Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.108 no.3 Madrid mar. 2016
ORIGINALS PAPERS
Using autologous platelet-rich plasma for the treatment of complex fistulas
Empleo de plasma autólogo rico en plaquetas en el tratamiento de fístulas perianales complejas
Almudena Moreno-Serrano1, Juan José García-Díaz1, Manuel Ferrer-Márquez1, Raquel Alarcón-Rodríguez2, Antonio Álvarez-García1 and Ángel Reina-Duarte1
1Hospital Torrecárdenas, AGSN. Almería, Spain.
2Universidad de Almería. Almería, Spain
ABSTRACT
Objective: This study aims to demonstrate the effectiveness and safety of autologous fibrin gel rich in platelet growth factors for the treatment of complex perianal fistulas.
Material and methods: Prospective epidemiological study. Patients with complex perianal fistula or perianal fistula mere alteration of continence are included. identification of both holes and the journey, curettage of it and instillation of Vivostat PRF® in the way it is done to observe excess material by OFE. The variables analyzed were: age, sex, use of prior Seton clinic prevalent type of fistula, postoperative complications, fistula closure and impaired quality of life using the SF-36 test (v2).
Results: From January 2011 to May 2013 have involved 23 patients, 12 men and 11 women, with an average age of 49 years and a minimum follow-up of 12 months. Two dropped out. 17 patients had low transsphincteric fistulas, 2 and 2 high transsphincteric intersphincteric with impaired continence. The most common symptom is the discharge. Twelve patients had a loose seton (62%), of which nine cured. Of all the patients we have operated the success rate is 62%. No patient developed incontinence after treatment. Only two reported a worse quality of life after surgery.
Conclusion: This study demonstrates that there is a clear benefit to the use of Vivostat PRF® as a treatment for complex perianal fistulas. It is a highly reproducible technique with acceptable results and does not produce impairment of continence.
Key words: Complex fistula. Autologous platelet-rich plasma. Quality of life.
RESUMEN
Objetivo: este estudio tiene como objetivo demostrar la efectividad y seguridad de un gel de fibrina autóloga rico en factores de crecimiento plaquetario para el tratamiento de las fístulas perianales complejas.
Material y métodos: estudio epidemiológico prospectivo descriptivo. Se incluyen pacientes que presentan fístula perianal compleja o fístula perianal simple con alteración de la continencia. Se realiza identificación de ambos orificios y del trayecto, legrado del mismo e instilación del Vivostat PRF® en el trayecto hasta observar exceso de material por el OFE. Las variables a analizar son: edad, sexo, uso de setón previo, clínica prevalente, tipo de fístula, complicaciones postoperatorias, cierre de la fístula y alteraciones en la calidad de vida mediante el test sf-36(v2).
Resultados: desde enero del 2011 hasta mayo del 2013 se ha intervenido a 23 pacientes, 12 hombres y 11 mujeres, con una media de edad de 49 años y un seguimiento mínimo de 12 meses. Dos abandonaron el estudio. 17 pacientes presentaban fístula transesfinteriana baja; 2, transesfinteriana alta, y 2, interesfinteriana con alteración de la continencia. El síntoma más frecuente es la supuración. Doce pacientes llevaban un setón laxo (62%), de los cuales curaron nueve. De todos los pacientes que hemos intervenido el porcentaje de éxitos es de un 62%. Ningún paciente desarrolló incontinencia después del tratamiento. Sólo dos refieren una peor calidad de vida después de la intervención.
Conclusión: este estudio demuestra que hay un claro beneficio con el uso de Vivostat PRF® como tratamiento para las fístulas perianales complejas. Es una técnica altamente reproductible con resultados aceptables y que no produce alteraciones de la continencia.
Palabras clave: Fístulas complejas. Plasma autólogo. Factores de crecimiento. Calidad de vida.
Introduction
Fistulous disease is one of the commonest anorectal conditions. Prevalence ranges from 8.6 to 10 per 100,000 people per year, with a difference of 8:1 between males and females (1). We know that most fistulas have a crypto-glandular etiology and result from infection in the intersphincteric space, although such theory has never been unequivocally demonstrated (2). The most appropriate surgical treatment remains controversial, primarily for complex perianal fistula.
