Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.106 no.2 Madrid feb. 2014
https://dx.doi.org/10.4321/S1130-01082014000200004
Dental erosion, an extraesophageal manifestation of gastroesophageal reflux disease. The experience of a center for digestive physiology in Southeastern Mexico
Erosión dental, una manifestación extraesofágica de la enfermedad por reflujo gastroesofágico. Experiencia de un centro de fisiología digestiva en el sureste de México
Laura Roesch-Ramos1, Federico Roesch-Dietlen2, José María Remes-Troche2, Graciela Romero-Sierra3, Carlos de Jesús Mata-Tovar1, Amyra Ali Azamar-Jácome2 and Antonia Barranca-Enríquez4
1Faculty of Dentistry
2Institute for Medical-Biological Research
3Faculty of Medicine, and
4Center for Health Studies and Services. University of Veracruz. Veracruz, Mexico
ABSTRACT
Background: Dental erosion (DE) is the loss of the hard tissues of the tooth produced by the action of gastric juice, pepsin and acid on the dental enamel, its frequency ranges from 5to 53.41%. In Mexico there are no reports on the frequency and possible association.
Objective: To establish the prevalence of dental erosion and its relationship to GERD.
Patients and methods: Prospective, observational, descriptive and comparative study was conducted in 60patients diagnosed with GERD and 60healthy patients at the Institute of Medical and Biological Research of the Universidad Veracruzana in Veracruz city. Anthropometric characteristics, dietary habits, oral hygiene, alcohol consumption, smoking, ED index and Index of decayed/missing dental pieces/sealed and correlation between severity of ED and GERD were analyzed.
Results: 78.67% of patients with GERD had ED, 23.33% corresponded to grade 0, 41.67% to N1, N2and 23.33% to 11.67% to N3. Predominance of females (2.3:1). The mean age was 50.92± 13.52years. The severity of dental erosion was significantly related to the severity of reflux, halitosis, CPO index and poor eating habits. There was no statistically significant difference in the other variables analyzed.
Conclusions: Dental erosion has a high frequency in patients with GERD and reflux characteristics are directly related to their severity and therefore should be considered as a manifestation of GERD extraesophageal.
Key words: Dental erosion. Gastroesophageal reflux disease. Prevalence.
RESUMEN
Antecedentes: la erosión dental (ED) es la pérdida de los tejidos duros del diente producida entre otras cosas por la acción del jugo gástrico, la pepsina y el ácido sobre el esmalte dentario, su prevalencia varía entre 5y 53,41%. En México no existen publicaciones sobre su frecuencia y posible asociación.
Objetivo: establecer la prevalencia de las erosiones dentales y su relación con la enfermedad por reflujo gastroesofágico (ERGE).
Pacientes y métodos: estudio prospectivo, observacional, descriptivo y comparativo realizado en 60 pacientes con diagnóstico de ERGE y 60sujetos sanos en el Instituto de Investigaciones Médico-Biológicas de la Universidad Veracruzana en la ciudad de Veracruz. Se analizaron las características antropométricas, hábitos alimentarios, higiene oral, consumo de alcohol, tabaquismo, índice de ED e índice de piezas dentales cariadas/perdidas/obturadas (CPO) y la correlación entre severidad de ED y la ERGE.
Resultados: El 78,67% de los pacientes con ERGE presentaron ED; el 23,33% correspondieron al grado N0, 41,67% al N1, 23,33% al N2 y 11,67% al N3. Predominó el género femenino (2,3:1). La edad promedio fue 50,92± 13,52 años. La gravedad de la erosión dental se relacionó significativamente con la severidad del reflujo, halitosis, índice CPO y con malos hábitos alimentarios. No hubo diferencia estadísticamente significativa en las demás variables analizadas.
Conclusiones: la erosión dental tiene una alta frecuencia en pacientes con ERGE y las características del reflujo se relacionan directamente con su severidad por lo cual debe ser considerada como una manifestación extraesofágica más de la ERGE.
Palabras clave: Erosión dental. Enfermedad por reflujo gastroesofágico. Prevalencia.
Background
Gastroesophageal reflux disease (GERD) is a common condition in adults, of chronic course, caused by the rise of gastric contents above the gastroesophageal junction (1).
