My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Gaceta Sanitaria
Print version ISSN 0213-9111
Gac Sanit vol.23 n.4 Barcelona Jul./Aug. 2009
Estimating the health and economic benefits associated with reducing air pollution in the Barcelona metropolitan area (Spain)
Estimación de los beneficios en salud y económicos asociados a la reducción de la contaminación atmosférica en el área metropolitana de Barcelona (España)
Laura Péreza, Jordi Sunyera and Nino Künzlia,b
aCentre de Recerca en Epidemiologia Ambiental (CREAL) Institut Municipal d'Investigació Mèdica (IMIM), Centro de Investigación Biomédica en Red de Epidemiología y Salud Pública (CIBERESP), Barcelona, Spain
bInstitut Català de Recerca i Estudis Avançats (ICREA), Barcelona, Spain
Dirección para correspondencia
ABSTRACT
Objectives: To estimate the health and economic benefits that would result from two scenarios of improved air quality in 57 municipalities of the metropolitan area of Barcelona.
Methods: We used attributable fractions and life tables to quantify the benefits for selected health outcomes, based on published concentration-response functions and economic unit values. The mean weighted concentration of PM10 for the study population was estimated through concentration surface maps developed by the local government.
Results: The annual mean health benefits of reducing the mean PM10 exposure estimated for the population in the study area (50µg/m3) to the annual mean value recommended by the World Health Organization (20µg/m3) were estimated to be 3,500 fewer deaths (representing an average increase in life expectancy of 14 months), 1,800 fewer hospitalizations for cardio-respiratory diseases, 5,100 fewer cases of chronic bronchitis among adults, 31,100 fewer cases of acute bronchitis among children, and 54,000 fewer asthma attacks among children and adults. The mean total monetary benefits were estimated to be 6,400 million euros per year. Reducing PM10 to comply with the current European Union regulatory annual mean level (40µg/m3) would yield approximately one third of these benefits.
Conclusions: This study shows that reducing air pollution in the metropolitan area of Barcelona would result in substantial health and economic benefits. The benefits are probably underestimated due to the assumptions made in this study. Assessment of the health impact of local air pollution is a useful tool in public health.
Key words: Air pollution. Health impact assessment. Preventive medicine. Attributable cases. Monetary evaluation.
RESUMEN
Objetivos: Se presenta una estimación de los beneficios para la salud y económicos de dos escenarios de mejora de la calidad del aire ambiental en 57 municipios del área metropolitana de Barcelona.
Métodos: Usando fracciones atribuibles y tablas de vida, se cuantificaron los beneficios para los indicadores de salud seleccionados basándose en funciones de concentración-respuesta y en unidades monetarias publicadas. La concentración media ponderada de PM10 para la población del estudio se obtuvo mediante mapas de concentraciones desarrollados por el gobierno local.
Resultados: Los beneficios anuales de reducir la exposición media a PM10 estimada para la población del área de estudio (50µg/m3) al valor anual medio recomendado por la Organización Mundial de la Salud (20µg/m3) se estiman en 3.500 muertes menos (representando en media un aumento de la esperanza de vida de 14 meses), 1.800 ingresos hospitalarios menos por causas cardiorrespiratorias, 5.100 casos menos de bronquitis crónicas en adultos, 31.100 casos menos de bronquitis agudas en niños y 54.000 crisis asmáticas menos en niños y adultos. Los beneficios económicos totales se estiman en una media de 6.400 millones de euros por año. Reducir las PM10 al nivel anual medio legislado por la Unión Europea (40µg/m3) representaría aproximadamente una tercera parte de estos beneficios.
Conclusiones: Este estudio indica que reducir la contaminación atmosférica en el aérea metropolitana de Barcelona resultaría en beneficios en salud y económicos muy sustanciales. Una infravaloración es probable debido al enfoque metodológico tomado. Las evaluaciones de impacto en salud son herramientas útiles en salud pública.
Palabras clave: Contaminación atmosférica. Evaluación impacto en salud. Medicina preventiva. Casos atribuibles. Evaluación monetaria.
Introduction
Experimental studies conducted in cellular models, animals and humans, and numerous epidemiological studies have shown that short-term and long-term exposure to current levels of anthropogenic air pollution lead to morbidity and mortality in humans1,2.