Fistulotomy is the most likely method to result in cure for simple fistula, reaching 90% of success with low risk for incontinence (3). These results are also offered by fistulectomy, but at the expense of a higher risk of sphincteric fiber injury and therefore increased risk for incontinence.
The main issue involves complex fistula management, as fistulotomy would represent an inacceptable risk of incontinence in these patients. To prevent this, many therapies have been posited over time. One option would be immediate sphincter reconstruction after fistulotomy. Despite the results achieved in various series (4) (recurrence rate around 15%, incontinence around 20%), the technique is envisaged with skepticism in surgical forums, even though it is a good option for selected cases.
During the last decade less aggressive therapies such as fibrin, plugs, etc., have been emphasized with disparaging results. Most studies usually include few patients, different assessment methods, and inappropriate methodologies (5,6).
Multiple clinical experiences have been collected with the use of autologous platelet-rich plasma (PRP), initially in the setting of maxillofacial surgery (7) for the repair of tumor resection-related defects; the technique was then endorsed in other specialties such as chest surgery (8), cardiovascular surgery (9), plastic surgery (10), traumatology (11), and nephrology (12).
PRP is defined as a portion of one's own plasma with above-baseline platelet and growth factor levels as obtained with centrifugation. Platelets have alpha granules that contain a number of proteins (PDGF, FCT-β, IL-1, FADP, VEGF, ECGF, osteocalcin, osteonectin, fibrinogen, fibronectin, and thrombospondin) with multiple actions on the various aspects of tissue repair. Platelets start secreting these substances 10 minutes after clot formation; following this massive release platelets synthesize proteins for 5 additional days until macrophages reach the site with vascular growth and take over tissue repair regulation. Among marketed PRP products we have used Vivostat® PRF, which includes three components including a disposable piece of equipment to directly collect 120 ml of blood from the patient to which citrate is added; a processing unit for the kit, which involves an automated biotechnological system that controls the biochemical process and provides the final product after 25 minutes; and a special delivery unit allowing administration at the fistulous tract.
No study of Vivostat PRF has been carried out to date for the stand-alone treatment of perianal fistula. Our goal was to assess the efficacy of growth factor-rich fibrin in the management of complex perianal fistula.
Patients and methods
Study design
A prospective, open-label, descriptive study that was approved by our site's Ethics Committee in October 2010. The study was carried out from January 2011 to May 2013 at the Departments of Colorectal Surgery, General Surgery and Digestive Diseases of the Hospital Torrecárdenas, in Almería, Spain. All patients were appropriately informed and their consent was obtained both for surgery and the various tests to be performed before and after the surgical procedure.
Patients
Patients diagnosed with perianal fistula at the aforementioned unit were included. Inclusion criteria were age between 18 and 80, diagnosis of perianal fistula (intersphincteric with impaired continence, transsphincteric, and suprasphincteric). Exclusion criteria included ASA-IV patients, presence of inflammatory bowel disease, fistulas with multiple tracts or active sepsis, and intersphincteric fistula with no continence impairment.
During the preoperative phase all patients underwent an in-depth perianal examination in the left lateral decubitus position (Sims). The external fistulous opening (EFO) was located, the fistulous tract was palpated, and internal fistulous opening (IFO) location was attempted using rectal palpation. Anoscopy is routinely performed in all patients at screening. We consider magnetic resonance imaging of choice in classifying fistula type.
Studied variables include age, sex, family history, personal history, prior anal surgery, and therapies undergone. Women are specifically asked about pregnancies and risk factors for fecal incontinence. We used Wexner's test to assess continence before and six months after surgery. We used manometry before surgery in those with incontinence manifestations, and 6 months thereafter in order to assess potential changes. We used the SF-36 (v2) questionnaire to assess quality of life at six months after the procedure. Patients were scheduled for a visit seven days preoperatively to be briefed on the self-care required before surgery, which includes a residue-free diet, washing with saline and iodinated povidone twice a day, and an enema the night before the procedure.
