Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.108 no.3 Madrid mar. 2016
REVIEW
Practical considerations in the management of proton-pump inhibitors
Consideraciones prácticas en el manejo de los inhibidores de la bomba de protones
Lara Aguilera-Castro, Carlos Martín-de-Argila-de-Prados and Agustín Albillos-Martínez
Department of Gastroenterology and Hepatology. Hospital Universitario Ramón y Cajal, IRYCIS. Universidad de Alcalá. Madrid, Spain
ABSTRACT
Proton-pump inhibitors (PPIs) are one of the most active ingredients prescribed in Spain. In recent decades there has been an overuse of these drugs in both outpatient clinics and hospitals that has lead to a significant increase in healthcare spending and to an increase in the risk of possible side effects. It is important for health professionals to know the accepted indications and the correct doses for the use of these drugs. On the market there are different types of PPI: omeprazole, pantoprazole, lansoprazole, rabeprazole and esomeprazole. Omeprazole is the oldest and most used PPI, being also the cheapest. Although there are no important differences between PPIs in curing diseases, esomeprazole, a new-generation PPI, has proved to be more effective in eradicating H. pylori and in healing severe esophagitis compared to other PPIs. In recent years the use of generic drugs has spread; these drugs have the same bioavailability than the original drugs. In the case of PPIs, the few comparative studies available in the literature between original and generic drugs have shown no significant differences in clinical efficacy.
Key words: Proton-pump inhibitor. PPI. Omeprazole. Pantoprazole. Lansoprazole. Rabeprazole. Esomeprazole.
RESUMEN
Los inhibidores de la bomba de protones (IBP) son uno de los principios activos más prescritos en España. En las últimas décadas se ha observado un sobreuso de estos fármacos tanto a nivel extrahospitalario como hospitalario que ha producido un aumento importante en el gasto sanitario y un incremento en el riesgo de posibles efectos adversos. Es importante que los profesionales sanitarios se ajusten a las indicaciones admitidas y a las dosis correctas para el empleo de estos medicamentos. Existen en el mercado diferentes tipos de IBP: omeprazol, pantoprazol, lansoprazol, rabeprazol y esomeprazol. El omeprazol es el más antiguo y utilizado, siendo también el más barato. Si bien en la mayoría de las indicaciones terapéuticas en las que se emplean estos medicamentos no se describen diferencias entre los distintos IBP en la curación de las enfermedades, el esomeprazol, IBP de última generación, ha demostrado mayor eficacia en la erradicación del H. pylori y en la curación de la esofagitis grave respecto al resto de IBP. En los últimos años el uso de los fármacos genéricos se ha extendido; este tipo de medicamentos presentan la misma biodisponibilidad que los medicamentos originales. En el caso de los IBP, los pocos estudios comparativos disponibles en la literatura entre los fármacos originales y los genéricos no han demostrado diferencias significativas en la eficacia clínica.
Palabras clave: Inhibidores de la bomba de protones. IBP. Omeprazol. Pantoprazol. Lansoprazol. Rabeprazol. Esomeprazol.
Introduction
Proton-pump inhibitors (PPIs) represent a family of drugs widely used in our country. The advent in the nineties of PPIs was a great revolution in the treatment of ulcer disease and gastroesophageal reflux. The main problems of PPIs at present time are its overuse and the errors in the therapeutic indication. This fact leads to a considerable health economic cost and the risk of long-term side effects. In this review the main problems of PPIs prescription, their indications, the differences between PPIs and tips on their use are discussed.
Brief historical memory: since alkaline diets until PPIs
The treatment of ulcer disease and gastroesophageal reflux disease (GERD) in the early twentieth century was the prescription of alkaline foods like milk, eggs and puree (1). Later, there were products as sodium bicarbonate that improved symptoms but did not prevent complications. Surgical treatment consisted of different types of gastric surgeries for peptic ulcer disease and Nissen fundoplication for GERD (2).
In the mid twentieth century the use of muscarinic antagonists was introduced, atropine being its main active ingredient. Acids secretion was partially inhibited by blocking the muscarinic receptor of the parietal cell. However, these drugs had little effectiveness and numerous adverse effects due to the amount of muscarinic receptors distributed throughout the body. E-type prostaglandin was discovered later, but, again, the limitations of these substances were their short half life and side effects. An important progress in gastric antisecretory therapy was the appearance of H2 receptors antagonists (H2 blockers) (3). The most used were ranitidine and famotidine. These drugs established a substantial change by blocking the action of histamine receptor in gastric parietal cells and achieving effective control in the production of hydrochloric acid (4). However, despite their greater clinical efficacy, they did not attain a complete healing of the esophageal and gastric lesions because they were unable to completely hold back the gastric acid secretion (5).
The revolution in the management of peptic diseases occurred in the mid seventies, using pyridine-2-thioacetamide, a compound that was being studied for its possible antiviral effect. Modifying this substance the timoprazole was created, the first PPI which had a gastric antisecretory effect. In 1979 omeprazole was synthesized, it had higher affinity for the proton pump of the parietal cell and fewer adverse effects (6). In subsequent years most clinical efficacy of PPIs relative to H2 blockers was demonstrated and quickly new fields of research were opened, leading to the synthesis of new PPIs (pantoprazole, lansoprazole, rabeprazole and esomeprazole) with differential features to omeprazole.
Overuse and its consequences
The prescription of PPIs has increased in recent years. In 2010, omeprazole became the most active ingredient consumed in packs in Spain, representing 5.51% of total drug packaging invoiced in that year (7). In the subgroup of ulcer drugs between 2000 and 2008, the use of PPIs increased 200.8% and at the same time, the prescription of H2-blockers decreased. Among the different PPIs, in 2007 esomeprazole became the PPI with higher growth rates (8). Regarding other European countries, Spain is the leader in the use of anti-ulcers, with a prescription of 70% above the average (9).
