INTRODUCTION
The pre-diabetic state is usually defined as having above-normal blood glucose concentrations or decreased glucose tolerance, but below the threshold for diabetes. This clinical condition of glycemic alteration represents a risk factor for the disease, a predictor of an increased likelihood of developing type-II diabetes mellitus (T2D). It may be associated with health risks and complications (1,2). T2D is considered a pandemic. It affects thousands of people worldwide. Its pathogenesis is linked to nutritional deficiencies such as vitamin D deficiency, which affects billions of people around the world, who have different degrees of deficiency (3-6).
Studies have shown that vitamin D is related to glucose tolerance and insulin resistance. Their authors report that in the presence of inadequate serum levels of vitamin D there is a change in glycemic control, with decreased glucose tolerance and predisposition to T2D. On the other hand, adequate concentrations of vitamin D, or its supplementation, have also been associated with a reduced risk of developing type-II diabetes mellitus (2,7,8).
Thus, vitamin D deficiency may favor changes in glycemic control, suggesting that supplementation of this vitamin could have a positive effect on the prevention of T2D in individuals with a high probability of developing this disease, such as pre-diabetic individuals. Considering that studies associating vitamin D with pre-diabetes are scarce, this study aims to systematically review the literature on the effects of supplementation of this vitamin on the glycemic control of pre-diabetic individuals.
METHODOLOGY
SEARCH STRATEGY
This is a systematic review based on the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) recommendations, which guide and improve the reporting of this study. The study search process was developed to answer a guiding question (does vitamin D supplementation improve glycemic control in pre-diabetic individuals?). Its elaboration followed the PICO (patient, intervention, comparison, and outcome) strategy (9).
We searched articles in the Science Direct, PubMed, and LILACS databases using the following health descriptors (DeCS): "Prediabetic," "Vitamin D," "Glucose," "Glycated hemoglobin A," "Insulin," and their translations into Portuguese and Spanish. The following combinations of descriptors were used in three languages (English, Portuguese, and Spanish): SEARCH 1 - "vitamin D AND pre-diabetic." SEARCH 2: "glucose OR glycated hemoglobin A AND vitamin D." SEARCH 3 - "insulin AND vitamin D." The search period ranged from February to April, 2020.
SELECTION OF STUDIES AND DATA EXTRACTION
We adopted the following inclusion criteria: articles published in English, Portuguese, or Spanish within the last ten years; randomized clinical trials conducted with individuals over 18 years of age with hypovitaminosis D, without restriction of race, gender, or ethnicity, and which included vitamin D supplementation in different chemical forms (D2-calciferol and D3-cholecalciferol), as well as an evaluation of different markers of glycemic control (fasting glucose, postprandial glucose, insulin, HOMA-IR index, among others). Studies that supplemented calcium and vitamin D together were also included.
We excluded any experimental studies carried out with animals and/or in vitro, studies that included pregnant women and/or individuals with a pathology that compromised vitamin D metabolism (such as chronic renal failure or celiac disease, among others), and studies published as abstracts in annals of scientific events.
All the studies retrieved in the search process were evaluated by two authors independently (C.H.R.L. and L.C.C.L.) according to previously established eligibility criteria. Initially, the titles and abstracts were read, which was followed by an analytical reading of the full text of the studies selected for inclusion in this review. Extracted data included author, year of publication, placebo and supplemented groups, gender, age, study location, nutritional status related to vitamin D, supplementation, chemical form, dose and duration of the intervention, variables analyzed, and the primary outcomes. Of note, data selection and extraction were also carried out by the same two independent evaluators (C.H.R.L. and L.C.C.L.).
ASSESSMENT OF THE RISK OF BIAS AND METHODOLOGICAL QUALITY
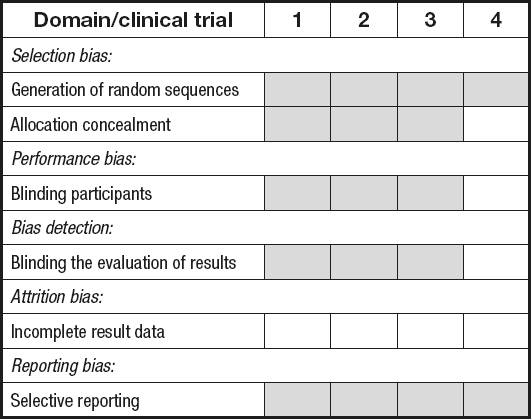
To assess the methodological quality of the studies included in this systematic review, we adopted the Cochrane collaboration tool. It is used for verifying potential biases in randomized controlled trials. The selection, performance, detection, attrition, and reporting biases were evaluated using this tool. Biases were classified as low, uncertain, or high risk (10).