Surgical technique
The surgical procedure is performed under intraspinal anesthesia in the prone decubitus (jack-knife) position. All fistulas are operated upon by the same surgeons. A complete anorectal exam is first performed to identify the fistula's external opening, tract, and internal opening. An attempt at stylet insertion is made. On occasion this maneuver may be facilitated by a seton already in place after a prior surgical procedure. The stylet acts as a guide for the introduction of a gauze into the tract for curettage. Saline and oxygenated water are used for washing. Skin and subcutaneous cellular tissue are debrided at the external opening, and a minimal resection (including the mucosa) is performed at the inner opening. The applicator is fixed to the gauze using a silk string. This guides the applicator through the tract to the IFO. The solution is applied in a controlled, precise manner on withdrawal, and the tract is totally filled until excess solution pours from the EFO. The IFO is subsequently closed with Vycril 2/0, and the EFO is left open for drainage (Fig. 1).
All patients are tested for tolerance on the evening following surgery, and then discharged on the same day if no complications arise. Postoperative care includes sitz baths using soap and water, and pain killers when needed.
Follow-up
A strict follow-up is performed at the colorectal surgery clinic at 1 week, at 1 and 6 months, and at 1 year after surgery, and then on a yearly basis. Patients are administered the Wexner's and quality of life (SF-36 v2) questionnaires on the sixth-month follow-up visit. An MRI is performed and patients with incontinence before surgery undergo manometry. The technique is deemed a failure when symptoms (suppuration, open EFO and/or pain) persist after six months following the procedure. Recurrence is diagnosed when healing criteria are met at follow-up but the patient presents at the ER for perianal abscess sometime afterwards, or when pain, suppuration or wound changes are back at a given follow-up visit. Asymptomatic patients are deemed a surgical success even when an MRI scan reveals a persistent tract (or there is doubt whether images correspond to a fistulous tract or postsurgical fibrosis). At the 12-month visit a complete clinical assessment is carried out. Asymptomatic patients have their next visit scheduled after one year. Patients with persistent manifestations are offered the option of re-intervention or of follow-up at the outpatient clinic.
Statistical analysis
All the variables obtained throughout the study via our surgical database will be fed to the SPSS v.2.0 for Windows software package for their processing and statistical analysis.
The process will commence with a univariate or descriptive analysis. In this study, qualitative variables will be expressed as frequencies with their corresponding percentages, and quantitative variables will undergo central tendency and dispersion measurements (mean, minimum, maximum, standard deviation).
Results
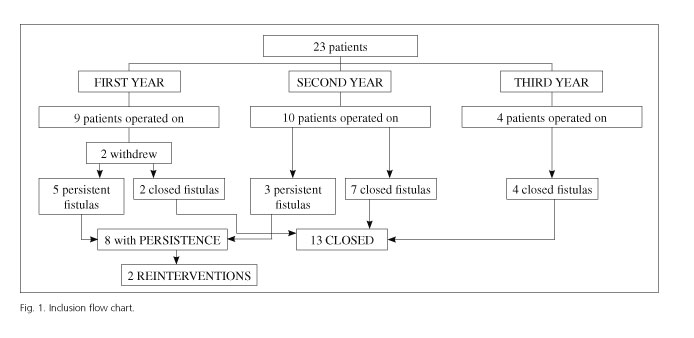
From January 2011 to May 2013 23 patients were operated upon; two of them withdrew from the study. The series comprises 21 patients (12 males and 9 females) whose characteristics are summed up in table I. Mean age was 49 (range: 30-80). Eighteen patients underwent a prior perianal abscess drain and eleven (52.4%) had a seton placed, which they kept until surgery. The most common symptom was discharge, present in 86% of subjects for over six months. Four patients had incontinence to some extent: one with a score of two, one with a score of four, and two with a score of six in Wexner's test. Only the latter two patients had manometry changes (one with a maximum baseline pressure [MBP] of 48.69 mmHg and a maximum voluntary contraction [MVC] of 75.62 mmHg; one with a MBP of 51.21 mmHg and a MVC of 72 mmHg). The diagnosis with perianal fistula according to the RMN classification was intersphincteric for three patients (14.2%), lower transsphincteric for seventeen (81%), and upper transsphincteric for one (4.8%). At the time of the procedure, perianal fistulas were diagnosed as lower transsphincteric for seventeen (81%), upper transsphincteric for two, and intersphincteric for two.
