Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.107 no.11 Madrid nov. 2015
Reasons for initiation of proton pump inhibitor therapy for hospitalised patients and its impact on outpatient prescription in primary care
Elena Villamañán1, Margarita Ruano1, Catalina Lara1, José Manuel Suárez-de-Parga2, Eduardo Armada3, Rodolfo Álvarez-Sala4, Ester Pérez1 and Alicia Herrero1
Departments of 1Pharmacy, 2Gastroenterology, 3Cardiology, and 4Neumology. Hospital Universitario La Paz. IdiPaz. Madrid, Spain
ABSTRACT
Background: Proton-pump-inhibitors are often prescribed unnecessarily in hospitals, which in turn induces their prescriptions after discharge.
Objective: To evaluate patients starting treatment with proton-pump-inhibitors during hospitalisation and proportion of inappropriate prescriptions. Patient risk factors and whether initiation in hospital induced their continuation in ambulatory care were also analyzed.
Methods: An observational, cross-sectional study in a tertiary hospital (1350 beds) was carried out on the first Tuesday in February 2015. Pharmacists screened admitted patients treated with proton-pump-inhibitors using an electronic prescription program (FarmaTools®-5.0). They also checked patients' home medications before admission by accessing a primary care program (Horus®). Authorized indications according to Spanish-Medicines-Agency and those recommended in Spanish-Clinical-Practice-Guidelines were considered appropriate. Hospital-medical-records were checked to know whether proton-pump-inhibitors were prescribed at discharge.
Results: Three hundred seventy nine patients were analysed. Two hundred ninety four of them were prescribed proton-pump-inhibitors (77.6%). Treatment was initiated during admission for 143 patients (48.6%, 95% CI: 42.8-54.5). Of them, 91 (63.6%, 95% CI: 55.2-71.5) were inappropriate, mainly due to its inclusion unnecessarily in protocols associated with surgeries or diseases (56 cases of 91, 61.5%). Additional inappropriate indications were surgical stress ulcer prophylaxis for surgeries without bleeding risks (19.8%) and polypharmacy without drugs that increase the risk of bleeding (18.7%). Of 232 discharge reports assessed, in 153 (65.9%, 95% CI: 59.5-72), proton-pump-inhibitor continuation was recommended, of them, 51 (33.3%) were initiated at admission.
Conclusion: In hospitalized patients there is a high prevalence of prescription of proton-pump-inhibitors unnecessarily. The superfluous use is often associated with the prescription of treatment protocols. Those treatments started in the hospital generally did not contribute to over-use existing primary care, most of them were removed at discharge.
Key words: Proton pump inhibitor. Hospitals. Overutilization.
Introduction
The release of the proton pump inhibitors (PPIs) in the early 80s represented a breakthrough in the treatment of digestive diseases related to gastric acid secretion. Currently, gastroesophageal reflux disease accounts for most of the approved indications for PPIs for long-term use, followed by prophylaxis against nonsteroidal anti-inflammatory drugs (NSAIDs) (1). The high prevalence of gastroesophageal reflux disease, its chronic nature and the concomitant presence of other risk factors such as the aging of the population, which is often associated with increased morbidity or treatments that increase the risk of bleeding such as anticoagulants or antiplatelets, contribute to a very high prescription of PPIs (2,3).
Due to the high prevalence of diseases that leads to PPI prescriptions, in recent years PPIs have become one of the most commonly used medicines in the developed world (4). Spain, for example, accounted for nearly 5% of global consumption of all medicines in 2010, making them in higher levels of pharmaceutical expenditure. However, Spain has a higher rate of prescription of PPIs than other neighbouring countries, which does not seem justified by our clinical needs (5).
PPIs have side effects that can have serious consequences for patients. Long-term use has been associated with hypergastrinemia and an increased risk of community-acquired pneumonia or vitamin B12 deficiency in the elderly and in patients with Zollinger-Ellison syndrome (6). Several epidemiological studies have shown associations between bone fractures and PPI use, mainly in long-term treatments at high doses (7,8). However, despite these adverse effects, their prevalence is not high and PPIs are considered safe drugs.
On the other hand, studies conducted on the use of PPIs in recent years have consistently found over-prescription. Clinicians often have a mistaken idea of the need for PPI prescriptions as gastric protectors for patients treated with polypharmacy, regardless of gastrolesive properties of the concomitant drugs, which generates a large number of unnecessary prescriptions for PPIs. The elderly, who generally have more comorbidity, are often treated with numerous drugs, thus they are particularly susceptible to inappropriate prescription of PPIs (9-11). Over-prescription is even greater in healthcare transitions. Many PPI treatments are initiated in hospital, sometimes without justification, and often the prescription remains after discharge from primary care, thus exposing patients to potential adverse effects and generating avoidable costs to the healthcare system.
