 3). Furthermore, the analysis was performed using data for each day of sleep restriction versus intake on the day of normal sleep, and found a higher difference using data from day one (mean 211.4 (95 % CI: 22.78-399.96); p = 0.03) (figure not showed).
MACRONUTRIENT AND MEAL INTAKE
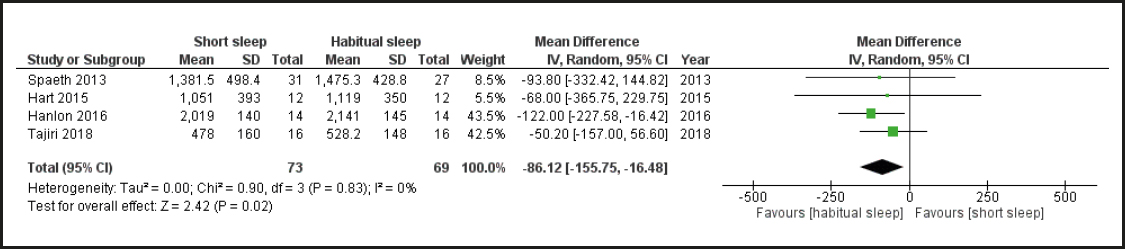
According to macronutrient intake, three articles had this information available (24,26,28). For this analysis, a higher intake of all macronutrients was generally found in the short sleep condition. The overall result shows a significant difference between study groups, and all results showed low heterogeneity, with I2 = 0 % (Fig. 4). Only four studies included energy intake per meal. Brondel et al. (24) reported the results of energy intake and macronutrient composition in four meals: breakfast, lunch, afternoon, and dinner. Data for the first meal, breakfast, on the second day of the intervention were used. Nedeltcheva et al., and Hanlon (25,31) reported the energy from snacks, meals, and total energy intake separately. Spaeth et al. (28) reported energy intake for three meals: 8: 00 am to 3: 00 pm, 3: 00 pm to 10: 00 pm, and after 10: 00 pm. They found that only the sleep restriction group consumed foods after 10 pm. Tajiri (29) reported the results for breakfast, lunch, and dinner, and they did not find any significant differences in intake for the different meal times.
There was not enough information for all mealtimes; however, it was possible to perform the analysis for the first mealtime.

Figure 4. Forest plot of the comparison of macronutrient intake (grams) for habitual sleep vs short sleep.
Figure 5 shows the results of energy intake for the first meal immediately after the nocturnal sleep restriction, compared to normal sleep. In contrast to what was expected regarding total energy and macronutrient intake, it appears that the first meal shows a lower intake in those subjects in the habitual sleep group. The results showed a low heterogeneity, with I2 at 0 %.
DISCUSSION
The relationship between sleep, appetite, and body weight has been studied for several years. However, the methods used in the different trials have been different. This is probably why it has been difficult to reach a conclusion on this subject. In this SR, we studied the relationship between habitual sleep (around 8 hours) versus short sleep (< 6 hours), and its effect on dietary energy intake in a healthy population.
In this SR, a positive relationship was found between total energy intake and short sleep, but not in a specific mealtime.
The most common outcome reported was total energy intake (24-27,29). In this meta-analysis we found a significant increase in total energy intake and macronutrient intake in the group with sleep restriction when compared to the control group. Similar to Khatib et al. (17), a statistically significant relationship was found between partial sleep deprivation and dietary energy intake in randomized and non-randomized trials. The difference in the first meal post-sleep deprivation between the groups was studied, and four articles (28 29 30-31) were analyzed. However, contrary to the expected, the results showed a higher energy intake in the group with habitual sleep.
