Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.103 no.5 Madrid may. 2011
https://dx.doi.org/10.4321/S1130-01082011000500003
Differences between pediatric and adult celiac disease
Diferencias entre la enfermedad celiaca infantil y del adulto
Luis Rodrigo Sáez1, D. Fuentes Álvarez1, I. Pérez Martínez1, N. Álvarez Mieres1, P. Niño García1, R. de Francisco García1, S. Riestra Menéndez1, C. Bousoño García2, R. Alonso Arias3 and A. López Vázquez3
Departments of 1Digestive Diseases, 2Pediatric Gastroenterology, and 3Immunology. Hospital Universitario Central de Asturias. Oviedo, Asturias. Spain
ABSTRACT
Introduction: celiac disease (CD) is a common autoimmune condition (involves 1-2% of the general population) that develops at any age in life but manifests differently in children and adults.
Objectives: to analyze clinical differences in disease expression between both groups, as well as findings at the time of diagnosis.
Methods: a retrospective study of a series of patients diagnosed with CD during childhood (< 14 years) versus a series of adult patients (> 14 years).
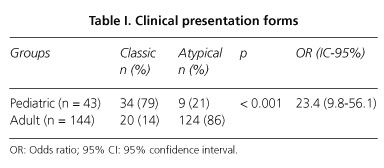
Results: a total of 187 patients were included, of which 43 were children and 144 were adults. Among clinical manifestations in children classic presentation forms predominated -34 patients (79%) versus 20 adult patients (14%) (p < 0.001) (OR = 23.4; 95% CI: 9.8-56.1). In contrast, atypical forms were predominant in the latter, and anemia was the most common finding in 61 patients (42%) versus 8 pediatric patients (19%) (p < 0.01).
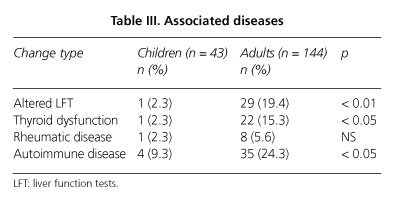
Adults had a greater diagnostic delay with a mean 10 ± 9 years versus 1 ± 2 years in children (p < 0.001). In adults, we found a higher frequency of associated autoimmune diseases (24.3 versus 9.3% in children) (p < 0.05).
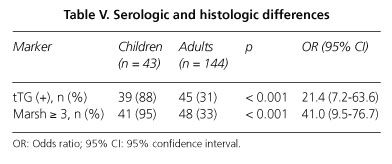
Regarding serum markers, TGt-2 was more commonly positive among children (88%) as compared to adults (31%) (p < 0.001); (OR = 21.4: 95% CI: 7.2-63.6). We found similar results with regard to the presence of villous atrophy, which was more common in children (95%) than in adults (33%) (p < 0.001) (OR = 41.0; 95% CI: 9.5-76.7).
As regards genetic markers, DQ2 was somewhat more common in children (97.7%) than in adults (90.3%) whereas DQ8 was less common in children (2.3%) than in adults (9.7%), with no significant differences between groups. Patients negative for both markers were not included.
Conclusions: pediatric CD has clear differences when compared to adult CD, with classic forms predominating in the former, who also display a higher occurrence of positive serology and villous atrophy, and less diagnostic delay. In contrast, atypical forms predominate in the adult, with a lower occurrence of positive serology and milder histological forms. In these patients associated autoimmune conditions are more common and diagnostic delay is longer.
Key words: Pediatric celiac disease. Adult celiac disease.
RESUMEN
Introducción: la enfermedad celiaca (EC) es un proceso frecuente (afecta al 1-2% en población general), de naturaleza autoimmune, que aparece a cualquier edad de la vida, pero que se presenta de forma diferente en el niño que en el adulto.
Objetivos: analizar las diferencias clínicas en las formas de expresión de la enfermedad entre ambos grupos, así como los hallazgos al momento del diagnóstico.
Métodos: estudio retrospectivo de una serie de pacientes diagnosticados, en la infancia (< 14 años), frente a una serie de adultos (> 14 años).