Objective
We developed the following study, whose hypothesis was that there is an unnecessary prescription of PPIs in hospitals, which in turn induces unnecessary PPI prescriptions after discharge. The main objective of the study was to evaluate the proportion of patients starting treatment with a PPI during hospitalisation and whether the prescription was appropriate or inappropriate. Additional objectives were to evaluate the clinical and pharmacological risk factors and whether PPI initiation in hospital induced the prescription of PPIs in ambulatory care. The PPIs used, medical specialties involved and the demographic data of hospitalised patients on PPIs were also assessed.
Methods
This was a descriptive, observational, cross-sectional study in a tertiary hospital of 1,350 beds. Pharmacotherapy prescribed to inpatients is registered in an electronic prescription program (FarmaTools® version 5.0), which is used by using clinicians to prescribe drugs to around 850 patients daily. The study was carried out on the first Tuesday in February of 2015. On this day, using this program, pharmacists screened all admitted patients who were being treated with a PPI.
To determine whether or not this treatment had been initiated in hospital, pharmacists checked hospital clinical medical records and the patients' home medications by accessing a primary care program (Horus®).
In those cases in which a PPI was prescribed at admission, pharmacists assessed the indication via the electronic prescription program, medical history record or by contacting the attending clinician.
Inclusion criteria
Patients who were on a PPI that was available in the hospital (omeprazole, pantoprazole or esomeprazole) the day the study was carried out; patients admitted to the following adult wards: Oncology, Internal Medicine, Pulmonology, Neurology, Cardiology, and General Surgery, Maxillofacial Surgery, Urology, Vascular Surgery, Neurosurgery and Traumatology; patients admitted to critical care units, Gastroenterology, the children's hospital and the maternity hospital were excluded.
Primary and secondary endpoints
The primary outcome variable measured was the number of patients who began treatment with a PPI at admission and whether this prescription was appropriate or not.
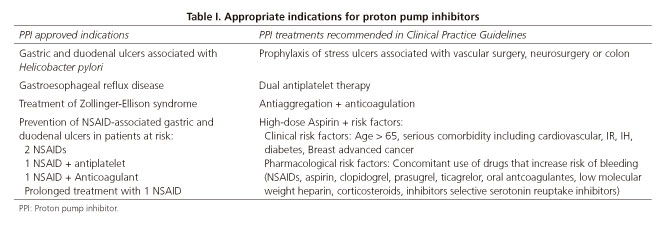
Approved indications according to the Spanish Agency of Medicines and Medical Devices (product characteristics) (12) and PPI treatments recommended in Clinical Practice Guidelines (CPG) (13-17) were considered appropriate (Table I). The rest were considered inappropriate and unnecessary prescriptions.
Secondary outcomes measured were clinical and pharmacological risk factors (13-17): History of a previously complicated ulcer, older than age 65, serious co-morbidity (cardiovascular, diabetes, renal or hepatic), smoker, history of gastrointestinal disease and concomitant use of drugs that increase risk of bleeding, demographic variables, available PPIs prescribed (omeprazole, pantoprazole, esomeprazole) and whether the PPI was prescribed at discharge and for how long.
The study was approved by the Clinical Research Ethics Committee of hospital.
Statistical analysis
Data management
A database was designed to reflect the Case Report Form's content, in which a data entry matrix with possible ranges or values was established, along with the various consistency rules between variables. The quality of information received through exploratory analysis aimed at detecting discrepancies in the values, out-of-range values or missing values. Exploratory analysis also provided information on the distribution of the main variables to be studied and provided guidance on possible transformations.
General considerations
The information included was: The mean, standard deviation, median, maximum, minimum and 25% and 75% quartiles. For categorical data, the frequency distributions (absolute and relative) were presented. In addition, the 95% confidence intervals were calculated, where appropriate. The statistical analysis was carried out using SAS 9.1 (SAS Institute Inc., Cary, NC, USA).
Sample size considerations
The sample size was calculated based on a pilot study in which a prevalence of PPI prescriptions was detected during the admission of 81.2%. It is estimated that in a cross section, the characteristics (secondary endpoints) of 400 patients could be analyzed and the percentages of each error could be estimated with an accuracy of at least 95%. According to data available from the Pharmacy Department, each patient admitted to the units studied receives approximately 10 medications daily.