Capers et al. (5), in a systematic review, found that the available experimental literature suggests that sleep restriction increases food intake and energy expenditure, but with inconsistent effects on energy balance. As we mentioned, a number of factors may contribute to these results, such as the method used for measuring diet and sleeping hours, and the ability to carry out randomized and blinded controlled clinical studies. However, despite the existing heterogeneity, there seems to be a clear trend, mainly for total energy intake, and it is important to know from which nutrients this energy is obtained. There is evidence that shows that dietary intake can significantly affect sleep when changing the amount of macronutrients consumed (32 33-34). According to the studies included in this analysis, the results show a higher intake of all macronutrients in the short sleep group; however, the information was available only in three studies, and this should be a limitation. Some studies have found that a diet high in carbohydrates and low in fat decreases slow-wave sleep (non-REM) as compared to a normal balanced diet or a diet low in carbohydrates and high in fat. Similarly, a rat-based study that fed the animals a protein-rich diet based on albumin, found that this diet significantly improved non-REM sleep (32,35).
A study carried out by Husain et al. on individuals without a previous history of sleep disorders found that those who consumed a diet rich in carbohydrates and low in fat spent less time in non-REM sleep when compared to those who consumed either a balanced diet or a low-carbohydrate, high-fat diet (34).
A study conducted by Hudson et al. (36) compared three groups that consumed test diets: 1) a bar-shaped source of tryptophan protein (i.e., husked pumpkin seed) and carbohydrates; 2) pharmaceutical grade tryptophan in combination with carbohydrates; and 3) only carbohydrates. They found that the protein bar was more effective in reducing waking time in the population with insomnia when compared to the subjects who consumed only carbohydrates or pharmaceutical grade tryptophan (36). Everything indicates that diet has a strong relationship with sleep, and different interventions should be carried out surrounding this topic. Unfortunately, the methods of measurement continue to be diverse, which may be the reason why there is still controversy surrounding this topic.
On the other hand, Zhu et al. (18) carried out an SR in which they included clinical trials, both randomized and non-randomized. They found a relationship between “hunger level” and partial or total sleep restriction; no differences in energy intake and weight change were reported.
Some studies have reported on energy intake as obtained through snacks (29,31) and during dinner (24,29,31); however, measurement times were so heterogeneous that it was not possible to draw any conclusions in this regard. It is important to point out that alterations in meal timing have been associated with inflammatory markers and changes in body weight (9,19).
In the SR conducted by Khatib et al. (17), an association was found between sleep restriction, increased total fat percentage, and decreased protein intake. Therefore, it is necessary to implement nutritional strategies focused on these food groups in order to prevent unfavorable outcomes in the population exposed to a shortening of night-time sleep.
One of the most important findings is that, regardless of the way sleep hours and diet were evaluated, the results supported the same conclusion: sleeping less than the recommended hours increases energy intake, regardless of the tool used to measure energy intake.
The results of the existing clinical trials regarding the effect of sleep deprivation and increase in ghrelin levels have been inconsistent and poorly targeted. The study by Spiegel et al. (37) showed that sleeping four hours for two consecutive nights induced a decrease in leptin by 18 %, and an increase in ghrelin by 28 %. However, these results are not consistent with experimental studies of sleep deprivation, probably because total energy intake was not duly considered, even though it influences the concentration of both hormones (37). A recent systematic review (5) whose aim was to find evidence of a relationship between sleep duration and body composition measurements, energy intake, and related biomarkers found that sleep deprivation did not affect ghrelin and leptin levels. However, their meta-analysis also found a high heterogeneity levels among the studies they selected (I2 = 77 %), which could be explained by the inclusion of different age groups.
One of the main reasons why the analysis was not carried out for this objective is that there is great heterogeneity in how these results are reported (e.g., time when the sample was taken, environmental conditions), even in the same report (e.g., ghrelin becomes reported as active or inactive). The study has some limitations that should be considered when interpreting the results. Despite having selected only randomized studies, one of the main limitations is intervention time, which can range from one to six days. In the study by Spaeth (28), who showed the intake for each day of sleep deprivation, the highest intake occurred after the first day of sleep restriction. Therefore, it is important to carry out more randomized studies in which intervention time, as well as the study conditions, are the same for all of the studies assessed.