Resultados: se incluyeron un total de 187 pacientes, de los cuales 43 eran niños y 144 adultos. En las manifestaciones clínicas de los niños, predominaron las formas de presentación clásicas, 34 casos (79%) frente a los adultos 20 casos (14%) (p < 0,001) (OR = 23,4; IC-95%: 9,8-56,1). Por el contrario, en estos predominaron las formas atípicas, siendo la anemia el hallazgo más frecuente en 61 casos (42%) frente a 8 casos (19%) en los niños (p < 0,01).
En los adultos existía un mayor retraso diagnóstico con una media de 10 ± 9 años, frente a los niños, que es de 1 ± 2 años (p < 0,001). Encontramos en los adultos una mayor frecuencia de enfermedades autoinmunes asociadas (24,3%), frente al 9,3% en niños (p < 0,05).
Respecto a los marcadores serológicos, la TGt-2 fue más frecuentemente positiva en los niños (88%), que en los adultos (31%) (p < 0,001); (OR = 21,4: IC-95%: 7,2-63,6). Resultados similares encontramos en relación con la presencia de atrofia vellositaria, que estuvo presente más frecuentemente en los niños (95%) que en los adultos (33%) (p < 0,001) (OR = 41,0; IC-95%: 9,5-76,7).
Respecto a los marcadores genéticos, el DQ2 fue algo más frecuente en niños (97,7%) que en adultos 90,3%, y el DQ8 ocurrió al contrario, siendo menos frecuente en niños (2,3%) que en adultos (9,7%), no encontrando diferencias entre ambos grupos. No se incluyeron pacientes negativos para ambos marcadores.
Conclusiones: la EC en el niño presenta claras diferencias con el adulto predominando en aquel las formas clásicas, con mayor positividad de la serología y atrofia vellositaria, con menor retraso diagnóstico. Por el contrario, en el adulto predominan las formas atípicas, con menor positividad de la serología y formas histológicas más leves. En ellos son más frecuentes las enfermedades autoinmunes asociadas y existe un mayor retraso diagnóstico.
Palabras clave: Enfermedad celiaca infantil. Enfermedad celiaca del adulto.
Introduction
Celiac disease (CD), also called "gluten-sensitive enteropathy", is a systemic chronic inflammatory condition of autoimmune origin derived from permanent gluten intolerance that develops in genetically predisposed individuals (1).
It is very common and affects 1-2% of the general population, including all races and countries, with quite a homogeneous distribution worldwide (2-4).
CD was traditionally thought to be a disease involving primarily children, uncommon in the adult. However, multiple studies in the last few decades have shown that adults are also involved even more commonly than children, since it may present at any age in life and around 20% of patients are older than 55 years at the time of diagnosis (5-7).
Pediatric CD usually develops in early years, when gluten-containing cereals are introduced, and usually displays the "classical" clinical picture characterized by predominant digestive features with a typical malabsorption syndrome associated with weight loss, florid diarrhea, and evidence of both nutritional and vitamin deficiencies (8).
In contrast, adult CD usually presents in a larvate manner with fewer digestive manifestations and absence of malabsorption, and displays "atypical" forms characterized by common extra-digestive complaints and various accompanying conditions, which renders diagnosis more challenging (9-11).
The fact that serum markers and duodenal biopsy findings are usually much milder in adults than in children with CD sure adds to these difficulties.
In the present paper we discuss a series of pediatric patients with CD regarding clinical features, and the prevalence of serum, genetic susceptibility, and duodenal histological markers, and compare them to a second series of adult patients under follow-up and with positive response to a gluten-free diet, in order to ascertain both shared and differential aspects in both age groups at the time of diagnosis.
Methods
A retrospective study was performed of patients diagnosed with CD at the Departments of Digestive Diseases and Pediatric Gastroenterology, HUCA, Oviedo, during the period between January 2000 to December 2006, with clinical and laboratory follow-up until June 2010. All had been diagnosed with CD according to internationally accepted criteria (modified ESPAGHAN and AGA for children and adults). All of them had responded favorably to gluten-free diet (GFD) after a protracted follow-up of at least 3 years. Pediatric cases were those pertaining to children and adolescents diagnosed before age 14, and adult cases included subjects older than 14 at the time of diagnosis. A total of 187 patients with CD were included, who were split into two age groups -43 children and 144 adults.