Results
A total of 379 patients admitted were analysed. Some 294 of them were prescribed a PPI (77.6%). Treatment was initiated during admission for 143 patients (48.6%, 95% CI: 42.8-54.5), the remainder had started treatment earlier and it was part of their usual medication.
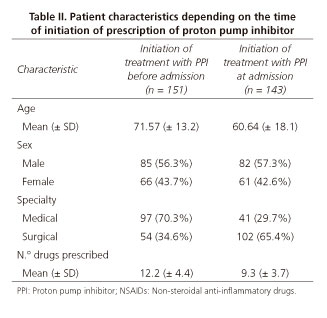
The characteristics of the patients receiving PPIs varied depending on the time from initiation of the prescription, whether it occurred at admission or was previously prescribed in primary care (Table II).
Of the three PPIs available at the hospital, 51% (n = 150) of the prescriptions corresponded to omeprazole, 48.3% (n = 142) to pantoprazole and 0.7% (n = 2) to esomeprazole. According to the prescribers, 55.9% of the PPIs were selected mainly due to a treatment protocols associated with a surgical procedure or pathology used in the hospital and 37.5% were selected without a specific reason. We also analysed the risk factors for gastric bleeding among the patients treated with PPIs (Table III). In terms of specialties, vascular surgery/neurosurgery, traumatology and neurology prescribed the most PPIs (Table IV).
Focusing on the main objective of our study, the reasons for PPI prescription were stratified into three categories: Approved indication when included in the product characteristics (12), unapproved but recommended by CPG (13-17) and unapproved and not recommended by CPG (13-17). The first two were considered appropriate indications and the last one inappropriate.
We found that of the 143 patients who were prescribed a PPI during hospitalisation, these prescriptions were inappropriate in 91 cases (63.6%, 95%CI: 55.2-71.5). The remaining PPIs prescriptions were considered appropriate, 26 of which (18.2%, 95% CI: 12.2-25.5) were prescribed for an approved indication and 26 of which (18.2%, 95% CI: 12.2-25.5) for an unapproved indication but were recommended by CPG. Figure 1 shows the proportion of appropriate and inappropriate indications.
The most inappropriate uses were made when a PPI was prescribed because of its inclusion unnecessarily in a protocol associated with a surgery, procedure or disease. Thus, of 91 cases of inappropriate prescription of PPIs, in 56 cases (61.5%), the prescription was induced by indication of any of these order sets used by the specialties included in the study. Among the various protocols that are commonly used in hospital, 36 correspond to the specialties included in the study, many of which unnecessarily include a PPI. As shown in figure 2, the specialty of general surgery and urology used a high number of PPIs.
Additional inappropriate indications were surgical stress ulcer prophylaxis for surgeries without bleeding risk, which occurred in 18 of 91 patients (19.8%) and polypharmacy without drugs that increase the risk of bleeding, which occurred in 17 of the patients (18.7%). Moreover, we detected a greater proportion of inappropriate prescriptions in surgical specialties than in medical specialties (73.6% vs. 26.4%), mainly in traumatology and general surgery (Table V).
We observed that most patients receiving PPIs for approved indications according to the product characteristics (12) were associations between an NSAID and another drug that increased the risk of bleeding. In 26 cases there were 24 appropriate indications (92.3%). Among the drugs that increased the risk of bleeding, low molecular weight heparins were the most frequently medicines associated with NSAIDs: 19 cases out of 24 (79.2%). As for unapproved but recommended in the CPG, the main indication was prophylaxis for surgical stress ulcers in surgeries with high bleeding risk, which occurred in 9 of the 26 cases (34.6%), and the association of an anticoagulant with antiplatelet drugs in 7 patients (26.9%).
We also found differences between the PPI prescribed in the hospital and their appropriate and inappropriate indications (Table VI).
Finally, access to discharge reports on patients included in the study allowed us to analyse whether or not PPI prescriptions continued after discharge. Unfortunately, of the 294 patients studied, we were unable to access reports on 62 patients (21.1%) because they were not available either in the hospital clinical records or in primary care, thus we could only evaluate reports on 232 patients.
Among the 232 discharge reports evaluated, in 153 (65.9%, 95% CI: 59.5-72) continuation of PPI treatment was recommended, of which 102 (66.7%) were patients receiving this treatment before admission, while 51 (33.3%) started this treatment at admission.
