My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.106 n.7 Madrid Jul./Aug. 2014
CLINICAL NOTES
Acute alithiasic cholecystitis: A not so rare disease
Colecistitis aguda alitiásica en pediatría. Una patología no tan rara
Javier Blasco-Alonso1, Eloísa Santiago-García-Caro2, Raquel Gil-Gómez3, Carolina Jiménez-Alcántara2, Pilar Sánchez-Yáñez2 and Guillermo Milano-Manso3
1Department of Gastroenterology and Children Nutrition, UGC Pediatrics.
2UGC Pediatrics.
3UGC Critical Care and Children Urgencies. Hospital Materno-Infantil.
Hospital Regional Universitario Carlos Haya. Málaga, Spain
ABSTRACT
Introduction: Acute acalculous cholecystitis (AAC) occurs more frequently in critically ill patients, in the immediate postoperative period, after trauma or extensive burns. It has a high rate of morbidity and mortality. Ischemia, infection and vesicular stasis are determinants in its pathogenesis.
Material and method: Retrospective study including all cases of AAC diagnosed in our pediatric intensive care unit between January 1997 and December 2012.
Results: We included 7 patients, all associated with viral or bacterial infection. All of them suffered from abdominal pain, mainly localized in the right upper quadrant, jaundice and dark urine. Abdominal ultrasound showed thickening and hypervascularity of the gallbladder wall in all cases. The outcome was satisfactory without surgery in all patients.
Conclusions: The clinical presentation is oligosymptomatic within severe systemic diseases. The AAC should be suspected in the appearance of any abdominal pain with jaundice/dark urine and hypertransaminasemia in patients suffering from critical or serious infections.
Key words: Acute acalculous cholecystitis. Ultrasound. Childhood. Critical Care. Hypertransaminasemia. Jaundice.
RESUMEN
Introducción: la colecistitis aguda alitiásica (CAA) se da con mayor frecuencia en pacientes críticos, en el periodo postoperatorio inmediato, tras traumatismos o quemaduras extensas. Tiene una alta tasa de morbimortalidad. La isquemia, la infección y la estasis vesicular son determinantes en su patogenia.
Material y método: estudio retrospectivo que incluye todos los casos de CAA diagnosticados en la unidad de cuidados intensivos de nuestro centro en el periodo comprendido entre enero de 1997 y diciembre de 2012.
Resultados: se incluyen a 7 pacientes, todos asociaron infección viral o bacteriana. Todos debutaron con dolor abdominal localizado en hipocondrio derecho, ictericia y coluria. La ecografía abdominal en todos los casos demostró engrosamiento e hipervascularización de la pared vesicular. La evolución fue satisfactoria en todos los casos sin necesidad de cirugía.
Conclusiones: la presentación del cuadro es oligosintomática en el seno de enfermedades sistémicas de gravedad variable. La CAA se debe sospechar ante todo cuadro de dolor abdominal con ictericia/coluria e hipertransaminasemia en pacientes críticos o que cursan infecciones graves.
Palabras clave: Colecistitis aguda alitiásica. Ecografía. Infancia. Cuidados intensivos. Hipertransaminasemia. Ictericia.
Introduction
Acute acalculous cholecystitis (AAC) represents an emerging disease (1) that can represent 30 to 50 % of cholecystitis in children (2). It usually occurs within systemic bacterial (gram-negative or anaerobic) or viral (EBV, hepatotropic virus...) infections (3), as well as secondary to dehydration or prolonged parenteral nutrition, appearing rarely as a complication of severe medical-surgical diseases (multiple trauma, burns, postoperative...), more common etiologies in adult. Regarding clinical manifestations (4,5), it is required a high suspicion, since the onset of unexplained fever, jaundice or vague abdominal discomfort in a critically ill patient, often intubated and sedated, may be the only track (6,7).
