Introduction
Obsessive-compulsive disorder (OCD) is characterized by obsessions (recurrent and intrusive thoughts) and/or compulsions (repetitive behaviors or mental acts) having distress and high levels of anxiety (American Psychiatric Association, 2013). Epidemiological studies have shown that OCD is relatively prevalent in children and adolescents, yielding similar rates -around 2% (Canals et al., 2012). Neuropsychology research has attempted to determine the degree of correspondence between neuroimaging data and neuropsychological test results, in order to identify a clinical endophenotype and predict and improve treatment outcomes (Subirà et al., 2016).
Executive function (EF) and the relationship between its different subdomains either as independent entities, or as part of a whole, has been the subject of debate. EF could be described as a set of high level control mechanisms whose main purpose is regulation of cognition, behavior and emotions to meet individual goals and aims (Miyake & Friedman, 2012). Some authors consider working memory, cognitive flexibility (CF) and inhibitory response (IR) as the main mechanisms responsible for executive control (Diamond, 2013).
Chevalier & Blaye (2008) considered CF to be the ability to change mental representation based on incoming information and to keep representation intact when changes are irrelevant. IR is the mental process responsible for intentional and voluntary control, or the ability to prevent inappropriate information from interfering with responses or response patterns in progress, and to suppress previously relevant information which is not useful (Carlson & Wang, 2007). IR is not considered a unitary function, as it includes motor (behavioral) IR and cognitive IR. WM is defined as maintaining or manipulating information across a short delay when that information is not available in the environment and involves both short-term storage of information and simultaneous manipulation of mental content (Harvey et al., 2004). Planning is the ability to identify and organize steps and elements necessary to carry out an intention (Soprano, 2003; Tsukiura et al., 2001). Decision making (DM) is defined as the ability to choose after analyzing both rational and emotional information (Bechara et al., 2000).
Several meta-analyses have attempted to assess neuropsychological deficits in adult patients with OCD. Thus, Abramovitz et al. (2013) presented the results of a meta-analytic study on different neuropsychological domains and subdomains supporting the existence of differences between OCD patients and healthy individuals in sustained attention, nonverbal memory, executive function, WM, etc., although effect sizes ranged from medium to moderate. Likewise, they reported that results found in Response Inhibition showed a smaller overall effect size than expected. Shin et al. (2014) reported smaller effect sizes in visuospatial memory, verbal memory, executive function, verbal fluency, and processing speed, with effect sizes of 0.7 and 0.3. Snyder et al. (2015) highlighted that effect sizes in IR were medium-medium low while medium in CF. Abramovitch et al. (2019) found that poorer performance of OCD patients in neuropsychological tests was related to response severity close to a mean effect size on CF tasks.
There are few studies focused on the evaluation of executive functions in children and adolescents with OCD. In a meta-analysis by Abramowitz et al. (2015) on 11 neuropsychological studies in pediatric OCD it was reported that pediatric OCD might not be associated with difficulties in executive function, although empirical studies had large limitations (small number of studies, different evaluation tests, comorbidity in samples, etc.).
Results of studies in the pediatric population have been discrepant. Some research has found that children/adolescents with OCD have performance problems in CF tasks (Ornstein et al., 2010; Shin et al., 2008; Taner et al., 2011) while others report they function similarly to healthy controls (Geller et al., 2017). Other studies indicate that CF in children and adolescents with OCD is modulated by the WM (Wolff et al., 2017). Ornstein et al. (2010) reported worse results in Planning and CF but not in IR and WM tasks, while Waters & Farrell (2014) indicated that children with OCD presented more failures in IR.
Study aim
Given these discrepancies, the overall aim of this study was to conduct a meta-analytic study to examine executive function performance in children/adolescents with OCD versus a healthy control group. The specific objectives were: 1) to know the descriptive characteristics of the studies; 2) to analyze its methodological quality; 3) to evaluate the difference in the performance of executive functions (decision making, problem solving, response inhibition and cognitive flexibility) in the OCD group compared to the healthy control group in the pediatric population.
