Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
The European Journal of Psychiatry
versión impresa ISSN 0213-6163
Eur. J. Psychiat. vol.28 no.4 Zaragoza oct./dic. 2014
https://dx.doi.org/10.4321/S0213-61632014000400006
A preliminary analysis of association between plasma microRNA expression alteration and symptomatology improvement in Major Depressive Disorder (MDD) patients before and after antidepressant treatment
Qiao-li Zhang MM1; Jim Lu, MD2; Xin-yang Sun, MM3; Wei Guo MM4; Lin Zhao, MM5; Hui-min Fan, MD6; Ai-fang Zhong, MM7; Wei Niu, MM8; Yun-hua Dai, MM9; Li-yi Zhang, MM3; Hong-tao Song, MM10; Liang Zhang, MM11
1Department of Neurology and Psychiatry, postgraduate school, Xuzhou Medical College. China
2GoPath Laboratories LLC, 1351 Barclay. USA
3Prevention and Treatment Center for Psychological Diseases, No. 102 Hospital of Chinese People's Liberation Army, Changzhou, Jiangsu; Department of Psychology and Psychiatry, Second Military Medical University, Shanghai. China
4Administration office, No. 102 Hospital of Chinese People´s Liberation Army, Changzhou, Jiangsu. China
5Department of Psychiatry, Suzhou Psychiatric Hospital, Suzhou, Jiangsu. China
6Department of Psychiatry and Psychology, Second Military Medical University, Shanghai. China
7Department of Laboratory, No. 102 Hospital of Chinese People´s Liberation Army, Changzhou, Jiangsu. China
8Department of Rehabilitation, No. 102 Hospital of Chinese People´s Liberation Army, Changzhou, Jiangsu. China
9GoPath Diagnostic Laboratory Co. Ltd, No. 801, Changwuzhong Road, Changzhou, Jiangsu. China
10Department of Psychiatric Medicine, Bengbu Medical College, Bengbu, Anhui
11First Center for Mental Health of Civil Affairs Bureau, Minhang District, Shanghai. China
USA
ABSTRACT
Background and Objectives: Currently, there is a serious need to find practical biomarker(s) for Major Depressive Disorder (MDD) therapeutic target(s). This study aimed to investigate the association between microRNA (miRNA, miR) expression level in Peripheral Blood Mononuclear Cells (PBMCs) and symptomatology improvement in MDD patients before and after six-week antidepressant treatment.
Methods: By using an Affymetrix array that covers 723 human miRNAs, 26 miRNAs were identified with significantly altered expression in PBMCs in MDD patients, of which 10 miRNAs were selected for quantitative real-time Reverse Transcription Polymerase Chain Reaction (RT-PCR) study. Twenty out of all the 81 MDD patients were selected for miRNA expression levels testing and symptomatology assessments before and after six-week treatment.
Results: Compared with the control group, the expression levels of miR-26b, miR-4743, miR-4498, miR-4485 and miR-1972 of the MDD group were significantly higher (P < 0.05); the changes of expression levels of miR-4743, miR-4498, miR-4485 and miR-1972 were positively related to retardation improvement (P < 0.05), and the change of expression level of miR-26b negatively to the improvement of day and night change (P < 0.05); regression analysis result demonstrated that the alteration of miR-4485 expression accounted for 28.8% of retardation improvement (P < 0.05).
Conclusions: These five miRNAs (miR-4743, miR-4498, miR-4485, miR-1972 and miR-26b) may serve as biomarker for MDD diagnosis and therapeutic targets for MDD treatment.
Key words: Major Depressive Disorder; microRNA; Antidepressant; Biomarker; Therapeutic target.
Introduction
Major Depressive Disorder (MDD) is a common mood disorder, affecting approximately 300 million people worldwide with an estimated lifetime prevalence rate of 5% to 19% in the general population1,2. Though its molecular and cellular pathophysiology has not been completely elucidated, it has been suggested that nearly 50% of the risk for depression is contributed to genetic factors3. Overwhelming evidences have indicated that depression is associated with altered neuronal and structural plasticity and neurogenesis4-7, or with an inability of the brain to make appropriate adaptive responses to environmental stimuli due to impaired synaptic plasticity8-10. Despite a tremendous amount of researches, the molecular and cellular mechanisms associated with the etiopathology of major depression are not clearly understood.