We detected that of 91 cases of inappropriate PPI prescriptions at admission, 29 (31.9%) were continued after discharge. Omeprazole and pantoprazole were prescribed at discharge almost equally (42.5% vs. 39.9%) and 16.3% of the PPIs were continued implicitly, due to the indication of maintaining their usual treatment. Only two reports recommended continuing esomeprazole treatment (1.3%).
The duration of treatment in the discharge reports was not usually specified (78.3%). Those reports in which the duration of treatment was specified mainly corresponded to patients whose onset of treatment with PPIs occurred at admission (75%). A 25% of those discharge reports on patients who had already started treatment before admission and continued after discharge made no reference to treatment duration.
Discussion
The prescription of PPIs in hospitalised patients is very high, in agreement with other authors (18), according to our study almost 80% of patients admitted received this treatment. Moreover, the number of patients receiving these treatments prior to admission was similar to those who started treatment at admission (51.4% vs. 48.6%).
Most of the PPIs prescribed by medical specialties were part of the patient's home medicines (70.3%). In surgical specialties, most prescriptions began during hospitalisation (65.4%) usually because PPIs were included in protocols associated with surgical procedures. Other studies, such as ours, have revealed an over-use of this type of drugs in hospitals (19-22).
Although these drugs are similar to each other in terms of efficacy and safety, they have important differences in their costs (5). However, this aspect seemed not to be taken into account when clinicians prescribed them. When asked about the reason for selecting one PPI or another, most prescribed the specific PPI included in a treatment protocol used in the hospital or they chose between the three available (omeprazole, pantoprazole and esomeprazole) without any specific criteria.
Although costs per day of treatment with omeprazole in Spain are significantly lower (5) than for the other PPIs, our results showed a similar rate of omeprazole and pantoprazole prescription (around 50%) and much a much lower rate of prescription of esomeprazole which were only prescribed when patient needed drug administration by nasogastric tube.
Most of the patients studied presented no comorbidity or risk factors that increased bleeding risk. However we found a high rate of PPI prescriptions concomitantly with other drugs that increase bleeding risk (84.7%), mainly low molecular weight heparin and antiplatelet agents (59.2% and 54%, respectively).
This high proportion would be explained by the high rate of prescription of PPIs in specialties such as orthopaedics, vascular surgery and neurology. However, although some authors recommend PPIs for concomitant prescription (22), others do not, such as when the antiplatelet is clopidogrel, because its interaction with PPIs can increase the risk of atherosclerotic complications (22,23). In agreement with other authors (1,19,24), treatment with PPIs is frequently initiated inappropriately on hospital admission. According to our study, more than half of the PPIs were prescribed for inappropriate indications (63.6%). In agreement, Walker et al. (25) found 67% inappropriate indications. In Spain, Martín-Echevarría et al. (26) detected a similar proportion (68.1%) and Batuwitage et al. (27), in England, observed inappropriate prescriptions for 56% of patients. It is noteworthy that in a high percentage (61.5%) of cases, the indication was induced because the PPIs were unnecessarily included in a treatment protocol.
Most of these protocols are associated with surgical procedures, which may explain in part the higher proportion of inappropriate prescription in surgical specialties compared with medical specialties (73.6% vs. 26.4%). As Sánchez Cuén et al. from Mexico (1), we found the most common inappropriate indications to be stress ulcer prophylaxis for surgery with a low risk of bleeding or polypharmacy that did not include drugs that increase bleeding risk. For PPIs with approved indications, the main reason for prescription was the use of NSAIDs concomitantly with other drugs (88.9%), primarily low molecular weight heparins (79.2%). Other studies agree with this finding (19). Regarding unapproved but recommended by CPG (13-17) we observed that the most frequent indication was related to stress ulcer prophylaxis for surgery (34.6%) and the association with anticoagulants and antiplatelets (26.9%). In our study, pantoprazol was improperly prescribed more often than the other available PPIs in the hospital (71%), probably because it was included in most treatment protocols used in the hospital for various surgical procedures.
In our study we found a low impact on primary care of PPIs prescriptions started in the hospital. Thus, in terms of continuing treatment after discharge, we detected that in more than half of discharge reports, physicians decided to maintain PPI treatments. However, most patients were already admitted on this treatment, thus only a third of the patients who started treatment during hospitalisation continued after discharge. Moreover, contrary to what is revealed in other studies (27), in our results the proportion of patients whose prescription of PPIs was inappropriate in the hospital was low (31.9%).
