INTRODUCTION
Coronaviruses usually infect humans and other mammals (1). Most coronavirus infections are mild.
In December 2019, some patients were admitted to hospitals in Wuhan City, China, with a diagnosis of pneumonia of unknown etiology (2). After laboratory analyses, a new coronavirus was isolated from the respiratory tract of patients with the disease, named Severe Acute Respiratory Syndrome-related Coronavirus 2 (SARS-CoV-2) (1). Since then, the new virus has spread to every country worldwide and caused Coronavirus 2019 Disease, i.e., COVID-19.
Patients contaminated with COVID-19 have had changes in their leukocyte counts, respiratory disorders and increase in cytokines, markers, and inflammatory substances in plasma (2). Among the inflammatory markers and pro-inflammatory substances elevated in bloodstream, one can mention C-reactive protein (CRP). Patients with higher serum levels of inflammatory markers worsen with greater severity (1,3).
Patients infected with SARS-CoV-2 might develop Acute Respiratory Distress Syndrome (ARDS). The ARDS caused by SARS-CoV-2 is the result of an inflammatory response, which, in turn, is based on the oxidative damage of proteases and oxygen free radicals released by leukocytes, and the excess of inflammatory mediators produced. This inflammatory cascade might be modulated through ingesting some nutrients (4). This type of modulation in immune system is called immunonutrition. Most used substrates for this purpose are arginine, glutamine, branched-chain amino acids, ω-3 fatty acids and nucleotides (5).
For managing nutritional therapy to patients with COVID-19, recommendations issued by international enteral and parenteral nutrition societies indicate that solutions containing ω-3 fatty acids might be an option to improve the immune response in these cases (6,7). Several studies have also brought the hypothesis that immunonutrition might be able to reduce risk of infection and change the course of the disease (8-11). Restoring arginine stores in those infected also seem to improve lymphocyte function, which prevents secondary infections (12). Unfortunately, clinical trials attesting to this possibility are not yet found in databases.
Considering the inflammatory reaction generated by COVID-19, this study aimed to evaluate the effectiveness of oral supplements with immunonutrients to improve serum levels of C-reactive protein and lymphocyte count in patients affected by SARS-CoV-2 infection.
METHODS
This is a phase-IV, longitudinal, prospective, analytical, controlled, randomized, 1:1 and double-blind clinical trial. The study was carried out at a hospital specialized in caring patients diagnosed with COVID-19 in the city of Salvador, Bahia, Brazil. All patients admitted to the ward with a diagnosis of COVID-19 were studied over a six-month period (from July to December 2020), who met the eligibility criteria. Patients were prospectively followed up to a maximum period of 8 days.
As there were no previous similar studies to support sample calculation, it was decided to evaluate the total number of hospital beds for recruiting patients. Adult patients aged between 18 and 65 years, diagnosed with COVID-19 through molecular examination (RT-CRP), with patent gastrointestinal tract, fed with oral diet, and not under mechanical ventilation or requiring hospitalization in an ICU.
The following patients could not participate in the study: pregnant women; patients submitted to artificial nutrition in the 15 days prior to inclusion in this study; patients allergic to any components of the diets used; individuals with severe hyperglycaemia (> 180 mg/L) or hypertriglyceridemia (> 400 mg/L); patients with previous gastrointestinal diseases (surgical resections, malabsorption syndromes, inflammatory bowel diseases, persistent paralytic ileus, upper gastrointestinal bleeding or severe acute pancreatitis); patients with immunosuppression defined by neutropenia, myelodysplastic syndromes, congenital immunodeficiency or acquired immunodeficiency syndrome (AIDS), submitted to immunosuppressive therapies and systemic chemotherapy in the last 3 months; patients submitted to autologous bone marrow transplant last year, halogen bone marrow transplant in the last 2 years, and patients with graft-versus-host disease (GVHD); individuals with advanced chronic diseases (Child-Pugh stage C, grade IV heart failure, functional stage-IV chronic lung failure, terminal degenerative neurological processes, neoplasms in remission or progressing under treatment); individuals with processes of short life expectancy including end-stage chronic kidney disease; individuals with acute processes of short survival, such as shock of any etiology with multiple organ dysfunction refractory to therapy in the first 48 hours, or individuals submitted to post-cardiopulmonary resuscitation with severe neurological damage within 72 hours.
To analyze the evaluation error, a kappa coefficient of < 0.80 was used. It was considered a good correlation (13).