MicroRNAs (miRNAs, miR) regulate gene expression at the post-transcriptional level, playing a significant role in coordinating nervous system development and neuronal function in the mature brain. Amounting evidences have suggested that a relatively large number of miRNAs are expressed in the brain11,12, and that they regulate gene expression during brain differentiation and development13, and in processes such as the establishment and maintenance of dendrites14 and neurite outgrowth15. Altered expressions of miRNAs have been demonstrated to contribute to genetic and biological basis of neuropsychiatric disorders, particularly Huntington disease16,17, Parkinson disease18,19, and Tourette's syndrome20. Expression profiling studies in postmortem gray matter and peripheral blood samples of individuals with MDD have implicated numerous miRNAs21-23. It has been increasingly acceptable that aberrant expressions of certain miRNAs have a significant role in underlying pathophysiology of MDD. There have been a number of studies investigating into the role of altered miRNA expression in the molecular mechanism of depression. Xu et al. conducted an association analyses in 1088 MDD patients and 1102 control subjects from Chinese Han Population, and found that a polymorphism within miR-30e was positively correlated with depression and its symptomatic onset21. In a microarray screening study, Maussion et al. observed that an increase of miR-185 expression levels regulates, at least in part, the TrkB-T1 decrease observed in the frontal cortex of suicide completers, which was later validated by a larger sample of 38 suicides and 17 controls24. In another study, Smalheiser et al. measured the expression of miRNAs in prefrontal cortex of 18 antidepressant-free depressed suicide and 17 well-matched non-psychiatric control subjects using multiplex RT-PCR plates, and found widespread changes in miRNA expression that were likely to participate in pathogenesis of major depression and/or suicide25. These studies are suggesting that miRNAs are not only involved with the pathophysiology of depression, but also with the symptoms of depression. By far, the only symptom of depression patients that has been noted was suicidal behaviors. There has been a lack of evidences supporting the view that certain miRNAs correlate with certain symptomatologies.
We thus hypothesize that certain miRNAs correlate with certain symptomatologies in depression patients, and covariation exists between corresponding miRNA and symptomatology along the antidepressant treatment course. In this study, a total of 81 depression patients and 46 normal controls were enrolled for the microarray analysis, and 20 out of the 81 patients and their corresponding controls were selected for antidepressant treatment course to investigate into the alteration of miRNA expression and symptomatology improvement, in an effort to clinically validate certain miRNAs as specific blood-based therapeutic targets in depression patients.
Methods and materials
Participants
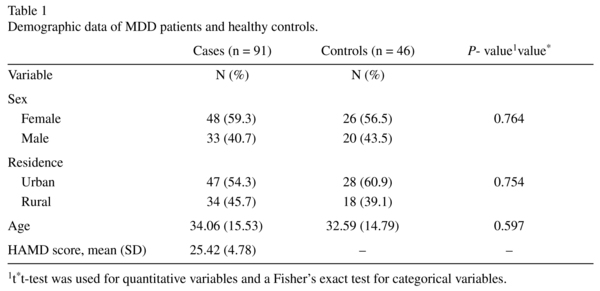
Ninety-one major Major Depressive Disorder (MDD) patients, aged from 15 to 68, fulfilling the criteria as defined by the Diagnostic and Statistical Manual 4th edition (DSM-IV), were enrolled from No.102 Hospital of the Chinese People's Liberation Army (PLA) from Aug. 2012 to Oct. 2013. Clinical diagnoses of the patients were made by at least two psychiatrists, and the diagnoses were further confirmed by an additional experienced clinical psychiatrist. All patients were prior to any antidepressant or antipsychiatric treatment, or in the absence of antidepressant or antipsychiatric medication within at least 3 months. No patients had history of severe medical diseases, other psychiatric disorders, structural brain disorders, mental retardation, unstable psychiatric features and movement disorders. Also, patients who had brain injury causing traumatic amnesia longer than 24 hours and who received blood transfusion within a month or electroconvulsive therapy within 6 months, were excluded from the study. In addition, 46 healthy controls, aged from 15 to 68, without any family history of major psychiatric disorders (SZ, bipolar disorder and MDD) within the last three generations were recruited. Similarly, all healthy controls were without any history of blood transfusion or severe traumatic event within a month. All individuals enrolled in the study provided written informed consent. The study was approved by local Institutional Review Board.