The overuse of PPIs has been demonstrated in several studies (10-12). In a study conducted at the Hospital de Guadalajara, Martín-Echevarría et al. found that among patients taking PPIs at admission, 73% had an inadequate indication and 38% of patients were discharged with this medication even without correct indication (13). In outpatient clinics, a cross-sectional study conducted in the IV area of Madrid showed that only 36.4% of PPIs consumers meet the criteria of adequate indication of PPIs (14). In patients hospitalized in the US treated with gastric antisecretory there has been an inadequate indication in 60% of the cases; the most frequent prescription error was the prophylaxis of gastrointestinal lesions in patients with low gastrolesive risk (15,16).
Risk of side effects of PPIs increases with the overuse of PPIs. The most common are headache, abdominal pain, diarrhea, bloating, nausea and constipation. They usually are mild and reversible upon discontinuation of the drug. On the other hand, security with long-term use has been controversial in recent years.
Gastric acid is one of the barriers against infections of the upper gastrointestinal tract, for this reason hypochlorhydria has been associated with increased of enteric and respiratory infections caused by microaspiration. Enteric Campylobacter and Salmonella infections, sensitive acid bacteria, have been linked to the use of PPIs (17,18). Furthermore, it has been suggested that this drug could also increase the risk of Clostridium difficile colitis. One meta-analysis and one systematic review have both found this association, despite the high heterogeneity of the studies that did not include any clinical trial (19,20). Several epidemiological studies have linked PPIs to increased risk of community-acquired pneumonia, especially during the first days of prescription (21,22); in 2011 a meta-analysis confirmed the association with an OR of 1.27 (95% CI 1.11 to 1.26) (23). However, in more recent studies, this association has not been observed (24,25).
A recent case-control study describes that PPIs may increase the risk of cryptogenic liver abscesses (26). Bone fractures have also been linked in several observational studies (27). A crossover study analyzed bone mineral density of the hip and lumbar vertebrae, and it showed no association between PPI use and osteoporosis (28).
The absorption of vitamin B12 and magnesium requires gastric acid and PPIs could be responsible for producing deficiencies of these elements. In the case of vitamin B12, most of the studies are case-control and many of them have been questioned because the method of measurement of vitamin B12 is not taking into account the patient's diet (29,30). Furthermore, studies of low methodological consistency have associated PPI for over a year with hypomagnesaemia (31,32). The Food and Drug Administration (FDA) has issued a warning about it and recommends applying serum magnesium levels before starting long-term treatment with PPIs and monitoring magnesemia in patients with risk factors (33).
Recently, two multicenter clinical trials with 812 patients have been published. They were randomized to patients with GERD taking PPIs (esomeprazole or omeprazole) long-term or undergo a laparoscopic antireflux surgery with a follow-up of 5 and 12 years (34). Adverse hematologic effects were analyzed and analytical studies were performed with measurements of vitamin B12, homocysteine, folate, iron, ferritin, calcium and vitamin D. No relevant clinical and analytical differences between the two groups were observed.
Mechanism of action of PPIs
Gastric acid production is controlled by a series of endogenous mediators. Somatostatin and prostaglandins are the main inhibitors of the secretion, while the most important stimulators are acetylcholine, gastrin and histamine. The latter bind to a specific G protein-coupled receptor in the basolateral membrane of the parietal cell and by different routes, they activate ATPase-dependent proton pumps. This pump exchanges potassium with hydrogen and it is located in the apical membrane of the parietal cell (35).
The various drugs used to achieve an increase in the intragastric pH are based on the inhibition of this proton pump, binding to different receptors of the parietal cell. The differential and main characteristic of PPIs is their ability to bind directly and irreversibly to this proton pump. This pump is the last metabolic step in the production of hydrochloric acid and it is effectively inhibited. Despite being an irreversible bond, trying to reach 100% inhibition, high doses in short time intervals or continuous intravenous infusions of these drugs are needed. This is due to several factors: the short plasma half-life, the continuous synthesis of new proton pumps by the parietal cell and the fact that only active pumps that are exposed to canalicular lumen are blocked (36).
Through this mechanism, PPIs achieve inhibit basal gastric acid secretion and stimulation induced which appears with intake; reduce secreted pepsinogen and pepsin proteolytic activity. It has not been shown in vivo models that PPIs are able to eradicate Helicobacter pylori. However, in vitro studies have shown that these drugs inhibit the enzyme urease which protects the microorganism from an acidic pH (37). Furthermore, it has also been observed that PPIs can stimulate the bactericidal action of antibiotics such as clarithromycin (15).
To prevent inactivation of PPI in the stomach acid medium, these drugs require an enteric coated. After absorption in the duodenum, they reach the peak plasma concentration in 30 minutes-3 hours. Intake with food delays absorption. After a standard dose of PPI, the acid secretion is fully recovered after 72-96 hours and its bioavailability increases with repeated doses (36). The short plasma half-life of PPIs results in a low incidence of toxicity, even in situations of reduced clearance such as the elderly or patients with liver and/or kidney failure. Only in cases of marked decrease in liver function it will be necessary to reduce the dose of these medications (38).
PPIs are metabolized primarily in the liver by the action of cytochrome P450, mainly by CYP2C19 and CYP3A4 isozymes (39). Most PPIs have higher affinity for CYP2C19 than CYP3A4; this last is reserved when the main route is saturated. They have demonstrated that CYP2C19 polymorphisms produce alterations in the metabolism and life variations in drug plasma (40).
Depending on CYP2C19 phenotypes, there are fast or slow metabolizers. The so-called "fast metabolizers" are CYP2C19 homozygous and heterozygous mutation without a single mutation; and the so-called "poor metabolizers" are CYP2C19 with two copies of the mutated gene (41). Fast metabolizers correspond to 1% of the black population, 2-6% of the Caucasian population and 15-23% of the Asian population (42).
This group of patient metabolizes the drug faster, with a significant decrease in PPI blood concentration and the therapeutic response (40). It has been observed that poor metabolizers have higher H. pylori eradication rates compared to fast metabolizers. Moreover, in non-erosive GERD, reflux esophagitis and duodenal ulcer a variation in the therapeutic results has also been observed in function of the metabolizer phenotype. In our country, there are few fast metabolizers; however, in areas of East Asia they can reach 23% of the population and adjusting the dose according to the detected polymorphism could be an option, offering a more personalized therapeutic prescription (43).
