INTRODUCTION
Chronic kidney disease (CKD) has a high prevalence among children, with an increase in incidence in recent years (1). Cardiovascular risk factors (CRFs), including dyslipidemia, are the main causes of morbidity and mortality in patients with CKD. This elevated cardiovascular risk begins in the early stages of CKD and is independent of age, ethnicity and sex (2).
CRFs are defined as factors that increase the risk for the development of atherosclerotic cardiovascular disease and diabetes mellitus (3). Patients receiving kidney replacement therapy (KRT) have an increased risk of CKD due to high prevalence rates of hypertension, dyslipidemia, obesity and sedentary lifestyle, in addition to factors related to uremia such as hypervolemia, anemia, alterations in calcium-phosphorus metabolism, hyperparathyroidism and accumulation of endogenous inhibitors of nitric oxide synthesis, among others (2,4).
As the deterioration of glomerular filtration progresses in patients, the lipid profile is altered, and the atherogenic risk index increases (5). This increase is due to the increased activity of cholesterol ester transfer protein, whose function is to transfer cholesterol esters from high-density lipoprotein cholesterol (HDLc) to low-density lipoprotein cholesterol (LDLc); to inhibit certain mediators that promote a decrease in HDLc; and to downregulate the action of lipoprotein lipase (LPL), which causes an elevation of very low-density cholesterol lipoprotein (VLDLc) and triglycerides (TGLs) (6,7).
Adipose tissue is considered an endocrine organ that produces multiple adipocytokines, and in recent years, two circulating adipokines, leptin and adiponectin, have been identified as mediators of inflammation and may be important markers of chronic systemic inflammation (8,9). Leptin is a peptide hormone produced by adipocytes that is correlated with the proportion of body fat stores (10). Additionally, leptin exhibits proinflammatory actions, including upregulating the phagocytic function of macrophages, increasing the production of proinflammatory cytokines, and stimulating reactive oxygen species (11). Adiponectin is produced by the mitochondria of adipocytes and acts as an anti-inflammatory factor, inhibiting the production of proinflammatory cytokines, especially with regard to atherosclerosis (12). Measuring the level of leptin or adiponectin in the peripheral circulation is useful in identifying the state of systemic inflammation; however, it has been shown that the leptin/adiponectin ratio (LAR) may be more valuable because high values of this index indicate an imbalance in pro- and anti-inflammatory conditions (13).
Since patients with KRT have high cardiovascular risk, the early identification and treatment of some risk factors, such as dyslipidemia, may reduce cardiovascular complications (14). In developing countries, such as Mexico, kidney transplantation is performed at a lower rate than in developed countries, and several years may pass before a pediatric patient undergoes kidney transplantation. Therefore, in these patients, strategies to keep long-term metabolic conditions under control are required, and it is important to identify cases that require greater vigilance and probably more aggressive treatment. The objective of our study was to determine the predictive value of the leptin/adiponectin index for the development of dyslipidemia after 1 year of follow-up in children with KRT.
METHODS
SUBJECTS
A prospective cohort study was carried out from January 2018 to December 2019 at two tertiary pediatric care centers in Mexico City: Hospital de Pediatría (Mexican Institute of Social Security) and Hospital Infantil de México Federico Gómez (Mexico Ministry of Health). In both centers, all pediatric KRT patients are usually cared for by a multidisciplinary team that includes pediatric nephrologists, pediatric endocrinologists, psychologists, and nutritionists.
Children aged between 8 and 17 years with stage V CKD according to the Kidney Disease: Improving Global Outcomes (KDIGO) staging scale (15) who were receiving peritoneal dialysis or hemodialysis prior to the start of follow-up were considered eligible to participate in the study. All patients included were selected using the consecutive sampling technique. Patients who were scheduled for kidney transplantation in the next 12 months, were diagnosed with diabetes mellitus, did not agree to participate or had incomplete clinical and biochemical evaluation data were excluded. The cohort follow-up duration was 12 months.
Of the 137 potential candidates, 59 were excluded (38 were scheduled for kidney transplantation, six were less than 8 years old, five had secondary diabetes mellitus and ten refused to participate in the study). An analysis of 70 patients is presented. The causes for dropout during follow-up were deaths (2 patients, secondary to heart failure), and loss of medical insurance (6 patients); accordingly, these patients did not attend hospital follow-up.
