INTRODUCTION
Obesity is a disease of rising prevalence and is considered one of the major public health issues in modern society. Over the last 20 years female obesity showed a rise in its prevalence with values that ranged from 8 to 16.9 % (1). It is estimated that 65 % of the world’s population live in countries where overweight and obesity kill more people than malnutrition (2).
Obesity predisposes to non-transmissible chronic diseases, which greatly impacts on health conditions in the economically active population. An increase in body mass index (BMI) may lead to congestive heart failure, pulmonary embolism, thromboembolism and diabetes mellitus type 2 (DM2) in a 20 times greater proportion, significantly increasing the risk of death from cardiovascular diseases (3).
Evidence points to the involvement of vitamin A in the regulation of fat mass, demonstrating that vitamin A deficiency (VAD) brings about an increase in the recruitment of pre-adipocytes to become adipocytes, inhibition of apoptosis, and reduction in adaptive thermogenesis (4,5). In addition to these occurrences, there are findings in the literature that show that the higher the BMI, the lower the serum concentrations of vitamin A, suggesting that VAD may contribute to an increase in obesity (6,7).
The negative association between retinol and BMI has been described in literature (8), with an important role of vitamin A on body adiposity (9). Minor serum concentrations of retinol and carotenoids have been verified in individuals with obesity when compared to eutrophic individuals, although no significant difference has been statistically found in the dietary intake of sources of these nutrients as calculated by food surveys (10,11). Thus, the higher serum vitamin A inadequacy associated with increased BMI may also be attributed to an increased metabolic use of this nutrient against oxidative stress, to which individuals with higher body adiposity (12-14) are more exposed.
Considering that the association between serum vitamin A inadequacy and body adiposity, decreased antioxidant capacity, and increased cardiovascular risk factors has been widely discussed, the present study aims to investigate the nutritional status of vitamin A and its relation with these risk factors in women showing different BMI ranges and recommended dietary intake of vitamin A.
MATERIAL AND METHODS
This is an analytical, cross-sectional study in adult women scheduled for nutritional care in a Nutrition Ambulatory at the Municipal Health Center of the city of Rio de Janeiro in the period from January 2016 to October 2018. All women who met inclusion criteria were invited to participate in the study. The research was previously approved by the Research Ethics Committee of Hospital Universitário Clementino Fraga Filho of the Federal University of Rio de Janeiro (Protocol of Research nº 011/05).
After the initial approach and clarifications about the goals and procedures of the study, all the women who agreed to participate in the study gave a formal authorization by signing an informed consent form, thus joining the first stage of the research that comprised an anthropometric assessment and a food intake survey.
The inclusion criteria were: female gender (aged 20-59 years), any ethnicity and social class, scheduled for nutritional care at the Health Unit. The following exclusion criteria were applied in the first phase of the research: women with inadequacy of vitamin A intake (evaluated in the first phase of the research), pregnant women and/or nursing mothers, liver diseases except non-alcoholic liver disease, malabsorptive syndromes, acute infection, alcoholism, kidney insufficiency, HIV carriers, cancer, and use of vitamin A supplements over the last 6 months.
After the first phase the sample was divided into 4 groups according to BMI range (15): eutrophic group (E), overweight group (OW), obesity class I group (OI), and obesity class II (OII) group.
SAMPLE SIZE
A sample size calculation was performed on the basis of the study that estimated the prevalence of Brazilian adults with inadequate intake of micronutrients (POF 2008-2009), which demonstrated an elevated inadequacy of vitamin A intake in women at about 78.5 % in the urban and rural areas studied (16). In relation to these findings, the prevalence of adequacy was 21.5 %.
To obtain a sample size with 95 % confidence interval, considering an adequacy prevalence of 21.5 % with a sampling error of 6 %, 155 women with a recommended dietary intake of vitamin A, according to the Institute of Medicine (17) would be needed at the end of the research. Based on this calculation, attendance, evaluated in a period of 18 months, considering the average weekly attendance in the six months prior to the present research, was 1,080 women (Fig. 1).
