My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.107 n.4 Madrid Apr. 2015
CASE REPORTS
Is the sinusoidal obstructive syndrome post-liver transplantation a pathologic entity with a multifactorial etiology?
¿Es el síndrome de obstrucción sinusoidal asociado al trasplante hepático una entidad patológica con una etiología multifactorial?
Luis Miguel Marín-Gómez1, José María Álamo-Martínez1, Gonzalo Suárez-Artacho1, José Ramírez-Santos2, Carmen Bernal-Bellido1, Lydia Barrera-Pulido1, Juan Serrano-Díaz-Canedo1, Francisco Javier Padillo-Ruiz1 and Miguel Ángel Gómez-Bravo1
1Liver Transplantation Unit and 2Department of Pathology. Hospital Universitario "Virgen del Rocío". Sevilla, Spain
ABSTRACT
The sinusoidal obstructive syndrome is a complication typically associated with hematopoietic stem cell transplantation. This syndrome, more commonly known as veno-occlusive disease, has also been described after liver transplantation. It can have a life-threatening course. Herein, we describe the hepatic graft loss secondary to the development of a sinusoidal obstructive syndrome after a severe acute cellular rejection and toxic levels of once daily modified released tacrolimus (TAC). We discuss the role of the endotheliitis of acute rejection and toxic metabolites of some immunosuppressants such as azathioprine and TAC. Based on the current scientific evidence, we contemplate the possibility that the etiology of sinusoidal obstruction syndrome post-liver transplantation is multifactorial.
Key words: Sinusoidal obstructive syndrome. Drug toxicity. Graft rejection.
RESUMEN
El síndrome obstructivo sinusoidal hepático, anteriormente conocido como enfermedad veno-oclusiva, es una complicación típicamente asociada al trasplante de precursores hematopoyéticos. También se ha descrito su asociación con el trasplante hepático. Puede llegar a ser una causa de mortalidad. En este documento se describe la pérdida de un injerto hepático como consecuencia del desarrollo de un síndrome de obstrucción sinusoidal, insistiendo en la evolución del mismo y su relación con el rechazo agudo celular y los niveles tóxicos de tacrolimus en plasma. Basándonos en la evidencia actual, discutimos sobre la posibilidad de que la etiología del síndrome de obstrucción sinusoidal postrasplante hepático sea multifactorial, puesto que se relaciona con la endotelitis del rechazo agudo y los metabolitos tóxicos de algunos inmunosupresores, como la azatioprina y el tacrolimus.
Palabras clave: Síndrome de obstrucción sinusoidal. Toxicidad por fármacos. Rechazo al trasplante.
Introduction
Sinusoidal obstructive syndrome (SOS) is a potentially fatal complication of myeloablative treatment before hematopoietic stem cell transplantation (SCT). SOS is a syndrome that is characterized by hyperbilirubinemia (> 2 mg/dl), painful hepatomegaly and weight gain secondary to fluid retention. Histological features are characterized by an initial acute injury to the endothelial cells, followed by detachment and embolization in the central area of the hepatic lobule, leading to post-sinusoidal outflow block. Afterwards, there is sub-endothelial deposition of fibrous tissue in sinusoids and in central and sub-lobular veins (1). SOS has also been described after liver transplantation (LT) (2), but only a few articles have been published because SOS after LT is relative rare, approximately 2%. The endothelial damage that occurs during the process of acute cellular rejection (ACR) or the administration of certain cytotoxic chemotherapeutic agents is known to facilitate the eventual development of SOS. The prognosis of SOS varies from complete resolution after withdrawal of the offending drugs to fatal outcome resulting from hepatic failure and death (1,3,4).
Herein, we describe a case of liver re-transplantation due to SOS after a severe ACR and toxic levels of once daily modified released TAC (TAC-MR).