According to the Declaration of Helsinki, the protocol was evaluated and approved by the ethics and research committee of the hospital under registry number R-2018-3603-075 & HIM-2017-117. A parent or legal guardian signed an informed consent form, and each child provided written assent according to the recommendations of the Declaration of Helsinki.
PROCEDURES
At the beginning of follow-up, for all patients: the lipid profile, adiponectin and leptin levels, and somatometry measurements, including body fat percentage, were determined. A serum TGL level higher than 250 mg/dl was an indication for treatment with benzafibrate.
Nutritional assessment and indications: the calorie requirements of each participant were estimated based on resting energy expenditure (calculated using the equation from the Institute of Medicine to predict dietary reference intake) and physical activity levels. Nutritionists designed patient diets according to daily calorie dietary requirements. The diets consisted of 50 % carbohydrates, 20 % protein and 30 % total fat. The protein intake differed according to replacement treatment and age (hemodialysis: 4-13 years, 1.05 g/kg/day and 14-18 years, 0.95 g/kg/day; peritoneal dialysis: 4-13 years, 1.1 g/kg/day and 14-18 years, 1.0 g/kg/day) (16). Additionally, consumption of low-fat dairy products was encouraged, and low consumption of high-cholesterol foods and refined grains was recommended. In addition, nutritionists provided nutrition lectures, and participants were given manuals with examples of serving sizes for the 7 food groups (fruits, vegetables, legumes, cereals/tubers, foods of animal origin, oils/fats, and dairy) so that the patients and their caregivers could identify the appropriate serving size of each food (16,17). All patients included were recommended physical activity for at least 30 minutes daily.
Follow-up visits: the patients were followed biweekly for the first 3 months and then once a month until completion of the one-year follow-up to evaluate nutritional indications.
Final assessment: the lipid profile was reassessed at the one-year follow-up. The presence of dyslipidemia was evaluated among the participants; in the case of patients who already had dyslipidemia, those who maintained their dyslipidemia status and who experienced exacerbation of their dyslipidemia were identified. Considering these data, at the 12-month follow-up, the patients were divided into three groups: those without dyslipidemia (WOD), those who developed or experienced worsening of their dyslipidemia (DWD) and those with persistent dyslipidemia (PD).
ANTHROPOMETRY
The anthropometric indicators of each patient were recorded by a certified nutritionist. Height was measured to the nearest 0.1 cm with a SECA model 769 stadiometer (SECA 769, SECA Corp. Oakland Center Columbia, MD, USA). Weight and body fat percentage measurements were conducted using the bioimpedance method (Tanita BC-568 Segmental Body Composition Monitor, Tokyo, Japan) with the patients barefoot and wearing only underwear.
SERUM HORMONES AND CHEMISTRY LEVELS
Blood samples were obtained from the forearm of each subject via the antecubital vein between 7:00 and 8:00 a.m. after a minimum of 12 hours of fasting during the baseline visit. Serum aliquots were separated (centrifuged at 4 °C; 3000 rpm; 15 min) and frozen at -80 °C until biochemical analysis.
Insulin was measured by chemiluminescence (Roche-Hitachi Modular P and D). Leptin and adiponectin levels were measured using an enzyme-linked immunosorbent assay (ELISA) (Human Leptin DuoSet (DY398) and Human Adiponectin DuoSet (DY1065), R&D Systems, Minneapolis, MN, USA). Plates were read using an ELISA microplate reader (Labsystems Multiskan EX, MTX Labsystems Inc., Vienna, VA, USA) and were determined in duplicate according to the manufacturer’s instructions. The LAR was obtained by dividing the serum concentrations of leptin by those of adiponectin.
Hemoglobin, urea, creatinine and parathyroid hormone levels were measured by a colorimetric enzymatic method (IN-REACT, SPIM120). All electrochemiluminescence immunoassays (ECLIAs) were performed using a COBAS 6000 e601 (Roche Diagnostics GmbH, Indianapolis, IN, USA) in duplicate according to the manufacturer’s recommendations. Glucose, TGLs, and HDLc were determined by colorimetric enzymatic methods (Bayer Diagnostics, Puteaux, France). Intra- and interassay coefficients of variation < 7 % were considered acceptable. A standard curve was also generated for each assay. The homeostatic model assessment for insulin resistance (HOMA-IR) was calculated according to the formula: HOMA-IR = fasting glucose (mg/dl) x fasting insulin (µU/ml) / 405 (18).