All patients were followed up for at least 12 months, with a mean of 22 months (12-35). At six months after surgery, patients underwent a clinical and RMN assessment. Thirteen patients (61.9%) had no clinical symptoms and eight still experienced discharge. MRI revealed a fibrous tract in nine patients (42.9%) and a persistent fistula in twelve (57.1%). Among the latter, four were asymptomatic, and were therefore considered as a successful cure. We considered recurrence at 31% because of persistent symptoms despite MRI revealing active sepsis in four subjects. At 1-year follow-up, only two patients were symptomatic, and underwent reoperation with a different technique. The remaining six symptomatic patients chose conservative management and outpatient follow-up.
Regarding patients with incontinence, those two with a Wexner score of six clinically improved to a score of four. Manometry, in contrast, remained unchanged in terms of MBP and MVC. Patients with a score of four also reported fewer manifestations with a Wexner score of two. The fourth patient remained the same. Mean pre-surgical Wexner score was 0.85, 0.57 after the procedure.
As regards quality of life, nineteen patients (80%) reported improvements following surgery. Only in two patients with recurrence did quality of life worsen after the procedure.
Postoperative complications included a perianal abscess in one patient two weeks after surgery, and postoperative pain requiring augmented analgesia in another patient.
Discussion
Fistula surgery remains an issue for colorectal surgeons and an exhausting process for patients, as concerns persist regarding cure rates, sequels, or quality of life impairment.
The treatment of simple fistula is usually based on fistulotomy, which provides healing for about 90% of patients and a low risk of incontinence (3). The main problem lies in the management of complex fistula, as fistulotomy represents an unacceptable risk of incontinence in these patients. Various therapies have been suggested over time (fibrin, plugs, etc.), but results are controversial (5,6).
In our study, we used growth factor-rich fibrin (Vivostat PRF®) in monotherapy, and obtained cure rates of 62%. As regards incontinence, our results show no incontinence at all (only patients with prior incontinence had incontinence after treatment) and a satisfactory quality of life in 80%. The primary limitation of our study was the low number of included patients. Intersphincteric fistula represented 60% according to the Parks classification system, but it was an exclusion criterion of ours for patients without incontinence, as fistulotomy solves it in over 90% of cases. Another limitation is our dealing with just one prospective cohort, with no comparisons between two groups undergoing different treatments. This paves the way for future comparative, properly designed, possibly multicenter studies including a greater number of patients.
Vivostat PRF® contains a potent, highly varied concentration of growth factors to stimulate fibroblast proliferation and tissue regeneration. Furthermore, fibrin provides a scaffold for cell migration, and structure during regeneration. Numerous in vitro studies recognize the unique characteristics of PRF® and its ability to stimulate cell growth. This being an autologous product, the risk for viral infection and autoimmune response is averted. Its usefulness has been demonstrated in several specialties (urology, thoracic and cardiac surgery, and plastic surgery) (7-13).
In the field of proctology, a recent paper shows the usefulness of PRF® for pilonidal sinus. It reports a clinical trial (14) in 100 patients undergoing clinical excision and randomized into two groups; group A received autologous platelet gel, and group B had PRF® injected. Results show a shorter healing period, fewer infections, and earlier return to work in group-B patients.
In the management of fistulas, to date only one multicenter study has been reported (15), which includes the experience of some hospitals in this setting. The remaining studies (16,17) are to demonstrate the closure of upper perianal fistulas with a technique combining advancement flaps and platelet-rich plasma.
Setons are used in a number of ways. This may explain variability in results obtained from the literature (18-20). The "snug seton" concept by Hammond et al. refers to a seton that is closely fit without excessive tightness (21). Section is slowly gradual, and the stitch comes off spontaneously within 6 months in 52% of cases; in the remaining patients, it is completed with minimal fistulotomy, with only minor continence changes reported.
Setons at present represent a bridge to definitive surgery, and result in lower recurrence and incontinence rates (22) in fistulizing Crohn's disease patients, where the incidence of fistula oscillates between 21% and 23% (23). We considered Crohn's disease an exclusion criterion. Eighteen patients had a history of perianal abscess. Eleven underwent drainage and had a seton placed in one surgical procedure.