Method
This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA 2020, Page et al., 2021) guidelines.
Selection Criteria
For inclusion in this research, studies had to fulfill the following criteria based on PICOS statement (Moher et al., 2009): a) studies must exclusively include children and adolescents with OCD, excluding those that include other clinical diagnoses (PANDAS, ADHD, Tourette's, etc.) versus a healthy control group; b) they must include the evaluation of the EF and other neuropsychological variables; c) the sample size in the posttest must comprise more than four participants; therefore, single-case designs were excluded; e) statistical data reported in the study must allow us to compute effect sizes, and f) the study must be written in English or Spanish.
Search strategy
Several literature search procedures were used to locate studies which met our selection criteria. Several electronic databases were first consulted: Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, CINAHL, and PsycINFO. The following keywords were combined, in English and Spanish in the electronic searches: ((obsessive-compulsive) or (OCD)) and ((child* or adolesc* or pediatric) and ((neuropsych) or (neurocog) or (executive function) or (processing speed) or (visuospatial) or (decisión making) or (response inhibition) or (planning) or (st-shifting)) which should be in the title or abstract. Second, the references of some meta-analyses and systematic reviews were consulted (Abramovitch et al., 2013; 2015; 2019; Bragdon et al., 2018; Shin et al., 2014; Snyder et al., 2015). Third, the references of the located studies were also reviewed. Finally, emails were sent to 10 experts in this area to locate unpublished studies. A literature flow chart of the search process is shown in Figure 1. The search strategy produced a total of 3132 references. Twenty articles met the selection criteria.
Variables recorded from studies
The coded variables in each study were: mean age (years), percentage of women, diagnosis time in OCD group, mean Verbal IQ, quality of the study adapted from the NOS scale (adequate definition of cases, adequate representativeness of cases, adequate selection of controls, adequate definition of controls, matching of cases and controls in age, matching of cases and controls in IQ and at least one other variable, evaluator blinding, comparable loss rates, instruments validated, measurement methods), total sample size, comorbidity, drug consumption, country and continent where study was conducted, and year of publication.
Computation of effect sizes
The main purpose of this meta-analysis was to search for neuropsychological variables of the OCD group-control group. The effect size index was the standardized mean difference, defined as the difference between the paediatric OCD and control means divided by a pooled standard deviation. Hedges’ correction for small sample sizes was also applied to each effect size: d = c(m)((MOCD - MControl)/S), with d being Hedges’ standardized mean difference, MOCD and MControl the OCD and control means, S a pooled estimate of the standard deviation of the two groups, and c(m) a correction factor for small sample sizes (Botella & Sánchez-Meca, 2015). Negative effect sizes indicated a poorer performance for the OCD group in comparison with the control group, and viceversa. Separate d indices were calculated for each executive function or neuropsychological outcome reported in the studies. For reliability assessment of effect size calculations, the same random sample of studies used in the coding reliability study was subjected to a double process of effect size calculations, obtaining excellent inter-coder reliability, with intra-class correlations over 0.90.
Statistical Analysis
Separate meta-analyses were carried out with the effect sizes for each outcome measure. Random-effects models were assumed in order to accommodate variability exhibited by the effect sizes (Borenstein & Hedges, 2019). To assess heterogeneity of effect sizes, the Q statistic and the I2 index were calculated. I2 indices around 25%, 50%, and 75% were interpreted as reflecting low, moderate, and large heterogeneity, respectively (Higgins et al., 2003). For each outcome measure, a weighted mean effect size with its confidence interval was calculated. To assess publication bias on the CF and RI, a funnel plot was constructed, the Egger test was calculated, and the trim-and-fill method for imputing missing effect sizes was applied (Rothstein et al., 2005). The moderator analysis was required to meet two conditions. First, the meta-analysis had to include at least 15 studies. Second, there had to be heterogeneity (I2 > 25%). The IR meta-analysis met the first condition but not the second. Accordingly, moderator analysis was not performed. Statistical analyses were conducted with the statistical program Comprehensive Meta-analysis 3.3, CMA 3.3 (Borenstein et al., 2014).