In the last decade, scientists and public health agencies have translated these research findings into quantities that reflect the burden on health attributable to air pollution in a given region, country, or city3-5. This process is important because it directly informs policy makers and the public about the current situation or the impact of future or past air pollution policies. Regional studies have also been used as tool for prioritizing national regulation and policy development. Given that every society has limited resources to allocate, decision makers request that health quantities be transformed into monetary valuations to be compared with mitigation investments. Thus, valuation of health effects is a critical component of health impact assessment (HIA)6.
Air quality characterized by the inhalable fraction of particulate matter (PM10), nitrogen dioxide (NO2) and other pollutants is poor in the Barcelona metropolitan area, with trends in recent years showing progressive degradation7. In 2007, when the local government initiated actions to reduce air pollution, questions arose about the potential health and economic benefits of abatement strategies. Air pollution burden for Spain and Barcelona has been partially estimated by different European studies8-10. An assessment of 26 European cities estimated that reducing PM2.5 levels in the city of Barcelona would decrease the numbers of deaths among people aged 30 years and over from 1.5% (95% confidence interval [CI]: 0.4-3.0%) to 5.4% (95%CI: 1.4-10.4%)8. In Spain, only one study has incorporated cost into this type of evaluation10. Comparing year 2000 emission levels to current legislation, this study estimated that air pollution caused morbidity diseases and approximately 22,000 premature deaths. This impact translated into a total cost of 400 to 1,000 euros per year per capita, depending on the method of calculation selected.
This article estimates the health and economic benefits associated with reducing air pollution in the Barcelona metropolitan area from current levels to the European Union (EU) standards and World Health Organization (WHO) targets. Few HIAs have analyzed air pollution burden at the community level, although local analysis might lead to better decision making. This study demonstrates how HIA may support public health policy at a scale that reflects conditions for the community for which policies will be designed or implemented.
Methods
This study follows standard methodology to derive the risk attributable to outdoor air pollution10. The methods require the following information: selection of study area, exposure assessment, health-outcomes, concentration-response functions, scenario of change in exposure considered, and unit monetary values.
Study area
We selected as area of study a continuous geographic area constituted by a total of 57 municipalities, including the municipality of Barcelona. For the purpose of our study, we denominated this area «Barcelona metropolitan area». This study area includes a total population of 3,868,663 inhabitants (year 2004). The municipality level reflects the smallest political unit for which routine health data (i.e. mortality data and hospital admission data) were available. This area was selected for its geographical continuity and potential uniformity in terms of air pollution exposure. figura 1 presents the 57 municipalities included in the study area with population per 1,000 for 2004.
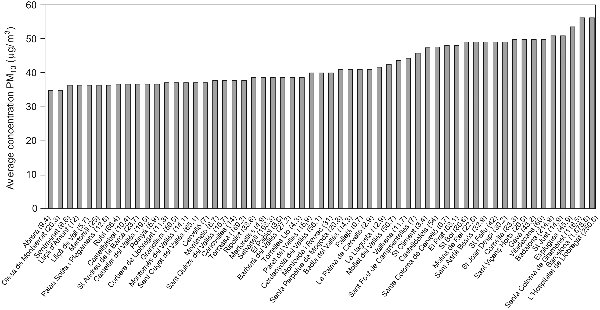
Figure 1. Average PM10 concentrations in urbanized areas of the 57 municipalities
included in the area of study (population per 1000 for 2004 in parenthesis).
Characterizing pollution
Air pollution is a complex mixture of often highly correlated constituents and pollutants2. Health impact assessments of air pollution rely on epidemiological studies, using one marker of air quality because it is not appropriate to sum the risk for correlated pollutants11. For this study, we selected PM10 as marker of air pollution to follow the approach used in other HIAs4,12 and based on the availability of exposure data for the area of study.
To derive benefits for a specific change in air pollution is necessary to determine the level of exposure of the population before changes occur. We considered the current levels as the point of reference for future changes. The population exposure for PM10 was represented by an average population-weighted concentration derived from a PM10 concentration surface map developed by local authorities. This map estimated 2004 spatial concentration of PM10 through dispersion models that took into account emission sources. Modeled surfaces were validated by comparing concentration levels at fixed monitoring sites with predicted concentrations7. This modeled surfaces are used by local authorities to develop air pollution mitigation strategies in the study area. In the context of this study, the population exposure is understood as the average concentration representative of people's residence. Several approaches are available to determine population exposures depending on the level of detail of the data available. No detailed population distribution was available for our area of study. The estimated population exposure was thus obtained by calculating municipality mean concentrations in urbanized areas based on the government surface map. These means were weighted by the population of each municipality to obtain an average concentration interpreted as the average exposure concentration for the area of study.