Case report
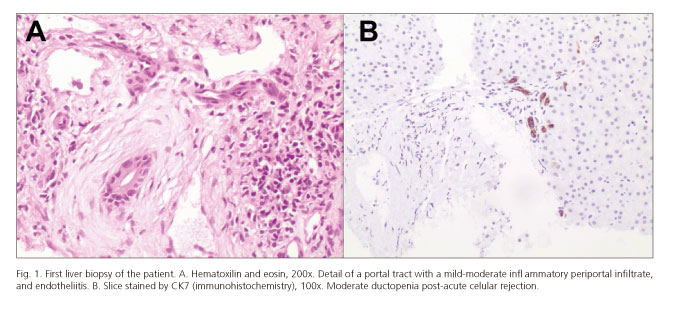
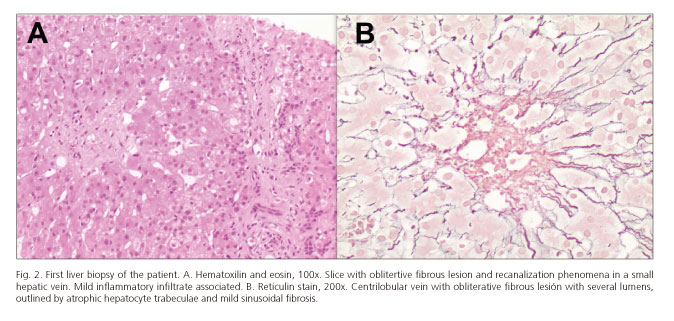
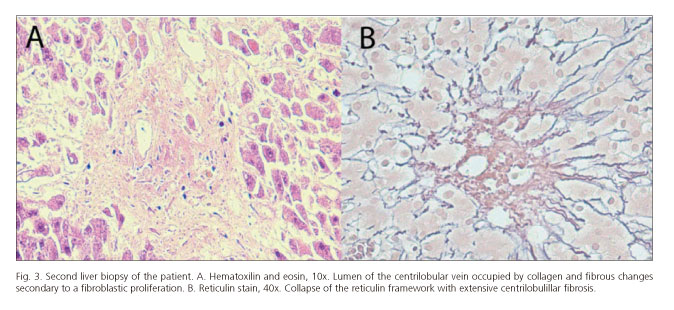
A 43-year-old woman underwent deceased donor LT for end-stage liver disease (MELD score 19) secondary to alcoholic hepatitis. The donor was a 54-year-old man and the liver was recovered using standard technique. It was preserved in Wisconsin University solution. A wedge biopsy before procurement showed 30% macro-steatosis without fibrosis. The cold ischemia time was 210 min. The recipient hepatectomy included preservation of the caval vein for the piggy-back technique. No transfusional requirement was needed during the surgery. The immunosuppression regimen consisted of triple therapy with TAC (Advagraf®, Astellas Pharma US, Il), mycophenolate mofetil, and prednisone. Initially, her post-transplantation course was marked by a mild graft dysfunction treated favorably with 0.03 µg/kg/min Alprostadil for 3 days. As the donor was cytomegalovirus (CMV) seropositive and the recipient CMV seronegative, we started CMV infection prophylaxis with 450 mg valganciclovir/12 h. Three weeks after LT she presented jaundice, abdominal pain, ascites and hepatomegaly. The blood test showed cholestasis associated with infratherapeutic levels of TAC (5.50 ng/ml). Analytical values were: Total bilirubin: 3.47 mg/dL; aspartate aminotransferase: 61 IU/L; alanine aminotransferase: 74 IU/L; gamma-glutamyl transpeptidase: 563 IU/L; alkaline phosphatase: 312 UI/L; INR: 5.24 mg/dL. Doppler ultrasound showed mild ascites and patency of the hepatic artery (30 cm/s), and portal (25 cm/s) and hepatic veins. Histological examination by transjugular biopsy carried out 24th post-transplantation day PTD revealed post-ACR ductopenia with moderate inflammatory signs. Centrilobular sclerosis hyaline compatible with SOS (Figs. 1 and 2). The rejection activity index (RAI) = 7 (3+2+2). We started treatment of the cellular rejection with three endovenous bolus of 1 g methyl-prednisolone. Simultaneously, we optimized the plasmatic levels of TAC and we administered three endovenous bolus of 1 g methyl-prednisolone / 24 h. However, we observed an increasing ascites. TAC reached toxic plasmatic levels (between 15 and 10 for 18 days), in spite of consecutive dose-decreasing of TAC-MR. She needed repeat large-volume paracentesis, volume expansion with intravenous albumin, and diuretic treatment. The study of the ascitic liquid retrieved a transudate with negative cultures (included mycobacteria after 90 days) and a serum-ascites albumin gradient > 1.1 g/dl, consistent with portal hypertension. The liver function was impaired and associated acute renal failure (total bilirubin: 1.23 mg/dl; aspartate aminotransferase: 100 IU/l; alanine aminotransferase: 149 IU/l; gamma-glutamyl transpeptidase: 1346 IU/l; alkaline phosphatase: 504 IU/l; creatinine: 5.24 mg/dl; Cokcroft-Gault ClCr: 11.37 ml/min). The patient had a comprehensive cardiac evaluation that excluded right-sided heart failure. At this point an angio computed tomography excluded venous and arterial flow obstruction again. We decided to quantify pressures and the portal gradient to define the SOS severity and prognosis. Unfortunately, a deterioration of her clinical status caused by a hemoperitoneum secondary to a paracentesis was diagnosed. An emergency laparotomy was made. The bleeding point was not identified, but the biopsy (Fig. 3) carried out confirmed an aggravation of the SOS, with extended and severe centrilobular fibrosis and partial resolution of the acute cellular rejection (RAI = 4). Retransplantation was performed 9 days after hemoperitoneum (51st PTD) without complications. The new immunosuppressive regimen was basiliximab, mycophenolate-mofetil, steroids and delayed introduction of TAC-MR. The pathologic study of the explanted allograft revealed striking bridging fibrosis between centrilobular veins and the centrilobular portal space. Some centrilobular veins had been totally replaced by a fibrous mass. Several portal venules showed remarkable multiplications with fibrous change, indicative of severe portal hypertension. Her post-transplantation course was complicated by a mild ACR and an acute renal failure. She was discharged at 29th PTD. At day, she is in study for biliary stenosis.