Acid inhibition powerful and pharmacological differences between PPI
Depending on the therapeutic indication, the potency of inhibition of gastric acid secretion required will be different. Thus, although in some patients with GERD the levels of hydrochloric acid in the distal esophagus are within normal range, for symptomatic GERD a higher acid inhibition to achieve therapeutic success is required in comparison with peptic ulcer (44). In the treatment of upper gastrointestinal bleeding, gastric pH must be as high as possible to avoid degradation of the clot and accelerate healing and ulcer cicatrization (45).
There are different parameters that must be taken into consideration when the available PPIs are compared. Among them, the average time during which intragastric pH is greater than 4 is relatively sensitive to changes in pH within 24 hours and easily understood (45). In cases such as GERD or upper gastrointestinal bleeding it is necessary to obtain an intragastric pH above 4 for at least 16 hours a day (46).
Furthermore, inhibition of gastric acid secretion by PPIs is influenced by individual characteristics and external factors. Individual factors include interpersonal variation in metabolism of PPIs regarding polymorphisms and P2C19 cytochrome CYP3A4, as discussed previously. Among the external factors H. pylori infection is one of them. It has been observed that the degree of inhibition of gastric secretion with PPIs is greater in patients infected with H. pylori (47). It is thought that gastritis caused by H. pylori induces an overproduction of gastrin to activate parietal cells and proton pumps, increasing the action of PPIs. Furthermore, the ammonia produced by H. pylori could help PPIs to neutralize gastric acid (48).
Regarding the pharmacological differences between other gastric antisecretory, anti-H2 have a faster onset of pharmacological effect than PPIs. However, these drugs develop early tachyphylaxis and their antisecretory capacity decreases after the first 12-24 hours after administration (49). Currently, the use of anti-H2 is restricted to the management of mild non-erosive GERD, in some patients with functional dyspepsia, in patients with hypersensitivity to the PPIs and in patients who need to perform a diagnostic test for H. pylori infection and they require a sustained gastric antisecretory (36).
The different types of PPIs currently available have similar pharmacological characteristics. However, they can be differentiated by the equivalent dose, route of administration available, price or drug interactions. The equivalent dose, route of administration and duration of acid inhibition of different PPIs are shown in table I. Regarding the route of administration, in Spain omeprazole, pantoprazole and esomeprazole are marketed for intravenous use. While the price of the drug should not be the highest priority, it should be a factor to be considered. Using data from 2014, omeprazole is the cheapest PPI in the Spanish pharmaceutical market, followed by lansoprazole, pantoprazole, rabeprazole and esomeprazole. With regard to the differences in time action, it has been found that rabeprazole, compared with omeprazole, has a greater efficacy in reducing symptoms of severe GERD in the first three days of treatment (49), these differences disappear from the fourth day of administration. Regarding the duration of gastric acid inhibition, in a randomized crossover study by Miner et al. it was found that the average hours intragastric pH above 4 was 14 h with esomeprazole, 12.1 h with rabeprazole, 11.8 h with omeprazole, 11.5h with lansoprazole and 10.1 h with pantoprazole. Esomeprazole has a longer duration of gastric acid inhibition statistically significant than the other drugs (50).
Drug interactions with PPIs are few and clinically irrelevant. The affinity for both cytochrome P450 (CYP2C19 and CYP3A4) differs between the types of PPIs and this could theoretically produce slight differences in the interactions. Pantoprazole and rabeprazole have a non-enzymatic metabolism pathway (39) and for this reason, it has been suggested that these two drugs could have less interaction than omeprazole or lansoprazole. However, in clinical practice the risk of drug interaction is extremely low (51).
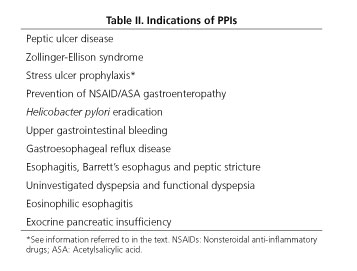
Indications of PPIs
Table II shows the indications of PPIs in Spain (52).
Gastroenteropathy by nonsteroidal antiinflammatory drugs (NSAIDs) prophylaxis is probably the PPI indication that produces most confusion among prescribers of this medicine. PPIs indications in these patients are shown in table III (53). Recently, in a meta-analysis with 8,780 participants of 10 clinical trials, it was confirmed that the use of PPIs decreases the risk of ulcers and gastrointestinal bleeding associated with the use of low-dose aspirin (54). This was also demonstrated with dual antiplatelet therapy with clopidogrel without causing an increase in cardiovascular events.
The use of PPIs in the prophylaxis of stress ulcer is limited to patients who are in intensive care and present risk factors for peptic ulcer (history of ulcer, acute renal failure, liver cirrhosis, coagulopathy, shock, severe sepsis, multiple organ failure, need for mechanical ventilation, head trauma, neurosurgery or mild burns) (52).
In upper gastrointestinal bleeding due to peptic ulcer with high risk of rebleeding, after endoscopic effective treatment, continuous infusion PPIs have been shown to reduce recurrent bleeding compared to a placebo (55). In major clinical guidelines, the use of one or two intravenous bolus dose of PPIs followed by a continuous infusion of 8 mg/h has been recommended. This pattern has been questioned following a recent meta-analysis comparing intermittent administration of PPIs versus continuous perfusion. It shows that intermittent dose is comparable to continuous infusion in relation to the risk of rebleeding, mortality and need for transfusion (56). However, this study has methodological limitations and heterogeneity in the frequency and dose of drug with comparative studies (57). Furthermore, it has been also reported that administration of oral and intravenous PPIs achieved similar rates of rebleeding, mortality, need for hospitalization and surgery after bleeding for high risk peptic ulcer (58).
