Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.107 no.11 Madrid nov. 2015
The intestinal barrier function and its involvement in digestive disease
Función barrera intestinal y su implicación en enfermedades digestivas
Eloísa Salvo-Romero1, Carmen Alonso-Cotoner1,2, Cristina Pardo-Camacho1, Maite Casado-Bedmar1 and María Vicario1,2
1 Neuro-Immuno-Gastroenterology Laboratory. Unit of Physiology and Digestive Physio-Pathology. Vall d'Hebron Institut de Recerca. Department of Digestive Diseases. Hospital Universitari Vall d'Hebron. Universitat Autònoma de Barcelona. Barcelona, Spain.
2 Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas (CIBERehd). Spain
Support: Fondo de Investigación Sanitaria, Instituto de Salud Carlos III, Subdirección General de Investigación Sanitaria, Ministerio de Economía y Competitividad: FI12/00254 (ES-R); PI12/00314 (CA); CP10/00502 & PI13/00935 (MV); Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas (CIBERehd): CB06/04/0021 (CA, MV).
ABSTRACT
The gastrointestinal mucosal surface is lined with epithelial cells representing an effective barrier made up with intercellular junctions that separate the inner and the outer environments, and block the passage of potentially harmful substances. However, epithelial cells are also responsible for the absorption of nutrients and electrolytes, hence a semipermeable barrier is required that selectively allows a number of substances in while keeping others out. To this end, the intestine developed the "intestinal barrier function", a defensive system involving various elements, both intra- and extracellular, that work in a coordinated way to impede the passage of antigens, toxins, and microbial byproducts, and simultaneously preserves the correct development of the epithelial barrier, the immune system, and the acquisition of tolerance against dietary antigens and the intestinal microbiota. Disturbances in the mechanisms of the barrier function favor the development of exaggerated immune responses; while exact implications remain unknown, changes in intestinal barrier function have been associated with the development of inflammatory conditions in the gastrointestinal tract. This review details de various elements of the intestinal barrier function, and the key molecular and cellular changes described for gastrointestinal diseases associated with dysfunction in this defensive mechanism.
Key words: Intestinal barrier. Tight junctions. Intestinal immune.
RESUMEN
La superficie de la mucosa del tracto gastrointestinal está revestida de células epiteliales que establecen una barrera efectiva, mediante uniones intercelulares, entre el medio interno y el medio externo, impidiendo el paso de sustancias potencialmente nocivas. Sin embargo las células epiteliales también son responsables de la absorción de nutrientes y electrolitos, por lo que se requiere una barrera semipermeable que permita el paso selectivo a ciertas sustancias, mientras que evite el acceso a otras. Para ello, el intestino ha desarrollado la "función barrera intestinal", un sistema defensivo compuesto por diferentes elementos, tanto extracelulares como celulares, que actúan de forma coordinada para impedir el paso de antígenos, toxinas y productos microbianos y, a la vez, mantiene el correcto desarrollo de la barrera epitelial, el sistema inmunitario y la adquisición de tolerancia hacia los antígenos de la dieta y la microbiota intestinal. La alteración de los mecanismos que componen la función barrera favorece el desarrollo de respuestas inmunitarias exageradas, y, aunque se desconoce su implicación exacta, la alteración de la función barrera intestinal se ha asociado al desarrollo de enfermedades inflamatorias en el tracto digestivo. En esta revisión se detallan los diferentes elementos que componen la función barrera intestinal y las alteraciones moleculares y celulares más características descritas en enfermedades digestivas asociadas a la disfunción de este mecanismo de defensa.
Palabras clave: Barrera intestinal. Uniones estrechas. Sistema inmunitario intestinal.
Abbreviations
IBD, inflammatory bowel disease;
GI, gastrointestinal;
GALT, gut-associated lymphoid tissue;
IFN-γ, interferon γ;
Ig, immunoglobulin;
IL, interleukin;
JAM, junctional adhesion molecules;
MLCK, myosin light-chain kinase;
NOD, nucleotide-binding oligomerization domain receptor;
PAMP, pathogen-associated molecular pattern;
PRR, pattern recognition receptor;
IBS, irritable bowel syndrome;
CNS, central nervous system;
ENS, enteric nervous system;
TGF-β, transforming growth factor β;
Th, T helper cell;
TLR, toll-like receptor;
TNF-α, tumor necrosis factor α;
AJ: adherens junction;
TJ, tight junction;
VIP, vasoactive intestinal peptide;
ZO, zonula occludens.
Introduction
The human body is exposed to potentially harmful substances and infectious agents, which threaten the balance between health and disease, on a daily basis. The gastrointestinal (GI) tract is one of the regions subjected to greater antigen loads because of its role and its having the largest contact surface with the outer environment, with an approximate surface area of 250 m2 (1). To ensure inner homeostasis the GI tract has a digestive role - nutrient digestion and absorption, water and electrolyte transport, and water and protein secretion into the intestinal lumen. Also, a defensive role is needed to prevent potentially harmful substances, including pathogens, antigens and proinflammatory factors, from reaching the inner environment from the intestinal lumen, while allowing the selective passage of substances favoring the development of the intestinal immune system and immune tolerance (2). In fact, the intestinal mucosa is particularly adapted to colonization by commensal bacteria that play a role in digestive processes and decisively influence the development and function of the intestinal immune system (3). Both these functions, digestive and defensive, take place based on the peculiar anatomy of the intestinal mucosa and, most particularly, on the so-called "intestinal barrier function", where various immune and non-immune mechanisms converge and act in a coordinated manner to ensure its functioning (4). Changes in the defense mechanisms making up this barrier function favor the passage of normally excluded luminal substances to the inner body environment, thus giving rise to exaggerated immune responses that, in turn, may amplify the barrier's dysfunction and perpetuate the inflammatory condition. While its exact involvement remains unknown, changes in intestinal barrier function have been associated with the development of inflammatory diseases in the GI tract (celiac disease, inflammatory bowel disease, irritable bowel syndrome) and also extradigestive conditions such as schizophrenia, diabetes and sepsis, among others (5,6). This paper describes the elements that the intestinal barrier function is comprised of under homeostasis and during GI conditions associated to barrier dysfunction.