DEFINITIONS
Dyslipidemia was defined as the presence of one or more of the following criteria: a) hypertriglyceridemia (for children < 10 years old: TGLs ≥ 90th percentile for age and sex and for children 10 years old: TGLs ≥ 150 mg/dl (3); b) low HDLc (for children < 10 years old HDLc < 10th percentile for age and sex and for children > 10 years old HDLc < 40 mg/dl in males and < 50 mg/dl in females); c) elevated LDLc (LDLc > 130 mg/dl according to the International Diabetes Federation (IDF) definition) (3,19).
Insulin resistance was defined as a HOMA-IR score higher than 3.16 (20). Obesity was indicated by a BMI ≥ the 95th percentile, and overweight was indicated by a BMI ≥ the 85th percentile for age and sex according to the 2000 CDC Growth Charts (21). For patients with < 2 standard deviations of height for age, BMI was calculated considering the age that corresponds to the 50th percentile of actual height. Hyperglycemia or elevated fasting plasma glucose was considered a fasting glucose level ≥ 100 mg/ dl (3).
STATISTICAL ANALYSES
The Shapiro-Wilk test was used for quantitative variables, and a nonparametric distribution was observed. We calculated the medians and ranges of quantitative variables.
To determine differences in quantitative basal variables among the three groups (WOD, DWD and PD), the Kruskal-Wallis test was used. To detect differences between serum glucose and lipid profiles at the baseline visit and the 12-month follow-up, the Mann-Whitney U-test was performed. Changes in HDLc, LDLc, TGLs, BMI Z-score, and body fat (ΔHDLc, ΔLDLc, ΔTGLs, ΔBMI Z-score, Δbody fat) were calculated by subtracting the HDLc, LDLc, TGLs, BMI Z-score, and body fat at 12 months from those at baseline. The correlation of the LAR with ΔHDLc, ΔLDLc, ΔTGLs and body fat percentage were determined using Pearson’s correlation coefficient.
In the multivariate analysis, the DWD and PD groups were combined to produce 2 groups: the WOD group and the present, persistent or worsened dyslipidemia (PPWD) group. Logistic regression analysis was performed to determine the relationship between LAR and PPWD at the 12-month follow-up; the analysis was adjusted for body fat percentage, BMI Z-score, ΔBMI Z-score, Δbody fat, replacement therapy, age at study onset, serum urea and bezafibrate use.
A receiver operating characteristic (ROC) curve was generated for the LAR to identify the cutoff point with the best sensitivity and specificity that allowed the correct classification and a positive likelihood ratio (LR+) to predict PPWD at the 12-month follow-up. With the results of the ROC curve, considering a cutoff point of 0.85, logistic regression analysis was performed to determine the relationship between a LAR > 0.85 and PPWD.
Differences were considered significant at p < 0.05. STATA v.11.0 was used for the statistical analyses.
RESULTS
BASELINE
The general characteristics of the patients are shown in table I. Seventy patients with KRT (37 male, 33 female) were included. The median age was 13 years (range, 8-17 years). The most frequent CKD etiology was congenital anomalies of the kidney and urinary tract (CAKUT) (45.7 %), followed by glomerulopathy (27.1 %) and indeterminate etiologies (21.4 %). At the time of evaluation, 35 patients (50 %) were receiving hemodialysis as a replacement therapy, and the remaining patients were receiving peritoneal dialysis with a median of 16 months of substitution treatment.
Table I. General characteristics of patients receiving kidney replacement therapy at the beginning of follow-up.
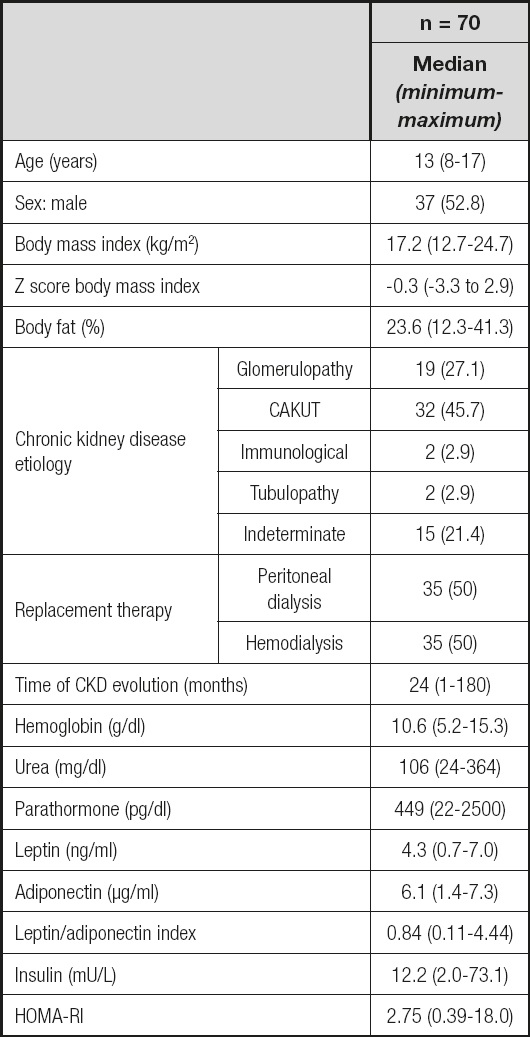
CAKUT: congenital anomalies of the kidney and urinary tract; CKD chronic kidney disease; HOMA-IR: insulin resistance index.