It has been shown that gastric juice, pepsin and acid can attack tissues not adapted to these substances, causing laryngitis, contact ulcers, subglottic stenosis, laryngospasm, dysphonia, pharyngitis, asthma, pneumonia, and dental diseases (2-4).
Dental erosion is defined as the loss of tooth structure by a chemical process with no involvement of the bacterial flora. Its etiology is associated with intrinsic and extrinsic factors, among which GERD is one of them.
In patients with GERD, the irreversible effect of acid produced by gastric juice on dental enamel can be recognized by the presence of dental erosion, which depends on the duration of the disease and the severity of reflux (5-7).
The demineralization of dental hard tissue by dissolution of apatite crystals can cause the total destruction of the teeth. The location of this damage in patients with GERD is more common in the occlusal and palatal surfaces of the upper teeth and in the buccal and lingual/occlusal surfaces of the lower teeth, because the position of the tongue leads the acid to these surfaces (8-10).
The pattern of severity of erosion is established by assessing the extent of the loss of tooth substance; the most widely used classification is the Erosion Index proposed by Eccles and Jenkins (11), which considers four grades:
-Grade 0: No erosion.
-Grade 1: Loss of enamel without exposure of dentin.
-Grade 2: Loss of enamel with exposure of dentin in less than a third of the tooth surface.
-Grade 3: Loss of enamel with exposure of dentin in more than a third of the tooth surface (11).
The lesions progress slowly and often go unnoticed in the early stages, so it is recommended that patients with GERD are adequately evaluated to establish preventive measures.
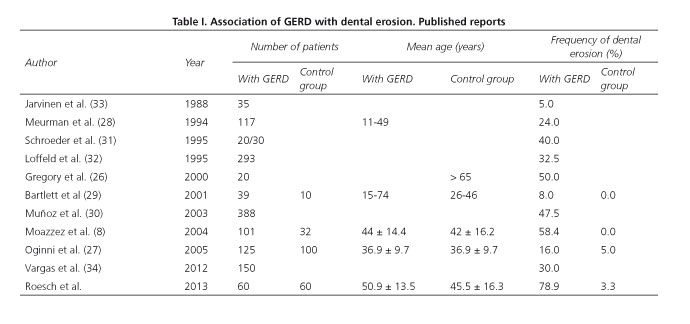
Different population groups have reported an association of GERD with dental erosion, the prevalence of which ranges from 5% to 58.41%, as shown in table I (8,26-33). In 2012, Vargas et al. published a series of 150 cases of Latin American patients and the two conditions were associated in 30% (34) of them.
There are no reports for Mexico, so we considered it of interest to carry out the present study, the aim of which was to establish the prevalence of dental erosion in patients with GERD diagnosis, and to determine its possible association and determinant factors.
Patients and methods
A prospective, observational, descriptive and comparative study approved by the ethics committee of the area of Health Sciences at the University of Veracruz, performed in patients who underwent a gastrointestinal examination at the Institute of Medical and Biological Research of the city of Veracruz (Mexico) during the period between January 2011 and December 2012. All patients who agreed to participate after signing an informed consent were asked to complete a validated Spanish version of a questionnaire about laryngeal symptoms for GERD (reflux symptoms index), which consists of 9 questions with 5 possible answers ranging from 0 to 4. It was considered positive when a score of more than 5 was obtained (36).
Two study groups were formed: Group A included patients of both sexes aged over 18 years of age, with no prior drug treatment and a positive questionnaire. They were subjected to endoscopy using the Los Angeles classification to assess the severity of reflux oesophagitis; 24 h stationary pH-metry and impedance were performed using an esophageal catheter and two sensors located at 5cm and 10 cm from the lower esophageal sphincter (LES) (ZepHr team, Sandhill, Colorado, USA); esophageal manometry was carried out with nasogastric catheter through the LES and subsequent administration of 10 swallows of 5 mL of saline solution (normal value 4.8-32 mm Hg) (Sierra-Given, Los Angeles, Ca , USA); a 14C urea breath test was done to detect Helicobacter pylori. Group B included volunteer patients of both sexes over 18 years of age, with a negative result on the GERD questionnaire.