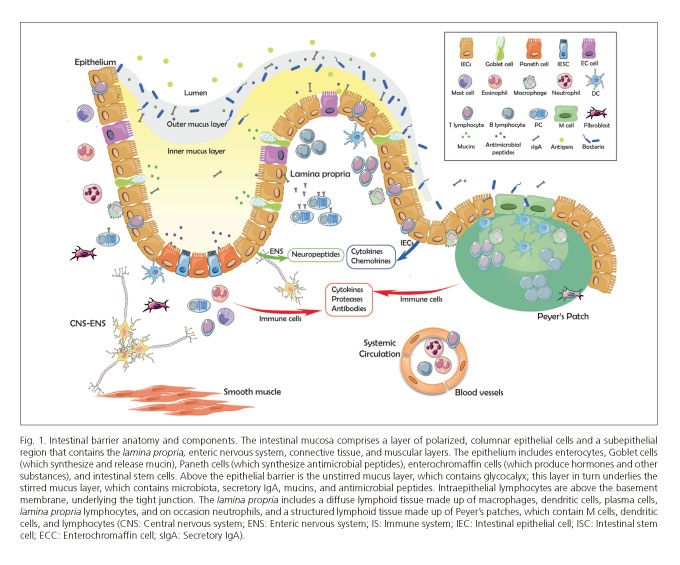
Intestinal barrier anatomy
The elements that make up the intestinal barrier are categorized into various protection levels, which are further split into extracellular and cellular components according to their nature and anatomical location (Fig. 1).
Extracellular components
The first line of defense in the gastrointestinal tract is found at the intestinal lumen, where microorganisms and antigens are degraded in a nonspecific fashion by pH and gastric, pancreatic and biliary secretions. Digestive enzymes, primarily proteases, lipases, amylases and nucleases, exert a toxic action on microorganisms via the destruction of their cell wall (7), and thus manage to initially clear a major portion of dietary organisms. The intestinal epithelium is lined with a microclimate comprising mucus, water and glycocalyx layer approximately 100 microns in thickness, which is primarily secreted by Goblet cells with hydrophobic and surfactant properties that prevent enteric bacteria from adhering to the intestinal epithelium (8). Within the mucous layer itself an outer layer (stirred mucus layer) may be distinguished, which contributes to the retention of antibacterial peptide-rich mucus and prevents mucosal adhesion and subsequent transepithelial invasion by microorganisms (9,10). This layer contains secretory immunoglobulin A (sIgA) (11), synthesized by plasma cells in the lamina propria, and antimicrobial products secreted by Paneth cells, including phospholipids, negatively charged mucins and peptides active against bacteria, yeasts, fungi, viruses and even tumor cells, such as trefoil factor family (TFF) peptides, cathelicidins, ribonucleases and defensins (9). Antimicrobial peptides induce bacterial lysis following the formation of membrane pores, although some of them, including cryptidines 2 and 3, can induce increased water secretion into the intestinal lumen, thus washing away bacteria on the epithelial surface (12,13) (Fig. 2). Defensins also play a role in microbiota modulation, and consequently in the setup of the adaptive immune response (14). Adhered to the epithelium is the denser, non-stirred mucus layer, also called glycocalyx, which facilitates nutrient absorption, preserves epithelial hydration, and protects the epithelial lining from luminal shear stress and digestive enzymes (15). Glycocalyx also plays a role in epithelial renewal and differentiation, as well as in the maintenance of oral tolerance, thus limiting intestinal antigen immunogenicity via tolerogenic signaling (16). Furthermore, chlorine and water secretion into the bowel lumen primarily by enterocytes blocks bacterial colonization and slows down antigen translocation to the lamina propria via a dilutional effect on intestinal contents (17). Finally, peristaltism, as induced by the intestine's muscle layers, evacuates luminal contents and reduces retention time, hence shortening the presence of potential toxic or pathogenic substances within the intestinal lumen.
Cellular components
Cells may act in either a specific or a nonspecific manner. The intestinal flora or microbiota is found in the outermost portion, and represents a key component of the intestinal barrier influencing epithelial barrier metabolism, proliferation and maintenance (18). The commensal flora, in addition, restricts colonization by pathogens by competing for nutrients and the ecological niche, by modifying pH, and by releasing antimicrobial substances that allow inter-species communication and optimizing the amount of beneficial organisms (18). The intestinal microbiota offers the host other crucial functions, including nutrient acquisition and energy regulation (19), and also influences processes such as inflammatory response, epithelial repair, and angiogenesis (20). The intestinal epithelium has a single layer of specialized, polarized epithelial cells that is continuously renewed every 3 to 5 days. Intestinal epithelial pluripotent stem cells reside deep within the crypts (Lieberkühn crypts) and generate cells that migrate to the upper villi where final differentiation takes place (21). Although most cells in the monolayer are enterocytes (around 80%), the diverse roles played by the intestinal epithelium derive from the presence of other cells specialized in mucus secretion (Goblet cells), defensin secretion (Paneth cells), hormone and neuropeptide secretion (enterochromaffin cells), and antigen uptake from the bowel lumen, the latter on the surface of lymphoid aggregates (M-cells) (22). Enterocytes are key elements in the epithelial lining, and are adapted to exert digestive, metabolic, and barrier physical integrity maintenance dunctions. They also play a role in the development of immune activity as they express receptors involved in the innate immune response (23), act as non-professional antigen-presenting cells, and release several cytokines and chemokines, including thymic stromal lymphopoietin, transforming growth factor β1 (TGF-β1) (24), interleukin (IL) 25 (25), a proliferation inducing ligand (APRIL) and B-cell activating factor (BAFF) (26,27), thus being involved in leukocyte recruitment and activation, and in the regulation of local immune responses. The subepithelial region includes the lamina propria, which harbors immune cells, the enteric nervous system, and connective tissue. GI tract immune cells make up the so-called gut-associated lymphoid tissue (GALT), which comprises two compartments: Organized GALT, which induces immune responses, and diffuse GALT, effector of immune responses. Organized GALT includes lymphoid structures, mainly lymphoid follicles, Peyer's patches, and mesenteric nodes (28). The epithelium lining Peyer's patches contains M-cells, specialized epithelial cells that play a role in monitoring the intestinal lumen and in maintaining the intestinal barrier function, as its unique physical characteristics, including microfold formation and reduced mucous layer, facilitate antigen and luminal organism uptake and their presentation to underlying immune cells (29,30). In turn, diffuse GALT includes two WBC populations on either side of the basement membrane. Intraepithelial lymphocytes, mostly CD8+ T-cells, are found among epithelial cells, above the basement membrane, and their primary function is monitoring and responding to bacteria and other luminal antigens. Lamina propria lymphocytes share their dwelling with eosinophils, dendritic cells, mast cells, and macrophages mainly. These lymphocytes are a heterogeneous population with 50% plasma cells and 30% T-cells, which in turn may be subcategorized according to their cytokine secretion pattern (31). Connective tissue is the tissue adjacent to the epithelium, and the abode of immune cells, neurons, blood vessels, and fibroblasts. Fibroblasts maintain the extracellular matrix, primarily by secreting collagen and metalloproteinases, and play a key role in the proliferation of the intestinal epithelium in response to liver cell growth factor (32), thus actively contributing to the maintenance of the intestinal barrier function.
