Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.107 no.3 Madrid mar. 2015
LETTERS TO THE EDITOR
Clinical presentation of anal duct adenocarcinoma as benign anal stenosis
Presentación clínica de adenocarcinoma de ductos anales como estenosis anal benigna
Key words: Anal cancer. Laparoscopic surgery. Anal stricture. Anal radiotherapy.
Palabras clave: Cáncer anal. Cirugía laparoscópica. Estenosis rectal. Radioterapia anal.
Dear Editor,
Anal cancer represents 2 % of digestive tumors, with anal duct cancer making up fewer than 10 % of anal malignancies (1). This is a rare neoplasm of CK7 (+) cells from the anal ducts. Since its initial description by Hermann in 1880 (2), 5-year survival rates have been estimated around 5 %-40 % (3-5), and one reason for this poor prognosis is delayed diagnosis due to overlapping with benign conditions.
Case report
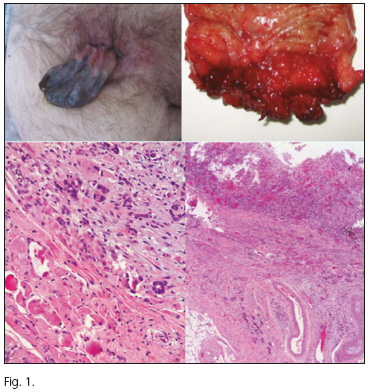
We report the case of a 53-year-old male with anal pain, anal bleeding, and fecal volume changes for 6 months who was studied in another institution, where he underwent a colonoscopy and magnetic resonance scan that revealed an anorectal mass, and then an endoscopic ultrasonography that showed a stenotic ring with significant inflammatory changes leading to loss of wall structure with uncertain infiltration; biopsies found chronic inflammation without dysplasia, consistent with solitary rectal ulcer. Two anorectoplasties, a Z-flap, and then dilatations were performed in that center, with persistent chronic pain. In our institution the patient presented with poorly controlled severe pain despite high-dose opioids, severe rectal incontinence, and a stricture 1 cm away from the pectineal line that hardly allowed a finger through it. In view of the above a laparoscopic ultralow anterior resection was decided upon. An endonal pulltrough is performed intraoperatively at the expense of definitive histology, with a result of CD7(+)/CD20(-)/PSA(-) adenocarcinoma of the anal glands with no mucinous areas, T3 N0, with involved radial and distal margins, which led to complete therapy by means of laparoscopic cylindrical abdominoperineal amputation. The patient remains alive and disease-free at 12 months following the procedure (Fig. 1).
Discussion
Two theories exist on the origin of these tumors: They either occur at the dentate line transitional epithelium or derive from perianal apocrine or intramuscular glands. Diagnosis requires: Localization in the area where glands are usually present; exclusion of a different origin; transition from normal gland to carcinoma, preferrably with dysplastic transitional tissue and typical microscopic appearance (6).
Clinical manifestations include pain crises, bleeding, incontinence, weight loss, and/or sphincter induration for long periods of time, which makes differentiation from other benign conditions challenging.
The presence of lymphatic spread varies among series from 13.5 % to 62 % (3-5).
The ideal therapy is controversial, with abdominoperineal amputation being most common. Tarazi and Nelson have shown a good response to chemotherapy followed by surgery (7). Unfortunately, most patients present with advanced disease, with adjuvancy providing no clear benefits (3,8).
Overall survival is poor despite aggressive management: From 5 % to 21 % at 5 years (3,8-10).
These neoplasms are highly aggressive and can only be cured when a high index of suspicion and an early diagnosis occur.
Iago Justo-Alonso1, María García-Nebreda1,
Carmelo Loinaz-Segurola1, Óscar Caso-Maestro1,
Félix Cambra-Molero1, Alejandro Alberto Marcacuzco-Quinto1,
Begoña Casis-Herce2 and Carlos Jiménez-Romero1
1 Department of General Surgery, Digestive Diseases and
Transplant of Abdominal Organs2 Department of Digestive
Diseases. Hospital Universitario 12 de Octubre. Madrid, Spain
References
1. Jemal A, Siegel R, Xu J, Ward E. Cancer statistics 2010. CA Cancer J Clin 2010;60:277-300. [ Links ]
2. Herrmann G. Sur la structure et le developement de la muuqueuse anale. J de lánat et physiol 1880;16:434-72. [ Links ]
3. Abel ME, Chiu YS, Russell TR, Volpe PA. Adenocarcinoma of the anal gland. Results of a survey. Dis Colon Rectum 1993;36:383-7. [ Links ]
4. Behan WM, Burnett RA. Adenocarcinoma of the anal glands. J Clin Pathol 1996;49:1009-11. [ Links ]
5. Jensen SL, Shokough-Amiri MH, Hagen K, Harling H, Vagn Nielsen O. Adenocarcinoma of the anal ducts. A series of 21 cases. Dis Colon Rectum 1988;31:268-72. [ Links ]
6. Fenger C, Filipe IM. Pathology of the anal gland with special reference to their mucin histochemistry. Acta Pathol Microbiol Immunol Scand 1977;85:273-85. [ Links ]
7. Tarazi R, Nelson RL. Anal adenocarcinoma: A comprehensive review. Semin Surg Oncol 1994;10:235-40. [ Links ]
8. Cabrera A, Tsukada Y, Pickren JW. Adenocarcinomas of the anal gland and peri-anal tissues. Ann Surg 1966;164:152-6. [ Links ]
9. Meriden Z, Montgomery E. Anal duct carcinoma: A report of 5 cases. Human Pathology 2012;43:216-20. [ Links ]
10. Lee Bigs R, Lucha P, Martin Stoll Ph. Anal duct carcinoma: Report of case and a survey of the experience of the American Osteopathic College of Proctology. JAOA 2001;101:450-3. [ Links ]











 texto en
texto en