Results
Study characteristics
The sample size of the 20 studies comprised 1450 participants (minimum = 19, maximum = 263, mean = 72), 685 groups with OCD (minimum = 10, and maximum = 102), with a mean sample size of 34 participants. The remaining 765 participants were part of the control groups (minimum = 9, maximum = 161), with a mean sample size of 38 participants. Table 1 presents a description of the characteristics of a quantitative nature.
Table 1: Descriptive characteristics of the methodological and participant quantitative variables.

Note:k = number of studies. Min and Max = minimum and maximum values. Mdn = median value.
For other participant variables, clinical participants (80%) were found to have no comorbid conditions and 55% were drug free. As for context variables, 73.7% of studies were performed in universities, with the rest in a hospital context. Studies were conducted between 1999 and 2020, with 65% published between 2009 and 2018.
Methodological quality of studies
Assessment of the methodological quality of studies was through an adaptation of the NOS scale. Table 2 shows scores obtained by every study in each of the 9 items of the quality scale. Items 3 (100%, adequate selection of controls), 2 (95%, adequate representativeness of cases) and 1 (90%, adequate definition of cases) were those most completed by studies. The total score on the quality scale ranged between 3 and 8.5 points (on a scale of 0 to 9), with a mean of 6.6.
Table 2: Assessment of the methodological quality of the studies with NOS scale.

Note:I1: Adequate definition of cases. I2: Adequate representativeness of the cas-es. I3: Appropriate selection of controls. I4: Adequate definition of controls. I5: Matching of cases and controls in age. I6: Matching cases and controls in IQ and at least one other variable. I7: Evaluator masking. I8: Comparable loss rates. I9: Validated measurement instruments. T: Total quality score. a Mean of the total scores.
In order to verify the extent to which the OCD and control groups were matched within the studies in the most relevant sociodemographic characteristics, meta-analytical syntheses of several of these were performed. The age variable revealed no heterogeneity (Q (17) = 13.75, p = .685, I2 = 0%), groups with OCD and CG being equal. The same occurred with the gender variable (Q (14) = 17.48, p = .231, I2 = 19.9%). Figure 2 presents a forest plot with these mean differences in age.
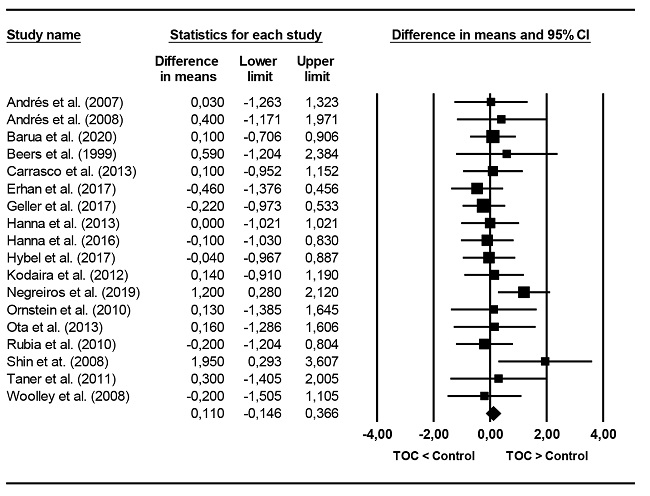
Figure 2. Forest plot of the differences between the mean ages of the OCD and control groups. Mean differences with a positive sign indicate a higher mean age in the group with OCD than in the control group; and vice versa.
Figure 3 presents a forest plot with the odds ratios (ORs) of these studies by gender. Odds ratios greater than 1 indicate a higher percentage of women in the OCD group than in the control group; and vice versa.

Figure 3. Forest plot of the odds ratios between the proportions of women in the OCD and control groups.