At enrollment, 80 % (n = 56) of the children were classified as having a normal nutritional status, eleven (15.7 %) were overweight, and three (4.3 %) were obese. In addition, 40 patients (57.1 %) had dyslipidemia, and 41.4 % (n = 29) had insulin resistance. Among the dyslipidemia patients, hypertriglyceridemia was most common (40.7 %), followed by decreased HDLc (27.1 %) and increased LDLc (7.1 %); of the 40 patients, 27 had combined dyslipidemia.
FOLLOW-UP
During the follow-up, among all the children, there were important changes in dyslipidemia status (Fig. 1). Of the 30 patients who did not present with dyslipidemia at the beginning of follow-up, only 13 remained without dyslipidemia (43.3 %); among the 17 who developed dyslipidemia, the most frequent types of dyslipidemia were reduced HDL and hypertriglyceridemia (n = 11). Of the 40 patients who had dyslipidemia at baseline, 14 (35 %) had normal lipids at the end of follow-up. In 11 patients (27.5 %), dyslipidemia worsened, with hypertriglyceridemia (n = 8) being the most frequent type, followed by increased LDLc (n = 7) and reduced HDLc (n = 6). It is worth mentioning that nine patients were indicated benzafibrate and only 2 improved dyslipidemia, in 4 cases they maintained hypertriglyceridemia and 3 patients, another dyslipidemia was added.
At the end of follow-up, 27 patients did not have dyslipidemia (WOD), 15 patients maintained their dyslipidemia status (persistent dyslipidemia [PD]), and 28 patients experienced worsening of their dyslipidemia or developed another type of dyslipidemia (DWD). At the end of the follow-up period, in the PD and DWD groups, there were significant increases in LDLc and TGs and a significant decrease in HDLc, while in the woD group, there were statistically significant improvements in serum concentrations of total cholesterol, HDLc, and TGs (Table II).
Table II. Comparison of the biochemical profile of all patients, and by groups at 12 months of follow-up.

LDL-c: low density cholesterol; HDL-c: high density cholesterol.
Regarding physical activity, at the interrogation, only 10 % (n = 7) of the included patients followed the indication (WOD n = 3 and PPWD n = 4).
Table III. Comparison of general characteristics of patients receiving kidney replacement therapy at the beginning of follow-up according to the study groups.

CAKUT: congenital anomalies of the kidney and urinary tract; CKD: chronic kidney disease; HOMA-IR: insulin resistance index; BMI: body mass index.
COMPARISONS AMONG CKD PATIENTS WITHOUT DYSLIPIDEMIA (WOD), WITH PERSISTENT DYSLIPIDEMIA (PD), AND WITH WORSENED DYSLIPIDEMIA (DWD) AT THE END OF FOLLOW-UP
When comparing age, CKD progression time, renal replacement time, and replacement treatment, no differences were identified among the WOD, PD and DWD groups; however, a lower body fat percentage was observed in the WOD group (WOD 19.5 % vs PD 25.3 vs DWD 28 %, p < 0.001). Regarding biochemical studies, no differences were identified among the three groups, but when the leptin, adiponectin, insulin, and HOMA-IR measurements were compared, serum leptin concentrations (median 5.6 ng/ml vs 5.8 ng/dl vs 3.6 ng/ml p < 0.001) and the leptin/adiponectin index (median 0.95 vs 0.95 vs 0.67 p < 0.001) were higher in the PD and DWD group than in the woD group (Table I). There was also no difference in the proportion of insulin resistance among the groups.