AAC handling depends on the time of diagnosis, and thus in early stages of the disease exclusive medical treatment may be sufficient (8), reserving cholecystectomy for patients with vesicular gangrene or perforation (9). Mortality depends on the underlying medical condition, ranging from 90 % in critically ill patients, up to 10 % in the outpatient (10), but always greater than 1 % corresponding to gallstone cholecystitis.
Our goal is to review the cases of AAC in our pediatric center and compare with the existing literature.
Case reports
Retrospective review of hospital records of pediatric patients under 14 years diagnosed with AAC in our hospital from January 2001 to December 2013. We collected epidemiological (age and sex), clinical (underlying disease and clinical characteristics), diagnostic (special emphasis on ultrasound) and therapeutic (drugs employed, complications) data after informed consent was obtained.
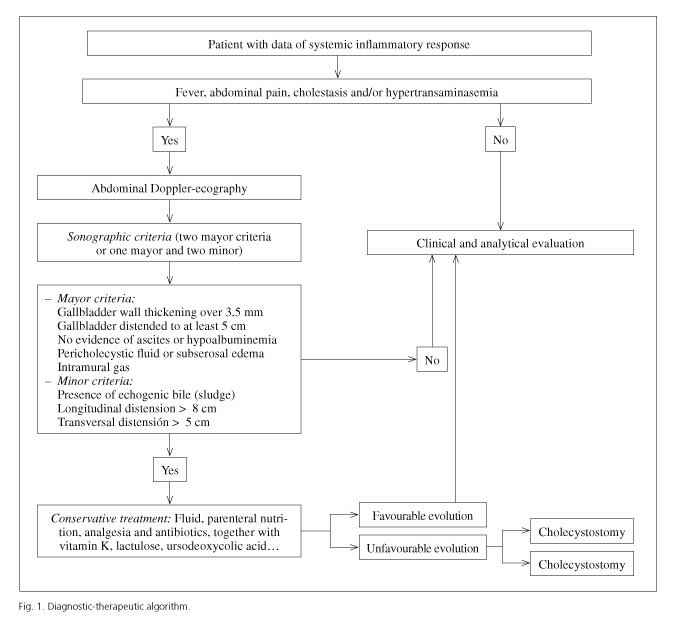
The AAC diagnosis was established by clinical suspicion (pain in the upper right quadrant, fever, leukocytosis, normal liver function) and confirmed by abdominal ultrasound (92 % sensitivity and 96 % specificity), not being necessary to perform any other imaging test. Ultrasound studies reviewed by two radiologists in all children who met clinical criteria. Ultrasonographic diagnostic criteria (8,11) were divided into major (gallbladder wall thickening over 3.5 mm, gallbladder distended to at least 5 cm in the longitudinal dimension and no evidence of ascites or hypoalbuminemia, the presence of pericholecystic fluid or subserosal edema, intramural gas) and minor criteria -presence of echogenic bile (sludge), distension greater than 8cm in the longitudinal or 5 cm in the transverse dimension-. Adiagnosis was considered positive if it included either a minimum of two major criteria or one major and two minor criteria, in the appropriate clinical setting.
Seven patients were included (Table I): 5 of them were women, ages distributed in two peaks (4 teenagers between 11 and 13 years and 3 infants between 2 and 24 months), with the first four cases diagnosed between 2001 and 2005 and the last three ones between 2011 and 2013. Five of them were previously healthy children, one suffered from acute lymphoblastic leukemia (ALL) and received a bone marrow transplant with suspected graft versus hostage disease (GVHD) and, the other case was a newborn with sepsis/meningitis by E. coli. The predisposing factor was a viral or bacterial infection (EBV, HAV, E. coli, Micrococcus, B. melitensis, P. pneumoniae) in 6 patients, highlighting positive urine culture only E. faecalis in the seventh case. All of them started with mild abdominal pain, right upper quadrant referral, and fever, and only the first four cases with cholestatic syndrome presented with jaundice and/or dark urine. Blood tests revealed moderate hypertransaminasemia, always greater than three times normal, in all patients (maximum levels 1,286 U/l AST and 2,798 ALT, with consistently high GGT), with hyperbilirubinemia in 4 cases. The ultrasound showed wall thickening and hypervascularity in the absence of lithiasis in all cases without pericholecystic fluid. Treatment was conservative with fluid therapy, parenteral nutrition, analgesia and antibiotics, adding vitamin K, lactulose and ursodeoxycholic acid in the patient with ALL and in neonates. The most used combination of antibiotics was third generation cephalosporin and antianaerobe agents (mainly metronidazole). The evolution was satisfactory without surgery in all patients.