For both groups, the following parameters were registered: Age, gender and body mass index (BMI); food habits were assessed using a questionnaire that included: Alcohol intake (≥ 30 g/day), excessive consumption of citrus (> 1 citrus fruit/day), carbohydrates (≥ 325 g/day), fat (≥ 78 g/day), intake of spicy food and coffee (≥3 cups/day). We also recorded smoking status (mild ≤5cigarettes/day, moderate 6-10 cigarettes/day, severe ≥11 cigarettes/day), dental hygiene habits, extraesophageal symptoms; a dental clinical examination with intraoral photography was performed; the dental erosion index and the decayed-missing-filled index (DMF) were recorded too, as well as tooth sensitivity, corroborated by a thermal test with exposure to cold and heat (37); salivary pH was measured using a test strip with a sensitivity range of 0-14. The pattern of severity of erosion was established according to the Erosion Index of Eccles and Jenkins (11).
The results were analyzed with descriptive statistics, χ2 test and Student's t, consisting in relative frequency and average central tendency measurements for obtaining percentages, means and standard deviation. A statistical significance level of p < 0.05 was used.
Results
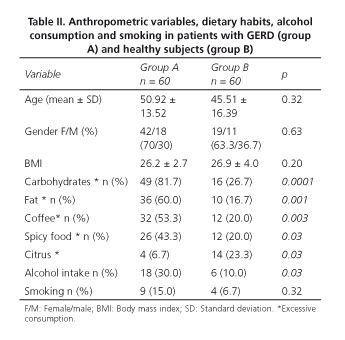
Group A was formed by 60 patients with GERD, and group B by 60 healthy subjects. The mean age for both groups was 48.21 (20-78 years of age) with predominance of female gender (male to female ratio 1:2.3) and no differences between groups. The anthropometric characteristics were similar in both groups and are shown in table II.
Regarding eating habits, we found a significantly higher, excessive intake of carbohydrate and fats, as well as coffee consumption, in group A. There was no difference in the consumption of spicy food and alcoholic beverages (beer intake 64.28% and liquor 35.71%), and in smoking (71.42% mild, moderate 14.28% and severe 14.28%), as shown in table II.
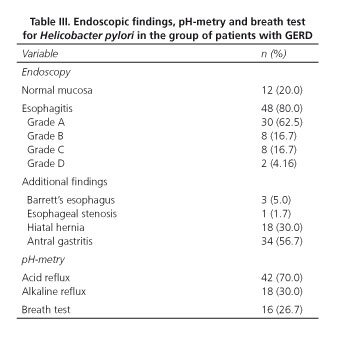
The time evolution of GERD was 50.09 ± 32.26 months (7-190 months). Esophagitis was identified in 48 patients (80.0%).Thirty cases (62.5%) were classified as grade A, 8 (16.67%) as grade B, 8 (16.67%) as grade C and 2 (4.16%) as grade D. The findings of endoscopy, pH-metry, pH-impedance, and the results of the breath test for Helicobacter pylori are shown in table III. Twenty patients (36.67%) showed extraesophageal manifestations (cough and hoarseness in 40.74%, cough and posterior nasal drainage in 20.00%).
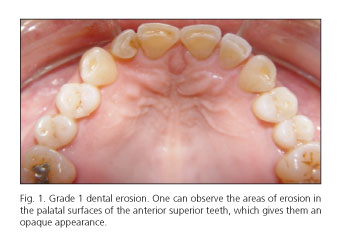
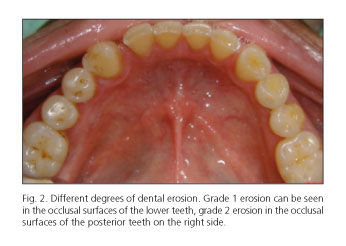
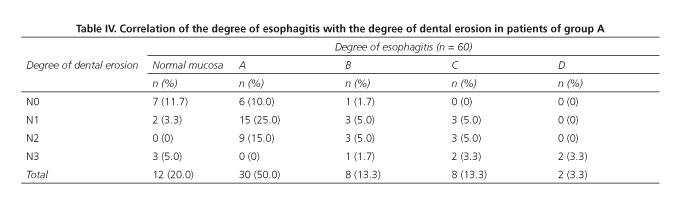
Dental erosion was identified in 46 cases (78.67%) in group A and in 1 case (3.3%) in group B (p = 0.0001); among patients with GERD, 23 cases (48.3%) were classified as N1, 15 (25.0%) as N2 and 8 (13.3%) as N3 (Figs. 1 and 2). The correlation between the degree of dental erosion and the extent of esophagitis is shown in table IV.