Finally, the central nervous system (CNS) and enteric nervous system (ENS), coordinate digestive functions and intestinal homeostasis maintenance via the release of neurotransmitters and, indirectly, via neuro-immune interactions. The ENS constitutes an intertwined network of neurons and glial cells that gather in the ganglia at two major plexuses: The myenteric (Auerbach's) plexus and submucosal (Meissner's) plexus. The ENS is in close contact with intestinal epithelial cells and neuroendocrine cells, modulates inflammatory responses, and collaborates with the immune system's response to pathogens. The ENS includes sensorial neurons, interneurons and motor neurons, which control peristaltism, local blood flow changes, and fluid and electrolyte secretion (33). It also includes enteric glial cells that make up a large network across GI tract layers, and act as intermediaries in the processing of neurotransmission and enteric information (34). ENS involvement in the barrier function is key because of its control of both motor and secretory activities, and of microcirculation and immune actions, which grants it a role in the monitoring of intestinal homeostasis. This communication takes place through chemical mediators such as neuropeptides, neurohormones, neurotransmitters, cytokines, chemokines, growth factors, and other regulatory molecules (35).
The intestinal epithelium
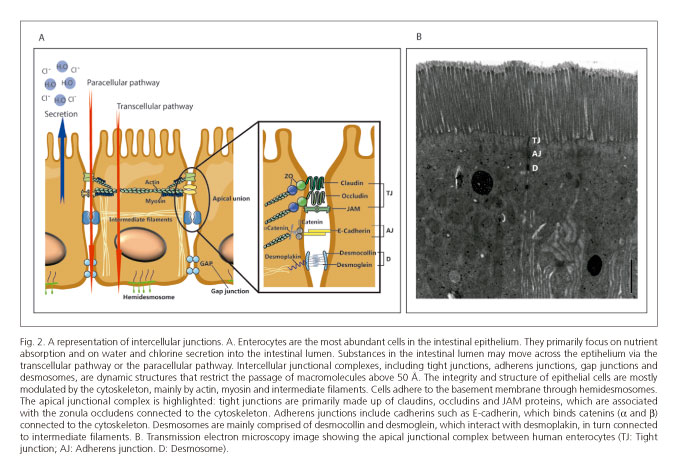
Epithelial cells make up a continuous polarized monolayer where individual cell membranes are interconnected and connected to the basement membrane by protein complexes that provide the epithelium with the structural integrity and cell activity needed to exert its specific functions. These intercellular junctions are classified into three functional groups - tight junctions, anchoring junctions and communicating junctions.
Tight junctions
The passage of small water-soluble molecules across the epithelium takes place through tight junctions, which seal spaces between epithelial cells. Tight junctions (TJs) are the most apical intercellular junctions and their function is key for barrier and epithelial polarity maintenance, limiting ion diffusion and luminal antigen (organisms and toxins) translocation from the apical to the basolateral membrane region (36,37). They are made of multiprotein complexes involving four classes of transmembrane proteins: Occludin, claudins, junctional adhesion molecules (JAM) and tricellulin (Fig. 2), and are predominantly expressed in epithelial and endothelial cells, but may also be found astrocytes, neurons, macrophages, dendritic cells and/or leukocytes (38,39).
- Occludin plays a role in the assembly and disassembly of TJs, and its membrane location is regulated by phosphorylation of Ser, Thr and Tyr residues. Highly phosphorylated occludin is seen in TJs whereas dephosphorylated occludin is found in the cytoplasm, hence changes in this protein's phosphorylation patterns may render TJs unstable and increase paracellular permeability (40,41).
- Claudins are the primary factor determinant of TJ barrier function - they control ion passage through the paracellular space (42) and are also regulated by specific Ser and Thr residue phosphorylation. These proteins conform channels with biophysical properties similar to ion channels, which preferentially permit the passage of specific ions (42). Claudin composition is significantly variable among intestinal segments because of their specific physiological function, with overall reduced permeability in distal gastrointestinal tract portions (43).
- JAMs are a subfamily of immune globulins expressed by epithelial and endothelial cells, as well as leukocytes and platelets. JAM proteins in epithelial TJs include JAM-A, JAM-C, CAR, ESAM, and JAM4, all of them laterally associated with other proteins in intercellular contacts in order to facilitate the assembly and formation of functional, polarized TJs (44). JAMs play a role in the regulation of intestinal permeability and inflammation (45).
- Tricellulin is found at intercellular contacts between three adjacent cells where it facilitates epithelial barrier stability and formation, and specifically seal epithelial cell sheets against macromolecules without significantly impairing ion permeability (46,47).
Transmembrane TJ proteins, claudins, occludin and JAMs, are linked to cytoskeletal actomyosin fibers by members of the zonula occludens protein family - ZO-1, ZO-2 and ZO-3. These proteins play a fundamental role in cellular permeability as well as in the regulation of adhesion, TJ formation and stabilization, and signal transmission from intercellular junctions to the inner cell in order to regulate processes such as cell migration (48).
Anchoring junctions
Anchoring junctions connect each cell's cytoskeleton to that of two neighboring cells or the extracellular matrix, which results in enduring structural units.
- Adherens junctions regulate adhesion between adjacent cells via transmembrane adhesion receptors and their actin-associated regulatory proteins. This connection between cell cytoskeletons occurs via transmembrane adhesion molecules in the cadherin and catenin superfamily, and their associated protein complexes, which link the cytoskeleton. They are necessary for TJ assembly and maintenance, and several regulatory proteins may affect this structural component, including growth factors (EGFR) and actin-regulating proteins (Rho, GTPases, myosin) (49).
- Desmosomes, mainly comprised of desmoglein, desmocollin and desmoplakin, are intercellular junctions that provide strong adhesion between cells even though they are dynamic structures whose adhesivity may change from high to low affinity states during processes such as embryo development and wound healing. By also linking intermediate filaments in the cytoskeleton of neighboring cells, they form a transcellular network that confers mechanical resistance to tissues and sustains cell morphology. Furthermore, they are signaling foci that play a role in various cell processes, including differentiation, proliferation and morphogenesis (50).
Communicating junctions
Communicating junctions (GAP junctions) allow communication between the cytoplasms of neighboring cells by setting up a channel through their membranes. They are comprised of 6 transmembrane proteins, called connexins, which mediate reciprocal ion and small molecule (> 1 KDa) exchange. Connexins are also considered to play a crucial role in the development, growth and differentiation of epithelial cells in addition to their role in TJs and adherens junctions (51), hence they play a relevant role in barrier function maintenance.