The clinical evolution of the clinical case is resumed in the figure 4.
Discussion
Recently, it has been clarified that the initial trigger is a toxic injury at the level of the sinusoidal endothelial cell (SEC). It is characterized by the loss of sinusoidal wall integrity with consequent sinusoidal congestive obstruction and is occasionally associated with perisinusoidal fibrosis, centrilobular hepatic vein fibrotic obstruction, nodular regenerative hyperplasia or peliosos (6). This disease has long been recognized as a complication of SCT (7) and, recently, to the administration of oxaliplatine (8). Though SOS has been described after solid organ transplantation, there are only a few reports. In the case of LT, the reported incidence of clinical SOS is 1.9-2.9% (3,4). However, the real incidence of SOS after LT is unknown, because the clinical signs and symptoms are non-specific and relatively frequent in the postoperative course of a LT. Probably, the majority of diagnosed cases are considered "Severe" or "Moderate", and we do not study self-limited mild forms. Therefore, we cannot estimate the real incidence and consequences of SOS in the outcome of LT at day. In addition, the available scientific evidence about the SOS management is mostly related to the SCT.
It is currently recognized that the development of SOS after LT is frequently associated with ACR. However, the reason why some individuals with severe ACR went on to develop SOS and others did not is unclear. There have been presented some retrospective series (3,4,9,10) with a significant component of endothelial damage between the findings of the ACR in patients with histologic changes of SOS (9,10). Our clinical case was diagnosed after a biopsy proven acute rejection with endothelitis in postoperative day 24th and ruling out several pathologies.
Moreover, the pathologic study of our donor liver just prior to procurement showed 30% macro-steatosis. The detoxification of metabolites that precipitate oxidative stress carries out into the hepatocyte by means of the glutathione. The availability of glutathione in fatty liver mitochondria, such as in this donor, is diminished. Although the SEC also has the function of detoxification, the SEC intracellular glutathione concentrations are less than half that of hepatocytes and the glutathione detoxification capacity of the SEC is therefore much less than that of hepatocytes (10). It is suggested that this weak glutathione detoxification capacity of the SEC predisposes and exacerbates sinusoidal injury. Moreover, genetic polymorphism all appears to increase the risk of developing SOS. A specific polymorphism of the glutathione-S-transferase gene was found to be more frequent among patients who developed SOS (11).
Our recipient was also at risk of suffering a CMV-related vascular complication because of the CMV seromismatched transplantation. Vascular injury caused by CMV infection is well known regarding, in particular, the incidence of hepatic artery thrombosis after LT. In response to the infection, intercellular adhesion molecule 1, which is not detected on normal SEC, is induced by monokines such as interleukin-1 and tumor necrosis factor 1 alpha in the sinusoidal endothelium, leading to marked disturbance of the sinusoidal microcirculation. The injury also occurs in the sinusoidal endothelium (12) and it may increase the severity of the SOS.
Finally, there is another factor that we consider very important for the SOS development in this particular case. SOS has been associated to the administration of immunosuppressant agents used after LT, as azathioprine or TAC (2,13). Increasing concentrations of these specific metabolites might account for the appearance of sinusoidal endothelial injury. The hepatotoxicity caused by active metabolites is aggravated in situations where TAC half life of the drug (V1/2) increases and clearance impair, as consequence of the reduction of the absolute hepatocyte mass (hepatocyte dysfunction, post-reperfusion damage, reduced CYP (cytochrome P450) 450 enzymatic concentration or activity, and decreased sinusoidal perfusion). TAC is primarily metabolized by the cytochrome P450 (CYP) 3A subfamily (mainly, CYP 3A4 and CYP 3A5) in liver microsomes. The zone 3 of the liver acinus has the highest levels of CYP and is the most affected by the SOS (13,14). However, this has not been confirmed, and further study is needed.