Health-outcomes and concentration-response functions
Three main families of health outcomes were evaluated: mortality, morbidity including chronic bronchitis and asthma related symptoms, and health care use represented by hospital admissions for cardiovascular and respiratory diseases. The association between outdoor air pollution and health-outcome frequency is described with a concentration-response function (CRF). We selected CRF from peer-reviewed literature used in other HIAs4,12-17. Some of this CRF were expressed for other size fractions of particulate matter (PM), mainly PM2.5 or Total Suspended Particles, rather than PM10. When needed, conversion factor of 1/0.6 and 0.6 was used to transform levels of PM2.5 or Total Suspended Particles into PM10, respectively4. Demographic and baseline frequencies of health outcomes were obtained from local health and statistics authorities or, when needed, extrapolated from studies conducted in Europe (tabla 1). Characteristics and limitations of the input information are discussed later in the text.
Scenario of change in exposure
We provided risk estimates for two scenarios to reflect the stepwise improvements of air quality expected in the area. The main scenario estimated the ultimate benefits for health if air quality characterized by PM10 was in compliance with the air quality guidelines recommended by the WHO. WHO recommends a mean annual level for PM10 of 20µg/m3 or less to protect human health2. The second scenario estimated health benefits for an intermediate step of air pollution abatement, namely reducing current PM10 levels to the EU air quality standard. The EU regulation states that PM10 levels should not have exceeded a mean annual level of 40µg/m3 since 2005. This is the year 2010 target of current local policy efforts because the current EU limit has not yet been met in several areas of Catalonia. In the context of this study, the WHO and the EU limit values are interpreted as average population exposure since limit values apply to any point of the territory.
Derivation of attributable number of cases
We expressed results as the number of attributable cases for the change of exposure under consideration. Within this evaluation, attributable cases are interpreted as the number of health events that could be prevented per year if air pollution was reduced. Attributable cases were derived from attributable population fractions applied to number of outcome cases in the population. All calculations were conducted at the aggregated level of the study area. Attributable fractions were derived with the standard formula AFpop=[pp (RR-1)]/[pp (RR-1)+1], where pp represents the fraction of the population exposed to air pollution and assumed as one, and RR represents the relative risk (RR) of the CRF for the change in PM10 exposure considered. RR is derived as RR=exp (ßxΔC) with ß the slope of the CRF expressed for 1µg/m3 and ΔC the change in exposure considered in µg/m3. The statistical models used to derive CRFs most often provide odds ratios (OR) from logistic regression models rather than RR. For rare events or small effects, RR and OR are similar. However, for frequent events and when the OR is large, OR may overestimate the true RR. To take this into account, we corrected OR with a standard formula18.
Derivation of life years lost
The concept of attributable or preventable death is conceptually flawed as death is ultimately not preventable but can only be postponed. Those exposed to lower pollution would in fact have, in average, a longer life expectancy due to reduction in death rates. We estimated average increase in life expectancy for our study population using standard life table methods19.
Monetary valuation of health benefits
Economists in different settings have attempted to develop alternative measures of values for benefits arising from clean air20. Ideally these measures should represent all the losses to individuals and to society that result from adverse health effects, and reflect preferences and decision-making processes similar to those of daily life20. In this evaluation, the value to attach to a reduction of the risk of death and other end points were based on the Willingness-To-Pay (WTP) approach. Unit monetary values based on WTP are derived from market choices that reduce risk to health or life indirectly20. We used same monetary values as those proposed in the air pollution European cost-benefit analysis and derived from empiric studies10. In this evaluation, a mean and median value are proposed for some outcomes. Since no consensus has been reached as to which value reflects better the value of health,10 we used a single mid monetary unit in the core estimates and discussed the impact of the variability of values in the sensitivity analysis. Values were transformed to 2006-price year using the real gross domestic product average annual increase for Spain21. Economic benefits were calculated multiplying the unit monetary value by the number of attributable cases obtained for each scenario.