Intestinal immune system activation
The epithelial barrier, water and antimicrobial secretion, and intestinal motility restrict the passage of antigens and microorganisms from the intestinal lumen to the body's inner environment. However, these nonspecific mechanisms do not always suffice and a monitoring system, namely the immune system, is required to mount fast, coordinated responses. Thus the immune system, while acquiring tolerance to harmless antigens such as those from the diet or the commensal microbiota, targets harmful agents and remains constantly activated in a state called "physiological inflammation" (52). The first immune response to become activated is nonspecific and depends upon the innate immune system, widely represented in the gastrointestinal tract by epithelial cells, dendritic cells, macrophages, and natural killer (NK) cells. These cells recognize pathogen-associated molecular patterns (PAMPs) in specific components of bacteria, fungi and viruses, including pattern recognition receptors (PRRs) such as toll-like receptors (TLRs) and nucleotide-binding oligomerization domain (NOD) receptors, amongst others. These receptors have a dual function and, because of the polar nature of the intestinal epithelium, allow anatomical segregation; both in vitro and in vivo, they provide epithelial cells with differential responses to apical and basolateral stimulation (53,54). Under normal conditions, the apical activation of PRRs by commensal bacteria helps in the secretion of antibacterial substances and the maintenance of some tolerance to inflammation (55). When structural changes develop in the epithelial barrier, bacteria may come in and a proinflammatory response is required via the activation of basolateral PRRs and of PRRs on innate immune system cells (56). Dendritic cells constantly detect bacteria and other antigens, which they internalize in phagosomes and process for presentation using the class II major histocompatibility complex (MHC). This determines the development of both cellular and humoral immune responses by the adaptive immune system (57). Therefore they must differentiate between signals from pathogenic microorganisms and signals from commensal organisms and harmless antigens before mounting an adequate inflammatory response locally (58). PAMPs in symbiotic organisms are thought to possess less affinity for PRRs when compared to pathogenic organisms, or they might need the convergence of another pathogenic signal -such as toxins- to trigger an immune response (59). The innate immune system comprises several cell subpopulations, and activation depends on the cytokines present in their environment. However, specific subpopulations, including innate NKT cells, respond to glycolipids such as the intracellular lipids of apoptotic enterocytes released during inflammation, dietary glycolipids modified by non-physiological enzymes, and glycolipids from bacteria present in the intestinal lumen (60).
The adaptive immune system includes T and B lymphocytes, and induces specific, memory responses to certain antigens. Under homeostatic conditions, bacterial diversity remains balanced by complex mechanisms involving the production of various IgA repertoires, selected at germinal centers by antigens and T-cells. These IgA+ B-cells become plasma cells that secrete large amounts of IgA, which in the intestinal lumen coat bacteria in order to control their expansion or invasion beyond the epithelium (61). CD4+ T-cells or Th (T-helper) cells arrange themselves in the lamina propria and lymphoid follicles of both the small and large intestine, and both Th1 and Th17 effector cells may be found in the bowel under homeostatic conditions (62). Their proinflammatory properties are usually countered by T regulatory (Treg) cells expressing Foxp3, which help in keeping physiological inflammation under control. In response to different signals and stimuli, the adaptive immune system will develop an appropriate specific response. Lymphocytes responsible for these responses may be classified into various groups according to their cytokine profile: Th1, Th2, Th17, Th25 and/or Treg (63).
Intestinal permeability
Intestinal permeability may be defined as the ability of the mucosal surface to be penetrated by specific substances. The passage of nutrients and the absorption of water and ions through the intestinal epithelium take place as a result of both active (transporters) and passive (diffusion) processes between the intestinal lumen and the mucosa, whence they may reach the blood stream. Enterocytes are highly active in the transport role because of their ion channels, transporters and pumps at the apical and basolateral membrane. Net transport results from balanced absorption and secretion. It is a selective process using two major pathways - the paracellular and transcellular routes (Fig. 2).
- The paracellular route allows 85% of the total passive transepithelial flow of molecules across the space between two adjacent epithelial cells, and is regulated by tight junctions, which display differently sized pores to limit particle entry. This route is an effective barrier against luminal antigens, and a determinant of intestinal permeability (64).
- The transcellular route allows solute transportation across the enterocyte's membrane. Various mechanisms mediate the passage of molecules through the transcellular route. Smaller hydrophilic and lipophilic compounds diffuse by passive means through the enterocyte membrane's lipid bilayer. Furthermore, epithelial permeability is conditioned by active transport mechanisms as mediated by transporters and various endocytosis, transcytosis and exocytosis mechanisms for ions, amino acids, and specific antigens. Larger substances such as proteins and bacterial byproducts are captured by cells using endocytosis, and actively transported by vectorial transcytosis across the cytoplasm for subsequent processing and presentation, which is part of the intestinal immune response (65). Bacteria, viruses and other particles take advantage of these host entry mechanisms by endocytosis or phagocytosis, involving the binding of molecules to the cell membrane via receptors (66).
Intestinal permeability regulation
The intestinal barrier is no static structure but is regulated by various physiological, drug-related, and disease-related stimuli. Particle permeability depends on particle size, charge and nature. While permeability varies between proximal and distal regions, as well as between crypts and villi, molecular mechanisms regulating the passage of substances through the epithelium are similar along the bowel and include intercellular protein interactions, the actin cytoskeleton, endocytosis and intracellular signaling. Rapid permeability changes occur via the cytoskeleton and are regulated by myosin light chain kinase (MLCK) phosphorylation and TJ protein endocytosis (6,67). Longer-term changes involve TJ protein expression regulation, epithelial cell apoptosis, and the development of epithelial structural changes (68,69).
Barrier control results from interactions between the microbiota, epithelial cells, immune system, and ENS. Thus, for instance, under homeostatic conditions, apical PRR activation by commensal bacteria also promotes epithelial cell proliferation and survival (70). The immune system, via diverse cytokines such as tumor necrosis factor alpha (TNF-α), gamma-interferon (IFN-γ), IL-8 or IL-10, also regulates the intestinal barrier through TJ rearrangement (71). An increased or inappropriate cytokine profile will increase permeability (72,73). On the other hand, enteric neurons play a role in the management of paracellular permeability and epithelial cell proliferation. For example, the release of vasoactive intestinal peptide (VIP) by enteric neurons inhibits epithelial cell proliferation and keeps epithelial barrier integrity by inducing ZO-1 expression (74). ENS glial cells seem to regulate the intestinal barrier function by releasing S-nitrosoglutathione, which regulates TJ protein expression (75). Vagus nerve activity may also modulate this defensive function via the release of neuropeptides such as acetylcholine and VIP. Furthermore, innate IS cells express a wide range of neuropeptide receptors, and the direct, bidirectional interaction between nerves and cells such as mast cells and eosinophils modulates intestinal permeability both under homeostatic and pathologic conditions.