A specific medical history was obtained from all patients together with a number of laboratory tests including serum and genetic markers of CD, plus a gastroscopy exam with multiple duodenal biopsies.
All patients underwent a complete blood count with ESR measurement using a Cell-DYN 3500 R (Lab. Abbott) automated analyzer, and complete coagulation tests using a ACL 3000 (Lab. Menarini) autoanalyzer. WBC counts were considered normal for values of 4-10 x103/L, and normal platelet counts were in the range of 130-400 x 103/L.
An extensive biochemistry profile was performed including the following parameters: iron metabolism including sideremia levels, transferrin saturation index (TSI) and serum ferritin. Normal sideremia was 60-140 mcg/ml, and normal ferritin was 13-150 ng/ml. TSI was normal for values in the range of 25-45%.
We also routinely measured the so-called liver function tests (LFT) including AP, AST, ALT, GGT, and bilirubin, as well as serum calcium, folic acid and vitamin B12, plasma creatinine, total cholesterol (normal, 150-240 mg/dl), HDL, LDL and triglycerides, urea, glycemia, total protein and albumin, and acute phase reagents such as CRP (C-reactive protein). Normal AST and ALT values were 1-31 U/L. All laboratory measurements were performed with a modular automated Hitachi SXA DPPP autoanalyzer (Lab. Roche) using enzymatic or kinetic methods.
Immunoglobulins were measured using nephelometry techniques. For serum CD screening a single marker was used, namely IgA tissue anti-transglutaminase 2 antibodies (tTG-2), which were measured using a commercial ELISA kit (Phadia Diagnostics, Uppsala, Sweden). We considered the test positive for values > 10 U/ml.
For the study of genetic susceptibility two markers were measured -HLA-DQ2 (DQA1*0501 and DQB1*0201) and HLA-DQ8 (DQA1*0301 and DQB1*0302)- using a PCR technique with DNA sequence specific primers (SSP) based on the commercial kit Protrans® Domino System HLA Celiac Disease (Protrans, Ketsch, Germany).
An upper digestive endoscopic study with multiple duodenal biopsy collection (4-6) was also performed for all study patients at the time of diagnosis. Obtained samples were routinely stained with hematoxylin-eosin (H-E) and CD3-specific immunohistochemical modalities to ascertain the presence of intra-epithelial lymphocytes (IELs), counted per one hundred epithelial cells.
Duodenal biopsies were examined by two expert pathologists and categorized as follows -stage 0 = histologically normal duodenum; stage I = increased IELs, > 25% of epithelial cells; stage II = crypt hyperplasia and/or chronic diffuse inflammatory infiltration at the lamina propria; stage III = villous atrophy, subdivided into three categories: a) mild; b) moderate; and c) severe, according to the pathological classification for CD screening as described by Marsh in 1992 (12) and later modified by Oberhuber et al. in 1999 (13).
All patients received a gluten-free diet for a mean 5.8 ± 2.6 years (range, 3-10 years), and showed a favorable clinical and laboratory response.
Statistical analysis
Initially, a descriptive analysis of the variables included in the study was performed. The normal distribution of quantitative variables was analyzed using the Kolmogorov-Smirnov test. Non-parametric variables were studied using the Mann-Whitney U-test.
For between-group comparisons we used Student's t-test for continuous variables in non-paired groups, and the chi-squared test for categorical variables, with Fisher's test being used when deemed necessary. For significant differences odds ratios (OR) were estimated for a 95% confidence interval (CI). A value of p < 0.05 was considered statistically significant.
Results
Among the 187 celiac patients in the study 144 were adults (71%) and 43 were children (29%), with 2.4 adults per child.
Mean age was 36 ± 19 years for adults (15-69) and 5 ± 4 years for children (1-12). Regarding sex, there were 115 women (80%) and 29 men (20%) in the adult group, with a female/male ratio of 4/1. Among children there were 25 girls (58%) and 18 boys (42%), with a female/male ratio of 1.4/1.
Classic presentations were clearly predominant among children (79%), whereas atypical forms predominated among adults (86%), with statistically significant differences between groups (p < 0.001) (Table I).