As for how long need the patients to take the PPI, in line with other studies (27), we found that few prescriptions specified treatment duration. This finding is relevant because it can contribute to the use in primary care of these drugs in the long term, with the accompanying risks of adverse effects and economic costs to the healthcare system. Unfortunately, there is a lack of guidelines or international consensus on PPI discontinuations (28).
Limitations
This study is limited by weaknesses inherent in a cross-sectional study. Other limitations may be that the treatment of patients who met inclusion criteria were not assessed for omitted prescriptions in electronic prescriptions program o handwritten PPI medication prescriptions in the administration nursing sheet. While this is an observational study, the sample of patients evaluated did not reach the number of patients initially estimated.
In hospitalized patients there is a high prevalence of prescription of PPIs unnecessarily. The superfluous prescription is often associated with the prescription of treatment protocols used in hospitals that include these drugs inappropriately. For inpatients treated with PPIs, concomitant treatment with low molecular weight heparins is the main risk factor of gastrointestinal bleeding. However, those treatments with PPIs started in the hospital generally did not contribute to over-use existing primary care and that most of them were removed at discharge.
On the other hand, patients whose prescription of a PPI began during hospitalization and continued after discharge, did so under an inadequate indication and also, in many cases it was not specified how long the patient should take them, which could perpetuate the treatment with a consequent increase in the risk of adverse effects and drug expenditure.
References
1. Sánchez Cuén JA, Irineo Cabrales AB, Bernal-Magaña G, et al. Indicaciones inadecuadas en el consumo crónico de inhibidores de bomba de protones en un hospital de México. Estudio transversal. Rev Esp Enferm Dig 2013;105:131-7. [ Links ]
2. Ponce J, Vegazo O, Beltrán B, et al. Prevalence and factors associated with gastro-oesophageal reflux disease (GORD) in Spain. Aliment Pharmacol Ther 2006;23:175-84. DOI: 10.1111/j.1365-2036.2006.02733.x. [ Links ]
3. Ponce J, Esplugues JV. Racionalizar el uso de IBP: una asignatura pendiente. Rev Esp Enferm Dig 2013;105:21-4. DOI: 10.4321/S1130-01082013000300001. [ Links ]
4. Bashford JN, Norwood J, Chapman SR. Why are patients prescribed proton pump inhibitors? Retrospective analysis of link between morbidity and prescribing in the General Practice Research Database. BMJ 1998;317:452-6. DOI: 10.1136/bmj.317.7156.452. [ Links ]
5. García del Pozo J. Estudio de utilización de antiulcerosos en España (2000-2008). IT del Sistema Nacional de Salud 2009;33(2). [ Links ]
6. Thomson A, Sauve M, Kassam N, et al. Safety of the long-term use of proton pump inhibitors. World J Gastroenterol 2010;16:2323-30. DOI: 10.3748/wjg.v16.i19.2323. [ Links ]
7. Yang YX, Lewis JD, Epstein S, et al. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA 2006;296:2947-53. DOI: 10.1001/jama.296.24.2947. [ Links ]
8. Eom CS, Park SM, Myung SK, et al. Use of acid-suppressive drugs and risk of fracture: A meta-analysis of observational studies. Ann Fam Med 2011;9:257-67. DOI: 10.1370/afm.1243. [ Links ]
9. Pasina L, Nobili A, Tettamanti M, et al. Prevalence and appropriateness of drug prescriptions for peptic ulcer and gastro-esophageal reflux disease in a cohort of hospitalized elderly. Eur J Intern Med 2011;22:205-10. DOI: 10.1016/j.ejim.2010.11.009. [ Links ]
10. Cahir C, Fahey T, Teeling M, et al. Potentially inappropriate prescribing and cost outcomes for older people: A national population study. Br J Clin Pharmacol 2010;69: 543-52. DOI: 10.1111/j.1365-2125.2010.03628.x. [ Links ]
11. Hanzat H, Sun H, Ford JC, et al. Inappropriate prescribing of proton pump inhibitors in older patients. Effects on an educational strategy. Drugs Aging 2012;29:681-90. DOI: 10.2165/11632700-000000000-00000. [ Links ]