The patients were randomized to receive a standard high-protein, normocaloric supplement (control) or a supplement enriched with immunonutrients (experiment) in a 1:1 ratio for 7 days. Randomization was performed on an Internet website called Research Randomizer (14). Supplements were blinded to patients and investigators. They were provided by the clinical nutrition team in disposable cups, similarly to the groups and without product identification.
Participants in the control group received two 200 mL units of a normocaloric, high-protein nutritional supplement without the addition of any immunonutrition component (Nutren Senior®, Nestlé) distributed over 24 hours. Every 100 mL of this supplement provided 98 kcal, 8.0 g of protein, 9.4 g of carbohydrate, and 3.2 g of fat, without fiber and lactose.
Participants in the experiment group received two 200 mL units of a normocaloric, high-protein nutritional supplement with some L-arginine, nucleotides, and ω-3 essential fatty acids (Impact®, Nestlé) distributed over 24 hours. Every 100 mL of this supplement provided 109 kcal, 6.5 g of protein, 14 g of carbohydrate, and 2.8 g of fat, without fiber and lactose.
Participants were monitored by researchers for 8 days, involving risk assessment and nutritional status, collection of blood samples for haematological (complete blood count) and biochemical (CRP) exams, as well as recording possible clinical complications.
The Nutritional Risk Screening 2002 (NRS 2002) tool (NRS 2002) was used for assessing nutritional risk (15). To classify subjects as having high nutritional risk, a score ≥ 3 was considered. Those with a score < 3 were classified as with low nutritional risk. In addition, all participants were weighed and had their heights recorded, they were barefoot and wore light clothing. The individuals were weighed in a digital scale (Welmy-W300A®, São Paulo, Brazil).
Participants underwent a blood sample collection in a peripheral vein (basilic or cephalic vein) on D0 and after the supplement use period on the 8th day (D8). These samples were identified and sent to the clinical analysis laboratory for total lymphocyte count and CRP measurement.
The main outcome of the study was to assess changes in total lymphocyte count and serum CRP level. The secondary outcomes of the study were related to patients' nutritional risk and nutritional status according to the body mass index (BMI).
The efficacy criterion for an oral supplement with immunonutrients was to promote an increase in total lymphocyte count and a reduction in CRP levels greater than or equal to 30 %.
The data analysis was performed through descriptive and analytical analyses according to relative and absolute frequency. A Shapiro-Wilk test and histogram inspection were applied to verify the normality of continuous variables. According to type of variable and normality, statistical tests were used to verify the difference between groups.
Student's t-test was used for normal variables and the Mann-Whitney U-test was applied for non-parametric variables through mean and standard deviation to verify the difference between comparison groups. Categorical covariates were evaluated by using Pearson's chi-square test.
To quantify the probability of effectiveness for the proposed treatment (CRP reduction and lymphocyte increase in 30 % or more), relative risk (RR) and relative risk reduction (RRR) were calculated. For calculating the RR, the p-value was obtained by using Fisher's exact test. For all analyses a confidence level of 95.0 % (p < 0.05) was adopted. The study power was 99.6 %.
Data were organized and analysed by using Stata/MP 16.0 for Windows (StataCorp LLC®, Texas, USA). The software was licensed to the State University of Bahia (UNEB) and linked to the Teaching, Research and Extension Laboratory in Public Health (LEPESC/UNEB).
This research project was registered at Research Ethics Committee (CEP/UNEB) on CAAE no. 31801820.0.0000.0057 and approved by opinion no. 4.031.187. The clinical trial was also registered in the Brazilian Registry of Clinical Trials (REBEC) under UTN No. U1111-1252-3270.
At all research stages, Resolution 466/2012 regarding research involving human beings and the principles of the Declaration of Helsinki were respected. The informed consent form (ICF), in two copies, was used immediately before data collection for the research.
RESULTS
From July to December 2020, a total of 70 patients were included in the study. Of these, 64 were randomly allocated to receive either an immunonutrient diet or a standard control diet (Fig. 1). After randomization and start of diet administration, 10 and 11 patients, respectively, could not be followed-up in the control and experiment groups. No patient withdrew consent or experienced a serious adverse event during the follow-up period of the study.
Patient characteristics were well distributed among groups (Table I). Mean age was 41.5 (± 1.9) years and 39.5 % of patients were women. Most participants declared themselves as black and brown (79.1 %). The average body mass index (weight in kilograms divided by the square of height in meters) was 27.6 (± 0.8) kg/m². Nutritional risk was considered low in almost half of the studied subjects (58.1 %). All patients included in the statistical analysis received a diet for 7 days as established in the research protocol.
