INTRODUCTION
Nutritional status assessments of newborns (NBs) use weight as an initial parameter, as it is easy to measure and provides a global assessment of body composition; however, this measure does not distinguish between types of body tissues, e.g., fat-free mass (FFM) and fat mass (FM). An adequate birth weight may be indicative of water retention, as newborns have a high metabolic rate; thus, fluid balance needs to be assessed (1).
In this perspective, the Bioelectrical Impedance Analysis (BIA) test is an alternative method to assess body composition and estimate total body water, as well as to analyze changes in body hydration. It is a simple, safe and non-invasive method based on the passage of a low-intensity alternating electrical current, which flows through the human body owing to the movement of ions. The ease of electrical conduction is directly proportional to the amount of body water and electrolytes in the body tissues (2-4).
BIA enables the measurement of raw bioelectrical values of resistance (R) and reactance (Xc) and the calculation of the phase angle (AF). Resistance refers to the opposition to the passage of electric current through the body; it is inversely proportional to the amount of intra and extracellular fluids. FFM, for example, has high conductivity of electric current, as it has a large amount of water and electrolytes, thus presenting low resistance. Bone and adipose tissue, on the other hand, show high resistance because they have a low amount of water and electrolytes; therefore, they are not good electrical conductors. Reactance, in turn, indicates cell membrane integrity, its value is directly related to the presence of a healthy or diseased membrane, nutritional status or hydration (5).
Based on the values of R and Xc, the values of FFM, FM and total body water (TBW) can be estimated using validated and age group-specific formulas. Phase angle (PA) can also be calculated; it reflects the size, integrity, and quality of the cell membrane. Phase angle can be used as a prognostic marker in different clinical situations; abnormal PA values are related to nutritional status. Individuals with low values may have a deficient nutritional status (6,7).
In NBs, BIA can be used to assess hydration, FFM and FM; it is more accurate for TBW assessment, since water is the main conductor in humans and almost all body water volume is located in FFM. Therefore, TBW and FFM can be estimated by means of sex- and age-specific equations that use impedance values in combination with height and weight (8).
However, there are gaps and challenges to an accurate evaluation of body composition by BIA in younger children — for example, a lack of prediction equations for different pediatric populations; overestimation of TBW; the impossibility of determining FM distribution; and the small number of studies that have addressed the use of BIA in this public (8,9). Therefore, the objective of this study was to describe the use of bioelectrical impedance in newborns.
METHODS
This was an integrative review of the literature, based on the analysis of articles that used BIA to assess newborns. This is the main research question: "How is BIA used for assessing newborns?". This study followed the steps proposed for the development of an integrative review, as discussed by Soares et al. (2014) theme selection; formulation of research question; definition of inclusion and exclusion criteria; search for articles in the databases; data analysis and interpretation; presentation, interpretation, and discussion of results (10).
Original articles were considered as eligible when they presented data on fat-free mass, fat mass, resistance, reactance, phase angle or hydration; were written in Portuguese, English and/or Spanish; and were published in the last 10 years. The following types of publications were excluded from this review of the literature: articles, dissertations, theses, articles published in non-indexed journals, animal studies, in vitro studies; genetic, speech-language, cardiac, respiratory studies; studies with schoolchildren or with children in early childhood; studies with pregnant women; studies that used electrical impedance tomography or cardiographic bioimpedance as an evaluation method.
The article search step was carried out in March 2022 in the electronic databases PubMed/MEDLINE, EMBASE, Web of Science and the Virtual Health Library, through the Portal de Periódico Capes platform. The search used the descriptors ‘bioelectrical impedance' and newborns and their variations according to the databases (DECS - Bireme, MESH TERMS and EMTREE TERMS), combined with the Boolean operators AND and OR, which resulted in the following search strategies (Table I).
The results were imported into the Mendeley® reference manager and, after checking for duplications, they were exported to an Excel® spreadsheet. The studies were analyzed by 3 independent evaluators, starting with the analysis of the title and then of the abstract, using a standardized eligibility form, according to the model proposed by the Ministry of Health (11). For this analysis, the following evaluation criteria were established: type of study, use of BIA, age group, description of resistance/reactance/phase angle values and the purpose of using BIA.
Subsequently, the studies selected by the 3 researchers were compared to check for agreement before inclusion in this review. In the case of differences, they were discussed on the basis of the inclusion criteria established and described previously. After this step, the articles were read in full. The methodological approach, data extraction and writing of findings followed PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines (12). The data were organized and summarized in tables with information about the studies, the participants, and the method.