Discussion
As for the typical age of presentation, according to Imamoglu (8), it predominates at school age (mean 7.8 years), not fully coinciding in our series (median 11.5 years), although cases have also been reported in neonates, infants and preschool (in our work appearing in two infants, aged 2 and 12 months).
AAC has been associated with intercurrent infections, metabolic disorders, vascular problems, burns, injuries and malignancies in children. Our patients presented within systemic diseases of varying severity (5 of them stayed in the pediatric intensive care unit at some point), as published in the literature (9), 5 of them with different blood cultures and/or seropositive to different infectious agents, as in the seventh patient, despite being a positive urine culture to E. faecalis, the clinical picture was most consistent with a viral hepatitis untested by serology. This demonstrates that most AAC in children occur during the course of serious infectious diseases (10), 10% of which are in the form of sepsis, as published by Shu-Ching Huang in 2011 (11), and which is also reflected in our work with the 2 month-old infant suffering from AAC within E. coli sepsis/meningitis. Furthermore, Shu-Ching Huang also said that less than 10 % of the AAC are produced in the course of neoplastic diseases, as our 12year old patient with a history of ALL and transplanted with suspected GVHD, treated cyclosporine, which developed Micrococcus AAC. Moreover, the latter patient is the first pediatric case reported in the literature by Micrococcus CAA, after a thorough review using MEDLINE + EMBASE (key words: AND Micrococcus cholecystitis), which showed only one previous case of acute gallstone cholecystitis by Kocuria kristinae, belonging to genus Micrococcus, a 56year old Chinese woman. It must be also pointed out the case of the AAC as a complication of systemic brucellosis, since only 21 cases have been described to date after a MEDLINE search from 1934 to 2012, with one pediatric patient, being, therefore, our case number 22 worldwide, the second pediatric patient (12).
The clinical presentation of the AAC is variable, and depends on the predisposing conditions (13,14). In all of our patients, at the onset of the disease, there was fever and abdominal pain (88 % and 40 % respectively in the work of Huang Shu-Ching), signs that often go unnoticed in critically ill patients, or make it difficult to classify abdominal pain. Cholestasis is another common sign in patients with AAC (5/7 of our patients) and, together with the hypertransaminasemia not justified for other reasons (all cases), are alarm data that must be then confirmed by imaging. In this respect, the ultrasound technique is more sensitive (92 %) and specific (96 %) for the diagnosis of CAA (8), besides being a non-invasive and having the possibility of being realized bedside. In all of our patients sonographic criteria were met: Gallbladder distension with wall thickening > 3.5 mm, without lithiasis although biliar sludge (8).
Although for many years the standard treatment was cholecystectomy, this technique is not free of morbidity and mortality, ranging between 9 and 66 %, and it is now accepted that in children with AAC that is recognized early, initial conservative treatment (15) is safe and effective, as described in our series, reserving surgical treatment for cases of gangrene or perforation (none in our case). Conservative treatment would be adequate hemodynamic stabilization, suppression of drugs that might hinder the gallbladder emptying, fluid therapy, parenteral nutrition, analgesia and use of antibiotics active on gram negative, anaerobes and Enterococci, which in turn reach therapeutic concentrations in the biliary tract. So, all of our patients were treated conservatively, adding vitamin K, lactulose and ursodeoxycholic acid in the patient with ALL and in neonates who attended E. coli sepsis/meningitis.