Table I. Demographic and clinical characteristics of participants with a molecular diagnosis of COVID-19, Salvador, Brazil, 2020

NRS 2002: Nutritional Risk Screening 2002; BMI: body mass index.
The results of the main outcome are shown in figure 2. Serum CRP levels on D0 were, on average, 28.6 (± 12.2) mg/L and 34.0 (± 8.0) mg/L in the control and experimental group, respectively. There was a statistical difference between these dosages in the groups (p < 0.05). On D8, mean serum CRP was 13.7 (± 3.1) mg/L and 10.4 (± 2.4) mg/L in the control and experimental group, respectively. There was no difference between the means of the groups (p = 0.585).

Figure 2. Measurement of serum C-reactive protein (CRP) levels and lymphocytes on baseline (D0) and after the supplement use period on the 8th day (D8) of the control and experimental groups.
The lymphocyte count on D0 showed an average of 2,555 (± 442.4) cells/mm³ and 1,696.5 (± 435.0) cells/mm³ for the control and experimental groups, respectively. There was a statistical difference between these dosages in the groups (p < 0.05). On D8, mean lymphocyte count was 2,272.2 (± 210.1) cells/mm³ and 2,064.0 (± 158.1) cells/mm³ in the control and experimental groups, respectively. In this case, there was no statistical difference between groups (p = 0.258).
The difference in mean serum levels between D0 and D8 for each group is shown in table II. In the experimental group, there was a mean reduction of 23.6 (± 7.5) mg/L in CRP, while in the control group the decrease was 14.8 (± 12.1) mg/L. There was statistical significance in the reduction differences (p = 0.002). On the other hand, there was a mean increase in lymphocytes for the experimental group (+367.5 ± 401.8 cells/mm³) and a reduction in this count in the control group (-282.8 ± 327.8 cells/mm³). Despite this, there was no statistical significance when comparing mean differences between the groups.
Table II. Main outcome (difference between CRP and lymphocyte measurements in the follow-up period of participants with molecular diagnosis of COVID-19), Salvador, Brazil, 2020
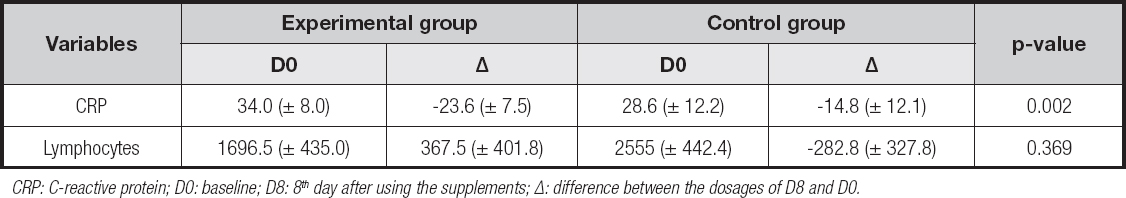
CRP: C-reactive protein; D0: baseline; D8: 8th day after using the supplements; Δ: difference between the dosages of D8 and D0.
Table III shows the results regarding effectiveness of the diet with immunonutrients to reduce serum CRP levels and to raise lymphocyte count by 30 % or more in comparison to the control diet. The reduction in CRP occurred in 80.9 % of those who received a diet with immunonutrients, while only 18.1 % of those who received a standard diet showed the same decrease.
Table III. Effectiveness in reducing the incidence of primary outcomes among participants with a molecular diagnosis of COVID-19, Salvador, Brazil, 2020

RR: relative risk; RRR: relative risk reduction; CI: confidence interval; CRP: C-reactive protein.
Thus, the group that received immunonutrition had 4.45 (95 % CI, 1.79-11.07) times more risk of reducing the CRP within the established limit than the control group. In other words, the use of a high-protein supplement enriched with arginine, ω-3 and nucleotides allowed a 345 % increase in CRP reduction when compared to the control group (p < 0.001).
As for lymphocyte count, 11 (52.4 %) participants in the experiment group increased lymphocyte rates, while 9 (40.9 %) of those who received the control diet had the same outcome. Therefore, the use of immunonutrients was represented 1.28 (95 % CI, 0.67-2.45) times more risk of increasing the amount of these cells in the bloodstream when compared to the use of a standard supplement. However, this result was not statistically significant (p = 0.327).