In addition, the level of agreement between the two independent evaluators (C.H.R.L. and L.C.C.L.) was assessed by applying the kappa coefficient at the study selection stage. For this purpose, we used the classification of Landis and Koch (11), considering kappa values as follows: < 0 = without agreement; 0-0.19 = poor agreement; 0.20-0.39 = fair agreement; 0.40-0.59 = moderate agreement; 0.60-0.79 = substantial agreement; and 0.80-1 = almost perfect agreement.
RESULTS
GENERAL CHARACTERISTICS
Based on the eligibility criteria, we identified 309 articles in the databases ScienceDirect (255), PubMed (53), and VHL (01). After reading the articles in full, we considered 4 studies to be eligible, as shown in figure 1. The articles included in the last stage of selection were consensual among researchers. The value of the kappa index was 1.0 (95 % CI: 0.0-0.0), revealing an almost perfect agreement between authors, according to the classification of Landis and Koch (11).

Figure 1. Flowchart of the identification, eligibility, and selection of the articles included in the systematic review. PRISMA flow diagram.
Table I shows the characteristics of the studies included in this review. The studies are from four different continents — 1 study was carried out in the European continent, 1 in Africa, 1 in Asia, and 1 in the Americas.
Table I. Studies on the effects of vitamin D on the glycemic profile of prediabetic individuals: characteristics of the studies included in the systematic review

M: male; F: female; IU: international unit; HOMA-IR: Homeostatic Model Assessment for Insulin Resistance; 2hPP glucose: two-hour postprandial glucose; %HbA1c: percentage of glycated hemoglobin; QUICKI: quantitative insulin sensitivity check index.
In the present review, 491 pre-diabetic individuals were analyzed, of which 247 belonged to the placebo group and 244 belonged to the group supplemented with vitamin D. They were males and females with a mean age of 51.3 ± 5.3 years in the placebo group, and 50.9 ± 4.2 in the supplemented group.
All studies evaluated vitamin D supplementation in comparison with placebo, and the duration of the intervention was between 16 weeks and 12 months with vitamin D supplementation in two chemical forms: D2 (calciferol) and D3 (cholecalciferol). Studies evaluated the homeostasis of the insulin resistance model (HOMA-IR), fasting blood glucose, fasting insulin, insulinogenic index, two-hour postprandial glucose (2hPP glucose), glycated hemoglobin (HbA1c), oral glucose tolerance, and quantitative insulin sensitivity check index (QUICKI) (Fig. 1).
ASSESSMENT OF THE RISK OF BIAS
Figure 2 shows the methodological quality of the studies included in this review according to the Cochrane collaboration tool. Figure 3 shows the classification of the risk of bias through the authors' judgment of each risk (presented in percentages).
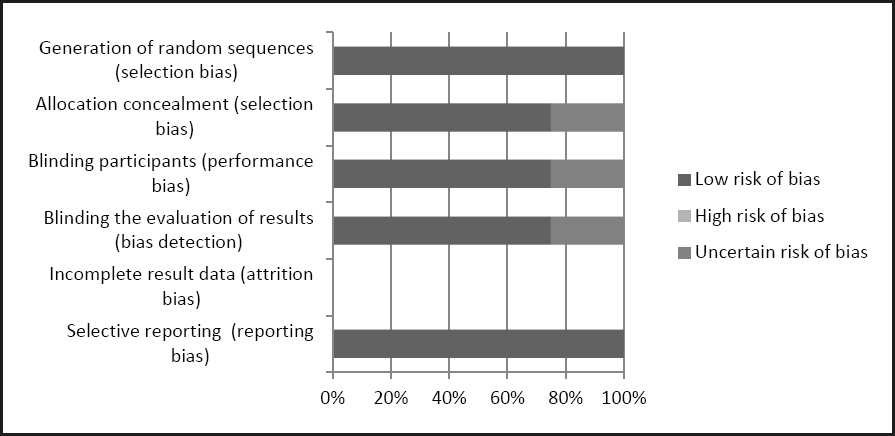
Figure 3. Risk of bias: authors' judgment of each risk of bias in all studies (presented as percentage).