Blood collection and RNA extraction
Whole blood (5 ml) was collected from each subject using EDTA anticoagulant tube and processed within 3 hours. Peripheral Blood Mononuclear Cells (PBMCs) were isolated from the blood by Ficoll density centrifugation, then transferred into fresh RNase/DNase-free 2 ml microcentrifuge tube, and stored at -80oC until use. Total RNAs were extracted from the PBMCs with the mirVanaTM PARISTM Kit (Applied Biosystems, p/nAM1556) according to the manufacturer's protocol, and were quantified by the NanoDrop ND-2100 (Thermo Scientific). The RNA integrity was assessed using Agilent 2100 (Agilent Technologies). To ensure a robust analysis for the following procedures, samples with an RNA integrity number (RIN) inferior to 8 were excluded.
MicroRNA microarray expression profiling
Total RNAs from three MDD patients (male, 30 years; male, 31 years; female, 28 years) and three controls (male, 30 years; male, 31years; female, 27 years) were used for miRNA microarray profiling. miRNA expression was measured by Affymetrix miRNA 3.0 array (Affymetrix, Santa Clara, CA, USA) containing probes for a total of 723 human miRNAs. The sample labeling, microarray hybridization and washing were performed based on the manufacturer's standard protocols. Briefly, total RNA were tailed with Poly A and then labeled with Biotin. After, the labeled RNAs were hybridized onto the microarray. Having washed and staining the slides, the arrays were scanned by the Affymetrix Scanner 3000 (Affymetrix). The scanned images were analyzed using Expression Console software (version1.3.1, Affymetrix).
Real-time quantitative reverse-transcription PCR (qRT-PCR)
According to microarray results and literature, candidate miRNAs were chosen for further validation with real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR). Blood samples from 91 MDD patients were used to validate the findings from microRNA profiling. Total RNAs were isolated from the PBMCs using Trizol reagent (Invitrogen®, USA) for quantitative detection of miRNA. Complementary DNA was synthesized using the Reverse Transcription TaqMan MicroRNA Reverse Transcription Kit (Applied Biosystems, inc., USA, P/N:4366596) according to the manufacturer's instructions. Each RT reaction consisted of 5μl of RNA, dNTP mix (100mM) 0.15μl, Multiscribe RT enzyme (50U/μl) 1.00ul, 10×RT Buffer 1.5μl, RNase Inhibitor 0.19ul, Nuclease free water 4.16μl, TaqMan MicroRNA Assays 3μl, in a total volume of 15μl. Reactions were performed using the following conditions: 30 min at 16oC, 30 min at 42oC, 5 min at 85oC, and held at 4oC. Real-time PCR was performed using Applied Biosystems 7900HT Real-Time PCR System (Applied Biosystems, Inc., USA), with 10μl PCR reaction mixture that included 2μl of the cDNA, 5μl of 2 × TaqMan Universal Master Mix II (Applied Biosystems, Inc.), 0.25μl of miRNA-specific primer/probe mix (Applied Biosystems, inc.), 0.5μl of TaqMan MicroRNA Assays (Applied Biosystems, CA) and 2.25μl of double distilled water. PCR reactions in a 384-well plate were run at 95oC, for 10 min, followed by 40 cycles of 95oC, for 15 s, 60oC, for 1 min. Each sample was run in triplicate for analysis. The miRNA-specific stem-loop primers were supplied by the TaqMan MicroRNA Assays (Applied Biosystems, Inc.) based on the microRNA sequences obtained from the miRBase database. Data were collected using the SDS 2.3 software (Applied Biosystems, Inc.). After normalized to RNU48, the expression levels of microRNAs were calculated using the 2-ΔCt method26.
Clinical symptomatology assessment instrument
Altogether three psychiatrists were responsible for scale assessment. Hamilton rating scale for depression (HAMD) and Clinical Global Impression (CGI) were used to assess the clinical symptoms of all patients before and six weeks after medication intervention. All three psychiatrists were collectively trained for unified procedure.