DISCUSSION
According to the knowledge of the authors of this article, with regard to the use of immunonutrient supplements in patients with COVID-19, this seems to be the first clinical trial carried out, as there are no others described in the literature so far. In this randomized, double-blind, placebo-controlled clinical trial involving patients hospitalized with pneumonia due to COVID-19, the use of an oral supplement with immunonutrients (arginine, ω-3 and nucleotides) was higher than the use of an oral high-protein supplement to reduce CRP.
ω-3 fatty acids are well established in the literature as important nutrients to prevent and treat several diseases, including coronary artery disease, diabetes, hypertension, arthritis and other inflammatory and autoimmune diseases (16). Studies previously conducted in diabetic patients (17,18) and in early-stage sepsis (19) showed that the use of these fats as a supplement was able to reduce serum CRP levels and other inflammatory markers. These data corroborate the findings of this study regarding the possibility of reducing CRP through ω-3 supplementation.
A recently published clinical trial carried out in Iran in 2020 evaluated 128 critically ill patients infected with COVID-19 (20). The participants were randomized to receive a formula fortified with ω-3 fatty acids or a control. The authors showed that omega-3 supplementation improved respiratory and renal function in these patients. This is probably due to the improvement in the levels of inflammatory cytokines induced by using this immunonutrient.
One of the hypotheses that could explain how ω-3 fatty acids can interfere with the inflammation caused by SARS-CoV-2 is that this substance could bind to the three-dimensional conformation of the spike protein (21) and modify it. This protein is used by the coronavirus to enter cells and infect them. Therefore, ω-3 could help reduce the complications of COVID-19 by changing the structure of this protein and decreasing viral load.
Despite not showing statistical significance, supplementation with immunonutrients promoted an increase in lymphocyte count when compared to the control group. The pro-inflammatory cascade caused by SARS-CoV-2 infection seems to promote lymphopenia and lymphocyte dysfunction, which may increase the risk of contracting secondary infections, septic shock, and multiple organ dysfunction (22).
A study carried out in France (12) in 2020 analyzed 26 patients diagnosed with COVID-19 and admitted to an ICU. The authors found that these patients had an increased activity of myeloid-derived suppressor cells (MDSC). This expansion of MDSCs was related to occurrence of lymphopenia and exacerbated arginase activity. That said, the work places the hypothesis that lymphocyte function could be restored through arginine supplementation. This seems to corroborate the findings found in this study since patients supplemented with this nutrient showed an improvement in the mean lymphocyte count. Possibly, due to the number of patients analysed, this result was not statistically significant. Papers that study a larger population may be able to find significance.
Some studies have shown how the interaction of arginine with the virus could change infectivity of SARS-CoV-2. A study conducted in 2020 (23) found that replacing isoleucine with arginine at position 407 in the spike protein could be crucial to destabilize the viral interaction with the human Angiotensin-Converting Enzyme 2 (ACE2) (functional SARS-CoV-2 receptor for entry into cells) (24), hindering viral entry into cells. Another group of researchers analysed that this same substitution could stabilize the spike protein structure (25). However, the authors were unable to determine if this characteristic would favour the interaction of this molecule with ACE2.
After having observed the possible effects of immunonutrient supplementation on CRP levels in this study, perspectives emerged for future work. New research could be conducted to confirm the findings presented and analyze the impact of immunonutrition on producing immunological markers of inflammation, such as interleukins and TNF-α. New evidence in modulating the inflammatory cascade could help to minimize the impacts of COVID-19 on people's health worldwide. As a result, several economic and social problems could be mitigated globally.
This study had some limitations. Losing track of 34.4 % and 31.2 % of participants in the experiment and control groups, respectively, may have altered the study result. However, this discontinuity of follow-up was proportionally similar between groups. Another limitation was the impossibility of removing all medications used by patients during the intervention. The impact of this was mitigated by maintaining the same drug treatment protocol instituted for all study participants. It is also noteworthy that this study was carried out only in one hospital, which may compromise the generalization of the observations made to other patient profiles.
This appears to be the first double-blind, randomized, controlled clinical trial to demonstrate the efficacy of immunonutrient supplementation in patients with COVID-19 to reduce serum CRP levels and increase lymphocyte counts. This article showed the potential use of these diets as adjuvant therapy for these patients.












 Curriculum ScienTI
Curriculum ScienTI