Among the 4 randomized clinical trials included in this review, 100 % had a low risk of bias in relation to the generation of random sequences (bias selection). In relation to allocation concealment (selection bias), 75 % of the studies also presented a low risk of bias.
Twenty-five percent of the studies showed an unclear risk of bias in relation to blinding participants (performance bias) and blinding the evaluation of results (detection bias). Regarding the attrition bias, no study presented incomplete results, showing a low risk of bias for this item. Likewise, all studies (100 %) showed a low risk of bias in selective reports (reporting bias).
EFFECTS OF VITAMIN D SUPPLEMENTATION ON THE GLYCEMIC CONTROL OF PRE-DIABETIC INDIVIDUALS
The results of 3 clinical trials revealed that vitamin D supplementation, in its chemical forms calciferol and cholecalciferol, does not significantly interfere in the control of fasting glucose and fasting insulin. They also show no changes in %HbA1c and HOMA-IR, both at high doses (50,000 IU/week) and low doses (1,200 IU/day).
Barengolts et al. (12) carried out a study with African males with hypovitaminosis D and dysglycemia to verify the effects of vitamin D2 supplementation using a super dosage of 50,000 IU/week.
After supplementation, the reduction in the levels of 25-hydroxy- vitamin D improved in 76 % of the supplemented individuals, reaching levels of vitamin D sufficiency with values of 30 ng/mL or higher. However, they did not find a significant relationship with the indexes oral glucose intolerance, HOMA-IR, HbA1c, insulin secretion, and insulinogenic index even after 12 months of intervention.
Oosterwerff et al. (13) used a dose lower than that of the study above. The authors used 1,200 IU/day of cholecalciferol over a period of 16 weeks in Dutch, pre-diabetic, and obese individuals with hypovitaminosis D. In that study, the authors reported no positive effects on insulinogenic index, HOMA-IR, glucose in fasting, fasting insulin, and oral glucose tolerance, revealing that supplementation with cholecalciferol in this period does not improve insulin resistance or pancreatic B-cell function.
Based on this study, it was possible to verify that supplementation had altered the levels of 25-hydroxyvitamin D in the blood of the participants. After supplementing, their blood levels reached 60 nmol/L, that is, levels that were still inadequate; however, the status of this vitamin was improved in supplemented individuals. It is important to highlight that in this study there was also calcium supplementation for all research participants, both in the control group and in the supplemented group; however, despite supplementation with this mineral, there were no significant improvements in the glycidic profile of the participants in this study (13).
Moreira-Lucas et al. (14) found no effects on the glycemic profile of the participants in their study, most of whom had obesity and hypovitaminosis D, and were on average 45.6 years old. Although the duration of supplementation with a cholecalciferol dose (28,000 IU/week) was longer than twenty weeks, there was no improvement in both oral glucose tolerance and other markers of glycemic control in pre-diabetic individuals.
Regarding their vitamin D status after the intervention, it was possible to observe an increase in blood 25-hydroxyvitamin D levels to 98.7 nmol/L. As a result, these individuals who received supplementation left their deficiency state and progressed to a state of vitamin D sufficiency (14).
Kuchay et al. (15) performed an intervention that lasted 12 months, and reported that supplementation with cholecalciferol at a megadose of 60,000 IU/month resulted in the reaching of adequate levels of vitamin D (> 30 ng/mL) in the blood of 68.2 % of the study participants. This study also revealed that the effect of supplementation positively interfered in improving glycemic control in pre-diabetics with vitamin D deficiency. The researchers analyzed megadoses in Indian men and women, and found significant improvements in fasting glucose, %HbA1c, and 2hPP glucose.