HAMD is a classic medical scale used for measuring symptom severity of MDD patients. It was published with three versions in 1960, namely the 17-item version, the 21-item version and the 24-item version27. The 24-item version of HAMD was used in this study. It covered 7 dimensions, including anxiety/somatization, body weight, cognitive disorder, day and night change, retardation, sleep disorder and hopelessness. In this study, we adopted the Chinese version of this scale, which has been naturalized and verified for its reliability and validity28. The total score and symptomatology scores were used for statistical analysis.
CGI was designed by WHO for measuring clinical treatment effects of any kind of psychiatric diseases29. Its reliability and validity also have been naturalized and verified30, applicable to Chinese people.
Clinical data collection and scales assessment
General information (including name, gender, age, ethnicity, education level, occupation, income level, marital status, substance abuse history and family history of psychotic diseases) from all subjects in the MDD group were collected. One attending doctor and two resident psychiatric doctors participated in evaluating illness severity and treatment effects, plus recording dosage information (including medication, dosage, side effects and treatment course) using HAMD and CGI. Likewise, the general information of healthy controls was also recorded. Procedures for data collection were standardized to ensure the accuracy of the collected data. The total score and factors score of HAMD and global improvement (GI) of CGI scale were used for statistical analysis.
Medication intervention
Twenty out of all MDD patients were selected by systematic random sampling for clinical intervention observation. A combination of medication were applied, including venlafaxine with dosage range from 150 mg to 225 mg, sertraline with dosage range from 50 mg to 150 mg and mitrazapine with dosage range from 22.5 mg to 45 mg, all of which are new-type antidepressants, inhibiting the reuptake of serotonin receptors and norepinephrine receptors and triggering less side effects.
Statistical analysis
All data were processed by SPSS v17.0. Real-time quantitative PCR data were collected by SDS software and relative levels of the tested miRNA in plasma were calculated based on the following formula using RNU48 as an internal reference: 2-ΔCt(ΔCt = CtmiRNA - CtRUN48). Wilcoxon rank sum test was used to test the differences of 10 miRNA expression levels between MDD group and control group. Paried sample t test was used to test the differences of assessment scores by HAMD and CGI. Pearson correlation test was carried out for testing the correlation of miRNA expression level change with HAMD and CGI scores changes. Finally, logistic regression analysis was performed to determine the accountability of miRNA for symptomatology improvements. All statistical tests were two-tailed, and P < 0.05 was considered statistically significant.
Results
Demographic data of the MDD patients
Of the 91 MDD patients, 10 of them had hemolysis and showed a extremely high CT value of RNU48 (the internal normalization control), thus were removed for further analysis. So there were 81 cases left in MDD group for final analysis. The demographic data of the 81 MDD patients is shown in Table 1. All the patients and controls were all of Han nationality. As is shown in Table 1, there were no differences in age, sex or residential locations between MDD patients and healthy controls.
Microarray analysis
Microarray analysis result showed that a total of 26 miRNAs were identified as significantly different from the control group, with 5 increased and 21 decreased at P = 0.05 or better. According to Allison31, we selected the group of miRNAs by the means of microarray analysis. Ten of the 26 miRNAs were selected by the following standards: miRNAs with higher fold change in micro-array results; miRNAs that had been reported in Public biological Biological databasesDatabases, such as GO, KEGG and BioCarta Pathways; miRNAs that could be detected in every sample.
Comparison of miRNA expression levels between MDD group and control group before medication
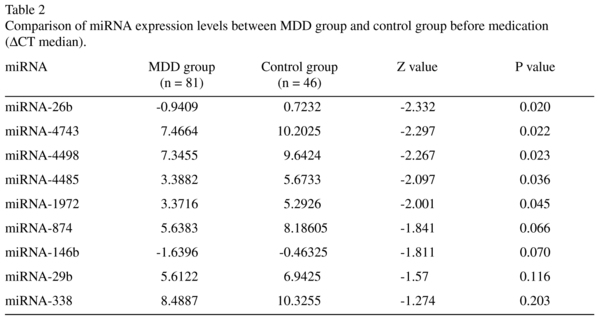
As is shown in Table 2, wilcoxon rank sum test result revealed that the ΔCT values of 5 miRNAs (miR-26b, miR-4743, miR-4498, miR-4485, miR-1972) in MDD group were significantly lower than those of the control group (P < 0.05), which indicated the expression levels of these 5 miRNA were significantly higher than those of the control group as was computed by 2-ΔCT.