We also found clinical differences regarding predominant complaints at diagnosis in each group, with diarrhea being the most common symptom in children (p < 0.001); in contrast, anemia was the most common finding among adults (p < 0.01) (Table II).
Differences were also seen in the frequency and type of associated diseases, including altered liver function tests, thyroid dysfunction, and autoimmune disorders in general, these been clearly more common and predominant in the adult (p < 0.05) versus children (Table III).
There was a clear diagnostic delay which was much longer in adults with a median of 4 years; children had a median delay of 1 year, and differences were also significant between both age groups (p < 0.001) (Table IV).
We also saw pronounced differences in the frequency of serum and histologic CD marker positivity (tTG2), particularly regarding villous atrophy (Marsh 3 3); both were more common in children with statistically significant differences (p < 0.001) (Table V).
Regarding the frequency of genetic susceptibility markers DQ2 was more common in children -42 cases (97.7%)- than in adults -130 cases (90.3%) (NS). In contrast, DQ8 was more common in adults, with 14 cases (9.7%), than in children, with 1 case (2.3%) (NS). No cases negative for both markers were included.
Discussion
In this study we analyzed differences between pediatric and adult CD, and our findings clinically confirm the well-known fact that classic presentation forms predominate in the former, characterized by florid digestive symptoms such as recurrent diarrhea, while atypical forms are more common in the latter, with predominant non-digestive symptoms such as anemia (most common sign at diagnosis) as well as a higher frequency of associated conditions, mainly autoimmune in nature. This greatly accounts for diagnostic delay, commonly around a decade in adults, a few months up to one year in children.
Regarding serum marker frequency, this is noticeably higher in children versus adults, as is the presence of villous atrophy (Marsh 3 3). This highlights a pronounced linear correlation between both, with higher serum anti-tTG antibodies in patients with versus without villous atrophy, as conclusively demonstrated by a number of studies (14-16).
As regards known genetic susceptibility markers for CD development, we found a higher prevalence of HLA-DQ2 in children, with HLA-DQ8 being more common in the adult (17).
The pathogenesis of CD at any age is related to continuous gluten exposure, as patients are permanently intolerant to gluten. Several observational studies suggest that the risk of CD development during childhood may be reduced by prolonging breast-feeding and delaying or gradually introducing cereals with gluten during the first year of life. In contrast, other studies show that these approaches only delay rather than reduce CD development (18-20).
Some of these factors are thought to have been involved in an epidemic of CD in Sweden between 1984 and 1996, where the frequency of CD developing in children younger than 2 years experienced a four-fold increase (21). Both the start and end of this epidemic were sudden and consistent with specific dietary changes; prevalences as high as 3% were seen in 12-year-olds as a result of this epidemic (22).
From a clinical perspective adult CD is considered a highly "chameleonic" process, as it varies greatly in its various presentation forms and commonly shows a wide spectrum of manifestations going from subclinical, aymptomatic cases through to florid malnutrition. Most common symptoms include abdominal (usually colic) pain in association with fluctuating bloating, dyspepsia, gastro-esophageal reflux, and intestinal habit changes such as diarrhea and constipation, or both in an alternating pattern. Symptoms such as anemia are also common, as seen in our series, in association with asthenia, moderate activity dyspnea, sleep disorders, weight loss, widespread bone pain due to common osteoporosis, and character disorders with irritability, headaches, and depression among others.
The presence of diarrhea was considered a common complaint until recently; however, it is not necessarily present in adults, and up to 50% of patients have constipation predominantly, often refractory to treatment; weight loss is also uncommon in adults, and up to 30% exhibit obvious overweight signs. Other changes are also common, including elevated liver function markers, thyroiditis and hypothyroidism, as well as various autoimmune conditions as in our series.
Primary diagnostic criteria for CD rely on the presence of consistent symptoms together with serum markers, duodenal biopsy findings, and associated susceptibility genes; these and a positive response to gluten-free diet are confirmatory. Markers are not always present and a high index of suspicion is needed. Family studies are also useful since the prevalence of CD among first-degree relatives is high -10-20% (23).