Expression of uncertainty
The various steps described above come with a range of assumptions and uncertainties, which differ for the different outcomes. To reflect these uncertainties results are presented with a point estimate as well as an upper and lower bound given by the 95%CI of the CRF. For other uncertainties and assumptions, a series of sensitivity analyses were conducted to assess the differences between our mean results and results under alternative assumptions. Figura 2 summarizes the percent change that would have applied to our central benefits of the different health point studied if alternative assumptions had been selected. Alternative assumptions were grouped into four: use of the upper and lower bound of the 95%CI of CRF; use of other CRFs; assuming some error in the baseline frequencies used; and use of alternative monetary values or approach.
Results
Based on the 2004 concentration surface maps, PM10 mean average concentrations for the study area was 43µg/m3, ranging between 35 and 56µg/m3 depending on the municipality. The highest mean concentrations were observed for municipalities with the highest number of inhabitants (i.e municipality of Barcelona) (figura 1). For the study area, we estimated a PM10 average exposure concentration of 50µg/m3 representative for a population of approximately four million inhabitants (all ages).
Following the traditional approach of reporting the burden of disease of air pollution by attributable deaths, we estimated that approximately 3,500 (95%CI: 2,200-4,800) annual deaths, about 12% (95%CI: 7-16) of all natural deaths, could be reduced if outdoor air pollution was reduced to levels recommended by the WHO (tabla 2). We estimated that this translates to an average 14 months increase in life expectancy (95%CI: 6-22 months). In addition, we estimated that there could be nearly 1800 (95%CI: 670-2,100) fewer hospitalization for cardio-respiratory diseases; 5,100 (95%CI: 550-8,500) fewer cases of chronic diseases in adults; 31,100 (95%CI: 17,500-40,500) fewer cases of acute bronchitis in children; and approximately 54,000 (95%CI: 27,400-75,700) fewer asthma attacks in adults and children if exposure to air pollution was reduced to the levels recommended by the WHO. One third of the benefits of the WHO scenario could already be obtained if air pollution was decreased to EU regulatory levels. Because the associations between air pollution and the health outcomes considered in this study are linear, these latter benefits can also be interpreted as the intermediate benefits that would be obtained per each decrease of 10µg/m3 of ambient concentrations in the area of study.
Results of the monetary valuation (tabla 3) showed that, the estimated total aggregated economical benefits were 6,400 million Euros (95%CI: 3,500 to 9,000 million Euros) translating to per capita benefits of 1,600 euros (95%CI: 870-2,300) per year for the WHO scenario. Total economical benefits would amount to 2,300 million euros (95%CI: 1,200-3,300) per year if air pollution was decreased to EU regulatory levels. More than 80% of the economical benefits were due to the reduction of long-term attributable deaths.
Discussion
This assessment indicates that health and economical benefits would be substantial if air pollution was reduced in the Barcelona metropolitan area. HIA provides a framework to evaluate the relevance of community-based policy decisions to public health. However, assumptions and uncertainty concerning methods must be considered when interpreting results.