On the other hand, LAR was correlated with serum ∆HDLc, ∆LDLc and ∆TGs, and a positive correlation of LAR with ∆LDLc was identified (r = 0.306, p = 0.013). Likewise, a positive correlation of percentage of body fat with the LAR was observed (r = 0.438, p < 0.001) (Fig. 2).

Figure 2. Leptin/adiponectin ratio correlation with serum low-density lipoprotein cholesterol delta, triglycerides delta, high-density lipoprotein cholesterol delta, and body fat percentage.
Considering that the LAR levels were significantly different between the groups, multivariate logistic regression analysis was performed to identify the factors related to PPWD in patients with KRT, and it was determined that LAR (OR, 15.36; 95 % CI, 1.05 to 224.3; p = 0.046) and body fat (OR, 1.30; 95 % CI, 1.11 to 1.53; p < 0.001) were independent risk factors for PPWD, while the level of urea, BMI Z-score, treatment with benzafibrate, CKD progression time, delta BMI Z-score, delta body fat, and replacement treatment were not (Table IV). Other logistic regression models were constructed, and leptin and adiponectin were included in the independent models; there was no statistical significance for the risk of PPWD in patients with KRT (Tables V and VI).
Table IV. Logistic regression to identify factors that affect persistence, development or worsening of dyslipidemia in pediatric patients receiving kidney replacement therapy at one year of follow-up.

Table V. Logistic regression to identify factors that affect persistence, development or worsening of dyslipidemia in pediatric patients receiving kidney replacement therapy at one year of follow-up.
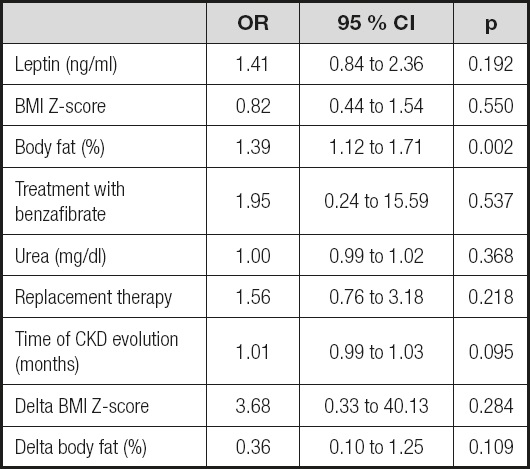
Table VI. Logistic regression to identify factors that affect persistence, development or worsening of dyslipidemia in pediatric patients receiving kidney replacement therapy at one year of follow-up.
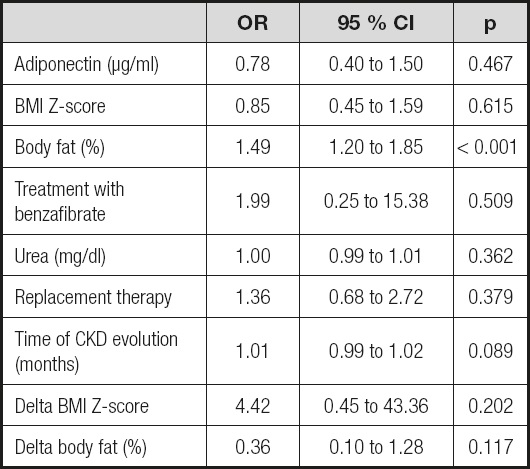
A ROC curve analysis was performed to identify the best LAR cutoff value for the prediction of PPWD in patients with KRT; the best LAR cutoff value was 0.85 (sensitivity of 72.09 %, specificity of 85.19 %, correct classification: 77.14 %, LR+, 4.8) (Fig. 3). Using this cutoff point, a multivariate logistic regression analysis was performed to identify the factors related to PPWD in patients with KRT, and it was determined that a LAR > 0.85 (OR, 16.7; 95% CI, 2.8 to 99.9; p = 0.002) and body fat percentage (OR, 1.46; 95 % CI, 1.16 to 1.84; p = 0.001) were independent risk factors associated with PPWD in patients with KRT.
DISCUSSION
In the present study, in pediatric patients with KRT, the body fat percentage and LAR were associated with worsened or persistent dyslipidemia at the 12-month follow-up. Our findings suggest an imbalance between leptin and adiponectin and that the percentage of fat plays an important role in dyslipidemia, considering that these factors are markers of metabolic risk (22,23).
Currently, it is recognized that regardless of the characteristics of the patient, renal failure produces changes in the lipid profile (14).