Comparison of miRNA expression levels between different genders, ages and residential locations in the MDD group before medication
The demographic data, including gender, age and residential locations, was also considered to exclude their influence upon our further analysis. The 81 SZ patients were further classified into higher and lower age subgroups according to age percentile, different gender subgroups, namely male subgroup and female subgroup, and different residential locations subgroups, namely urban subgroup and rural subgroups. Patients with ages above 42 were classified into the higher age subgroup (P75, n = 21) and those with ages below 21 into the lower age subgroup (P25, n = 21). As is shown in Table 3, no significant differences of expression levels of selected miRNAs were found between different genders, age and residential locations subgroups.
Comparison of HAMD score and its factor scores between before and after antidepressants treatment in MDD group
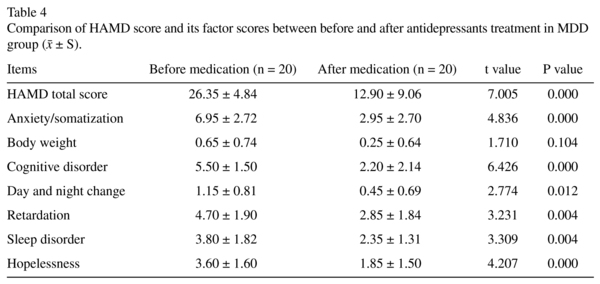
As is shown in Table 4, paired sample t test result demonstrated that the total score and most factor scores except body weight were significantly lower than those of control group (P < 0.05).
Correlation of miRNA expression alteration and HAMD and GI score changes before and after antidepressants treatment
As is shown in Table 5, the ΔCt changes of miR-1972, miR-4485, miR-4498 and miR-4743 were positively correlated with the improvement of retardation (P < 0.05); the ΔCt changes of miR-26b was negatively correlated with the improvement of day and night change symptom (P < 0.05). The ΔCt changes of any miRNA was not significantly correlated with GI improvement (P < 0.05).
Logistic regression analysis of the effects of miRNA expression alteration upon certain symptomatology improvement
Taking the expression alteration of miRNA as independent variable, and the treating effect of retardation and day and night change as dichotomous dependent variable, logistic regression analysis was carried out to verify the association between miRNA expression alteration and symptomatology improvement. The treating effect was determined by score reduction rate (Spre-Spost/Spre, Spre represents pre-medication score, Spost represents post-medication score). Score reduction rate less than 0.5 was taken as low treating effect, and score reduction rate more than 0.5 as high treating effect. As Table 6 indicates, ΔmiR-4498 was entered into the regression function with retardation factor as independent variable, accounting for 28.8% of retardation improvement (P < 0.05). On the other hand, the analysis result of logistic regression with day and night change factor as independent variable was insignificant (P > 0.05).
Discussion
Currently, pharmacological treatments for MDD mainly involve SSRI, pharmacological mechanism of which remains elusive, which makes it impossible to customize treatment and predict treatment effects. In recent years, the emerging trend of miRNA research had made it possible to tap the potential of manipulating miRNAs as a strategy for novel antipsychotic development32,33. Some studies have reported that certain drugs could alter miRNA expression. Zhou et al. investigated hippocampal miRNA expression changes following chronic treatment with lithium and valproate, and found 37 differentially expressed miRNAs in lithium group and 31 differentially expressed miRNAs in valproate group34. Kocerha et al. found that miR-219 levels were significantly reduced by 50% in the prefrontal cortex of mice 15 min after injection of dizocilpine (0.5 mg/kg), and miR-219 expression levels returned to basal levels by 120 min after treatment35. Chen et al. quantitatively analyzed the expression patterns of 13 miRNAs in 20 lymphoblastoid cell lines (LCLs) with or without Li treatment in culture, identified significant changes in 7 of the 13 miRNAs tested in LCLs sampled at treatment day 4. Four of the 7 significant miRNAs, miR-34a, miR-152, miR-155, and miR-221 consistently changed expression in the same LCLs at a longer treatment time point day 1636. Bocchio-Chiavetto et al. conducted a whole-miRNome quantitative analysis with qRT-PCR of the changes in the blood of 10 depressed subjects after 12 weeks of treatment with escitalopram, and found that 30 miRNAs were differentially expressed after the AD treatment: 28 miRNAs were up-regulated, and 2 miRNAs were strongly down-regulated37. However, there is hardly any study so far that focuses on the association between the changes of miRNA expression and the symptomatology improvement in MDD patients.