In our experience, in cases of SOS associated to ACR we try to minimize the dose of TAC to avoid perpetuating the sinusoidal (14). The optimization of dose to low levels, within the therapeutic range, is possible because we start early treatment with endovenous methyl-prednisolone boluses. We believe that the promptness in the decrease of the dose is more important in cases in which we use once daily modified released TAC (Advagraf®, Astellas Pharma US, Il). The TAC-MR V1/2 is more sustained than in the traditional formula (TAC V1/2: 12 h vs. TAC-MR V1/2: 30 h) (15,16).
As conclusion, SOS is a complex pathologic entity with a probably multifactorial etiology. In our clinical case, we interpret that the ACR that did not respond to the treatment acted as a precipitating factor of sinusoidal endothelial damage, while the altered metabolism of TAC associated to the hepatic dysfunction contributed to perpetuate and aggravate the SOS through its own toxic metabolites.
References
1. DeLeve LD, Valla DC, Garcia-Tsao G; American Association for the Study Liver Diseases. Vascular disorders of the liver. Hepatology 2009;49:1729-64. [ Links ]
2. Sterneck M, Wiesner R, Ascher N, et al. Azathioprine hepatotoxicity after liver transplantation. Hepatology 1991;14:806-10. [ Links ]
3. Sebagh M, Debette M, Samuel D, et al. Silent presentation of veno-occlusive disease after liver transplantation as part of the process of cellular rejection with endotelial prediction. Hepatology 1999;30:1144-55. [ Links ]
4. Sebagh M, Azoulay D, Roche B, et al. Significance of isolated hepatic veno-occlusive disease/sinusoidal obstruction syndrome after liver transplantation. Liver Transpl 2011;17:798-808. [ Links ]
5. Demetris AJ, Batts KP, Dhillon AP, et al. Banff schema for grading liver allograft rejection: An international consensus document. Hepatology 1997;25:658-63. [ Links ]
6. Rubbia-Brandt L. Sinusoidal Obstruction Syndrome. Clin Liver Dis 2010;14:651-68. [ Links ]
7. Shulman HM, Fisher LB, Schoch HG, et al. Venoocclusive disease of the liver after marrow transplantation: Histological correlates of clinical signs and symptoms. Hepatology 1994;19:1171-80. [ Links ]
8. Rubbia-Brandt L, Audard V, Sartoretti P, et al. Severe hepatic sinusoidal obstruction associated with oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer. Ann Oncol 2004; 15:460-6. [ Links ]
9. Sanei MH, Schiano TD, Sempoux C, et al. Acute cellular rejection resulting in sinusoidal obstruction syndrome and ascites postliver transplantation. Transplantation 2011;92:1152-8. [ Links ]
10. DeLeve LD, Shulman HM, McDonald GB. Toxic injury to hepatic sinusoids: Sinusoidal obstruction syndrome (veno-occlusive disease). Semin Liver Dis 2002;22:27-41. [ Links ]
11. DeLeve LD. Glutathione defense in non-parenchymal cells. Semin Liver Dis 1998;18:403-13. [ Links ]
12. Beutler E, Gelbart T, Kondo T, et al. The molecular basis of a case of gamma-glutamylcysteine synthetase deficiency. Blood 1999;94:2890-4. [ Links ]
13. Madalosso C, de Souza NF Jr, Ilstrup DM, et al. Cytomegalovirus and its association with hepatic artery thrombosis after liver transplantation. Transplantation 1998;66:294-7. [ Links ]
14. Nakazawa Y, Chisuwa H, Mita A, et al. Life-threatening veno-occlusive disease after living-related liver transplantation. Transplantation 2003;75:727-30. [ Links ]
15. Mori T, Shimizu T, Yamazaki R, et al. Altered metabolism of tacrolimus in hepatic veno-occlusive disease. Transpl Int 2005;18:1215-7. [ Links ]
16. Barraclough KA, Isbel NM, Johnson DW, et al. Once- versus twice-daily tacrolimus. Are the formulations truly equivalent? Drugs 2011; 71:1561-77. [ Links ]
![]() Correspondence:
Correspondence:
Luis M. Marín-Gómez.
Liver Transplantation Unit.
Hospital Universitario Virgen del Rocío.
Avda. Manuel Siurot, s/n. 41013 Sevilla, Spain
e-mail:
luism.marin.sspa@juntadeandalucia.es
Received: 09-04-2014
Accepted: 20-07-2014











 text in
text in