Intestinal barrier dysfunction in gastrointestinal diseases
The intestinal barrier is a dynamic system involving a number of factors, and an increased passage of substances due to increased permeability does not necessarily imply dysfunction. Progression from increased intestinal permeability to disease development implies an imbalance in barrier function-sustaining factors, and the immune system is likely a key player given the association between inflammation and barrier dysfunction in several gastrointestinal diseases. Under normal conditions, increased permeability does not suffice as a cause of intestinal disease since the epithelial barrier may recover once the stimulus subsides. However, under certain pathological conditions this self-regulating capacity may be lost and contribute to increased permeability, thus facilitating chronic intestinal inflammation (Table I).
While the etiology of inflammatory bowel disease (IBD) remains unknown, patients have increased intestinal permeability when compared to healthy subjects. This has been identified to result from structural changes in TJ proteins, primarily reduced claudin-3, 4, 5 and 8 and occludin expression, as well as increased claudin-2 expression and MLCK phosphorylation, which faiclitates cytoskeleton contraction (68,76,77). However, these junction changes in active IBD are not seen in remitted IBD patients (76), which suggests that TJ changes are a consequence of disease. An exaggerated inflammatory response would presumably be responsible for these changes given the increased IFN-γ and TNF-α levels in these patients (78) and the effect these cytokines have on the epithelial barrier in vitro (79). Therefore, a convergence of genetic and environmental factors, as well as barrier function defects, ultimately leads to an abnormal immune response and higher susceptibility to intestinal inflammation. In fact, IBD development has been associated with the presence of mutated proteins such as X-box binding protein 1 (XBP1) or NOD-2 gene mutations related to reduced IL-10 production or inadequate immune tolerance to luminal antigens and microbial products (80-82).
Celiac disease is an autoimmune condition of unknown etiology that results from gluten intolerance and usually develops in genetically susceptible individuals. Celiac disease may result in bowel disease because of an abnormal response of intestinal T-cells to gliadin, to which the innate immune system contributes by actively collecting and processing gluten antigens by dendritic cells (83). This response results in TJ structural changes, allowing the entry of this protein in the mucosa, and inducing a sustained immune response that contributes to increased intestinal permeability. These patients have been seen to display rearranged actin filaments and impaired occludin, claudin-3, claudin-4, ZO-1 and E-cadherin expression (84,85). Increased permeability has been shown to be associated with higher zonulin levels, which induce cytoskeleton rearrangement via PKC, ZO-1 and occludin downregulation, and TJ complex integrity disruption (86,87).
Impaired intestinal barrier function has also been involved in the pathophysiology of food allergy, as patients display increased intestinal permeability even in the absence of food allergens (88). While not posited as the primary cause of allergy, the presence of certain environmental factors (infection, stress) increases intestinal permeability and the passage of substances that, under normal conditions, would never penetrate the epithelial barrier. This may favor an allergic response to food antigens in susceptible individuals (89).
Irritable bowel syndrome (IBS) is a chronic functional disorder whose pathophysiological mechanisms remain unknown. Despite different clinical subtypes according to stool pattern (diarrhea, constipation, mixed), all patients share an increase in intestinal permeability (90). This barrier dysfunction has been associated with TJ integrity changes that in turn are related to intestinal mucosal mast cell activation and symptoms, mainly in IBS with predominant diarrhea (91). Most relevant changes in TJ protein expression include reduced ZO and occludin, and increased claudin-2 and myosin light chain phosphorylation (92,93).
Conclusions
The intestinal barrier function is key for the maintenance of intestinal homeostasis and the prevention of exaggerated immune responses that facilitate chronic bowel inflammation. This defensive role is undertaken by numerous elements of diverse nature and anatomical location with the purpose of preserving intestinal integrity. Tight junctions are critical determinants of barrier function, hence knowledge regarding their regulation and modulation of epithelial cell changes is critical to understand their contribution to the pathogenesis of gastrointestinal conditions associated with barrier dysfunction. Therefore, therapy strategies targeted at restoring this defensive role show promise for recovering intestinal homeostasis and overall health.
References
1. Artis D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat Rev Immunol 2008;8:411-20. DOI: 10.1038/nri2316. [ Links ]
2. Turner J. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol 2009;9:799-809. DOI: 10.1038/nri2653. [ Links ]
3. Caricilli A, Castoldi A, Cámara N. Intestinal barrier: A gentlemen's agreement between microbiota and immunity. World J Gastrointest Pathophysiol 2014;5:18-32. DOI: 10.4291/wjgp.v5.i1.18. [ Links ]
4. Rescigno M. The intestinal epithelial barrier in the control of homeostasis and immunity. Trends Immunol 2011;32:256-64. DOI: 10.1016/j.it.2011.04.003. [ Links ]