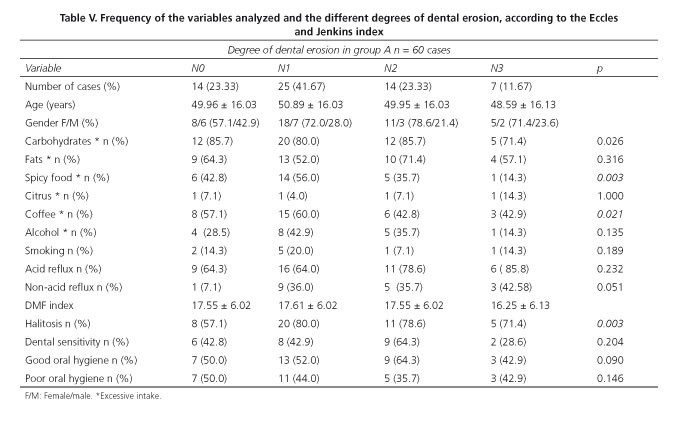
The mean age of patients with dental erosion was 49.84 years (48-51 years) and the female gender was predominant with a ratio of males to females of 1:2.3. The excessive intake of carbohydrates and fats did not vary significantly in the subgroups. Intake of spicy food and excessive coffee consumption predominated in the N0 and N1 subgroups. Excessive consumption of citrus was recorded only in 4 cases (6.6%). Alcohol intake was predominant in the N1 and N2 subgroups, and smoking in the N0 and N1 subgroups (Table V).
Oral hygiene habits showed no statistically significant difference (p = 0.82) between both groups. The additional use of mouthwashes and dental floss was reported by 52% in group A and 39% in group B. There were more patients with halitosis in group A (73.3%) compared with group B (30.0%) (p = 0.01).
The dental sensitivity test was positive in 25 cases (41.67%) in group A and in 16 cases (50.0%) in group B (p = 0.37), with predominance in the N2 subgroup (64.3%). The DMF index in group A was 17.61 ± 5.70, and 11.93 ± 5.22 in group B (p = 0.0001). Salivary pH was 6.14 ± 0.65 in group A and 6.26 ± 0.61 in group B. In both groups, the pH oscillated between 5.0 and 8.0 (p = 0.97) (Table VI).
Discussion
Dental erosion is a multifactorial disease that occurs more frequently in elderly patients, associated with bad hygiene/dietary habits, but especially with exposure to aggressive agents, among which acid plays a major role. It has been reported that the prevalence of dental erosion in patients with GERD varies from 5.00% to 58.41% (8,24-34), which we corroborated in our series of patients, were a prevalence of 78.9% was found, higher than the prevalence reported by other authors (8,28,30,31). This is directly related to the severity and duration of the disease; in our cohort, the average time of evolution was 50.09 ± 32.26 months, and the presence of acid reflux was demonstrated in 70.0% of cases by pH-metry (8,19-22,24,34). By comparing the degree of dental erosion with the severity of esophagitis, we found that 75.0% of the patients with grade N0 and N1 had normal mucosa or grade A esophagitis, whereas patients with N2 and N3 DE were associated with greater frequency to esophagitis stages C and D; the difference was statistically significant (p = 0.021).
The intake of certain foods has been linked to the presence of reflux because they decrease the tone of the lower esophageal sphincter and slow gastric emptying. Regarding dietary habits, we found that excessive consumption of fats, carbohydrates, spicy food and coffee intake in the group of patients with GERD was higher than in the control group (p = 0.0001 and 0.03 respectively) .Citrus consumption was higher in group B (p = 0.03) but no dental erosion was found in them.
Group A had an average salivary pH of 6.14 ± 0.65, and group B 6.26 ± 0.61 (p not significant). However, this cannot be considered a valid measurement because it was performed during a reflux episode.
Although the relationship between other dental diseases and GERD has not been accurately documented, 73.3% of our patients had halitosis, a higher frequency than in controls (p = 0.001) and than the frequency reported in the general population. Thus, the DMF index and dental sensitivity were higher in group A, which may have been due to poor hygiene habits in our population.
The results of our study suggest the involvement of acid reflux in the etiology of dental erosion; however, we believe that the number of cases studied is too small and that it should be expanded to confirm our findings.