The "gold standard" for the treatment of complex fistula is likely the advancement flap. In most series recurrence rates range from 0% to 33% (24), and the scarce interest of some authors (around 13%) in the assessment of postoperative continence is outstanding (25). Result differences suggest that technique represents a key factor. The controversy over which type of flap (full or partial thickness flap) is best remains ongoing (26). In their paper, Roig et al. (27) show that continence changes range from 51% with full thickness flaps to 33% with partial thickness flaps (p = 0.08) (26).
Fibrin sealants seem to behave similarly. Initial findings were encouraging, but later papers reported long-term relapses in up to 90% of cases (28-30). They may be indicated for patients with sphincteric function impairment or high surgical risk, having in mind what may be expected though.
Ligation of intersphincteric fistula tract (LIFT) represents a recently emerged technique often described as simple and reproducible. Thus far, results are insufficient to draw solid conclusions, but no continence impairment has been reported following the procedure (31). Further studies comparing LIFT versus other techniques are needed.
An anal fistula plug was launched 8 years ago as described by Johnson et al. in 2006 (32). A success rate of 87% was initially reported. However, subsequent reports (33-35) with a longer follow-up found a cure rate of 28%. Approximately, 19% of failures dropped their plugs due to inadequate fixation or excessive tension. Postoperative abscesses occur in 4-29% of cases (36).
Therapy with adult stem cells is promising for many a medical setting. A recent study was performed by Herreros et al. (37), which was designed based on the encouraging results obtained by two prior trials, a phase-I trial and a phase-II trial (38), assessing the management of perianal fistula. The phase-II trial in 49 patients found a cure rate of 71% with almost no risk for incontinence and a low recurrence rate (12%). Thus, a phase-III trial was carried out to assess the effectiveness and safety of stem-cell therapy in 200 patients at 19 sites, using 20 million stem cells from adipose tissue (group A, 64 patients), stem cells plus fibrin sealant (group B, 60 patients) or fibrin sealant alone (group C, 59 patients). Results are similar to prior studies; group B obtained a higher healing rate, but differences were not statistically significant.
Conclusion
The use of Vivostat PRF® for the closure of perianal fistula is a novel alternative that may offer significant benefit. It achieves a cure in over 60% of cases without impairing continence. As this is an autologous product, its use precludes rejection risk and minimizes infection risk. Notwithstanding this, further properly designed, comparative studies (possibly multicenter studies in order to achieve an adequate number of patients) are warranted to obtain solid conclusions.
References
1. Sainio P. Fistula in ano in a defined population. Incidence and epidemiological aspects. Ann Chir Gynaecol 1984;73:219-24. [ Links ]
2. Philips RKS, Lunnis P. Anal fistula: Surgical evaluation and management. Londres; 1996. [ Links ]
3. Van Onkelen RS, Gosselink MP, Van Rosmalen J, et al. Different characteristics of high and low transsphincteric fistulae. Colorectal Dis 4. 2014;16:471-5. DOI: 10.1111/codi.12578. [ Links ]
4. Roig JV, García-Armengol J, Jordán J, et al. Immediate reconstruction of the anal sphincter after fistulectomy in the management of complex anal fistulas. Colorectal Dis 1999;1:137-40. DOI: 10.1046/j.1463-1318.1999.00021.x. [ Links ]