ANTHROPOMETRIC MEASURES
In this stage of the research, the weight and height of the women participating in the study were measured and their BMI was calculated according to the cutoff points recommended by WHO (18). Additionally, waist circumference (WC) was measured using a cutoff of > 80 cm for women, as proposed by the International Diabetes Federation (IDF) (19).
FOOD FREQUENCY QUESTIONNAIRE FOR VITAMIN A
Women involved in the study were asked separately to recall all the dishes, snacks, or other foods eaten in the previous 24 hours prior to the survey, regardless of whether the food was eaten inside or outside the house. During data collection, each woman was prompted to make sure that no meal or snack was forgotten. Next, a detailed list of all the ingredients of the dishes, snacks, or other foods mentioned in the reported consumed foods were collected from each woman via an interview. The study participants were then asked to recall how many days they had consumed each of the 15 locally available vitamin A-rich foods in the past seven days (20). During the training of data collectors, it was stressed that small quantities of food, smaller than 1 tablespoon, should be excluded. This was important as foods eaten in amounts smaller than the aforementioned threshold would not contribute significantly to nutrient adequacy but would however inflate the score. A minimum consumption of 1 tablespoon of food was better correlated with adequacy probability (20). Food frequency questions were double-coded and entered into a custom-built spreadsheet, which calculated the overall mean daily intake of vitamin A using the vitamin A content of foods as published in the Table published by the Instituto de Nutrición de Centro América y Panamá (INCAP) (21). Portion size was assessed using the Photographic Atlas of Food Portion Sizes (22).
Vitamin A intake was compared to the values of daily intake recommended by the Institute of Medicine in 2001. The cutoff point adopted for the recommended dietary intake of vitamin A was 700 μg/day.
A month after the first procedure the totality of women participating in the first stage of the study were again scheduled to receive information about their nutritional diagnosis, including their intake of vitamin A, in addition to the relevant dietary guidelines. Women who had reached the recommended dietary intake of this vitamin were invited to follow through the previously established research protocol and, to this end, they were instructed to visit for a blood draw after fasting for 12 hours.
In the second phase of the research only women who showed the recommended dietary intake of vitamin A were included in accordance with the results of the food intake survey conducted in the previous phase.
BIOCHEMICAL MEASURES OF VITAMIN A AND DIAGNOSIS OF NIGHT BLINDNESS (NB)
The nutritional status of vitamin A was assessed according to biochemical and functional parameters. For the biochemical assessment of vitamin A, serum concentrations of retinol and β-carotene were quantified by high-performance liquid chromatography with ultraviolet detector (HPLC-UV), and the following cutoff points were used: < 1.05 μmol/L and 40 μg/dL to indicate inadequacy of retinol and β-carotene, respectively (23,24).
In the functional assessment of the nutritional status of vitamin A, the presence of NB was investigated through an interview standardized by the World Health Organization (WHO) (25).
The interview was composed of the following questions:
1. Do you have difficulty to see during the day?
2. Do you have difficulty to see in low light or at night?
3. Do you have NB?
Cases of NB were considered when the answer to question 1 was “No” and, at least, one answer to questions 2 or 3 was “Yes”. If the subject interviewed had any ophthalmic problem corrected by glasses or contact lens, she was asked about her ability to see with them.
DIAGNOSIS OF DM2
The diagnosis of DM2 took place in accordance with the American Diabetes Association (ADA) (26) guidelines previously endorsed by the Brazilian Society of Diabetes (Sociedade Brasileira de Diabetes), which classifies the levels of blood glucose into ≥ 126 mg/dL (fasting) or > 200 mg/dL (one-off).