GERD symptoms are often more difficult to control in patients without esophageal lesions (59). In many patients with erosive GERD, PPIs fail to achieve complete healing of esophageal lesions. It has been described that over 20% of patients with GERD have an inadequate response to taking omeprazole 20 mg twice per day (60). A high dose of omeprazole (80 mg/day) improves the symptoms on refractory patients (60). One of the probable causes of failure in the effectiveness of standard dose is the night event of acid escape, when a suitable acid inhibition does not occur for long periods (61). However, even today it remains unclear the clinical significance of this phenomenon and whether it is related to the lack of therapeutic response to these medications. Other causes of refractoriness to the treatment include poor compliance, hypotensive lower esophageal sphincter and ineffective esophageal peristalsis (62).
One of the tested treatments to treat dyspepsia (not investigated and functional) is the use of PPIs (63). Several systematic reviews have shown that PPIs are superior to a placebo in the disappearance or improvement of symptoms of dyspepsia (64). The highest efficacy was observed in patients with ulcer or gastroesophageal reflux symptoms. No differences were shown in the group with dysmotility-like dyspepsia (65). Although clinically relevant outcomes do not exist, the use of PPIs is recommended in these patients for several reasons: they are drugs with few side effects and patients misclassified as functional dyspepsia that really present illness peptic ulcer or GERD can benefit from this treatment (36,66).
In eosinophilic esophagitis, PPIs are useful for the differential diagnosis of GERD and PPI-responsive esophageal eosinophilia (67,68). In exocrine pancreatic insufficiency, the use of these drugs has increased due to the refractoriness of enzyme replacement therapy. Associating a PPI to enzymatic treatment, the pH increases and lipase degradation decreases secondarily (69).
Clinical differences between PPIs
Although there are, as mentioned above, certain pharmacological disparities between PPIs, the impact of these differences in their effectiveness for healing various diseases is difficult to establish by the scarcity of quality comparative studies. However, there are data that suggest under certain diseases the choice of either PPI.
In H. pylori infection it has been shown that PPIs increase eradication rates of this microorganism (70). The potent inhibition of gastric acid secretion induces antibiotics stability in acidic gastric environment and increases the sensitivity of some antibiotics against the bacteria (45). Next-generation PPIs, rabeprazole and esomeprazole, have shown greater H. pylori inhibitory potential than first-generation ones (71-73). A recent meta-analysis shows that the use of next-generation PPIs (rabeprazole and esomeprazole) in standard triple therapy gets higher eradication rates than first generation PPIs (74). In addition, this superiority was higher when comparing esomeprazole managed to double dose with first generation PPIs. However, no statistically significant differences between rabeprazole and esomeprazole were observed. Moreover, in this same study, rates of eradication of the new generation PPIs were not affected by the phenotype of CYP2C19, while the first generation had a tendency to lower rates eradication in fast metabolizers of PPI. This suggests that any standard dose PPI produces maximal inhibition in poor metabolizers, while fast metabolizers high doses of PPIs are required to achieve the same degree of inhibition (74). For this reason, new generation PPIs can be an attractive option in countries with high rate metabolizers.
In the treatment of GERD in patients with typical symptoms of the disease and no evidence of clinical alarm, empirical treatment with standard dose PPI for 2-4 weeks is recommended (63). However, approximately 75% of patients with typical symptoms of GERD do not have erosive reflux disease and severe symptoms are not predictive of erosive esophagitis (75). In patients with GERD, there has been seen a faster clinical improvement during the first five days with esomeprazole 40 mg versus omeprazole 20 mg, lansoprazole 30 mg and pantoprazole 40 mg; however, these differences are not maintained from the fifth day of treatment (76). Eggleston et al. compared rabeprazole 20 mg, esomeprazole 20 mg and 40 mg in the treatment of GERD. No significant differences in resolution of symptoms between rabeprazole 20 mg, esomeprazole 20 mg and 40 mg esomeprazole were identified (75). Regarding the refractory GERD, a recent study evaluates the effect of the change of PPI, noting that 28.4% of patients had no improvement after administration of omeprazole 20 mg daily for 8 weeks and when in this group of patients omeprazole was replaced by rabeprazole 20 mg daily, clinical improvement was observed in only 23.5% (77).
In relation to the erosive GERD, in a recent systematic review different types of PPI were compared in patients with severe erosive esophagitis grades C and D of the Los Angeles classification, esomeprazole 40 mg showed a substantial advantage over four PPIs (omeprazole 20 mg, omeprazole 40 mg, pantoprazole 40 mg and lansoprazole 30 mg) for cure rates at 4 and 8 weeks (78). Low doses of PPI compared with standard doses are not equally effective in healing erosive esophagitis or symptomatic GERD (79). However, in maintenance treatment of GERD there is similar efficacy.
In the maintenance treatment of peptic ulcer it has been found that low doses of PPI have similar effects to standard doses (79). In healing rates of ulcers there are not statistically significant differences between omeprazole, pantoprazole, lansoprazole and rabeprazole (80). There are no studies comparing esomeprazole with other PPIs.
Original, generic, copy and similar drugs
In the pharmaceutical market there are many commercial names for PPI: original, generic and similar PPI. Subjective clinical differences observed by some clinicians in daily clinical practice with these different types of PPI have questioned the use of generic drugs. It is important to clarify the conceptual differences between the various types of pharmaceutical preparations of PPI:
- Original drugs are new molecules that have passed through all stages of research required for the development of a new active substance.
- Generic drugs have the same active ingredient, dose and pharmaceutical form as the original drug and have proved therapeutic equivalence, that is, to be bioequivalent to the original medicine. Bioequivalence studies analyze the area under the curve (AUC), and to be considered bioequivalent, the AUC of the new drug compared to the reference product should be between 80 and 125%. In the Spanish market, generics are distinguished by presenting the initials EFG (Generic Pharmaceutical Specialty) in the container and general labeling (81).
- Another specialty is called copies, drugs with the same active ingredient as the original but that have not proved therapeutic equivalence and, therefore, excipients or differences in the process of development can determine differences in the behavior of the drug. The royal decree of 1993 governing the registration of medicines demanded proof that any drug that registers must show therapeutic equivalence to the reference product. Omeprazole patent remained in force in Europe until 1999, so all registered drugs have had to demonstrate bioequivalence and theoretically there should not be copies but PPI called similar. Similar drugs have demonstrated bioequivalence but do not have the initials EFG.