Regarding serologic approaches to the diagnosis and follow-up of CD, it should be remembered that anti-gliadin IgA antibodies are no longer used in clinical practice given their low sensitivity and specificity, both around 50%, which renders them unsuitable for diagnosis. In 1997 Dieterich et al. reported the use of tTG2, the autoantigen against which anti-endomysial antibodies (EMA) reacted and whose measurement is as useful as that of EMA (24).
Since then tTG2 is the most commonly used serum marker for CD in clinical practice -it is easily measured using a commercially available ELISA test, and has shown a sensitivity and specificity nearing 90% in several studies performed when it was first marketed over a decade ago, clinical trials in rigorously selected patients with significant villous atrophy. There was some variability according to the various substrates used (25), and early studies validating its diagnostic effectiveness have become noticeably devalued over time. Several studies in extensive series of celiac patients have clearly shown that tTG2 sensitivity varies depending on duodenal damage severity, and is almost 100% in the presence of complete villous atrophy, 70% for subtotal atrophy, and up to 30% when normal mucosal architecture is preserved and only an increase in intra-epithelial lymphocytes may be identified (26).
On the other hand, gluten-sensitive enteropathy (GSE) with a significant clinical impact also develops in patients with mucosal preservation. In this respect a study performed at Servicio de Digestivo, Hospital Mutua de Tarrasa, by Esteve et al., where serology and genetic tests were used for the diagnosis of GSE in a recognized high-risk population (first-degree relatives), demonstrated that their diagnostic approach (genetic tests followed by intestinal biopsy in positive cases) could diagnose up to three-fold as many patients with GSE when compared to serology alone. Even more significant is the fact that patients with lymphocytic enteritis (LE), type I according to Marsh's classification, were diagnosed -most of these would have remained undiagnosed by serology alone, and most had the same clinical manifestations seen in celiac patients with villous atrophy (27).
This fact, reported by many authors, is of great clinical importance, and invites to follow this same diagnostic strategy in other groups at risk such as type-1 diabetics, subjects with hypothyroidism, patients with altered LFTs, and individuals with other autoimmune conditions (28-30).
The use of HLA-DQ2 and HLA-DQ8 haplotype typing in the diagnostic approach to patients with suspected CD helps identify individuals susceptible of developing CD, hence eligible for duodenal biopsy, in groups at risk. This is based not only on the findings of studies performed in our setting, but also on results from other geographical areas that confirm that around 90% of celiac patients are HLA-DQ2-positive versus 20% in the control population (31).
Most HLA-DQ2-negative celiac patients have HLA-DQ8 positivity in association with DR4. Therefore, a DQ2-positive status usually delimits the population for follow-up. It should be highlighted that this strategy allows to identify patients with seronegative CD, and thus to go deeper into the "celiac iceberg", possibilitating the identification of many Marsh-I CD patients with the so-called "lymphocytic enteritis" and no villous atrophy, and a change in the current "gluten-sensitive enteropathy" concept to be treated with gluten-free diet (32). The same possibly occurs in patients with a histologically normal duodenal mucosa (Marsh type 0), but few studies support this as of today.
This fact has led to new diagnostic strategies such as genetic testing or tTG measurement is duodenal aspirates in cases with negative serology (33), and also to a lower positivity threshold for tTG -2 U/ml, as recommended by Mariné et al. after screening studies in the general population (34).
Nevertheless, despite clear limitations, measuring anti-tTG IgA antibodies is usually the only serum marker in clinical practice, both for the screening of celiac patients and their follow-up, to confirm patient compliance with gluten-free diet despite its frequent negativity in adults. Currently, a serologic diagnosis of CD based exclusively on serum marker positivity is not acceptable, even though highly elevated levels -tTG > 100 U/ml- may predict with adequate reliability the presence of villous atrophy and, at least in children, may render upper endoscopy unnecessary, which in such cases usually requires deep sedation or even general anesthesia, and includes multiple biopsies for diagnostic confirmation, which is relevant at least from a clinical standpoint (15).