We can conclude that in our population dental erosion has a high frequency in patients with GERD and that the characteristics of reflux are directly related to the severity of DE and, therefore, that it should be considered as an extraesophageal manifestation of reflux. We recommend that DE is detected in a timely manner in order to control it and prevent it from causing extensive dental damage.
References
1. Vakil N, van Zanten SV, Kahrilas P, Dent J, Jones R. The Montreal definition and classification of gastroesophageal reflux disease: A global evidence-based consensus. Am J Gastroenterol 2006;101:1900-20. [ Links ]
2. Pérez MJ, Serdio SM, Antonio MM, Valdéz LJ, Pérez TE. Guías clínicas de diagnóstico y tratamiento de enfermedad por reflujo gastroesofágico. Rev Gastroenterol Méx 2007;72:163-7. [ Links ]
3. Carmona SR. Enfermedad por reflujo gastroesofágico: síndromes extraesofágicos. Rev Gastroenterol Mex 2011;76:15-8. [ Links ]
4. Huerta IF. Enfermedad por Reflujo Gastroesofágico. En: Roesch DF, editor. Gastroenterología Clínica. 3a ed. México: Editorial Alfil; 2013. p. 67-82. [ Links ]
5. Barry DW, Vaezi MF. Laryngopharyngeal reflux: More questions than answers. Cleve Clin J Med 2010;77:327-34. [ Links ]
6. Ali DA, Brown RS, Rodriguez LO, Moody EL, Nasr MF. Dental erosion caused by silent gastroesophageal reflux disease. J Am Dent Assoc 2002;133:734-7. [ Links ]
7. Van Roekel Ned. Gastroesophageal reflux disease, tooth erosion, and prosthodontic rehabilitation: A clinical report. J Prosthodont 2003;12:255-9. [ Links ]
8. Moazzez R, Bartlett D, Anggiansah A. Dental erosion, gastro-oesophageal reflux disease and saliva: How are they related? J Dent 2004;32:489-94. [ Links ]
9. Oginni AO, Agbakwuru EA, Ndububa DA. The prevalence of dental erosion in Nigerian patients with gastro-oesophageal reflux disease. BMC Oral Health 2005;5: 1-11. [ Links ]
10. Pace F, Pallotta S, Tonini M, Vakil N, Bianchi PG. Systematic review: Gastro-oesophageal reflux disease and dental lesions. Aliment Pharmacol Ther 2008;27:1179-86. [ Links ]
11. Eccles JD, Jenkins WG. Dental erosion and diet. Journal of Dentistry 1974;2:153-9. [ Links ]
12. Holbrook WP, Furuholm J, Gudmundsson K, Theodors A, Meurman JH. Gastric reflux is a significant causative factor of tooth erosion. Journal of Dental Research 2009;88:422-6. [ Links ]
13. Wilder SC, Wilder SP, Kawakami WH, Voronets J, Osann K, Lussi A. Quantification of dental erosions in patients with GERD using optical coherence tomography before and after double-blind, randomized treatment with esomeprazole or placebo. Am J Gastroenterol 2009;104:2788-95. [ Links ]
14. Ranjitkar S, Smales R, Kaidonis J. Oral manifestations of gastroesophageal reflux Disease. Journal of Gastroenterology and Hepatology 2012;27:21-7. [ Links ]
15. Ranjitkar S, Smales R, Kaidonis J. Gastroesophageal reflux disease and tooth erosion. International Journal of Dentistry 2012;17:1-10. [ Links ]
16. Herbella FA, Patti MC. Gastroesophageal reflux disease: From pathophysiology to treatment. World J Gastroenterol 2010;16:3745-9. [ Links ]
17. Valdovinos DM. Avances en el diagnóstico de la enfermedad por reflujo gastroesofágico Rev Gastroenterol Mex 2011;76:11-5. [ Links ]
18. Fass R, Achem SR, Harding S, Mittal RK, Quigley E. Review article: Supra-oesophageal manifestations of gastro-oesophageal reflux disease and the role of night-time gastro-oesophageal reflux. Aliment Pharmacol Ther 2004;20:26-38. [ Links ]