5. Pascual S, Martínez J, Pérez-Mateo M. The intestinal barrier: Functional disorders in digestive and non-digestive diseases. Gastroenterol Hepatol 2001;24:256-67. DOI: 10.1016/S0210-5705(01)70167-7. [ Links ]
6. Shen L, Turner J. Role of epithelial cells in initiation and propagation of intestinal inflammation. Eliminating the static: Tight junction dynamics exposed. Am J Physiol Gastrointest Liver Physiol 2006;290:G577-82. DOI: 10.1152/ajpgi.00439.2005. [ Links ]
7. Sarker S, Gyr K. Non-immunological defence mechanisms of the gut. Gut 1992;33(7):987-93. DOI: 10.1136/gut.33.7.987. [ Links ]
8. Qin X, Caputo F, Xu D, et al. Hydrophobicity of mucosal surface and its relationship to gut barrier function. Shock 2008;29:372-6. [ Links ]
9. Bevins C, Salzman N. Paneth cells, antimicrobial peptides and maintenance of intestinal homeostasis. Nat Rev Microbiol 2011;9: 356-68. DOI: 10.1038/nrmicro2546. [ Links ]
10. Antoni L, Nuding S, Weller D, et al. Human colonic mucus is a reservoir for antimicrobial peptides. J Crohns Colitis 2013;7:652-64. DOI: 10.1016/j.crohns.2013.05.006. [ Links ]
11. Brandtzaeg P. Molecular and cellular aspects of the secretory immunoglobulin system. APMIS 1995;103:1-19. DOI: 10.1111/j.1699-0463.1995.tb01073.x. [ Links ]
12. Elphick D, Mahida Y. Paneth cells: Their role in innate immunity and inflammatory disease. Gut 2005;54:1802-9. DOI: 10.1136/gut.2005.068601. [ Links ]
13. Lence WI, Cheung G, Strohmeier G, et al. Induction of epithelial chloride secretion by channel-forming cryptdins 2 and 3. Proc Natl Acad Sci USA 1997;94:8585-9. DOI: 10.1073/pnas.94.16.8585. [ Links ]
14. Salzman N, Hung K, Haribhai D, et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat Immunol 2010;11(1):76-83. DOI: 10.1038/ni.1825. [ Links ]
15. Ugolev A, De Laey P. Membrane digestion. A concept of enzyme hydrolysis on cell membranes. Biochim Biophys Acta 1973;300:105-28. DOI: 10.1016/0304-4157(73)90001-4. [ Links ]
16. Shan M, Gentile M, Yeiser J, et al. Mucus enhances gut homeostasis and oral tolerance by delivering immunoregulatory signals. Science 2013;342:447-53. DOI: 10.1126/science.1237910. [ Links ]
17. Chang E, Rao M. Intestinal water and electrolyte transport: Mechanisms of physiological and adaptive responses. In: Johnson LR, ADH. Physiology of the Gastrointestinal Tract. 3rd ed. New York: Raven, Lippincott Williams & Wilkins; 1994. p. 2027-81. [ Links ]
18. Neish A. Microbes in gastrointestinal health and disease. Gastroenterology 2009;136:65-80. DOI: 10.1053/j.gastro.2008.10.080. [ Links ]
19. Palmer C, Bik E, DiGiulio D, et al. Development of the human infant intestinal microbiota. PLoS Biol 2007;5:e177. DOI: 10.1371/journal.pbio.0050177. [ Links ]
20. Tappenden K, Deutsch A. The physiological relevance of the intestinal microbiota-contributions to human health. J Am Coll Nutr 2007;26:679S-83S. DOI: 10.1080/07315724.2007.10719647. [ Links ]
21. Booth C, Potten C. Gut instincts: Thoughts on intestinal epithelial stem cells. J Clin Invest 2000;105:1493-9. DOI: 10.1172/JCI10229. [ Links ]
22. Van Der Flier L, Clevers H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu Rev Physiol 2009;71:241-60. DOI: 10.1146/annurev.physiol.010908.163145. [ Links ]
23. Pott J, Hornef M. Innate immune signalling at the intestinal epithelium in homeostasis and disease. EMBO Rep 2012;13:684-98. DOI: 10.1038/embor.2012.96. [ Links ]
24. Zeuthen L, Fink L, Frokiaer H. Epithelial cells prime the immune response to an array of gut-derived commensals towards a tolerogenic phenotype through distinct actions of thymic stromal lymphopoietin and transforming growth factor-beta. Immunology 2008;123:197-208. [ Links ]
25. Zaph C, Du Y, Saenz S, et al. Commensal-dependent expression of IL-25 regulates the IL-23-IL-17 axis in the intestine. J Exp Med 2008;205:2191-8. DOI: 10.1084/jem.20080720. [ Links ]
26. He B, Xu W, Santini P, et al. Intestinal bacteria trigger T cell-independent immunoglobulin A(2) class switching by inducing epithelial-cell secretion of the cytokine APRIL. Immunity 2007;26:812-26. DOI: 10.1016/j.immuni.2007.04.014. [ Links ]
27. Xu W, He B, Chiu A, et al. Epithelial cells trigger frontline immunoglobulin class switching through a pathway regulated by the inhibitor SLPI. Nat Immunol 2007;8:294-303. DOI: 10.1038/ni1434. [ Links ]
28. Cerutti A, Rescigno M. The biology of intestinal immunoglobulin A responses. Immunity 2008;28:740-50. DOI: 10.1016/j.immuni.2008.05.001. [ Links ]
29. Corr S, Gahan C, Hill C. M-cells: Origin, morphology and role in mucosal immunity and microbial pathogenesis. FEMS Immunol Med Microbiol 2008;52:2-12. DOI: 10.1111/j.1574-695X.2007.00359.x. [ Links ]
30. Miller H, Zhang J, Kuolee R, et al. Intestinal M cells: the fallible sentinels? World J Gastroenterol 2007;13:1477-86. [ Links ]
31. Gill N, Wlodarska M, Finlay B. Roadblocks in the gut: Barriers to enteric infection. Cell Microbiol 2011;13:660-9. DOI: 10.1111/j.1462-5822.2011.01578.x. [ Links ]
32. Göke M, Kanai M, Podolsky D. Intestinal fibroblasts regulate intestinal epithelial cell proliferation via hepatocyte growth factor. Am J Physiol 1998;274:G809-18. DOI: 10.1016/S0016-5085(98)70583-9. [ Links ]
33. Furness J. Types of neurons in the enteric nervous system. J Auton Nerv Syst 2000;81:87-96. DOI: 10.1016/S0165-1838(00)00127-2. [ Links ]
34. Rühl A. Glial cells in the gut. Neurogastroenterol Motil 2005;17:777-90. DOI: 10.1111/j.1365-2982.2005.00687.x. [ Links ]
35. Flemström G, Sjöblom M. Epithelial cells and their neighbors. New perspectives on efferent signaling between brain, neuroendocrine cells, and gut epithelial cells. Am J Physiol Gastrointest Liver Physiol 2005;289:G377-80. [ Links ]
36. Balda M, Matter K. Tight junctions at a glance. J Cell Sci 2008;121:3677-82. DOI: 10.1242/jcs.023887. [ Links ]