There are very few studies that have investigated the clinical differences between original and generic PPIs. The most notable one is a retrospective study with a sample of 450 patients conducted in Israel that compared the original omeprazole (Losec®) and generic (Omepradex®) in eradication of H. pylori infection with standard triple therapy that found no statistically significant differences (82). There are no comparative trials between original and similar drugs.
Tips to optimize ppi treatment
Despite the widespread use of PPIs, lack of knowledge in the administration dose or schedule may reduce its effectiveness. It is therefore important to explain to patients when and how to take PPIs.
It is recommended to administer PPI fasting and about 30 minutes before a meal, as the presence of food in the stomach reduces its absorption. Furthermore, fasting administration makes the peak plasma concentration match the maximum number of activated proton pump. In nocturnal GERD, it has been found that omeprazole administration before dinner reaches greater inhibitory levels than its administration before breakfast (83). Moreover, in patients with GERD administration twice daily is more effective than once, although it can hinder adherence.
In relation to the dose, PPI dose must be individualized according to clinical indication and therapeutic response. In GERD, doses up to 80 mg a day of omeprazole can be achieved depending on the intensity of symptoms (60). Esomeprazole 40 mg twice daily could achieve higher responses than omeprazole 80 mg/day (36). It has been suggested that monitoring gastric pH during treatment may help to identify non responders to the prescribed dose of PPI and to increase the dose to achieve adequate control, defined as a pH greater than 4 at least 16 hours. However, this clinical practice systematically is not recommended due to the discomfort of the test and the cost of it (36).
In H. pylori eradication it is currently recommended to use double doses of PPI to optimize the eradication efficacy (84). A recent meta-analysis has shown that double dose PPI increases cure rate by 8% compared with the use of standard dose (85). Another meta-analysis has shown that when PPI is administered every 12 hours cure rates are also higher than when administered once daily (86).
As already mentioned, interactions of PPIs with other drugs are scarce. Nevertheless, the prescription of PPIs with clopidogrel has been disputed due to suspicion of an increase in cardiovascular events (87). This fact conditioned sending of communications by the FDA and the European Medicines Agency (EMEA) warning about this risk and even discouraging the concomitant use of PPIs and clopidogrel in patients with cardiovascular risk. The Spanish Medicines Agency advises against using clopidogrel with omeprazole and esomeprazole, despite the lack of solid studies, and the FDA recommended pantoprazole as an alternative in these patients. In 2012 a meta-analysis found that concomitant use of PPIs with clopidogrel in patients with coronary disease was associated with an increase in acute coronary syndrome or major cardiovascular events. However, the study concluded that there was insufficient evidence to say anything for drug interactions (87). Given the absence of definitive evidence, it seems prudent to weigh the gastrointestinal and cardiovascular risks in each patient. When there is an obvious gastrointestinal risk, we must exert a prevention action with PPI. It must also perform other actions directed to minimize possible interactions such as spacing the administration of clopidogrel and PPI over time, tomorrow vs. night (88).
Conclusions
PPIs are a family of highly effective drugs for the treatment and prevention of common and significant gastrointestinal diseases related to gastric acid secretion. They have good pharmacological and safety profile even when administered for long periods of time. This profile has led to an overuse of these drugs both in ambulatory care and in hospital setting; therefore, it required to use conforms to the stated guidelines based on clinical evidence. While different molecules of PPI have shown high clinical efficacy in many indications, last generation of PPI has been more effective in eradication treatment of H. pylori infection and severe reflux esophagitis. There are very few studies that have examined the efficacy of the original PPI compared to generic preparations of these drugs and so far they have not shown differences in clinical use. In the coming years we will have new molecules of gastric antisecretory drugs that will further enhance the excellent pharmacological and clinical profile of PPIs.
References
1. Díaz-Rubio M. Historical perspective of gastric acid inhibition. Drugs 2005;65(Suppl1):1-6. [ Links ]
2. Banic M, Malfertheiner P, Babic Z, et al. Historical impact to drive research in peptic ulcer disease. Dig Dis 2011;29:444-53. DOI: 10.1159/000331512. [ Links ]
3. Black JW, Duncan WA, Durant CJ, et al. Definition and antagonism of histamine H2-receptors. Nature 1972;236:385-90. DOI: 10.1038/236385a0. [ Links ]
4. Martín de Argila de Prados C. Crónica de una revolución: Manejo de las enfermedades relacionadas con la secreción ácida del estómago. Línea de comunicación 2015, Madrid (en imprenta). [ Links ]