In this study, we have excluded potential confounding factors by making comparisons of the demographic data, including gender, age and residential locations, between the MDD group and control group, and then within the MDD group. There were no differences in age, sex or residential locations between MDD patients and healthy controls nor within the MDD group. However, we found that the changes of four differentially expressed miRNAs (miR-1972, miR-4485, miR-4498 and miR-4743) were positively correlated with the improvement of retardation after six-week antidepressant medication. As the HAMD score lowered along the medication course, the symptoms improved, and the expression levels of miRNAs down regulated. Logistic regression analysis further revealed that the expression of miR-4498 was apparently linearly related to retardation score. These five differentially expressed miRNAs have been scarcely reported before. According to Fan38, there was a significant enrichment in several pathways associated with brain functions and nervous system, supporting the hypothesis that differentially-regulated miRNAs may be involved in mechanism underlying development of MDD. On the other hand, the change of miR-26b expression was also found to be negatively correlated with the day and night factor score change, which indicated that the higher of the expression level of miR-26b was, the more severe the day and night change symptom would be. According to Absalon39, ectopic overexpression of miR-26b in rat primary postmitotic neurons led to the DNA replication and aberrant Cell Cycle Entry (CCE) and, in parallel, increased tau-phosphorylation, which culminated in the apoptotic cell death of neurons39. However, studies involving miR-26b in MDD patients have not seen so far. Another study demonstrated that the expression of only five malfunctioning host genes could result in esophageal cancer in comparison with cancer in other gastrointestinal organs and breast cancer. One of the five genes was S1PR2 with binding sites for miR-1972. The expression of these five target genes depends on the expression of 12 host genes encoding seven miRNAs, including miR-197240. As a transcription factor, NF-κB was demonstrated to regulate the expressions of miRNAs. But only a few miRNAs have been identified as its targets so far. By using ChIP-Seq, Gene-chip and miRNA-Seq techniques, Zhou et al. identified 14 NF-κB target miRNAs in TNFα-stimulated HeLa Cells, including miR-448541. On the other hand, miR-4498 was found to hyper-expressed in the BRCA1 breast cancer patients42. These studies have involved these five miRNAs in different diseases, but that how exactly are these five miRNAs involved in depression remains unclear. The newly discovered miRNAs that are potentially involved in the molecular and cellular mechanism of depression underscore the significance of this study. More studies are warranted to further explore the mechanism pathways of depression.
In conclusion, miR-4743, miR-4498, miR-4485, miR-1972 and miR-26b are potentially involved in pathogenesis of MDD, and may serve as therapeutic targets of antidepressant drugs. The specific mechanism underlying this interaction warrants further investigation.
Conflict of interest
None declared.
References
1. Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2013; 380 (9859): 2163-96. [ Links ]
2. Andrade L, Caraveo-Anduaga JJ, Berglund P, Bijl RV, De Graaf R, Vollebergh W, et al. The epidemiology of major depressive episodes: Results from the International Consortium of Psychiatric Epidemiology (ICPE) Surveys. Int J methods in Psychiatr Res. 2003;12 (1): 3-21. [ Links ]
3. Fava M, Kendler KS. 2000. Major depressive disorder. Neuron. 2000; 28 (2): 335-41. [ Links ]