OTHERS BIOCHEMICAL MEASURES
For biochemical evaluation, a blood sample was obtained by venipuncture after a 12-h fast period. Laboratory tests were conducted to evaluate: lipid profile [total cholesterol, triglycerides, high-density lipoprotein cholesterol (HDL-c), low density lipoprotein cholesterol (LDL-c)], and the following concentrations were considered normal values: total cholesterol < 200 mg/dL; triglycerides < 150 mg/dL; HDL-c > 50 mg/dL; LDL-c < 200 mg/dL (27). Determinations of triglycerides, total cholesterol, and HDL-c were performed by the enzymatic colorimetric method. Reagents for these biochemical evaluations were purchased from Labtest Diagnóstica S.A., Minas Gerais, Brazil. The LDL-c fraction was determined in accordance with Friedewald’s formula. Serum concentrations of glutathione peroxidase (GSH-Px) (28) and thiobarbituric acid reactive substances (TBARS, nmol/L) (29,30) were assessed, and serum concentrations of uric acid (mg/dL) for enzymatic reactions were analyzed through spectrophotometry (31).
STATISTICAL ANALYSIS
In the statistical analysis, the measures of central tendency and dispersion were calculated and expressed as means and standard deviations for clinical, dietary, and biochemical variables. We used the analysis of variance (ANOVA) and the Bonferroni test for multiple comparisons. Pearson’s correlation coefficient was applied for serum concentrations of retinol and β-carotene with variables related to markers of oxidative stress. Odds ratios were estimated to evaluate the strength of the association between serum concentrations of retinol and β-carotene with biochemical, clinical, and anthropometric variables, as well as the association of inadequacy of serum concentrations of both micronutrients according to BMI ranges. The significance level adopted was 5 % (p < 0.05). The statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) for Windows, version 17.
RESULTS
The sample comprised 200 adult women, 80 women in the eutrophic group (E), 40 in the overweight group (OW), 40 in the obesity class I group (OI), and 40 in the obesity class II (OII) group, according to their BMI classification (18). The general characteristics of the sample are shown in table I.
Table I. Clinical, dietary, and biochemical variables in all the group of women with recommended intake of vitamin A according to BMI
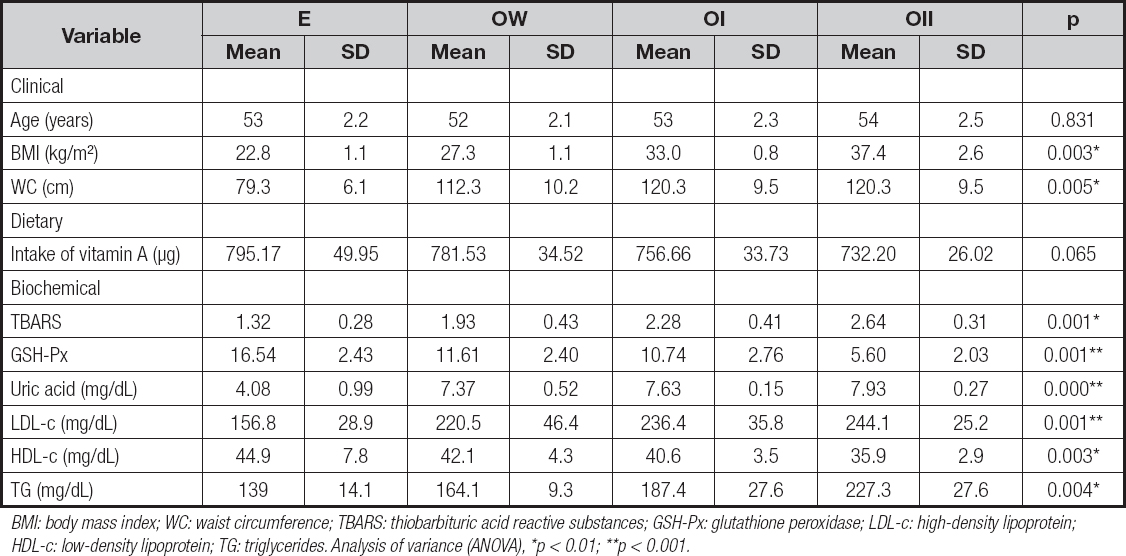
BMI: body mass index; WC: waist circumference; TBARS: thiobarbituric acid reactive substances; GSH-Px: glutathione peroxidase; LDL-c: high-density lipoprotein; HDL-c: low-density lipoprotein; TG: triglycerides. Analysis of variance (ANOVA), *p < 0.01; **p < 0.001.