RESULTS
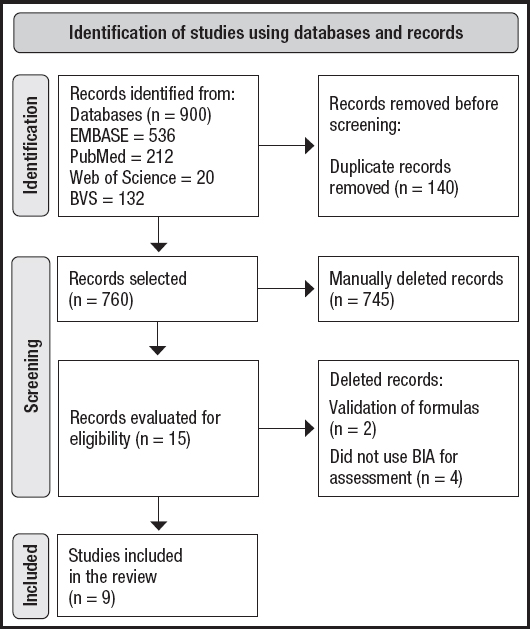
The initial search resulted in 900 articles, which totaled 760 after exclusion of duplicate documents. After the application of the eligibility criteria, 11 articles were selected for analysis, in addition to another 4 articles included from the reference list of selected articles; thus, there were 15 studies in total. However, after full reading, six documents were excluded, thus resulting in the final analysis of 9 articles (Fig. 1).
Table II shows the characteristics of the studies reviewed; objective, design and study location are highlighted. The studies were carried out in four countries: Australia, Brazil, Spain and Poland, with three longitudinal studies evaluating BIA at two or more times (13-15). When considering the objectives of the studies, only three compared body composition with other indicators, e.g., type of diet offered to NBs, which was evaluated in two studies (16,17) and analysis of the specific mortality indicator, which was evaluated in only one study (15).
Table II. Characteristics of the studies included in the review.

BIA: bioelectrical impedance analysis; ICU-Neo: neonatal intensive care unit; VLBW: very low birth weight; NBs: newborns; PTNBs: preterm newborns; TNBs: term newborns; BF: body fat; FFM: fat-free mass; PA: phase angle; SNAP: Score for Neonatal Acute Physiology; BIA: bioelectrical impedance analysis; BIVA: bioelectrical impedance vector analysis; Xc: capacitance; R: resistance.
All the characteristics of the population of the studies (Table III) were stable at the time of evaluation, and some newborns (n = 4) had VLBW (16-18). Three studies included term and preterm newborns (14,17,18), another three only full-term NBs (13,19,20) and three focused on premature infants (15,16,21). All of them were up to 15 days old at the beginning of the studies and the number of participants ranged from 11 to 154 NBs; there was a similar number of male and female infants.
Table III. Characteristics of the population evaluated in the studies.
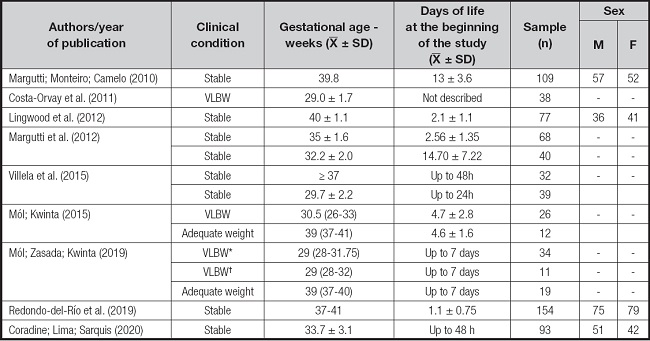
VLBW: very low birth weight; SD: standard deviation; n: number; F: female; M: male; h: hours. *Formula-fed VLBW;
†Breastfed very low birth weight infants.
Table IV shows that R was the most studied bioelectric indicator; it was not evaluated in only one study (17). Body composition, both FFM and FM were investigated in four of the studies (13,16-18). TBW was also an indicator addressed in these studies (n = 3) and Bioelectrical Impedance Vector Analysis (BIVA) was used in three studies (19-21).
Table IV. Summary of the variables analyzed in the studies.

BC: body composition; BIA: bioelectrical impedance analysis; BIVA: bioelectrical impedance vector analysis.
Table V shows the methodology and the main results of the studies, with the majority (n = 5) using a single-frequency bioimpedance analyzer (15,16,19-21). To analyze TBW, two studies used the formula by Tang (1997) (14,16), which includes the newborn's weight, foot length and the R value. Two studies found higher R and R/H among girls (19,20).
Table V. Methods and main results of the studies included in the review.

BIVA: bioelectrical impedance vector analysis; PA: phase angle; R: resistance; R/H: resistance/length; Xc: reactance; Xc/H: reactance/length; TBW: total body water; FFM: fat-free mass; FM: fat mass; VLBW: very low birth weight infants; Rinf: resistance at infinite frequency; Zc: resistance at the characteristic frequency; R0: resistance at zero frequency; %BF: percentage of body fat; PTNBs: preterm newborns; TNBs: term newborns.
DISCUSSION
The main findings in the reviewed articles showed that BIA can be used to assess the amount of TBW, body composition (FFM and FM), and the resistance, reactance and PA values in newborns; R was the most assessed indicator. However, there were some comparative limitations between the results of these studies, since each study uses a specific population of neonates with different gestational ages, and different BIA devices.