4. Horan WP, Pogge DL, Borgaro SR, Stokes JM, Harvey PD. Learning and memory in adolescent psychiatric inpatients with major depression: A normative study of the California Verbal Learning Test. Arch Clin Neuropsychol. 1997; 12 (6): 575-584. [ Links ]
5. Bearden CE, Glahn DC, Monkul ES, Barrett J, Najt P, Villarreal V, et al. Patterns of memory impairment in bipolar disorder and unipolar major depression. Psychiatry Res. 2006; 142 (2-3): 139-50. [ Links ]
6. Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993; 75 (5): 843-54. [ Links ]
7. Dwivedi Y, Rizavi HS, Zhang H, Mondal AC, Roberts RC, Conley RR, et al. Neurotrophin receptor activation and expression in human postmortem brain: Effect of suicide. Biol Psychiatry. 2009; 65 (4): 319-28. [ Links ]
8. Abelson JF, Kwan KY, O'Roak BJ, Baek DY, Stillman AA, Morgan TM, et al. Sequence variants in SLITRK1 are associated with Tourette's syndrome. Science. 2005; 310 (5764): 317-20. [ Links ]
9. Duman RS, Malberg J, Nakagawa S, D'Sa C. Neuronal plasticity and survival in mood disorders. Biol psychiatry. 2000; 48 (8): 732-9. [ Links ]
10. Duman RS. Pathophysiology of depression: The concept of synaptic plasticity. Eur psychiatry. 2002; Jul; 17 Suppl 3: 306-10. [ Links ]
11. Fossati P, Radtchenko A, Boyer P. Neuroplasticity: From MRI to depressive symptoms. Eur Neuropsychopharmacol. 2004; 14 Suppl 5: S503-10. [ Links ]
12. Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005; 120 (1): 15-20. [ Links ]
13. Sempere LF, Freemantle S, Pitha-Rowe I, Moss E, Dmitrovsky E, Ambros V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 2004; 5 (3): R13. [ Links ]
14. Krichevsky AM, King KS, Donahue CP, Khrapko K, Kosik KS. A microRNA array reveals extensive regulation of microRNAs during brain development. RNA. 2003; 9 (10): 1274-81. [ Links ]
15. Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M, et al. A brain-specific microRNA regulates dendritic spine development. Nature. 2006; 439 (7074): 283-9. [ Links ]
16. Vo N, Klein ME, Varlamova O, Keller DM, Yamamoto T, Goodman RH, et al. A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc Nati Acad Sci U S A. 2005; 102 (45): 16426-31. [ Links ]
17. Packer AN, Xing Y, Harper SQ, Jones L, Davidson BL. The bifunctional microRNA miR-9/miR-9* regulates REST and CoREST and is downregulated in Huntington's disease. J Neurosci. 2008; 28 (53): 14341-6. [ Links ]
18. Johnson R, Zuccato C, Belyaev ND, Guest DJ, Cattaneo E, Buckley NJ. A microRNA-based gene dysregulation pathway in Huntington's disease. Neurobiol Dis. 2008; 29 (3): 438-45. [ Links ]
19. Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E, et al. A MicroRNA feedback circuit in midbrain dopamine neurons. Science. 2007; 317 (5842): 1220-4. [ Links ]
20. Wang G, van der Walt JM, Mayhew G, Li YJ, Züchner S, Scott WK, et al. Variation in the miRNA-433 binding site of FGF20 confers risk for Parkinson disease by overexpression of alpha-synuclein. Am J Hum Genet. 2008; 82 (2): 283-9. [ Links ]
21. Abelson JF, Kwan KY, O'Roak BJ, Baek DY, Stillman AA, Morgan TM, et al. Sequence variants in SLITRK1 are associated with Tourette's syndrome. Science. 2005; 310 (5764): 317-20. [ Links ]
22. Xu Y, Liu H, Li F, Sun N, Ren Y, Liu Z, et al. A polymorphism in the microRNA-30e precursor associated with major depressive disorder risk and P300 waveform. J Affect Disord. 2010; 127 (1-3): 332-6. [ Links ]
23. Smalheiser NR, Lugli G, Rizavi HS, Torvik VI, Turecki G, Dwivedi Y. MicroRNA expression is down-regulated and reorganized in prefrontal cortex of depressed suicide subjects. PloS One. 2012; 7 (3): e33201. [ Links ]
24. Mouillet-Richard S, Baudry A, Launay JM, Kellermann O. MicroRNAs and depression. Neurobiol Dis. 2012; 46(2): 272-8. [ Links ]