According to biochemical and functional indicators, an increase in the inadequacy of the nutritional status of vitamin A was observed as BMI increased. Proportions of 5 % and 15 % of retinol and β-carotene deficiencies were found, respectively, in the E group, reaching 42.5 % and 47.5 % in the OW group, 65 % and 75 % in the OI group, and 77.5 % and 82.5 % in the OII group.
Inadequate serum concentrations of retinol and β-carotene showed a positive association with increased BMI. However, the occurrence of serum retinol deficiency enhanced the chances of body adiposity in a greater proportion when this deficiency was compared to the body adiposity that occurs in the presence of β-carotene deficiency (Table II).
Table II. Association between inadequate serum concentrations of retinol (μmol/L) and β-carotene (μg/dL) according to BMI range

OW/E’: overweight/eutrophy; OI/E’’: class I obesity/eutrophy; OII/E’’’: class I obesity/eutrophy; OR♦: odds ratio; 95% CI*: 95 % confidence interval.
A negative correlation was observed between serum concentrations of retinol and β-carotene and markers of oxidative stress in the groups studied (Table III). β-carotene showed a moderate correlation with these markers but a strong correlation with TBARS levels in the women in class I and class II obesity groups (Table III).
Table III. Correlation between serum concentrations of retinol and β-carotene and variables related to markers of oxidative stress in all the group of women with recommended intake of vitamin A

E: eutrophy; OI: class I obesity; OII’: class II obesity; TBARS: thiobarbituric acid reactive substances; GSH-Px: glutathione peroxidase; Pearson’s correlation, *p < 0.01; **p < 0.001.
As for parameters related to oxidative stress, we observed a rising exposure to high concentrations of TBARS and uric acid in women with retinol and β-carotene deficiencies when compared to women with adequate concentrations of vitamin A. Besides, the OII group stood out showing a chance of occurrence of high concentrations of TBARS that was 19.3 and 16.2 times greater, and of uric acid that was 16.2 and 16.0 times greater, respectively (Tables IV and V).
Table IV. Association between serum retinol deficiency with inadequacy of uric acid, TBARS, GSH-Px, LDL-c, HDL-c, triglycerides, WC, and the presence of DM2, according to the groups studied
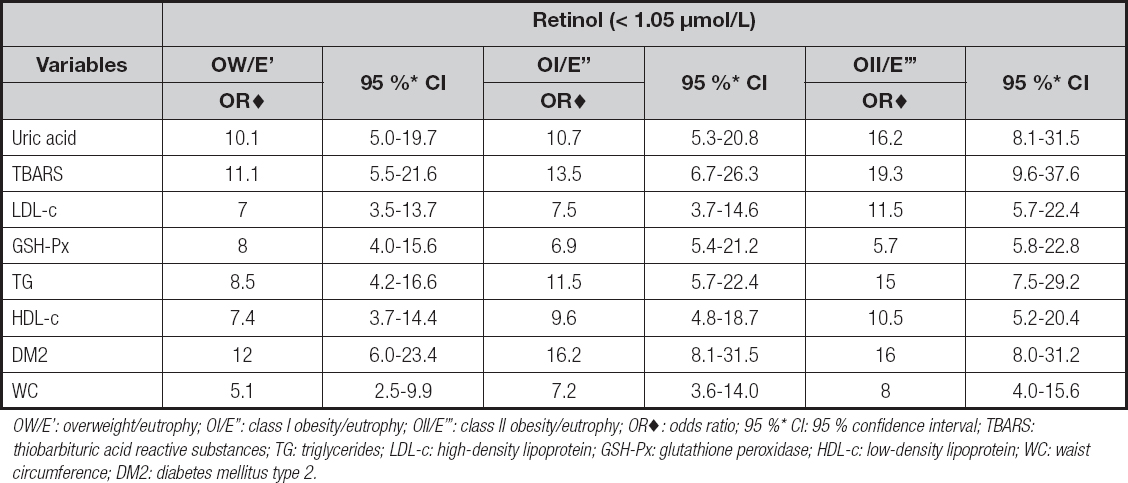
OW/E’: overweight/eutrophy; OI/E’’: class I obesity/eutrophy; OII/E’’’: class II obesity/eutrophy; OR♦: odds ratio; 95 %* CI: 95 % confidence interval; TBARS: thiobarbituric acid reactive substances; TG: triglycerides; LDL-c: high-density lipoprotein; GSH-Px: glutathione peroxidase; HDL-c: low-density lipoprotein; WC: waist circumference; DM2: diabetes mellitus type 2.