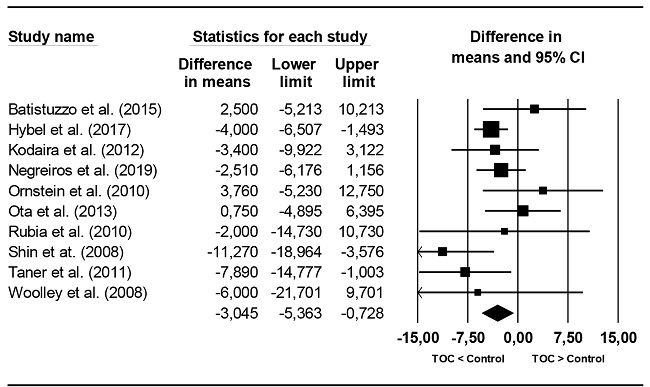
Regarding the Verbal IQ the 20 studies, 10 reported the mean Verbal IQ, observing a lower Verbal IQ in the OCD group compared to the CG. This mean difference was statistically significant (95% CI: -5.363, -0.728). Regarding heterogeneity, it did not reach statistical significance (Q(9) = 12.97, p = .164), although the I2 index showed low to moderate heterogeneity (I2 = 30.6%). Figure 4 presents a forest plot with these mean differences.
Distribution of effect sizes and heterogeneity
Separate meta-analyses were performed for each executive function. Table 3 shows the mean effects obtained with each outcome variable, together with a 95% confidence interval and the heterogeneity assessment tests (Q statistic, I2 index and inter-study standard deviation, ().
Table 3: Effects sizes in EF.

Note:k = number of studies. d+ = average standardized mean change. LL and UL = lower and upper limits of the 95% confidence interval for d+. +. Q = Cochran’s heterogeneity Q statistic. I2 = heterogeneity index. = tipic deviation inter-study.
*zp < .05.
**p < .01.
***p < .001.
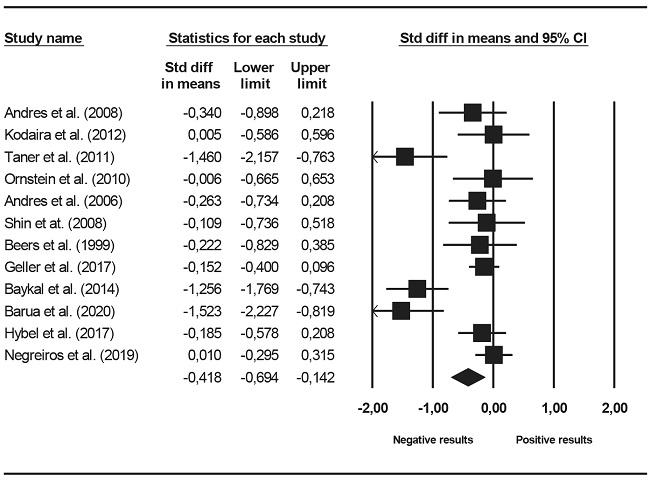
Four executive functions were assessed: RI, CF, DM and planning. Figure 5 presents a forest plot in RI. The mean effect was negative (d+ = -0.221) and statistically significant (95% CI: -0.335, -0.108) compared to OCD groups. According to Cohen's (1988) criteria, the magnitude of difference in inhibition between OCD and control groups was of low relevance (close to 0.20), but clinically significant. Heterogeneity statistics revealed no variability between study effect sizes (I2 = 0). Figure 6 presents a forest plot of effect sizes in CF. The average in cognitive flexibility were lower in the OCD groups (d+ = -0.418) and statistically significant. The magnitude between moderate and low (between 0.20 and 0.50) and clinically significant. Heterogeneity statistics revealed the presence of high variability between effect sizes (I2 = 74.7%). Effect sizes achieved in decision making were negative (d+ = -0.000, -0.169), indicating a certain tendency to a lower performance in the group with OCD but did not reach clinical significance. The average of the effect sizes in planning was negative, indicating a worse performance in groups with OCD (d+ = -0.319), but clinically significant.