DISCUSSION
Although vitamin D supplementation is unlikely to contribute to the prevention of type-II diabetes mellitus (16), several likely mechanisms of action may explain the possible role of vitamin D to help improve glucose metabolism, including its anti-inflammatory and immunomodulatory effects, induction of insulin secretion by pancreatic β-cells, indirect effects on the regulation of calcium concentration in pancreatic β-cells, and the subsequent insulin secretion, reduction of peripheral insulin resistance by vitamin D receptors in muscles and the liver, and influence of vitamin D on regulating genes for cell proliferation, differentiation, and apoptosis in metabolic pathways (17). In addition, individuals with vitamin D deficiency have a moderate elevation of parathyroid hormone, which can impair the release of insulin by pancreatic β-cells (18).
In the present study, only one article reported a beneficial effect of vitamin D3 supplementation on pre-diabetic Indian individuals with hypovitaminosis D (15). In that study, the participants were overweight (BMI = 25 kg/m2), whereas other studies reported that their participants had an average BMI of 30 kg/m2, that is, they were obese.
The molecule 1,25-dihydroxyvitamin D (1,25D) acts to limit the production of its precursor, 25-hydroxyvitamin D (25(OH)D). There is a hypothesis that 1,25D concentrations are high in obese individuals, and it is believed that this situation can reduce the levels of 25(OH)D. The adipose tissue of obese women expresses enzymes for the formation of 25(OH)D and its active metabolite (1,25D), and also for the degradation of vitamin D. The subcutaneous adipose tissue has a lower expression of one of the enzymes responsible for vitamin D 25-hydroxylation (CYP2J2), as well as a tendency to decrease the expression of 1-α hydroxylase. These data suggest that both 25-hydroxylation and 1-α hydroxylation are compromised in obesity (19).
In a meta-analysis study, a significantly inverse association was reported between percentage of body fat and total fat mass and levels of 25(OH)D regardless of age, latitude, and longitude (18). Likewise, a meta-analysis carried out by Li et al. (7), aiming to verify the effects of supplementation on glycemic control in type-2 diabetics, showed that non-obese individuals had a more significant improvement in glycemic markers when compared to obese individuals. This suggests that the obese population, even with an inadequate vitamin D status, no longer benefits from supplementation because of a possible retention of part of the supplemented doses in their fat mass (7).
However, according to Drincic et al. (20), a dilution of cholecalciferol would happen in the body tissue mass (in the fat mass and the extracellular fluid), instead of sequestration in the adipose tissue. Thus, the response to vitamin D doses in obese individuals is about 30 % lower than that observed in non-obese individuals, suggesting that vitamin D replacement therapy should be adjusted according to body size to achieve the desired serum 25(OH)D concentrations.
Thus, vitamin D supplementation, in most studies, may not have exerted the desired effects on the analyzed glycemic parameters due to differences in the metabolic processing of vitamin D, resulting from different levels of BMI and body fat.
Ethnic differences between the populations of the different studies may have influenced the results. In a study by Kuchay et al. (15), which presented favorable results for the glycemic parameters evaluated, the participating population was Indian. The populations of the other articles analyzed in this review were Europeans (13,16), North Americans (14), and Africans (12). It is known that black people have a higher risk for the development of type-2 diabetes mellitus, and a different homeostasis for vitamin D because their greater amount of melanin is correlated with a reduction in the capacity of the skin for synthesizing vitamin D. As the skin gets darker, vitamin D levels decrease; people with a lighter skin have higher overall levels of vitamin D (21,22).
In this context, the effect of ethnicity, regardless of other factors, is generally not considered prior to the preparation of strategies and recommendations for low-dose or high-dose vitamin D supplementation (21).
In a study carried out in England, that aimed to assess differences in cholecalciferol concentrations between healthy Caucasians and Asians, taking into account seasonal differences, the authors observed that serum levels of this vitamin were significantly lower (by more than 70 %) in Asian men and women as compared to Caucasian individuals in both the summer and the winter (23).
Gopal-Kothandapani et al. (24) investigated the effects of supplementation with 150,000 IU of vitamin D on the serum levels of free and total 25(OH)D in Caucasian and Asian individuals, and observed that Asian individuals had a greater levels of vitamin D deficiency at baseline when compared to those of Caucasians.