In this study, we made several assumptions that most likely have underestimated the benefits of reducing air pollution. First, we selected PM10 as a marker of air pollution. Recent European assessments have used smaller fractions of PM, specifically PM2.53,8. Although variations may occur depending on location and hour of the day, PM2.5 and PM10 in the Barcelona metropolitan area are relatively highly correlated,26 thus results of a PM2.5 based assessment are expected to be similar. However, we ignored effects and interactions with other pollutants that may have independent effects2. In particular, effects of ozone (O3) were not included. A recent report of the US Academy of Sciences recommended the use of acute effects of ambient O3 in risk assessment27. Also, recent research indicates that pollutants which occur in very high concentrations close to busy streets, e.g. the ultrafine fraction of PM, may have a particularly detrimental effect on health28. PM10 does not capture these traffic-related exposure differences within Barcelona, thus our estimation may not fully reflect these effects. The future availability of concentration surfaces for PM2.5 or even the finer fraction of PM such as ultrafine would be an important contribution to this health impact assessment. Second, air pollution has adverse health effects (i.e. self-medication, restricted activity days, cardiovascular diseases, etc) not included in this assessment due to incomplete data2. Third, we have assumed that air pollution is a risk factor for acute asthma attacks only. While not conclusive, several studies indicate that air pollution, in particular traffic emissions, may lead to onset of asthma in children28. Therefore, we may have underestimated the public health benefits of reducing both acute and chronic effects29. Fourth, recent studies indicate that the CRF for long-term effects of air pollution on mortality, which we used in this study, may be an underestimation30. Fifth, we estimated annual results assuming an hypothetical scenario where PM10 concentrations would have decreased during one year only. Sustained reduction of air pollution would result in larger cumulative benefits for the population. In addition to these assumptions, we used the current EU standard and WHO target as reference levels in our assessment. The EU standard, based on older evidence and confirmed by parliament in 2007, is high compared to standards adopted by several European countries, the United States, and the new WHO target. Although target values are regulatory instruments with compliance requirements that differ between countries, the revised values adopted by these countries reflect a more stringent vision of protecting public health31. Adopting this more stringent WHO target would yield benefits three times larger than those achieved by reducing air pollution to the current EU standard. For the purpose of this study, the WHO and EU ambient limit values are interpreted as target average population exposure. This is consistent with the compliance approach of local authorities that assumes that any location in the area of study area must reach annual concentrations levels below the EU limit value. The reference average population exposure concentration based on modeling approaches may present some error, but we consider the impact on the estimates low, as the same errors and uncertainties would apply in the measure of the compliance level.
Sensitivity analysis showed that alternative assumptions influence the central point estimate of the benefits to different degrees (figura 2). First, the 95%CI of the CRF is the source of the largest uncertainty. These uncertainty bounds are on average ±50% but range between 30% and 80% for the different health outcomes. The CRF for chronic bronchitis in adults presents the widest range. This CRF is based on only one cohort study from North America14. Chronic symptoms have been shown to be associated with air pollution in Switzerland32 and among women in Germany33 and Europe34. When additional cohort studies are available the uncertainty is likely to shrink. It should be emphasized that the HIA estimates within the 95%CI are not all equally likely. It is more probable that true health benefits lay closer to the point estimate than the limits of the CI. Second, choice of CRFs can represent large uncertainties. For example, we chose a CRF for mortality due to long-term exposure similar to the CRF used in other European HIAs,10,35 but lower estimates have been used in more conservative evaluations8. Third, errors in the frequency of respiratory outcomes lead in uncertainties for cardio-respiratory morbidity outcomes larger than for mortality and health care outcomes but smaller than for other sources of uncertainty. Errors could result from our choice of using frequencies from populations for which the CRF was derived to avoid mismatch in definitions and non transferability between populations. For example, due to the specificity of the population used to derive the CRF for chronic bronchitis, a cohort of non-smokers adults aged 25 years and above from the Seventh-Day Adventist community in the United States, we preferred using the occurrence of cases from this population. Similarly, due to the specificity in definitions used in the CRFs, the prevalence of acute bronchitis in children was based on a cross-sectional study conducted during 1992/1993 that investigated the association between long-term exposure to air pollution and respiratory health and allergy in Swiss children (study SCARPOL). The number of asthma attacks per asthmatic child was also obtained from this study. When outcome definitions in CRF studies matched more closely with definitions of data available at the local level, we preferred using the latter to minimize potential errors in health frequencies. The number of asthmatics and the number of asthma attacks per adults were obtained from the Barcelona data of the European Community Respiratory Health Survey (ECRHS), although the ECRHS study is limited to young adults (aged between 20 and 44 years). The underlying number of asthmatics was obtained from the cross-sectional Barcelona SARI study (Estudio sobre la Salud Respiratoria en la Infancia), that derived the prevalence of asthma based on 10,821 children aged between 7 and 8 years recruited from primary schools in the cities of Barcelona and Sabadell. Health monitoring projects could substantially enhance the ability to estimate public health risks of various exposures. Finally, WTP approaches rely on respondents' ability to answer when estimating how much they are willing to pay for small changes of risks. Analysis showed that uncertainty around these unit values are large, averaging 50%. Taken together, these alternative assumptions show that presenting a range of uncertainty around the mean estimate based only on the 95%CI of the CRF may be too simplistic. However, studies using probabilistic models have also shown that uncertainties associated with the exposure-response coefficient, and plausibility of the cardiopulmonary mortality are greater than when compared with other uncertainties36. Considering current knowledge, our evaluation presents a general estimate of benefits.