5. Vela MF. Medical treatments of GERD: The old and new. Gastroenterol Clin North Am 2014;43:121-33. DOI: 10.1016/j.gtc.2013.12.001. [ Links ]
6. Sachs G. Pump blockers and ulcer disease. N Engl J Med 1984;310:785-6. DOI: 10.1056/NEJM198403223101211. [ Links ]
7. Subgrupos ATC y principios activos de mayor consumo en el Sistema Nacional de Salud en 2010. Información terapéutica del Sistema Nacional de Salud 2011;35:124-128. [ Links ]
8. García del Pozo J. Estudio de utilización de antiulcerosos en España (2000-2008). Información terapéutica de la Seguridad Social 2009; 33:49-54. [ Links ]
9. Simo Miñana J. Utilización de medicamentos en España y en Europa. Aten Primaria 2012;44:335-47. DOI: 10.1016/j.aprim.2011.06.009. [ Links ]
10. Sánchez-Cuen JA, Irineo-Cabrales AB, Bernal-Magana G, et al. Inadequate prescription of chronic consumption of proton pump inhibitors in a hospital in Mexico. Cross-sectional study. Rev Esp Enferm Dig 2013;105:131-6. DOI: 10.4321/S1130-01082013000300003. [ Links ]
11. Ponce J, Esplugues JV. Rationalizing the use of PPIs: An unresolved matter. Rev Esp Enferm Dig 2013;105:121-4. DOI: 10.4321/S1130-01082013000300001. [ Links ]
12. Ksiadzyna D, Szelag A, Paradowski L. Overuse of proton pump inhibitors. Pol Arch Med Wewn 2015 (en imprenta). [ Links ]
13. Martín-Echevarría E, Pereira Juliá A, Torralba M, et al. Evaluación del uso de los inhibidores de la bomba de protones en un servicio de medicina interna. Rev Esp Enferm Dig 2008;100:76-81. DOI: 10.4321/S1130-01082008000200003. [ Links ]
14. De Burgos Lunar C, Novo del Castillo S, Llorente Díaz E, et al. Estudio de prescripción-indicación de inhibidores de la bomba de protones. Rev Clin Esp 2006;206:266-70. DOI: 10.1157/13088585. [ Links ]
15. Zink DA, Pohlman M, Barnes M, et al. Long-term use of acid suppression started inappropriately during hospitalization. Aliment Pharmacol Ther 2005;21:1203-9. DOI: 10.1111/j.1365-2036.2005.02454.x. [ Links ]
16. Eid SM, Boueiz A, Paranji S, et al. Patterns and predictors of proton pump inhibitor overuse among academic and non-academic hospitalists. Intern Med 2010;49:2561-8. DOI: 10.2169/internalmedicine.49.4064. [ Links ]
17. García Rodríguez LA, Ruigómez A, Panes J. Use of acid-suppressing drugs and the risk of bacterial gastroenteritis. Clin Gastroenterol Hepatol 2007;5:1418-23. DOI: 10.1016/j.cgh.2007.09.010. [ Links ]
18. Bavishi C, Dupont HL. Systematic review: The use of proton pump inhibitors and increased susceptibility to enteric infection. Aliment Pharmacol Ther 2011;34:1269-1281. DOI: 10.1111/j.1365-2036.2011.04874.x. [ Links ]
19. Janarthanan S, Ditah I, Adler DG, et al. Clostridium difficile-associated diarrhoea and proton pump inhibitor therapy: A meta-analysis. Am J Gastroenterol 2012;107:1001-10. DOI: 10.1038/ajg.2012.179. [ Links ]
20. Leonard J, Marshall JK, Moayyedi P. Systematic review of the risk of enteric infection in patients taking acid suppression. Am J Gastroenterol 2007;102:2047-56. DOI: 10.1111/j.1572-0241.2007. 01275.x. [ Links ]
21. Laheij RJ, Sturkenboom MC, Hassing RJ, et al. Risk of community-acquired pneumonia and use of gastric acidsuppressive drugs. JAMA 2004;292:1955-60. DOI: 10.1001/jama.292.16.1955. [ Links ]
22. Gulmez SE, Holm A, Frederiksen H, et al. Use of proton pump inhibitors and the risk of community-acquired pneumonia: A population-based case-control study. Arch Intern Med 2007;167:950-5. DOI: 10.1001/archinte.167.9.950. [ Links ]
23. Eom CS, Jeon CY, Lim JW, et al. Use of acid-suppressive drugs and risk of pneumonia: A systematic review and metaanalysis. CMAJ 2011;183:310-19. DOI: 10.1503/cmaj.092129. [ Links ]
24. Dublin S, Walker RL, Jackson ML, et al. Use of proton pump inhibitors and H2 blockers and risk of pneumonia in older adults: A population-based case control study. Pharmacoepidemiol Drug Saf 2010;19:792-802. DOI: 10.1002/pds.1978. [ Links ]
25. Filion KB, Chateau D, Targownik LE, et al. Proton pump inhibitors and the risk of hospitalization for community-acquired pneumonia: Replicated cohort studies with meta-analysis. Gut 2014;63:552-8. DOI: 10.1136/gutjnl-2013-304738. [ Links ]
26. Wang YP, Liu CJ, Chen TJ, et al. Proton pump inhibitor use significantly increases the risk of cryptogenic liver abscess: A population-based study. Aliment Pharmacol Ther 2015;41:1175-81. DOI: 10.1111/apt.13203. [ Links ]
27. Khalili H, Huang ES, Jacobson BC, et al. Use of proton pump inhibitors and risk of hip fracture in relation to dietary and lifestyle factors: A prospective cohort study. BMJ 2012;344:e372. DOI: 10.1136/bmj.e372. [ Links ]
28. Targownik LE, Lix LM, Leung S, et al. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology 2010;138:896-904. DOI: 10.1053/j.gastro.2009.11.014. [ Links ]
29. Lam JR, Schneider JL, Zhao W, et al. Gastric acid-inhibiting medications and vitamin B12 deficiency. JAMA 2013;310:2435-42. DOI: 10.1001/jama.2013.280490. [ Links ]
30. Jung SB, Nagaraja V, Kapur A, et al. Association between vitamin B12 deficiency and long-term use of acid-lowering agents: A systematic review and meta-analysis. Intern Med J 2015;45:409-16. DOI: 10.1111/imj.12697. [ Links ]
31. Danziger J, William JH, Scott DJ, et al. Proton-pump inhibitor use is associated with low serum magnesium concentrations. Kidney Int 2013;83:692-699. DOI: 10.1038/ki.2012.452. [ Links ]