These modifications in the lipid profile during the evolution of CKD are characterized by normal or slightly elevated LDLc levels, decreased HDLc levels, elevated TGLs and increased lipoproteins that are mainly related to the degree of kidney damage, age, the presence of proteinuria, the replacement therapy used and obesity (24-26). In our study, 68.5 % of the patients had dyslipidemia at the beginning of follow-up, which is similar to that reported by Bonthius M et al., who identified a 68 % dyslipidemia rate in 917 pediatric patients (27).
In general, the usual recommendations for the management of dyslipidemia in patients with KRT are based on lifestyle modification interventions (17). However, despite appropriate implementation of lifestyle changes, there is a high rate of dyslipidemia reduction failure, which is observed several months after initiating the interventions (28). At present, there is no marker that can help initially identify patients who have a high probability of failure to initiate more intensive treatment.
In CKD patients, the accumulation of TGs is caused by both the excess production of lipoparticles rich in TGs and a decrease in their catabolism due to a decrease in the activity of LPL and liver lipase. This outcome is due to an increase in apoC-III levels, which causes an increase in the apoC-III/apoC-II ratio, and a decrease in LPL synthesis, secondary to the presence of hyperparathyroidism, or a decrease in insulin levels. Alterations in the HDLc level in CKD patients decrease its atheroprotective properties and may contribute to excess cardiovascular mortality in such patients, although the effect of advanced CKD on the composition and function of HDLc has not been completely clarified (14). These conditions explain the high prevalence of dyslipidemia and indicate that lifestyle modifications, which in these patients were indirectly analyzed by delta body fat percentage and BMI Z-score, are not sufficient to improve dyslipidemia in patients with CKD.
Adipose tissue is metabolically active and secretes adipokines, such as leptin, which cause vascular inflammation and insulin resistance, and adiponectin inhibits adherence molecules and increases the production of anti-inflammatory cytokines, such as IL-10 (29-31). Analyzing the impacts of leptin and adiponectin on inflammatory conditions independently can give us an incomplete picture, as happened in this study; no statistical significance was shown for the prediction of PPWD at the 12-month follow-up when leptin and adiponectin were analyzed independently (ANNEX); however, the LAR was significant. This indicates that the imbalance of these adipokines is a prognostic factor in pediatric patients with KRT. The LAR in adults with CKD has been associated with the presence of cardiometabolic factors (32) and is considered a prognostic factor the presence of cardiometabolic factors (33). Although the LAR has not been evaluated as a prognostic factor for cardiometabolic factors in pediatric patients with KRT, this concept has been explored in another group of pediatric patients, showing that an elevated LAR is associated with the risk of metabolic syndrome (34) or the development of systemic arterial hypertension at the 2-year follow-up (35). The elevated LAR reflects an increased risk due to the proatherogenic effect of leptin; at the endothelial cell level, leptin induces oxidative stress, increases the production of endothelin-1, and enhances proliferation, while at the vascular smooth muscle level, leptin favors proliferation, migration and hypertrophy in addition to facilitating thrombosis by increasing platelet aggregation (36); low levels of adiponectin prevent this adipocin from performing cardioprotective functions (37,38) such as inhibiting C-reactive protein mRNA and stimulating nuclear factor-κB signaling and tumor necrosis factor-alpha secretion (39). Hence, it is important to measure the LAR, as LAR elevation reflects an increase in proatherogenic conditions and a decrease in cardioprotective conditions that counteract it.
With regard to body fat, in pediatric patients with KRT, at the one-year follow-up, it was shown that changes in abdominal fat were associated with changes in circulating levels of adipokines and an increase in serum cholesterol (40). However, as indirect causes, a decrease in the volume of muscle mass and an increase in fat mass have been reported in patients with KRT (30,31), with large impacts on cardiometabolic factors and mortality; this was supported by the association of leptin levels with lower muscle mass in subjects with KRT (32).
We recognize that there are some limitations in the study. The sample size was small and the follow-up time was relatively short; it is possible that during a longer follow-up period, the LAR might behave differently. In addition, we did not objectively measure the adolescents’ lifestyle modifications, and only 10 % performed physical activity for 30 minutes daily, so it is possible that the LAR and body fat percentage are more useful in those who follow the recommendations more closely.
According to the results obtained, we consider that a LAR > 0.85 and fat body percentage can serve as a prognostic indicator for persistent or worsened dyslipidemia in patients with KRT.
