As in other HIAs, the overall burden attributed to air pollution was heavily driven by mortality due to long-term exposure, thus this outcome requires further discussion4,8,10. Most of these studies express results in terms of attributable deaths, a common approach used for other risk factors37. However, expressing results in terms of life expectancy rather than attributable deaths is increasingly favoured38,39. Our study expresses results using both approaches. This change reflects the fact that the concept of «attributable deaths», and in particular the often interchangeably used term «preventable death», is misleading, because reducing pollution postpones rather than prevents death. As a cohort ages, and the benefits of reducing air pollution are realized, the number of attributable deaths will not remain constant, and gains will sum to zero when all members of the cohort are dead39. Long-term air pollution benefits are thus more appropriately expressed in terms of life years gained or life expectancy because life years accumulate independently of attributable deaths19,39 (figura 3), developed following methods in Miller and Hurley report,39 presents the evolution under WHO escenario.
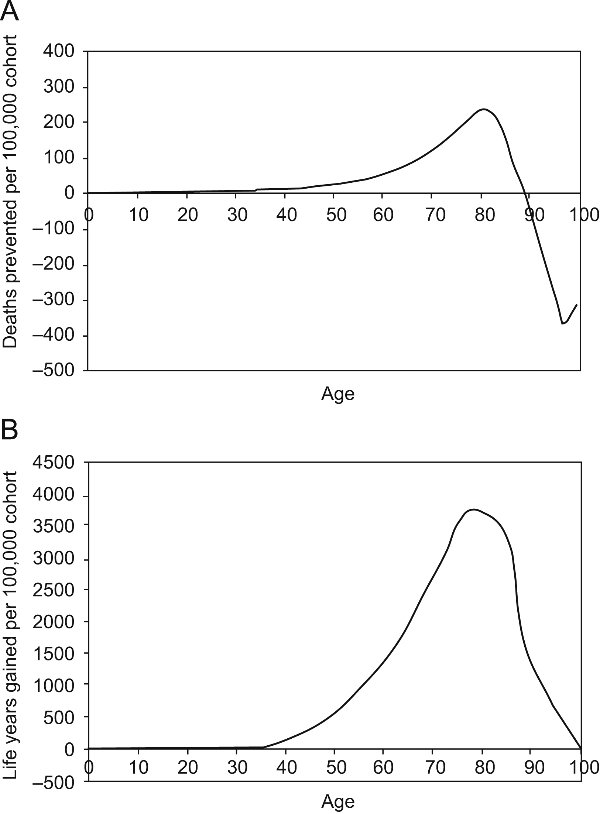
Figure 3. Evolution of the number of preventable deaths (A) and life years gained with time (B)
for the cohort of the 2004 population followed until extinction. Reduction in mortality rates is based
on the WHO air pollution reduction scenario39.
The use of life years has considerable impact on economic valuation. Using a published value of life year (VOLY),6,10 we estimated that, without discounting, the total annual monetary benefits would be approximately 50% less than benefits obtained using attributable death and associated monetary values. Similar differences have been obtained in other evaluations6. While expressing results in terms of life years gained may be more accurate, communicating these results to lay people is more complex than dealing with attributable deaths. In addition, many questions remain regarding the interpretation of long-term public health benefits of air pollution abatement strategies, including the integration of future populations and life-time dynamics in calculations, and the appropriateness of discounting and weighting38,39.
In conclusion, the results of this study indicate that the health and economic benefits of reducing air pollution in the Barcelona metropolitan area may be substantial. These benefits could only be estimated in crude terms, due to a range of inherent uncertainties and other methodological limitations. Despite these limitations, local air pollution HIAs are useful tools to translate research results into community-based evidence to protect public health.
Acknowledgements
The authors would like to thank all staff from the following Catalonian agencies that collaborated in this project: Departament de Medi Ambient i Habitatge de la Generalitat de Catalunya, the Departament de Salut de la Generalitat de Catalunya, the Registre del Conjunt Mínim Bàsic de Dades de l'Alta Hospitalària (CMBDAG), the Catalonian mortality registry, Agència de Salut Pública de Barcelona and Véase nota en servicios autores (CREAL).
Funding
The study was commissioned by the Departament de Medi Ambient i Habitage and the Departament de Salut de la Generalitat de Catalunya (Spain) and funded by the Center for Research in Environmental Epidemiology.