32. Sumukadas D, McMurdo MET, Habicht D. Proton pump inhibitors are associated with lower magnesium levels in older people with chronic kidney disease. J Am Geriatr Soc 2012;60:392-3. DOI: 10.1111/j.1532-5415.2011.03808.x. [ Links ]
33. FDA Drug Safety Communication. Low magnesium levels can be associated with long term use of proton pump inhibitor drugs (PPIs). Washington DC, US. Food and Drug Administration, 2011. [ Links ]
34. Attwood SE, Ell C, Galmiche JP, et al. Long-term safety of proton pump inhibitor therapy assessed under controlled, randomized clinical trial conditions: Data from the SOPRAN and LOTUS studies. Aliment Pharmacol Ther 2015;41:1162-74. DOI: 10.1111/apt.13194. [ Links ]
35. Esplugues JV. A pharmacological approach to gastric acid inhibition. Drugs 2005;65(Suppl1):7-12. DOI: 10.2165/00003495-200565001-00003. [ Links ]
36. Gomollon F, Calvet X. Optimizing acid inhibition treatment. Drugs 2005;65(Suppl1):25-33. DOI: 10.2165/00003495-200565001-00004. [ Links ]
37. Sjostrom JE, Larsson H. Factors affecting growth and antibiotic susceptibility of Helicobacter pylori: Effect of pH and urea on the survival of a wild-type strain and a urease-deficient mutant. J Med Microbiol 1996;44:425-33. DOI: 10.1099/00222615-44-6-425. [ Links ]
38. Robinson M, Horn J. Clinical pharmacology of proton pump inhibitors: What the practicing physician needs to know. Drugs 2003;63:2739-54. DOI: 10.2165/00003495-200363240-00004. [ Links ]
39. Martín de Argila C. Safety of potent gastric acid inhibition. Drugs 2005;65(Suppl1):97-104. DOI: 10.2165/00003495-200565001-00013. [ Links ]
40. Kuo CH, Lu CY, Shih HY, et al. CYP2C19 polymorphism influences Helicobacter pylori eradication. World J Gastroenterol 2014;20:16029-36. [ Links ]
41. Inaba T, Mizuno M, Kawai K, et al. Randomized open trial for comparison of proton pump inhibitors in triple therapy for Helicobacter pylori infection in relation to CYP2C19 genotype. J Gastroenterol Hepatol 2002;17:748-53. DOI: 10.1046/j.1440-1746.2002.02790.x. [ Links ]
42. Ishizaki T, Horai Y. Review article: Cytochrome P450 and the metabolism of proton pump inhibitors - Emphasis on rabeprazole. Aliment Pharmacol Ther 1999;13(Suppl3):27-36. DOI: 10.1046/j.1365-2036.1999.00022.x. [ Links ]
43. Furuta T, Sugimoto M, Shirai N, et al. CYP2C19 pharmacogenomics associated with therapy of Helicobacter pylori infection and gastro-esophageal reflux diseases with a proton pump inhibitor. Pharmacogenomics 2007;8:1199-210. DOI: 10.2217/14622416.8.9.1199. [ Links ]
44. Orlando RC. Why is the high grade inhibition of gastric acid secretion afforded by proton pump inhibitors often required for healing of reflux esophagitis? An epithelial perspective. Am J Gastroenterol 1996;91:1692-6. [ Links ]
45. Calvet X, Gomollon F. What is potent acid inhibition, and how can it be achieved? Drugs 2005;65(Suppl1):13-23. DOI: 10.2165/00003495-200565001-00004. [ Links ]
46. Hatlebakk JG. Review article: gastric acidity - Comparison of esomeprazole with other proton pump inhibitors. Aliment Pharmacol Ther 2003;17(Suppl1):10-5. DOI: 10.1046/j.1365-2036.17.s1.3.x. [ Links ]
47. Verdu EF, Armstrong D, Idstrom JP, et al. Intragastric pH during treatment with omeprazole: Role of Helicobacter pylori and H. pylori-associated gastritis. Scand J Gastroenterol 1996;31:1151-6. [ Links ]
48. Labenz J, Tillenburg B, Peitz U, et al. Efficacy of omeprazole one year after cure of Helicobacter pylori infection in duodenal ulcer patients. Am J Gastroenterol 1997;92:576-81. [ Links ]
49. Holtmann G, Bytzer P, Metz M, et al. A randomized, double-blind, comparative study of standard-dose rabeprazole and high-dose omeprazole in gastro-oesophageal reflux disease. Aliment Pharmacol Ther 2002;16:479-85. DOI: 10.1046/j.1365-2036.2002.01207.x. [ Links ]
50. Miner PJr, Katz PO, Chen Y, et al. Gastric acid control with esomeprazole, lansoprazole, omeprazole, pantoprazole, and rabeprazole: A five-way crossover study. Am J Gastroenterol 2003;98:2616-20. DOI: 10.1111/j.1572-0241.2003.08783.x. [ Links ]
51. Labenz J, Petersen KU, Rösch W, et al. A summary of food and drug administration-reported adverse events and drug interactions occurring during therapy with omeprazole, lansoprazole and pantoprazole. Aliment Pharmacol Ther 2003;17:1015-9. DOI: 10.1046/j.1365-2036.2003.01550.x. [ Links ]
52. Gisbert JP, Martín de Argila de Prados C. Tratamiento de las enfermedades gastroenterológicas. Asociación Española de Gastroenterología. En: Ponce García J, editor. Úlcera péptica e infección por Helicobacter pylori. Barcelona: Elsevier; 2011. p. 109-21. [ Links ]
53. Arroyo Villarino MT, Lanas Arbeloa A. Tratamiento de las enfermedades gastroenterológicas. Asociación Española de Gastroenterología, Ponce García J, editores. Gastroenteropatía por AINE. Barcelona: Elsevier; 2011. p. 123-31. [ Links ]
54. Mo C, Sun G, Lu ML, et al. Proton pump inhibitors in prevention of low-dose aspirin-associated upper gastrointestinal injuries. World J Gastroenterol 2015;21:5382-92. DOI: 10.3748/wjg.v21.i17.5382. [ Links ]
55. Sung JJ, Barkun A, Kuipers EJ, et al. Intravenous esomeprazole for prevention of recurrent peptic ulcer bleeding: A randomized trial. Ann Intern Med 2009;150:455-64. DOI: 10.7326/0003-4819-150-7-200904070-00105. [ Links ]