In summary, we conclude by pointing out that pediatric CD is very different from adult CD, with classic forms predominating and usually positive serology and duodenal biopsies, which seem consistent with greater severity and intestinal involvement. In contrast, atypical forms predominate in adults, with fewer positive serology and duodenal biopsy findings. All this suggests a milder intestinal involvement and greatly accounts for the longer diagnostic delay seen in adults.
References
1. Rodrigo L. Celiac disease. World J Gastroenterol 2006;12:6585-93. [ Links ]
2. Barada K, Bitar A, Mokadem MA, Harshash JG, Green P. Celiac disease in Middle Eastern and North African countries: a new burden? World J Gastroenterol 2010;16:1449-57. [ Links ]
3. Wu J, Xia B, von Blomberg BM, Zhao C, Yang XW, Crusius JB, et al. Coeliac disease emerging in China? Gut 2010;59:418-9. [ Links ]
4. Cummins AG, Roberts-Thomson IC. Prevalence of celiac disease in the Asia-Pacific region. J Gastroenterol Hepatol 2009;24:1347-51. [ Links ]
5. O'Leary C, Wieneke P, Healy M, Cronin C, O'Regan P, Shanahan F. Celiac disease and the transition from childhood to adulthood: a 28-year follow-up. Am J Gastroenterol 2004;99:2437-41. [ Links ]
6. Fernández Salazar L, de la Torre Ferrera N, Velayos Jiménez B, Nocito Colón M, González Hernández JM, Garrote Adrados JA. Diagnostic problems in adult celiac disease. Rev Esp Enferm Dig 2008; 100:24-8. [ Links ]
7. Vilppula A, Kaukinen K, Luostarinen L, Krekela I, Patrikainen H, Valve R, et al. Increasing prevalence and high incidence of celiac disease in elderly people: a population-based study. BMC Gastroenterol 2009;9:49. [ Links ]
8. Savilahti E, Kolho KL, Westerholm-Ormio M, Verkasalo M. Clinics of celiac disease in children in the 2000s. Acta Paediatr 2010;99: 1026-30. [ Links ]
9. Fernández A, González L, de la Fuente J. Coeliac disease: clinical features in adult population. Rev Esp Enferm Dig 2010;102:466-71. [ Links ]
10. Häuser W, Janke KM, Klump B, Gregor M, Hinz A. Anxiety and depression in adult patients with celiac disease on a gluten-free diet. World J Gastroenterol 2010;16:2780-7. [ Links ]
11. Thomson AB. Small intestinal disorders in the elderly. Best Pract Res Clin Gastroenterol 2009;23:861-74. [ Links ]
12. Marsh MN. Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue'). Gastroenterology 1992;102: 330-54. [ Links ]
13. Oberhüber G, Granditsch G, Vogelsang H. The histopathology of coeliac disease. Time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol 1999;11:1185-94. [ Links ]
14. Dahlbom I, Korponay-Szabó IR, Kovács JB, Szalai Z, Mäki M, Hansson T. Prediction of clinical and mucosal severity of celiac disease and dermatitis herpetiformis by quantification of IgA/IgG serum antibodies to tissue transglutaminase. J Pediatr Gastroenterol Nutr 2010;50:140-6. [ Links ]
15. Vivas S, Ruiz de Morales JG, Riestra S, Arias L, Fuentes D, Álvarez N, et al. Duodenal biopsy may be avoided when high transglutaminase antibody are present. World J Gastroenterol 2009;15:4775-80. [ Links ]
16. Donaldson MR, Book LS, Leiferman KM, Zone JJ, Neuhansen SL. Strongly positive tissue transglutaminase antibodies are associated with Marsh 3 histopathology in adult and pediatric celiac disease. J Clin Gastroenterol 2008;42:256-60. [ Links ]
17. van Heel DA, Hunt K, Greco L, Wijmenga C. Genetics in celiac disease. Best Pract Res Clin Gastroenterol 2005;19:323-39. [ Links ]
18. Ivarsson A, Hernell O, Stenlund H, Persson LA. Breast-feeding protects against of celiac disease. Am J Clin Nutr 2002;75:914-21. [ Links ]