Such differences between ethnicities regarding vitamin D concentrations may be the result of variations in the amount of melanin and in the type of clothing Asian individuals use. However, it is possible that genetic and/or biological causes are also involved.
In this context, in addition to environmental and nutritional factors, genetic factors can also influence vitamin D status. Broad-scope genomic association studies have already shown that single nucleotide polymorphisms in vitamin D binding protein (DBP), as well as in the enzymes necessary for the activation or degradation of vitamin D and its metabolites, affect the serum concentrations of 25(OH)D (25).
Regarding the effects of vitamin D supplementation on glycemic parameters, there were differences according to the type of vitamin D that was supplemented. While Kuchay et al. (15) obtained positive results regarding the parameters analyzed in patients receiving cholecalciferol supplements, Barengolts et al. (12) did not find significant results for supplementation with calciferol in the same period.
A randomized clinical trial that evaluated the efficacy of supplementation with calciferol and cholecalciferol overdoses to improve serum 25(OH)D concentrations reported that cholecalciferol increased both total and free 25(OH)D levels to a greater extent than calciferol (26). Likewise, a meta-analysis found that cholecalciferol is more effective for raising serum 25(OH)D concentrations than calciferol, and therefore vitamin D3 could potentially become the preferred choice for supplementation (27). However, results regarding the efficacy of cholecalciferol and ergocalciferol to improve 25(OH) D levels are still controversial.
Gender differences in the populations studied may also have influenced the studies' results. A study was carried out that sought to assess factors related to vitamin D deficiency in North American university students from two campuses in different states, and showed that women had consistently higher average levels of vitamin D than men in both campuses, although this difference was not significant. AlQuaiz et al. (28) studied the prevalence of vitamin D deficiency in adults in Saudi Arabia, and observed significantly lower levels of serum 25(OH) D in males as compared to women.
Likewise, Braun et al. (29) examined the relationship of vitamin D with cardiometabolic traits in Indian subjects, and observed that vitamin D levels remained significantly low in men, as compared to women, regardless of the presence of obesity or type-2 diabetes mellitus.
Vitamin D binding proteins are more prevalent in women than in men, including pre-menopausal women, post-menopausal women, pregnant women, women taking oral contraceptives, and women on post-menopausal hormone replacement therapy. Thus, the differences observed between genders may be related to high protein binding levels to vitamin D and rapid 25-hydroxylation in females, although a difference between genders in the rates of vitamin D absorption may also be considered (30).
In addition to gender, latitude can also cause changes in the nutritional status of vitamin D, as it influences the cutaneous production of vitamin D (31). Leary et al. (21) also observed that differences in latitude were significantly associated with the risk of vitamin D deficiency.
At higher latitudes, the serum levels of 25(OH) D show variability throughout the year, peaking in late summer and declining in late winter, due to the variation in UVB rays that reach the skin. However, there is a peculiarity in countries with a low solar incidence, such as Norway (60 ° N), which has a lower frequency of vitamin D deficiency when compared to countries with a higher solar incidence such as those of Mediterranean Europe. In the Middle East, which has lower latitudes, there is a high prevalence of vitamin D deficiency, which can be explained by the usual use of clothing that covers almost the entire body. These locations are generally in arid and semiarid areas with year-round sunshine and few rainfalls throughout the year (32).
In view of the above, we suggest that differences in the ethnicity, gender, nutritional status, seasonality, and nationality of the population to be supplemented, as well as the chemical form of vitamin D that will be used, should be taken into account when making decisions regarding doses and intervention times.
CONCLUSION
There is insufficient evidence to support the notion that vitamin D supplementation improves glycemic control in pre-diabetic individuals. Therefore, this review highlights the need to conduct additional randomized clinical trials with a large number of patients and with proper doses of vitamin D to corroborate or rule out a possible association.
It is important to emphasize that maintaining adequate concentrations of vitamin D is not enough to prevent chronic diseases. It is necessary to consider factors such as the individual's inflammatory state, polymorphisms in receptors, drug-nutrient interactions, food consumption, and calcium intake, among others. Studies evaluating all these factors are essential. Such studies should also establish the optimal supplementation periods and doses in larger samples, in an attempt to obtain such benefits.