Bibliografía
1. Pope 3rd CA, Dockery D.W. Health effects of fine particulate air pollution: lines that connect. J Air Waste Manag Assoc. 2006; 56:709-42. [ Links ]
2. WHO. World Health Organization. Air quality guidelines for particulate matter, ozone, nitrogen dioxide and sulfur dioxide. global update 2005. WHO/SDE/PHE/OEH/06.02. 2005. [ Links ]
3. Boldo E., Medina S., LeTertre A., et al. Apheis: Health impact assessment of long-term exposure to PM(2.5) in 23 European cities. Eur J Epidemiol. 2006; 21:449-58. [ Links ]
4. Kunzli N., Kaiser R., Medina S., et al. Public-health impact of outdoor and traffic-related air pollution: a European assessment. Lancet. 2000; 356:795-801. [ Links ]
5. Cohen A.J., Ross Anderson H., Ostro B., et al. The global burden of disease due to outdoor air pollution. J Toxicol Environ Health A. 2005; 68:1301-7. [ Links ]
6. Hurley F, Hunt A, Cowie H, et al. Methodology for the cost-benefit analysis fro CAFE. Vol. 2. Health Impact assessment, Didcot, Oxon, United Kingdom, AEA Technology Environment (AEAT/ED51014/Methodology Volume 2 Issue 1; http://europa.eu.int/comm/environment/air/cafe/pdf/cba_methodology_vol2.pdf, accessed 7 July 2007. 2005. [ Links ]
7. MediAmbient Dd. Departament de Medi Ambient. Govern de Catalunya (Spain). Pla d'actuació associat a la declaració de les zones de protecció especial de l'ambient atmosfèric pels contaminants diòxid de nitrogen i pertícules en suspensió. Available at http://mediambient.gencat.net/. 2006. [ Links ]
8. Ballester F., Medina S., Boldo E., et al. Reducing ambient levels of fine particulates could substantially improve health: a mortality impact assessment for 26 European cities. J Epidemiol Community Health. 2008; 62:98-105. [ Links ]
9. WHO. World Health Organization. Country profiles of Environmental Burden of Disease. Available at: www.who.org. 2007. [ Links ]
10. CAFE. Clean Air For Europe (CAFE). Commission staff working paper. The communication on Thematic on air pollution and the directive on "Ambient ait quality and cleaner air for Europe. Impact assessment. COM(2005)446 final. Available at: http://ec.europa.eu/environment/air/cafe/index.htm. 2005. [ Links ]
11. WHO. World Health Organization. Quantification of health effects of exposure to air pollution. Copenhagen: WHO Regional Office for Europe (EUR/01/5026342); 2001. [ Links ]
12. Medina S, Boldo E. and contributing members of the APHEIS group. APHEIS: Health Impact assessment of Air Pollution Communication Strategy. Third year report. Available at: http://www.apheis.net/. ISBN: 2-11-094838-6. 2005. [ Links ]
13. Ward D.J., Ayres J.G. Particulate air pollution and panel studies in children: a systematic review. Occup Environ Med. 2004; 61:e13. [ Links ]
14. Abbey D.E., Petersen F., Mills P.K., et al. Long-term ambient concentrations of total suspended particulates, ozone, and sulfur dioxide and respiratory symptoms in a nonsmoking population. Arch Environ Health. 1993; 48:33-46. [ Links ]
15. WHO. World Health Organization. Meta-analysis of time-series studies and panel studies of Particulate Matter (PM) and ozone (O3). EUR/04/5042688. 2004. [ Links ]
16. Lacasana M., Esplugues A., Ballester F. Exposure to ambient air pollution and prenatal and early childhood health effects. Eur J Epidemiol. 2005; 20:183-99. [ Links ]
17. Le Tertre A., Medina S., Samoli E., et al. Short-term effects of particulate air pollution on cardiovascular diseases in eight European cities. J Epidemiol Community Health. 2002; 56:773-9. [ Links ]
18. Zhang J., Yu K.F. What's the relative risk?: a method of correcting the odds ratio in cohort studies of common outcomes. JAMA. 1998; 280:1690-1. [ Links ]
19. Miller B.G., Hurley J.F. Life table methods for quantitative impact assessments in chronic mortality. J Epidemiol Community Health. 2003; 57:200-6. [ Links ]