37. Schulzke J, Fromm M. Tight junctions: Molecular structure meets function. Ann N Y Acad Sci 2009;1165:1-6. DOI: 10.1111/j.1749-6632.2009.04925.x. [ Links ]
38. Bauer H, Stelzhammer W, Fuchs R, et al. Astrocytes and neurons express the tight junction-specific protein occludin in vitro. Exp Cell Res 1999;250:434-8. DOI: 10.1006/excr.1999.4558. [ Links ]
39. Blank F, Wehrli M, Lehmann A, et al. Macrophages and dendritic cells express tight junction proteins and exchange particles in an in vitro model of the human airway wall. Immunobiology 2011;216:86-95. DOI: 10.1016/j.imbio.2010.02.006. [ Links ]
40. Rao R. Occludin phosphorylation in regulation of epithelial tight junctions. Ann N Y Acad Sci 2009;1165:62-8. DOI: 10.1111/j.1749-6632.2009.04054.x. [ Links ]
41. Dörfel M, Huber O. Modulation of tight junction structure and function by kinases and phosphatases targeting occludin. J Biomed Biotechnol 2012;2012:807356. DOI: 10.1155/2012/807356. [ Links ]
42. Hartsock A, Nelson W. Adherens and tight junctions: Structure, function and connections to the actin cytoskeleton. Biochim Biophys Acta 2008;1778:660-9. DOI: 10.1016/j.bbamem.2007.07.012. [ Links ]
43. Escaffit F, Boudreau F, Beaulieu J. Differential expression of claudin-2 along the human intestine: Implication of GATA-4 in the maintenance of claudin-2 in differentiating cells. J Cell Physiol 2005;203:15-26. DOI: 10.1002/jcp.20189. [ Links ]
44. Liu Y, Nusrat A, Schnell F, et al. Human junction adhesion molecule regulates tight junction resealing in epithelia. J Cell Sci 2000;113:2363-74. [ Links ]
45. Laukoetter M, Nava P, Lee W, et al. JAM-A regulates permeability and inflammation in the intestine in vivo. J Exp Med 2007;204:3067-76. DOI: 10.1084/jem.20071416. [ Links ]
46. Ikenouchi J, Furuse M, Furuse K, et al. Tricellulin constitutes a novel barrier at tricellular contacts of epithelial cells. J Cell Biol 2005;171:939-45. DOI: 10.1083/jcb.200510043. [ Links ]
47. Mariano C, Sasaki H, Brites D, et al. A look at tricellulin and its role in tight junction formation and maintenance. Eur J Cell Biol 2011;90:787-96. DOI: 10.1016/j.ejcb.2011.06.005. [ Links ]
48. Umeda K, Ikenouchi J, Katahira-Tayama S, et al. ZO-1 and ZO-2 independently determine where claudins are polymerized in tight-junction strand formation. Cell 2006;126:741-54. DOI: 10.1016/j.cell.2006.06.043. [ Links ]
49. Niessen C, Gottardi C. Molecular components of the adherens junction. Biochim Biophys Acta 2008;1778:562-71. DOI: 10.1016/j.bbamem.2007.12.015. [ Links ]
50. Garrod D, Chidgey M. Desmosome structure, composition and function. Biochim Biophys Acta 2008;1778:572-87. DOI: 10.1016/j.bbamem.2007.07.014. [ Links ]
51. Kojima T, Murata M, Go M, et al. Connexins induce and maintain tight junctions in epithelial cells. J Membr Biol 2007;217:13-9. DOI: 10.1007/s00232-007-9021-4. [ Links ]
52. Macdonald T, Monteleone G. Immunity, inflammation, and allergy in the gut. Science 2005;307:1920-5. DOI: 10.1126/science.1106442. [ Links ]
53. Rhee S, Im E, Riegler M, et al. Pathophysiological role of Toll-like receptor 5 engagement by bacterial flagellin in colonic inflammation. Proc Natl Acad Sci U S A 2005;102:13610-5. DOI: 10.1073/pnas.0502174102. [ Links ]
54. Lee J, Mo J, Katakura K, et al. Maintenance of colonic homeostasis by distinctive apical TLR9 signalling in intestinal epithelial cells. Nat Cell Biol 2006;8:1327-36. DOI: 10.1038/ncb1500. [ Links ]
55. Yan F, Polk D. Disruption of NF-kappaB signalling by ancient microbial molecules: Novel therapies of the future? Gut 2010;59:421-6. [ Links ]
56. Sansonetti P. War and peace at mucosal surfaces. Nat Rev Immunol 2004;4:953-64. DOI: 10.1038/nri1499. [ Links ]
57. Bernardo D. Human intestinal dendritic cells as controllers of mucosal immunity. Rev Esp Enferm Dig 2013;105:279-90. DOI: 10.4321/S1130-01082013000500006. [ Links ]
58. Blander J, Sander L. Beyond pattern recognition: five immune checkpoints for scaling the microbial threat. Nat Rev Immunol 2012;12:215-25. DOI: 10.1038/nri3167. [ Links ]
59. Sansonetti P. To be or not to be a pathogen: That is the mucosally relevant question. Mucosal Immunol 2011;4:8-14. DOI: 10.1038/mi.2010.77. [ Links ]
60. Montalvillo E, Garrote J, Bernardo D, et al. Innate lymphoid cells and natural killer T cells in the gastrointestinal tract immune system. Rev Esp Enferm Dig 2014;106:334-45. [ Links ]
61. Kato L, Kawamoto S, Maruya M, et al. The role of the adaptive immune system in regulation of gut microbiota. Immunol Rev 2014;260:67-75. DOI: 10.1111/imr.12185. [ Links ]
62. Maynard C, Weaver C. Intestinal effector T cells in health and disease. Immunity 2009;31:389-400. DOI: 10.1016/j.immuni.2009.08.012. [ Links ]
63. Khor B, Gardet A, Xavier R. Genetics and pathogenesis of inflammatory bowel disease. Nature 2011;474:307-17. DOI: 10.1038/nature10209. [ Links ]
64. Turner J. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol 2009;9:799-809. DOI: 10.1038/nri2653. [ Links ]
65. Keita A, Söderholm J. The intestinal barrier and its regulation by neuroimmune factors. Neurogastroenterol Motil 2010;22:718-33. DOI: 10.1111/j.1365-2982.2010.01498.x. [ Links ]
66. Conner S, Schmid S. Regulated portals of entry into the cell. Nature 2003;422:37-44. DOI: 10.1038/nature01451. [ Links ]
67. Utech M, Mennigen R, Bruewer M. Endocytosis and recycling of tight junction proteins in inflammation. J Biomed Biotechnol 2010;2010:484987. DOI: 10.1155/2010/484987. [ Links ]
68. Prasad S, Mingrino R, Kaukinen K, et al. Inflammatory processes have differential effects on claudins 2, 3 and 4 in colonic epithelial cells. Lab Invest 2005;85:1139-62. DOI: 10.1038/labinvest.3700316. [ Links ]