19. Bartlett DW, Evans DF, Anggiansah A, Smith BG. A study of the association between gastro-oesophageal reflux and palatal dental erosion. British Dental Journal 1996;181:125-31. [ Links ]
20. Richter JE. Ear, nose and throat and respiratory manifestations of gastroesophageal reflux disease: An increasing conundrum. Eur J Gastroenterol 2004;16:837-45. [ Links ]
21. Farrokhi F, Vaezi MF. Extra-esophageal manifestations of gastroesophageal reflux. Oral Dis 2007;13:349-59. [ Links ]
22. Frye JW, Vaezi MF. Extraesophageal GERD. Gastroenterol Clin North Am 2008;37:845-58. [ Links ]
23. Ranjitkar S, Smales RJ, Kaidonis JA. Oral manifestations of gastroesophageal reflux disease. J Gastroenterol & Hepatol 2012;27:21-7. [ Links ]
24. Schiffner U, Micheelis W, Reich E. Erosionen und keilförmige Zahnhalsdefekte bei deutsche Erwachsenen und Senioren. Dtsch Zahnärztl Z 2002;57:102-6. [ Links ]
25. Lussi A, Schaffner M, Hotz P, Suter P. Dental erosion in a population of Swiss adults. Community dentistry and oral epidemiology 1991;19(5):286-90. [ Links ]
26. Gregory HB, Curtis DA, Kim L, Cello J. Evaluation of dental erosion in patients with gastroesophageal reflux disease. The Journal of prosthetic dentistr 2000;83(6):675-80. [ Links ]
27. Oggini AO, Agbakwuru EA, Ndubbuba DA. The prevalence of dental erosion in Nigerian patients with gastroesophageal reflux disease. BMC Oral Health 2005;5:1. [ Links ]
28. Meurman JH, Toskala J, Nuutinen P, Klemetti E. Oral and dental manifestations in gastroesophageal reflux disease. Oral Surg Oral Med Oral Pathol 1994;78:583-9. [ Links ]
29. Bartlett DW, Anggiansah A, Smith BG, Kidd EA. The role of regurgitation and other symptoms of reflux disease in palatal dental erosion; an audit project. Annals of The Royal College of Surgeons of England 2001;83:226. [ Links ]
30. Munoz JV, Herreros B, Sanchiz V, Amoros C, Hernandez V, Pascual I, et al. Dental and periodontal lesions in patients with gastro-oesophageal reflux disease. Dig Liver Dis 2003;35:461-7. [ Links ]
31. Schroeder PL, Filler SJ, Ramirez B, Lazarchik DA, Vaezi MF, Richter JE. Dental erosion and acid reflux disease. Ann Intern Med 1995;122:809-15. [ Links ]
32. Loffeld RJ. Incisor teeth status in patients with reflux oesophagitis. Digestion 1996;57:388-90. [ Links ]
33. Jarvinen V, Meurman JH, Hyvarinen H, Rytomaa I, Murtomaa H. Dental erosion and upper gastrointestinal disorders. Oral Surg Oral Med Oral Pathol 1988;65:298-303. [ Links ]
34. Vargas TL, Torres VN, Vargas CG. Erosiones dentales en pacientes con diagnóstico de enfermedad por reflujo gastroesofágico en el Hospital Nacional Arzobispo Loayza. Rev Gastroenterol Perú 2012;32:343-50. [ Links ]
35. Seung H, Woon S, Sik H, Wook H, Hahm K. Halitosis - Could It Be Another Extra-Esophageal Symptom of GERD? The 2nd International Gastrointestinal Consensus Symposium (IGICS). Digestion 2009;79:53-77. [ Links ]
36. Belafsky, Postma GN, Koufman JA. Validity and reliability of the reflux symptom index (RSI). J Voice 2002;16:274-7. [ Links ]
37. Villavicencio JJ, Xaus G. Diagnóstico y tratamiento de la hipersensibilidad dentinaria. Revista Dental de Chile 2010;101:17-25. [ Links ]
![]() Correspondence:
Correspondence:
Laura Roesch Ramos
Coordinator of the Master in Comprehensive Rehabilitation
University of Veracruz. Juan Pablo II SN
Frac. Costa Verde
Boca del Rio Ver
Veracruz, México
e-mail: lroesch@uv.mx
Received: 09-09-2013
Accepted: 27-01-2014











 texto en
texto en