Figure 5. Forest Plot displaying the standardized mean changes for the RI. Hedges’ g coincides with the d index reported in the text.
Analysis of publication bias
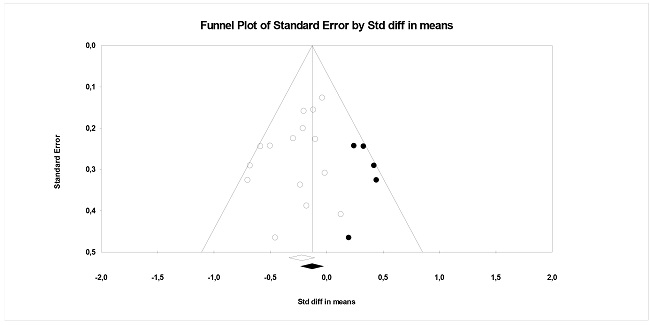
To assess publication bias as a threat against the average effect sizes for RI and CF, funnel plots were constructed (Figure 7 and 8). Ten studies are required for accurate results. The funnel plot for the effect sizes of the RI variable showed asymmetry. The application of the Egger test yielded a marginally significant result (b0 = -1.12, t (12) = 1.76, p = .103), indicating some suspicion of publication bias. Trim-and-fill imputed 5 additional effect sizes, resulting in an adjusted mean effect daj = -0.134 (95% CI: -0.267, 0.0003), indicating a clear effect of publication bias. As for the CF variable, Figure 8 presents a funnel plot of said meta-analysis. Egger's test reached statistical significance (b0 = -2.76, t(10) = 1.87, p = .091). Trim-and-fill did not impute any additional effects.
Discussion
There is abundant research on executive functions in adults with OCD, reaching smaller effect sizes in IR and CF. These data could be mediated by variables such as response severity (Abramovitch et al., 2019; Snyder et al., 2015). Studies in the pediatric population are fewer with somewhat inconsistent results (Geller et al., 2017; Ornstein et al., 2010; Shin et al., 2008; Taner et al., 2011). The aim of this work was to investigate performance in EF on pediatric OCD. A meta-analysis was conducted on 20 studies, of which fifteen evaluated IR, twelve CF, seven planning and three DM.
Our first aim was to analyze participant characteristics. We were able to observe that the mean ages of studies did not differ significantly between the OCD and control groups, except for studies by Negreiros et al. (2019) and Shin et al. (2008). Sex was well represented, apart from one study (Barua et al., 2020). Regarding verbal IQ, three studies presented a significantly lower mean difference in Verbal IQ for the group with OCD compared to CG (Hybel et al., 2017; Shin et al., 2008; Taner et al., 2011). This, together with the fact that only 10 studies presented this information might mean that participants are not equal in this variable. All this leads to the conclusión that studies were quite equal in terms of sociodemographic characteristics, sex and age, but not in verbal IQ, which could be a variable affecting results in performance of EF.
Our second aim was to analyze the methodological quality of studies. We consider that first meta-analysis in this field reports the quality of the studies in detail, being a relevant value for research. We observed an average quality of 6.6 (scale from 0 to 9). Studies scoring highest in quality were Batistuzzo et al. (2015), Andres et al. (2006), Geller et al. (2017), Kodaira et al. (2012), Ornstein et al. (2010), Ota et al. (2013) and Shin et al. (2008). All studies fulfilled item 1 (adequate selection of control groups). However, the item on masking was only taken into account by the evaluator for 5%, which could lead to overestimation of effects, that is, to obtain more marked deficits in participants with OCD compared to controls.
Our third aim was to analyze difference in performance of EF (DM, planning, RI and CF) in the OCD group versus the healthy control group.