5. Damin DC, Rosito MA, Contu PC, et al. Fibrin glue in the management of complex anal fistula. Arq Gastroenterol 2009;14:300-3. DOI: 10.1590/S0004-28032009000400010. [ Links ]
6. Parés D. Pathogenesis and treatment of fistula in ano. Br JSurg 2011; 98:2-3. DOI: 10.1002/bjs.7341. [ Links ]
7. Fernández López RG, López Buendía MC, Ruiz González E. Plasma rico en factores de crecimiento en cirugía bucal. Presentación de caso clínico. Revista Odontológica Mexicana 2005;9:141-6. [ Links ]
8. Méndez R, López-Cedrún JL, Patiño B, et al. Plasma enriquecido en plaquetas en la alveoloplastia de pacientes fisurados. Cir Pediatr 2006;19:23-6. [ Links ]
9. Tansley P, Al-Mulhim F, Lim E, et al. A prospective, randomized, controlled trial of the effectiveness of BioGlue in treating alveolar air leaks. J Thorac Cardiovasc Surg 2006;132:105-12. DOI: 10.1016/j.jtcvs.2006.02.022. [ Links ]
10. Rousou J. Use of fibrin sealants in cardiovascular surgery. J Card Surg 2013;28:238-47. DOI: 10.1111/jocs.12099. [ Links ]
11. Rodríguez Flores J, Palomar Gallego A, García-Denche J. Plasma rico en plaquetas: fundamentos biológicos y aplicaciones en cirugía maxilofacial y estética facial. Cirugía Oral y Maxilofacial 2012;34:8-17. DOI: 10.1016/j.maxilo.2011.10.007. [ Links ]
12. Frei R, Biosca FE, Handl M, et al. Conservative treatment using plasma rich in growth factors (PRGF) for injury to the ligamentous complex of the ankle. Acta Chir Orthop Traumatol Cech 2008;75:28-33. [ Links ]
13. Hevia M, Abascal-Junquera JM, Sacristán R, et al. Control de la hemostasia durante la nefrectomía parcial laparoscópica sin renorragia parenquimatosa: la experiencia vivostat. Actas Urol Esp 013;37:47-53. [ Links ]
14. Gipponi M, Reboa G, Testa T, et al. Tension-free primary closure with autologous platelet gel versus Vivostat for the definitive treatment of chronic sacrococcygeal pilonidal disease. In Vivo 2010;24:583-90. [ Links ]
15. Lara FJ, Serrano AM, Moreno JU, et al. Platelet-rich fibrin sealant as a treatment for complex perianal fistulas: A multicentre study. J Gastrointest Surg 2015;19: 360-8. DOI: 10.1007/s11605-014-2698-7. [ Links ]
16. Göttgens KW, Smeets RR, Stassen, et al. Treatment of Crohn's disease-related high perianal fistulas combining the mucosa advancement flap with platelet-rich-plasma: A pilot study. Tech Coloproctol 2015;19:455-9. DOI: 10.1007/s10151-015-1311-8. [ Links ]
17. Van der Hagen SJ, Baeten CG, Soeters PB, et al. Autologous platelet-derived growth factors (platelet-rich plasma) as an adjunct to mucosal advancement flap in high cryptoglandular perianal fistulae: A pilot study. Colorectal Dis 2011;13:215-8. DOI: 10.1111/j.1463-1318. 2009.01991.x. [ Links ]
18. Hasegawa H, Radley S, Keighley MR. Long-term results of cutting seton fistulotomy. Acta Chir Iugosl 2000;47(Suppl.1):19-21. [ Links ]
19. Chuang-Wei C, Chang-Chieh W, Cheng-Wen H, et al. Cutting seton for complex anal fistulas. Surgeon 2008;6:185-8. DOI: 10.1016/S1479-666X(08)80117-5. [ Links ]
20. Lykke A, Steendahl J, Wille-Jørgensen PA. Treating high anal fistulae with slow cutting seton. Ugeskr Laeger 2010;172:516-9. [ Links ]
21. Hammond TM, Knowles CH, Porrett T, et al. The snug seton: Short and medium term results of slow fistulotomy for idiopathic anal fistulae. Dis Colon Rectum 2006;8:328.37. DOI: 10.1111/j.1463-1318. 2005.00926.x. [ Links ]
22. Vial M, Pare's D, Pera M, et al. Faecal incontinence after seton treatment for anal fistulae with and without surgical division of internal anal sphincter: A systematic review. Colorectal Dis 2010;12:172-8. DOI: 10.1111/j.1463-1318.2009.01810.x. [ Links ]