20. Hall JV, Brajer V, Lurmann FW. The health and related economic benefits of attaining healthful air in the San Joaquin Valley. California State Fullerton, Institut for Economic and environmental Studies. Report. March 2006. [ Links ]
21. Eurostat. http://epp.eurostat.ec.europa.eu. 2007. [ Links ]
22. Abbey D.E., Ostro B.E., Petersen F., et al. Chronic respiratory symptoms associated with estimated long-term ambient concentrations of fine particulates less than 2.5 microns in aerodynamic diameter (PM2.5) and other air pollutants. J Expo Anal Environ Epidemiol. 1995; 5:137-59. [ Links ]
23. Dockery D.W., Speizer F.E., Stram D.O., et al. Effects of inhalable particles on respiratory health of children. Am Rev Respir Dis. 1989; 139:587-94. [ Links ]
24. Dusseldorp A., Kruize H., Brunekreef B., et al. Associations of PM10 and airborne iron with respiratory health of adults living near a steel factory. Am J Respir Crit Care Med. 1995; 152:1932-9. [ Links ]
25. Roemer W., Hoek G., Brunekreef B. Effect of ambient winter air pollution on respiratory health of children with chronic respiratory symptoms. Am Rev Respir Dis. 1993; 147:118-24. [ Links ]
26. Querol X., Alastuey A., Rodríguez S., et al. PM10 and PM2.5 source apportionment in the Barcelona Metropolitan Area, Catalonia, Spain. Atmospheric Environment. 2001; 35:6407-19. [ Links ]
27. NCR. Estimating mortality risk reduction and economic benefits from controlling ozone air pollution. National Research Council. The National Academies Press; 2008. [ Links ]
28. McConnell R., Berhane K., Yao L., et al. Traffic, susceptibility, and childhood asthma. Environ Health Perspect. 2006; 114:766-72. [ Links ]
29. Künzli N., Perez L., Lurmann F., et al. An attributable risk model for exposures assumed to cause both chronic disease and its exacerbations. Epidemiology. 2008; 19:179-85. [ Links ]
30. Jerrett M., Burnett R.T., Ma R., et al. Spatial analysis of air pollution and mortality in Los Angeles. Epidemiology. 2005; 16:727-36. [ Links ]
31. Annesi-Maesano I., Forastiere F., Kunzli N., et al. Particulate matter, science and EU policy. Eur Respir J. 2007; 29:428-31. [ Links ]
32. Zemp E., Elsasser S., Schindler C., et al. Long-term ambient air pollution and respiratory symptoms in adults (SAPALDIA study). The SAPALDIA Team. Am J Respir Crit Care Med. 1999; 159:1257-66. [ Links ]
33. Schikowski T., Sugiri D., Ranft U., et al. Long-term air pollution exposure and living close to busy roads are associated with COPD in women. Respir Res. 2005; 6:152. [ Links ]
34. Sunyer J., Jarvis D., Gotschi T., et al. Chronic bronchitis and urban air pollution in an international study. Occup Environ Med. 2006; 63:836-43. [ Links ]
35. Pope 3rd C.A., Burnett R.T., Thun M.J., et al. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. JAMA. 2002; 287:1132-41. [ Links ]
36. Tainio M., Tuomisto J.T., Hanninen O., et al. Parameter and model uncertainty in a life-table model for fine particles (PM2.5): a statistical modeling study. Environ Health. 2007; 6:24. [ Links ]
37. Ezzati M., Lopez A.D., Rodgers A., et al. Selected major risk factors and global and regional burden of disease. Lancet. 2002; 360:1347-60. [ Links ]
38. Brunekreef B., Miller B.G., Hurley J.F. The brave new world of lives sacrificed and saved, deaths attributed and avoided. Epidemiology. 2007; 18:785-8. [ Links ]
39. Miller B, Hurley F. Comparing estimated risks for air pollution with risks for other health effects. Research Consulting Services. Institute of Occupational Medicine. Research report TM/06/01. March. 2006. [ Links ]
![]() Dirección para correspondencia:
Dirección para correspondencia:
lperez1@creal.cat
(L. Pérez)
Received: 12 March 2008
Accepted: 22 July 2008