12. Product Characteristics. Spanish Agency of Medicines and Medical Devices. Available at: http://www.aemps.gob.es/cima/fichasTecnicas.do?metodo=detalleForm (accessed 08/05/2015). [ Links ]
13. Grupo de trabajo Sector Zaragoza I Salud. Empleo de los inhibidores de la bomba de protones en la prevención de gastropatías secundarias a fármacos. Guía de Práctica Clínica. Enero 2012. Available at: http://www.guiasalud.es/GPC/GPC_509_IBP_gastropatias_2rias_fcos_completa.pdf. [ Links ]
14. Vallés R, Franzi A, Ferro JJ. Condiciones clínicas y terapéuticas que requieren gastroprotección. FMC 2014;21:528-33. [ Links ]
15. National Institute for Health and Clinical Excellence. Guidance on the use of cyclo-oxygenase (Cox) II selective inhibitors, celecoxib, rofecoxib, meloxicam and etodolac for osteoarthritis and rheumatoid arthritis. July 2001. http://www.nice.org.uk/Guidance/TA27/Guidance/pdf/English (accessed 08/07/2015). [ Links ]
16. Lanza FL, Chan FK, Quigley EM. Practice Parameters Committee of the American College of Gastroenterology. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol 2009;104:728-38. DOI: 10.1038/ajg.2009.115. [ Links ]
17. Bhatt DL, Scheiman J, Abraham NS, et al. ACCF/ACG/AHA 2008 expert consensus document on reducing the gastrointestinal risks of antiplatelet therapy and NSAID use: A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents. Circulation 2008;118:1894-909. DOI: 10.1161/CIRCULATIONAHA.108.191087. [ Links ]
18. Solís-García-del-Pozo J, Pérez-Pinar M, Fernández-Jiménez P, et al. Inappropriate use of proton pump inhibitors, guidelines and clinical research. Rev Esp Enferm Dig 2013;105:373. DOI: 10.4321/S1130-01082013000600016. [ Links ]
19. Ahrens D, Chenot JF, Behrens G, et al. Appropriateness of treatment recommendations for PPI in hospital discharge letters. Eur J Clin Pharmacol 2010;66:1265-71. DOI: 10.1007/s00228-010-0871-9. [ Links ]
20. Ramirez E, Lei SH, Borobia AM, et al. Overuse of PPIs in patients at admission, during treatment, and at discharge in a tertiary Spanish hospital. Curr Clin Pharmacol 2010;5:288-97. DOI: 10.2174/157488410793352067. [ Links ]
21. Mat Saad AZ, Collins N, Lobo MM, et al. Proton pump inhibitors: A survey of prescribing in an Irish general hospital. Int J Clin Pract 2005;59:31-4. DOI: 10.1111/j.1742-1241.2004.00298.x. [ Links ]
22. Niklasson A, Bajor A, Bergendal L. Overuse of acid suppressive therapy in hospitalised patients with pulmonary diseases. Respir Med 2003;97:1143-50. DOI: 10.1016/S0954-6111(03)00187-2. [ Links ]
23. Ho PM, Maddox TM, Wang L, et al. Risk of adverse outcomes associated with concomitant use of clopidogrel and proton pump inhibitors following acute coronary syndrome. JAMA 2009;301:937-44. DOI: 10.1001/jama.2009.261. [ Links ]
24. Durand C, Willet K, Desilets A. Proton Pump Inhibitor use in hospitalized patients: Is overutilization becoming a problem? Clinical Medicine Insights: Clin Med Insights Gastroenterol 2012;5:65-76. DOI: 10.4137/CGast.S9588. [ Links ]
25. Walker NM, McDonald J. An evaluation of the use of proton pump inhibitors. Pharm World Sci 2001;23:116-7. DOI: 10.1023/A:1011278030001. [ Links ]
26. Martín-Echevarría E, Pereira A, Torralba M, et al. Evaluación del uso de los inhibidores de la bomba de protones en un servicio de medicina interna. Rev Esp Enferm Dig 2008;100: 76-81. DOI: 10.4321/S1130-01082008000200003. [ Links ]
27. Batuwitage BT, Kingham JG, Morgan NE, et al. Inappropriate prescribing of proton pump inhibitors in primary care. Postgrad Med J 2007;83:66-8. DOI: 10.1136/pgmj.2006.051151. [ Links ]
28. Haastrup P, Paulsen M, Begtrup L, et al. Strategies for discontinuation of proton pump inhibitors: A systematic review. Fam Pract 2014;6:625-30. DOI: 10.1093/fampra/cmu050. [ Links ]
![]() Correspondence:
Correspondence:
Elena Villamañán.
Departement of Pharmacy.
Hospital Universitario La Paz. IdiPaz.
Paseo de La Castellana, 261. 28046 Madrid
e-mail: evillabueno@telefonica.net
Received: 07-06-2015
Accepted: 27-07-2015