25. Maussion G, Yang J, Yerko V, Barker P, Mechawar N, Ernst C, et al. Regulation of a truncated form of tropomyosin-related kinase B (TrkB) by Hsa-miR-185* in frontal cortex of suicide completers. PloS One. 2012; 7 (6): e39301. [ Links ]
26. Smalheiser NR, Lugli G, Zhang H, Rizavi H, Cook EH, Dwivedi Y. Expression of microRNAs and Other Small RNAs in Prefrontal Cortex in Schizophrenia, Bipolar Disorder and Depressed Subjects. PloS One. 2014; 9 (1): e86469. [ Links ]
27. Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative CT method. Nat protoc. 2008; 3 (6): 1101-8. [ Links ]
28. Hamilton M. Development of a rating scale for primary depressive illness. Br J soc clin psychol. 1967; 6 (4): 278-96. [ Links ]
29. Zhang ZJ. Scale Manual of Behavioral Medicine. Beijing: Chinese Medicine Electronic Audiovisual Press; 2005. p. 77-485. [ Links ]
30. Guy W. ECDEU Assessment Manual for Psycho-patho-logy. Rockville, MD: U.S National Institute of Health, Psychopharmacology Research Branch; 1976: 218-221. [ Links ]
31. Wu WY. Clinical Global Impression(CGI). Shanghai Psychiatric Medicine, 1984; 2: 76-77. [ Links ]
32. Allison DB, Cui X, Page GP, Sabripour M. Microarray data analysis: From disarray to consolidation and consensus. Nat Rev Genet. 2006; 7 (1): 55-65. [ Links ]
33. Kosik KS. The neuronal microRNA system. Nat Rev Neurosci. 2006; 7 (12): 911-20. [ Links ]
34. Dwivedi Y. Evidence demonstrating role of microRNAs in the etiopathology of major depression. J chem neuroanat. 2011; 42 (2): 142-56. [ Links ]
35. Zhou R, Yuan P, Wang Y, Hunsberger JG, Elkahloun A, Wei Y, et al. Evidence for selective microRNAs and their effectors as common long-term targets for the actions of mood stabilizers. Neuropsychopharmacology. 2009; 34 (6): 1395-405. [ Links ]
36. Kocerha J, Faghihi MA, Lopez-Toledano MA, Huang J, Ramsey AJ, Caron MG, et al. MicroRNA-219 modulates NMDA receptor-mediated neurobehavioral dysfunction. Proc Nati Acad Sci U S A. 2009; 106 (9): 3507-12. [ Links ]
37. Chen H, Wang N, Burmeister M, Mclnnis MG. MicroRNA expression changes in lymphoblastoid cell lines in response to lithium treatment. Int J Neuropsychopharmacol. 2009; 12 (7): 975-81. [ Links ]
38. Bocchio-Chiavetto L, Maffioletti E, Bettinsoli P, Giovannini C, Bignotti S, Tardito D, et al. Blood microRNA changes in depressed patients during antidepressant treatment. Eur neuropsychopharmacol. 2013; 23 (7): 602-11. [ Links ]
39. Fan HM, Sun XY, Guo W, Zhong AF, Niu W, Zhao L, et al. Differential expression of microRNA in peripheral blood mononuclear cells as specific biomarker for major depressive disorder patients. J Psychiatr Res. 2014 Aug 24. Pii:S0022-3956(14)00243-X. (Epub ahead of print). [ Links ]
40. Absalon S, Kochanek DM, Raghavan V, Krichevsky AM. MiR-26b, upregulated in Alzheimer's disease, activates cell cycle entry, tau-phosphorylation, and apoptosis in postmitotic neurons. J Neurosci. 2013; 33 (37): 14645-59. [ Links ]
41. Berillo O, Régnier M, Ivashchenko A. Binding of intronic miRNAs to the mRNAs of host genes encoding intronic miRNAs and proteins that participate in tumourigenesis. Comput Biol Med. 2013; 43 (10): 1374-81. [ Links ]
42. Zhou F, Wang W, Xing Y, Wang T, Xu X, Wang J. NF-KappaB target microRNAs and their target genes in TNF-stimulated HeLa Cells. Biochim Biophys Acta. 2014; 1839 (4): 344-54. [ Links ]
43. Murria Estal R, Palanca Suela S, de Juan Jiménez I, Egoavil Rojas C, García-Casado Z, Juan Fita MJ, et al. Breast Cancer. Res Treat. 2013; 142 (1): 19-30. [ Links ]
![]() Correspondence:
Correspondence:
Dr. Li-yi Zhang
Prevention and Treatment Center for Psychological
Diseases, No.102. Hospital of Chinese People's Liberation
Army, North Peace Road 55. Changzhou
Jiangsu 213003
People's Republic of China.
Tel. 86 519 83064556
Fax: 86 519 83064560
E-mail: zly102@126.com
Received: 9 September 2014
Revised: 19 October 2014
Accepted: 21 October 2014