As described in the literature, BIA is a simple and non-invasive method to estimate TBW, FM and FFM both in term and preterm infants; it is a useful tool to assess abnormal values in TBW and body composition (18). Another way of evaluating BIA data is using BIVA, which allows detecting changes in hydration status and cell mass in newborns. Vector displacements along the major axis of the tolerance ellipses reflect variations in hydration status, and the displacements in relation to the minor axis show changes in body cell mass (20). The three studies that evaluated BIVA between 2010 and 2020 enable a better comparison of results, since the devices being used had the same frequencies (single frequency/50 kHz), and the NBs were the same age (in days of life) in the evaluation and showed similar gestational ages. In a study by Margutti et al. (2012) (21) PTNBs presented vector distribution above the 95 % tolerance intervals available in the literature for TNBs — equivalent to a state of lower TBW and lower FFM/FM ratio in comparison to TNBs.
Body water in neonates is variable and can account for 78 % of their birth weight. Loss of body water occurs in the NB's first weeks of life and the main reason is the mobilization of water from the extracellular space. In preterm infants, such loss may be greater, as breastfeeding is often not well established and the evolution of feeding is gradual (22,23). Mól & Kwinta (2015) (18) have corroborated these findings when they found that there was a decrease in TBW percentage during the first 3 months of life of TNBs and PTNBs; at the end of this period, the values were similar in both groups.
Lingwood et al. (2012) (13) stressed that the use of BIA to compare groups of neonates is adequate; however, caution is necessary in the analysis of the results, especially as regards FM, since it is more vascularized in this group, which results in a greater volume of water in fat cells and can interfere with resistance values.
Villela et al. (2015) (14) conducted a study to compare TNBs and PTNBs, and they found that body composition is different in the corrected gestational age of PTNBs, and that this group had a lower percentage of TBW, which suggests lower FFM percentage. Mól & Kwinta (2015) (18) also confirmed this finding: TBW was significantly lower in PTNBs in the first week of life compared to TNBs. In a study by Margutti et al. (2012) (21), the results were similar in the first weeks of life, as PTNBs had a lower absolute amount of TBW, FFM and FM when compared to TNBs.
The analysis of R and Xc data showed that the values were significantly higher in PTNBs in their first week of life; thus, it can be inferred that FFM and FM values are lower in these newborns. PA behaved differently, with a significant increase after 7 days of life in PTNBs, which may be indicative of improvement in clinical conditions (18,21). Coradine et al. (2020) (15) showed that decreases in PA values at different times of assessment were a good marker of severity. Vector analysis also showed significant differences in these data, as female infants showed higher values of R, R/C and Xc/C than male ones (19,20).
In addition to these results, the reviewed studies sought to analyze the relationship between body composition and the diet provided to neonates. Costa-Orvay et al. (2011) (16) found that the type of diet interferes with the body composition of VLBW infants; there was a greater increase in FFM in those who received a high-protein diet. Mól & Kwinta (2019) (17), when comparing the body composition of PTNBs fed with infant formula and TNBs at three months of age, found that the former had a significantly lower percentage of FFM and a higher percentage of FM. In that study, there was no difference in body composition between preterm infants fed with breast milk and infant formula; however preterm infants fed with breast milk had similar body composition to that of TNBs.
The comparative analysis between these two studies is limited because the former evaluated neonates using BIA in a single-frequency analyzer while the latter used a multi-frequency analyzer, which made up to 50 measurements. Currently, there are both single-frequency devices (50 kHz), which are the most used, and multi-frequency devices, whose frequencies range from 5 to 1,000 kHz. At different frequencies, an analyzer calculates the amount of total body water and its intra and extracellular distribution, assuming constant hydration, unlike the single-frequency analyzer (24).
Despite the articles found for this review, there were few studies on BIA in NBs, and they have limited practical applicability, as studies differ in terms of the formulas and devices that they use, BIA frequencies and the clinical conditions of the neonates. Therefore, an in-depth analysis should be made of body composition, using BIA in newborns in longitudinal, multicenter and representative research, because the assessment of body weight gain in NB, alone, is not enough to assess body composition. According to Lyons-Red et al. (2021) (25), the use of BIA in neonates needs to be standardized, since the existing equations are methodologically limited; thus, it cannot be inferred that using them is better than collecting data by means of anthropometric equations.
Knowing the body composition of NBs is important at different gestational ages at birth, as it can support interventions (for example, the type of diet offered) aimed at the clinical evolution of patients. It is also important to assess whether other variables e.g., genetics and environmental factors, are associated with changes in the body composition of neonates.
One of limitations of the present study is the delimitation of time — the most updated articles (last ten years) were included; the delimitation of language; and the proposal of a mini review, even though the researchers followed most of the steps of a systematic review.
CONCLUSION
BIA in newborns is commonly used to assess body composition and hydration and to compare these values between groups of neonates. The reviewed studies found that PTNBs have a lower amount of body water and FFM than TNBs. Studies using BIVA showed a statistically significant difference in body composition between male and female infants, with FM being higher among female ones. The diet provided to newborns interferes with their body composition, and some findings have shown that a high-protein diet can benefit PTNBs in FFM gain. The evaluation of the phase angle is still little explored in this population, but its increase after 7 days of life seems to be related to the improvement of clinical conditions. Another study found that its reduction was associated with the risk of mortality. There are still limitations for comparisons between studies, given the differences between the samples, and the adoption of different ways to characterize the results.