23. Hukkinen M, Pakarinen MP, Piekkala M, et al. Treatment of complex perianal fistulas with seton and infliximab in adolescents with Crohn's disease. J Crohn Colitis 2014;8:756-62. DOI: 10.1016/j.crohns.2014.01.001. [ Links ]
24. Soltani A, Kaiser AM. Endorectal advancement flap for cryptoglandular or Crohn's fistula-in-ano. Dis Colon Rectum 2010;53:486-95. DOI: 10.1007/DCR.0b013e3181ce8b01. [ Links ]
25. Whiteford MH, Kilkenny J 3rd, Hyman N, et al. Standards practice task force; American Society of Colon and Rectal Surgeons Practice parameters for the treatment of perianal abscess and fistula-in-ano (revised). Dis Colon Rectum 2005;48:1337-42. DOI: 10.1007/s10350-005-0055-3. [ Links ]
26. Dubsky PC, Stift A, Friedl J, et al. Endorectal advancement flaps in the treatment of high anal fistula of cryptoglandular origin: Full-thickness vs. mucosal-rectum flaps. Dis Colon Rectum 2008;51:852-7. [ Links ]
27. Roig JV, García Armengol. Tratamiento de las fístulas de ano complejas de causa criptoglandular. ¿Aún se requiere un cirujano con experiencia? Cir Esp 2013;91:78-89. [ Links ]
28. Haim N, Neufeld D, Ziv T. Long-term results of fibrin glue treatment for cryptogenic perianal fistula: A multicenter study. Dis Colon Rectum 2011;54:1279-83. DOI: 10.1097/DCR.0b013e318223c894. [ Links ]
29. Panidis S, Papadopoulous VN, Michalpoulos. The use of fibrin glue in the treatment of complex perianal fistulae: A case report. Tech Coloproctol 2010;14:19-20. DOI: 10.1007/s10151-010-0608-x. [ Links ]
30. Yeung JM, Simpson JA, Tang SW. Fibrin glue for the treatment of fistulae in ano - A method worth sticking to? Colorectal Dis 2010;12:363-6. DOI: 10.1111/j.1463-1318.2009.01801.x. [ Links ]
31. Aboulian A, Kaji AH, Kumar RR. Early result of ligation of the intersphincteric fistula tract for fistula-in-ano. Dis Colon Rectum 2011;54:289-92. DOI: 10.1007/DCR.0b013e318203495d. [ Links ]
32. Johnson EK, Gaw JU, Armstrong DN. Efficacy of anal fistula plug vs. fibrin glue in closure of anorectal fistulas. Dis Colon Rectum 2006;49:371-6. [ Links ]
33. Chung W, Kazemi P, Ko D, et al. Anal fistula plug and fibrin glue versus conventional treatment in repair of complex anal fistulas. Am J Surg 2009;197:604-8. DOI: 10.1016/j.amjsurg.2008.12.013. [ Links ]
34. El-Gazzaz G, Zutshi M, Hull T. A retrospective review of chronic anal fistulae treated by anal fistulae plug. Colorectal Dis 2010;12:442-7. DOI: 10.1111/j.1463-1318.2009.01802.x. [ Links ]
35. Garg P, Song J, Bhatia A, et al. The efficacy of anal fistula plug in fistula-in-ano: A systematic review. Colorectal Dis 2010;12:965-70. DOI: 10.1111/j.1463-1318.2009.01933.x. [ Links ]
36. Ellis CN, Rostas JW, Greiner FG. Long-term outcomes with the use of bioprosthetic plugs for the management of complex anal fistulas. Dis Colon Rectum 2010;53:798-802. DOI: 10.1007/DCR. 0b013e3181d43b7d. [ Links ]
37. Herreros D, García-Arranz M, Guadalajara H, et al. A phase III clinical trial. Autologous expanded adipose-derived stem cell for the treatment of complex cryptoglandular perianal fistulas. Dis Colon Rectum 2012;55:762-77. DOI: 10.1097/DCR.0b013e318255364a. [ Links ]
38. García-Olmo D, García-Arranz M, Herreros D, et al. A phase I clinical trial of the treatment of Crohn's fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum 2005;48:1416-23. DOI: 10.1007/s10350-005-0052-6. [ Links ]
![]() Correspondence:
Correspondence:
Almudena Moreno-Serrano.
Hospital Torrecárdenas, AGSN.
C/ Hermandad de Donantes de Sangre, s/n.
04009 Almería, Spain
e-mail: almumoreno84@hotmail.com
Received: 06-08-2015
Accepted: 15-11-2015











 texto en
texto en