19. Akobeng AK, Ramanan AV, Buchan I, Heller RF. Effect of breast-feeding on risk of celiac disease: a systematic review and meta-analysis of observational studies. Arch Dis Child 2006;91:39-43. [ Links ]
20. Farrell RJ. Infant gluten and celiac disease: too early, too late, too much, too many questions. JAMA 2005;293:2410-2. [ Links ]
21. Ivarsson A, Persson LA, Nyström L, Ascher H, Cavell B, Danielsson L, et al. Epidemic of celiac disease in Swedish children. Acta Paediatr 2000;89:165-71. [ Links ]
22. Myleus A, Ivarsson A, Webb C, Danielsson L, Hernell O, Högberg L, et al. Celiac disease revealed in 3% of Swedish 12-year-olds born during an epidemic. J Pediatr Gastroenterol Nutr 2009;49: 170-6. [ Links ]
23. Rodrigo L, Fuentes D, Riestra S, Niño P, Alvarez N, López-Vázquez C, et al. Increased prevalence of celiac disease in first and second-grade relatives. A report of a family with 19 studied members. Rev Esp Enferm Dig 2007;99:149-55. [ Links ]
24. Dieterich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken EO, et al. Identification of tissue transglutaminase as the auto-antigen of celiac disease. Nat Med 1997;3:797-801. [ Links ]
25. Fernández E, Riestra S, Rodrigo L, Blanco C, López-Vázquez A, Fuentes D, et al. Comparison of six human anti-transglutaminase ELISA-tests in the diagnosis of celiac disease in the Saharawi population. World J Gastroenterol 2005;11:3762-6. [ Links ]
26. Tursi A, Brandimarte G, Giorgetti GM. Prevalence of anti-tissue transglutaminase antibodies in different degrees of intestinal damage in celiac disease. J Clin Gastroenterol 2003;36:219-21. [ Links ]
27. Esteve M, Rosinach M, Fernández-Bañares F, Farré C, Salas A, Alsina M, et al. Spectrum of gluten sensitive enteropathy in first degree relatives of coeliac patients: clinical relevance of lymphocytic enteritis. Gut 2006;55:1739-45. [ Links ]
28. Plenge RM. Shared genetic risk factors for type 1 diabetes and celiac disease. N Engl J Med 2008;359:2837-8. [ Links ]
29. Elfström P, Montgomery SM, Kämpe O, Ekbom A, Ludvigsson JF. Risk of thyroid disease in individuals with celiac disease. J Clin Endocrinol Metabol 2008;93:3915-21. [ Links ]
30. Múgica F, Aranzadi MJ, Recasens M, Almagro F, Muñagorri A, Elósegui E, et al. Adult celiac disease and hypertransaminasemia. Rev Esp Enferm Dig 2000;92:78-85. [ Links ]
31 Vilppula A, Collin P, Mäki M, Valve R, Luostarinen M, Krekelä I, et al. Undetected celiac disease in the elderly. A biopsy-proven population-based study. Dig Liv Dis 2008;40:809-13. [ Links ]
32. Santaolalla R, Fernández-Bañares F, Rodríguez R, Alsina M, Rosinach M, Mariné M, et al. Diagnostic value of duodenal anti-tissue transglutaminase antibodies in gluten-sensitive enteropathy. Aliment Pharmacol Ther 2008;27:820-9. [ Links ]
33. Koskinen O, Collin P, Lindfors K, Laurila K, Mäki M, Kaukinen K. Usefulness of small-bowel mucosal transglutaminase-2 specific autoantibody deposits in the diagnosis and follow-up of celiac disease. J Clin Gastroenterol 2010;44:483-8. [ Links ]
34. Mariné M, Fernández-Bañares F, Alsina M, Farré C, Cortijo M, Santaolalla R, et al. Impact of mass screening for gluten-sensitive enteropathy in working population. World J Gastroenterol 2009;15:1331-8. [ Links ]
![]() Correspondence:
Correspondence:
Luis Rodrigo Sáez.
Servicio de Aparato Digestivo.
Hospital Universitario Central de Asturias.
c/ Celestino Villamil, s/n.
33006 Oviedo, Asturias. Spain
e-mail: lrodrigosaez@gmail.com
Received: 14-09-10.
Accepted: 10-02-11.











 texto en
texto en