Table V. Association between serum β-carotene deficiency with inadequacy of uric acid, TBARS, GSH-Px, LDL-c, HDL-c, triglycerides, WC, and presence of DM2, according to the groups studied

OW/E’: overweight/eutrophy; OI/E’’: class I obesity/eutrophy; OII/E’’’: class II obesity/eutrophy; OR♦: odds ratio; 95 % CI*: 95 % confidence interval; TBARS: thiobarbituric acid reactive substances; TG: triglycerides; LDL-c: high-density lipoprotein; GSH-Px: glutathione peroxidase; HDL-c: low-density lipoprotein; DM2: diabetes mellitus type 2; WC: waist circumference.
We observed a decrease in GSH-Px concentrations according to BMI among women with retinol and β-carotene deficiencies (Tables IV and V).
Women with inadequacy of serum retinol and β-carotene showed an increased chance of occurrence of DM2 diagnosis as BMI increased. The OII group is worth highlighting since in this group the inadequacy of serum retinol increased the chance of having a diagnosis of DM2 by 16 times, and the inadequacy of β-carotene by 20.7 times when compared to eutrophic women (Tables IV and V).
As for the assessment of the nutritional status of vitamin A according to the functional indicator, a significant difference was observed between groups (p < 0.001). In the E group no case of NB was detected; however, the number of cases increased as BMI increased, reaching 7.5 % of women in the OW group (n = 3), 17.5 % in the OI group (n = 7), and 25 % in the OII group (n = 10).
A significant negative correlation was found between serum concentrations of retinol and WC in the OW (r = -0.32), OI ((r = -0.56) and OII (r = -0.68) groups, and β-carotene and WC in the OW (r = -0.46), OI (r = -0.70) and OII (r = -0.85) groups, and a statistically significant difference was found between groups (p < 0.001).
Women with inadequacy of retinol and β-carotene presented a high risk of increase in triglyceride levels and LDL-c levels as BMI increased, in addition to a reduction in serum HDL-c levels (Tables IV and V).
The women in the OII group with inadequacy of serum retinol and β-carotene were 10.5 and 5.1 times more likely to present, respectively, serum HDL-c inadequacy when compared to those in the eutrophic group (Table V).
The chance of high serum triglyceride levels in women with inadequacy of retinol in the OI and OII groups was 11.5 and 15.0 times greater, respectively. However, in women with inadequacy of β-carotene in the same groups chances were 15.0 and 12.8 times greater, respectively (Table V).
DISCUSSION
The literature has suggested an inverse association between intake and serum concentrations of antioxidant nutrients such as vitamin A with the presence of obesity, DM2, and other cardiovascular risk factors (32-34).
Such a finding is consistent with the results presented here, as in our study an inverse relationship was found between increased BMI and changes in the nutritional status of vitamin A as assessed by a decrease in serum concentrations of retinol and β-carotene, and the diagnosis of NB. Besides, this inverse relationship was also verified between glycemic profile and some cardiovascular risk factors and increased BMI.
Results showed that overweight and obese women presented a compromised biochemical and functional vitamin A profile when compared to eutrophic women despite having their dietary intake of vitamin A restricted to the quantities recommended. The inadequacy of this nutrient’s nutritional status may be related to an increase in BMI and in body adiposity; consequently, the nutritional requirements of vitamin A may be greater than established by current recommendations.