69. Schulzke J, Bojarski C, Zeissig S, et al. Disrupted barrier function through epithelial cell apoptosis. Ann N Y Acad Sci 2006;1072:288-99. DOI: 10.1196/annals.1326.027. [ Links ]
70. Wells J, Rossi O, Meijerink M, et al. Epithelial crosstalk at the microbiota-mucosal interface. Proc Natl Acad Sci U S A. 2011;1:4607-14. DOI: 10.1073/pnas.1000092107. [ Links ]
71. Wang F, Graham W, Wang Y, et al. Interferon-gamma and tumor necrosis factor-alpha synergize to induce intestinal epithelial barrier dysfunction by up-regulating myosin light chain kinase expression. Am J Pathol 2005;166:409-19. DOI: 10.1016/S0002-9440(10) 62264-X. [ Links ]
72. Al-Sadi R, Ye D, Dokladny K, et al. Mechanism of IL-1beta-induced increase in intestinal epithelial tight junction permeability. J Immunol 2008;180:5653-61. DOI: 10.4049/jimmunol.180.8.5653. [ Links ]
73. Al-Sadi R, Boivin M, Ma T. Mechanism of cytokine modulation of epithelial tight junction barrier. Front Biosci 2009;14:2765-78. DOI: 10.2741/3413. [ Links ]
74. Neunlist M, Toumi F, Oreschkova T, et al. Human ENS regulates the intestinal epithelial barrier permeability and a tight junction-associated protein ZO-1 via VIPergic pathways. Am J Physiol Gastrointest Liver Physiol 2003;285:G1028-36. DOI: 10.1152/ajpgi.00066.2003. [ Links ]
75. Savidge T, Newman P, Pothoulakis C, et al. Enteric glia regulate intestinal barrier function and inflammation via release of S-nitrosoglutathione. Gastroenterology 2007;132:1344-58. DOI: 10.1053/j.gastro.2007.01.051. [ Links ]
76. Zeissig S, Bürgel N, Günzel D, et al. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn's disease. Gut 2007;56:61-72. DOI: 10.1136/gut.2006.094375. [ Links ]
77. Blair S, Kane S, Clayburgh D, et al. Epithelial myosin light chain kinase expression and activity are upregulated in inflammatory bowel disease. Lab Invest 2006; 86: 191-201. DOI: 10.1038/labinvest.3700373. [ Links ]
78. MacDonald T, Hutchings P, Choy M, et al. Tumour necrosis factor-alpha and interferon-gamma production measured at the single cell level in normal and inflamed human intestine. Clin Exp Immunol 1990;81:301-5. DOI: 10.1111/j.1365-2249.1990.tb03334.x. [ Links ]
79. Madara J, Stafford J. Interferon-gamma directly affects barrier function of cultured intestinal epithelial monolayers. J Clin Invest 1989;83:724-7. DOI: 10.1172/JCI113938. [ Links ]
80. Kaser A, Lee A, Franke A, et al. XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 2008;134:743-56. DOI: 10.1016/j.cell.2008.07.021. [ Links ]
81. Fisher S, Tremelling M, Anderson C, et al. Genetic determinants of ulcerative colitis include the ECM1 locus and five loci implicated in Crohn's disease. Nat Genet 2008;40:710-2. DOI: 10.1038/ng.145. [ Links ]
82. Watanabe T, Asano N, Murray P, et al. Muramyl dipeptide activation of nucleotide-binding oligomerization domain 2 protects mice from experimental colitis. J Clin Invest 2008;118:545-59. [ Links ]
83. Comino I, Suligoj T, Al-Hassi H, et al. Constitutive gut-homing capacity on circulating myeloid dendritic cells in coeliac disease. Rev Esp Enferm Dig 2014;106:64-5. DOI: 10.4321/S1130-01082014000100013. [ Links ]
84. Pizzuti D, Bortolami M, Mazzon E, et al. Transcriptional downregulation of tight junction protein ZO-1 in active coeliac disease is reversed after a gluten-free diet. Dig Liver Dis 2004;36:337-41. DOI: 10.1016/j.dld.2004.01.013. [ Links ]
85. Sander G, Cummins A, Henshall T, et al. Rapid disruption of intestinal barrier function by gliadin involves altered expression of apical junctional proteins. FEBS Lett 2005;579:4851-5. DOI: 10.1016/j.febslet.2005.07.066. [ Links ]
86. Fasano A, Not T, Wang W, et al. Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease. Lancet 2000;355:1518-9. DOI: 10.1016/S0140-6736(00)02169-3. [ Links ]
87. Drago S, El Asmar R, Di Pierro M, et al. Gliadin, zonulin and gut permeability: Effects on celiac and non-celiac intestinal mucosa and intestinal cell lines. Scand J Gastroenterol 2006;41:408-19. DOI: 10.1080/00365520500235334. [ Links ]
88. Ventura M, Polimeno L, Amoruso A, et al. Intestinal permeability in patients with adverse reactions to food. Dig Liver Dis 2006;38:732-6. DOI: 10.1016/j.dld.2006.06.012. [ Links ]
89. Heyman M. Gut barrier dysfunction in food allergy. Eur J Gastroenterol Hepatol 2005;17:1279-85. DOI: 10.1097/00042737-200512000-00003. [ Links ]
90. Camilleri M, Lasch K, Zhou W. Irritable bowel syndrome: methods, mechanisms, and pathophysiology. The confluence of increased permeability, inflammation, and pain in irritable bowel syndrome. Am J Physiol Gastrointest Liver Physiol 2012; 303: G775-85. DOI: 10.1152/ajpgi.00155.2012. [ Links ]
91. Martínez C, González-Castro A, Vicario M, et al. Cellular and molecular basis of intestinal barrier dysfunction in the irritable bowel syndrome. Gut Liver 2012;6:305-15. DOI: 10.5009/gnl.2012.6.3.305. [ Links ]
92. Martínez C, Lobo B, Pigrau M, et al. Diarrhoea-predominant irritable bowel syndrome: An organic disorder with structural abnormalities in the jejunal epithelial barrier. Gut 2013;62:1160-8. DOI: 10.1136/gutjnl-2012-302093. [ Links ]
93. Bertiaux-Vandaële N, Youmba S, Belmonte L, et al. The expression and the cellular distribution of the tight junction proteins are altered in irritable bowel syndrome patients with differences according to the disease subtype. Am J Gastroenterol 2011;106:2165-73. DOI: 10.1038/ajg.2011.257 [ Links ]
![]() Correspondence:
Correspondence:
María Vicario.
Neuro-Immuno-Gastroenterology Laboratory.
Unit of Physiology and Digestive Physio-Pathology.
Vall d'Hebron Institut de Recerca.
Hospital Universitario Vall d'Hebron.
Paseo Vall d'Hebron, 119-129.
08035 Barcelona, Spain
e-mail: maria.vicario@vhir.org
Received: 14-05-2015
Accepted: 25-05-2015











 texto en
texto en