According to the data of our study and the literature reviewed, we draw several conclusions: Although the AAC is a rare entity in children, it must be considered by the pediatrician among the causes of abdominal pain, especially in critically ill children that are often intubated and sedated, with detection of vague abdominal discomfort, fever and jaundice besides, and where the source of infection is not entirely clear. Ultrasound is the most reliable method for diagnosis (Fig. 1). In children, conservative treatment is effective in solving this disease, although it does require close clinical, analytical and ultrasound monitoring, able to detect complications.
References
1. Glenn F, Becker CG. Acute acalculous cholecystitis: an increasing entity. Ann Surg 1982;195:131-6. [ Links ]
2. Tsakayannis DE, Kozakewich HP, Lillehei CW. Acalculous cholecystitis in children. J Pediatr Surg 1996;31:127-30. [ Links ]
3. Souza LJ, Braga LC, Rocha N de S, Tavares RR, et al. Acute acalculous cholecystitis in a teenager with hepatitis A viral infection: A case report. Braz J Infect Dis 2009;13:74-6. [ Links ]
4. Venkataramani A, Strong RM, Anderson DS, Gilmore IT, Stokes K, Hofmann AF. Abnormal duodenal 1. zbile composition in patients with acalculous chronic cholecystitis. Am J Gastroenterol 1998; 93:434-41. [ Links ]
5. Ryu JK, Ryu KH, Kim KH. Clinical features of acute acalculous cholecystitis. J Clin Gastroenterol 2003;36:166-9. [ Links ]
6. Trowbridge RL, Rutkowski NK, Shojania KG. Does this patient have acute cholecystitis? JAMA 2003;289:80-6. [ Links ]
7. Yokoe M, Takada T, Strasberg SM, Solomkin JS, Mayumi T, Gomi H, et al. New diagnostic criteria and severity assessment of acute cholecystitis in revised Tokyo Guidelines. J Hepatobiliary Pancreat Sci 2012;19:578-85. [ Links ]
8. Imamoglu M, Sarihan H, Sari A, Black CT, Lally KP, Andrassy RJ. Acute acalculous cholecystitis in children: Diagnosis and treatment. J Pediatr Surg 2002;37:369-72. [ Links ]
9. Owen CC, Jain R. Acute acalculous cholecystitis. Curr Treat Options Gastroenterol 2005;8:99-104. [ Links ]
10. Unal H, Korkmaz N, Kirbas I, Selcuk H, Yilmaz U. Acute acalculous cholecystitis associated with acute hepatitis B infection. Int J Infect Dis 2009;13:e310-2. [ Links ]
11. Shu-Ching Huang and Yao-Jong Yang. Septic shock and hypofibrinogenemia predict a fatal outcome in childhood acute acalculous cholecystitis. JPGN 2011;53: 548-52. [ Links ]
12. Al Otaibi FE. Acute acalculus cholecystitis and hepatitis caused by Brucella melitensis. J Infect Dev Ctries 2010; 4: 464-7. [ Links ]
13. Ryu JK, Ryu KH, Kim KH. Clinical features of acute acalculous cholecystitis. J Clin Gastroenterol 2003; 36:166-9. [ Links ]
14. Huffman JL, Schwenker S. Acute acalculous cholecystitis: A review. Clin Gastroenterol Hepatol 2010;8:15-22. [ Links ]
15. Janowitz P, Kratzer W, Zemmler T, Tudyka J, Wechsler JG. Gallbladder sludge: Spontaneous course and incidence of complications in patients without stones. Hepatology 1994;20:291-4. [ Links ]
![]() Correspondence:
Correspondence:
Javier Blasco Alonso.
Department of Gastroenterology and Children Nutrition,
UGC Pediatrics.
Hospital Materno-Infantil.
Hospital Regional Universitario Carlos Haya.
Avda. Carlos Haya, s/n.
29010 Málaga, Spain
e-mail: javierblascoalonso@yahoo.es
Received: 15-11-2013
Accepted: 24-03-2014











 text in
text in