The present study corroborated the findings of other scientific studies (35,36) that reported a decrease in the serum concentrations of β-carotene as circulating retinol levels decreased, thus suggesting a greater mobilization of β-carotene for conversion to retinol, as β-carotene is recognized as the most powerful precursor of retinol (37). This finding is worth highlighting to the extent that the decrease in serum concentrations of β-carotene was associated with an increase in oxidative stress, as seen in the studies conducted by Shaker and co-workers (2011) and by Matos and co-workers (2012) (38,39), which show that this antioxidant can be diverted from other important functions, such as fighting oxidative stress, in order to keep an adequate nutritional status of retinol (40). Therefore, it is important to maintain serum retinol concentrations for preserving the function of β-carotene as antioxidant.
Individuals with DM2 present high levels of oxidative stress with an increase in the production of free radicals (29). Hyperglycemia, excess of free fatty acids, and insulin resistance in these individuals generate adverse metabolic events in endothelial cells, impairing their function (41). In this study, most diabetic women had VAD. In this sense, adequate serum concentrations of nutrients with antioxidant function, such as vitamin A, may contribute to preventing or reducing exposure to oxidative stress, thus minimizing endothelial dysfunction or even potential atherosclerotic issues.
The analysis of the lipid profile, considering the same BMI range in which women with inadequacy of serum retinol and β-carotene were compared to women with adequacy of these nutrients, showed greater chances of having high concentrations of triglycerides and LDL-c, and a reduction in serum HDL-c levels. These results point to an increased risk of cardiovascular diseases, which becomes progressively higher as BMI increases (42-44).
A cross-sectional study with 2,895 obese women showed that these individuals presented the smallest serum concentrations of carotenoids, these being inversely associated with LDL-c and serum concentrations of glucose and glycated hemoglobin, and positively associated with HDL-c, thus suggesting a relationship between glucose intolerance, increased activity of reactive oxygen species, and low concentrations of antioxidants (45).
Among cardiovascular risk factors, uric acid (46,47) stands out. As reported in the literature, hyperuricemia is strongly associated with insulin resistance and abnormal glucose metabolism (48,49). A study revealed that hyperuricemia increases the production of reactive oxygen species, inhibits the proliferation of pancreatic β-cells, and compromises stimulation of insulin secretion (50). In the present study a significant negative correlation was found between serum concentrations of retinol and β-carotene, and uric acid in overweight and obese women (51,52).
Corroborating this finding, a cross-sectional study analyzed a database of 14,349 adults in the United States where concentrations of vitamin A were inversely associated with concentrations of uric acid (53), and the authors suggested that a change in the action of xanthine oxidase, which converts retinol into retinoic acid and xanthines into uric acid, justifies the changes seen in serum vitamin A levels and impacts uric acid levels.
Retinol and β-carotene deficiencies were associated with a reduction in concentrations of GSH-Px, aggravated with the increase in BMI. Studies show that a reduction in GSH-Px levels accompanies increased insulin resistance, hyperinsulinemia, dyslipidemia, and a worsening of the vitamin A nutritional status (54,55).
Aspects reported so far seem to demonstrate that body adiposity and obesity represent an important cause of depletion of vitamin A and may be considered an aggravating factor. This deficiency, in turn, seems to be related to oxidative stress and to some cardiovascular risk factors, thus forming the following metabolic cycle: VAD → increase in body adiposity → increase in the metabolic use of vitamin A → VAD.
Given the limited number of studies relating obesity, DM2, and cardiovascular risk factors to vitamin A, in existing reports the consumption of this vitamin is invariably described as below daily recommendations. This study brings out an important aspect that needs to be valued as it contemplates individuals who have reached the recommended intake of vitamin A, and the relationship of this recommendation with the variables presented here.
The present study presents limitations among which we highlight the evaluation of food consumption. However, such limitation is present in studies using data from reports on consumption, particularly the issue of intake underestimation. We see no reason to believe under-reporting might have been different among the groups evaluated here.
CONCLUSIONS
The results showed that increased demand of vitamin A may be related to increased BMI, body adiposity, and oxidative stress even when the recommended intake of vitamin A is reached. Considering that retinol is a potent inhibitor of early-stage adipocyte differentiation, given its contribution to the metabolic regulation of adipose tissue, our findings suggest that the nutritional status of vitamin A should be closely monitored, and not measured to evaluate the recommended dietary intake.