Of the 15 studies evaluating IR, four presented a low but clinically significant effect size in comparison to groups with OCD (Andrés et al., 2006, 2008; Baykal et al., 2014; Taner et al., 2011), although this must be analyzed with caution due to the small sample size. Nevertheless, the magnitude of the effect size found in this meta-analysis was higher than in that by Abramowitz et al. (2015) which only reached an effect size close to zero (-0.07). Compared to adult-focused meta-analyses (Abramovitch et al., 2013; Shin et al., 2014; Snyder et al., 2015) that reported a medium magnitude (0.49 to 0.55), our effect sizes were minor. This may be due to several reasons. Firstly, perhaps this domain of EF deteriorates over time, being lower in childhood and adolescence than in adulthood (Beers et al., 1999). Nonetheless, it must be remembered that differences in ETs were not found in all studies with adults (Rosa-Alcázar et al., 2020). Another possible explanation would be selection of tasks to assess IR. Abramovitch and Cooperman (2015) pointed out differences in IR according to different paradigms. Thus, studies that used the Go-No-Go and Stroop interference paradigms did not find differences between groups, it being necessary to use stop tasks, as there may be different neuronal substrates (Eagle et al., 2008).In the present meta-analysis, most studies used the Stroop and Go-No-Go task, thus in future research it would be important to use tests associated with the three paradigms within each study (Abramovitch et al., 2015).
The effect size average in CF was lower in groups with OCD (d+ = -0.418) its magnitude moderate and low (between 0.20 and 0.50) and clinically significant. Three studies presented significantly lower mean levels of flexibility than the control groups (Barua et al., 2020; Baykal et al., 2014; Taner et al., 2011). Our results achieved worse results in CF in the OCD group than those in the meta-analysis by Abramowitz et al. (2015) whose mean ES was medium low (d = -0.26). In the meta-analysis by Snyder et al. (2015) and Shin et al. (2014) -focused on adults- effect sizes were similar to our data. The most used measure was the computerized version WCST the main signs being dysfunction, perseverative errors are the main signs of frontal dysfunction (Teubner-Rhodes et al., 2017). This version appears to be more sensitive than the classic method for identifying deficiencies in patients with OCD (Shin et al., 2014).
Results in DM reported a mean effect of -0.169, not considered relevant from a clinical viewpoint. However, only three studies reported this domain (Erhan et al., 2017; Hybel et al., 2017; Negreiros et al., 2019), with homogeneous results.
The mean effect in Planning was of low to moderate magnitude, indicating worse performance in groups with OCD (d+ = -0.319). Effect sizes for this variable exhibited high homogeneity but only 7 studies reported this domain. This result is close to that found in the meta-analysis by Abramowitz et al. (2015) which is also under-represented in studies.
Overall, effect sizes for all domains ranged from -0.169 to -0.418, with impairment clinically significant in CF, IR, and Planning. This does not mean, as indicated by Abramowitz et al. (2015) that there are children with OCD who may perform worse on these variables, but these results should be taken in the context of the limited sample size. Notably, the small number of effect sizes precluded any meaningful statistical analyzes to examine possible moderator variables. Interestingly, subdomains that produced the smallest effect sizes (Planning, RI, and DM) also turned out to be homogeneous. Furthermore, due to the small sample size, we decided not to examine possible moderating variables.
These findings reported that results in children/adolescents with OCD are quite smaller in magnitude compared to those obtained in meta-analyses in adult patients with OCD (Abramovitch et al., 2013, 2019; Shin et al., 2014).
The current study has important implications for clinical practice with pediatric OCD patients. Firstly, this study may suggest that incorporating a CF component into treatment could enable more effective interventions. The inclusion of specific modules on flexibility might potentially enhance the effectiveness of exposure with response prevention, improving adherence to treatment, and preventing desertion (Rosa-Alcázar et al., 2020).
This research is not without limitations. Firstly, the small number of studies, and low representation in the measure of some domains, together with certain publication bias, all restrict us from generalizing our results, therefore these must be interepreted with caution. This has also prevented analyzing possible influence of moderating variables in the different domains.
Future studies particularly longitudinal studies, that examine neural and clinical neurocognitive correlates of pediatric OCD are needed to confirm our findings and to determine the extent to which our results may have clinical relevance.