56. Sachar H, Vaidya K, Laine L. Intermittent vs. continuous proton pump inhibitor therapy for high-risk bleeding ulcers: A systematic review and meta-analysis. JAMA Intern Med 2014;174:1755-62. DOI: 10.1001/jamainternmed.2014.4056. [ Links ]
57. Hsu YC, Lin HJ. Intermittent bolus or continuous infusion of proton pump inhibitors for ulcer bleeding? JAMA Intern Med 2015;175:461. [ Links ]
58. Yen HH, Yang CW, Su WW, et al. Oral versus intravenous proton pump inhibitors in preventing re-bleeding for patients with peptic ulcer bleeding after successful endoscopic therapy. BMC Gastroenterol 2012;12:66. DOI: 10.1186/1471-230X-12-66. [ Links ]
59. Dean BB, Gano AD, Knight K, et al. Effectiveness of proton pump inhibitors in non-erosive reflux disease. Clin Gastroenterol Hepatol 2004;2:656-64. DOI: 10.1016/S1542-3565(04)00288-5. [ Links ]
60. Leite LP, Johnston BT, Just RJ, et al. Persistent acid secretion during omeprazole therapy: A study of gastric acid profiles in patients demonstrating failure of omeprazole therapy. Am J Gastroenterol 1996;91:1527-31. [ Links ]
61. Badillo R, Francis D. Diagnosis and treatment of gastroesophageal reflux disease. World J Gastrointest Pharmacol Ther 2014;5:105-12. DOI: 10.4292/wjgpt.v5.i3.105. [ Links ]
62. Subramanian CR, Triadafilopoulos G. Refractory gastroesophageal reflux disease. Gastroenterol Rep (Oxf) 2015;3:41-53. DOI: 10.1093/gastro/gou061. [ Links ]
63. Gisbert JP, Calvet X, Ferrandiz J, et al. Guía de práctica clínica sobre el manejo del paciente con dispepsia. Actualizacion 2012. Gastroenterol Hepatol 2012;35:725e1-38. DOI: 10.1016/j.gastrohep.2012.05.002. [ Links ]
64. Soo S, Moayyedi P, Deeks J, et al. Pharmacological interventions for non-ulcer dyspepsia. The Cochrane database of systematic reviews 2000; CD001960. [ Links ]
65. Talley NJ, Lauritsen K. The potential role of acid suppression in functional dyspepsia: The BOND, OPERA, PILOT, and ENCORE studies. Gut 2002;50(Suppl4):36-41. DOI: 10.1136/gut.50.suppl_4.iv36. [ Links ]
66. Mascort JJ, Marzo M, Alonso-Coello P, et al. Guía de práctica clínica sobre el manejo del paciente con dispepsia. Gastroenterol Hepatol 2003;26:571-613. DOI: 10.1016/S0210-5705(03)70414-2. [ Links ]
67. Dellon ES. Diagnosis and management of eosinophilic esophagitis. Clin Gastroenterol Hepatol 2012;10:1066-78. DOI: 10.1016/j.cgh.2012.06.003. [ Links ]
68. Molina-Infante J, Katzka DA, Dellon ES. Proton pump inhibitor-responsive esophageal eosinophilia: A historical perspective on a novel and evolving entity. Rev Esp Enferm Dig 2015;107:29-36. [ Links ]
69. Lindkvist B. Diagnosis and treatment of pancreatic exocrine insufficiency. World J Gastroenterol 2013;19:7258-66. DOI: 10.3748/wjg.v19.i42.7258. [ Links ]
70. Boparai V, Rajagopalan J, Triadafilopoulos G. Guide to the use of proton pump inhibitors in adult patients. Drugs 2008;68:925-47. DOI: 10.2165/00003495-200868070-00004. [ Links ]
71. Kirchheiner J, Glatt S, Fuhr U, et al. Relative potency of proton-pump inhibitors-comparison of effects on intragastric pH. Eur J Clin Pharmacol 2009;65:19-31. DOI: 10.1007/s00228-008-0576-5. [ Links ]
72. McKeage K, Blick SK, Croxtall JD, et al. Esomeprazole: A review of its use in the management of gastric acid-related diseases in adults. Drugs 2008;68:1571-607. DOI: 10.2165/00003495-200868110-00009. [ Links ]
73. Baldwin CM, Keam SJ. Rabeprazole: A review of its use in the management of gastric acid-related diseases in adults. Drugs 2009;69:1373-401. DOI: 10.2165/00003495-200969100-00007. [ Links ]
74. McNicholl AG, Linares PM, Nyssen OP, et al. Meta-analysis: Esomeprazole or rabeprazole vs. first-generation pump inhibitors in the treatment of Helicobacter pylori infection. Aliment Pharmacol Ther 2012;36:414-25. DOI: 10.1111/j.1365-2036.2012.05211.x. [ Links ]
75. Eggleston A, Katelaris PH, Nandurkar S, et al. Clinical trial: The treatment of gastro-oesophageal reflux disease in primary care - Prospective randomized comparison of rabeprazole 20 mg with esomeprazole 20 and 40 mg. Aliment Pharmacol Ther 2009;29:967-78. DOI: 10.1111/j.1365-2036.2009.03948.x. [ Links ]
76. Zheng RN. Comparative study of omeprazole, lansoprazole, pantoprazole and esomeprazole for symptom relief in patients with reflux esophagitis. World J Gastroenterol 2009;15:990-5. DOI: 10.3748/wjg.15.990. [ Links ]
77. Takeuchi T, Oota K, Harada S, et al. Characteristics of refractory gastroesophageal reflux disease (GERD) symptoms - Is switching proton pump inhibitors based on the patient's CYP2C19 genotype an effective management strategy? Intern Med 2015;54:97-105. [ Links ]
78. Edwards SJ, Lind T, Lundell L, et al. Systematic review: Standard and double-dose proton pump inhibitors for the healing of severe erosive oesophagitis - A mixed treatment comparison of randomized controlled trials. Aliment Pharmacol Ther 2009;30:547-56. DOI: 10.1111/j.1365-2036.2009.04077.x. [ Links ]
79. Vakil N, Fennerty MB. Direct comparative trials of the efficacy of proton pump inhibitors in the management of gastro-oesophageal reflux disease and peptic ulcer disease. Aliment Pharmacol Ther 2003;18:559-68. DOI: 10.1046/j.1365-2036.2003.01756.x. [ Links ]
80. Klok RM, Postma MJ, Van Hout BA, et al. Meta-analysis: Comparing the efficacy of proton pump inhibitors in short-term use. Aliment Pharmacol Ther 2003;17:1237-45. DOI: 10.1046/j.1365-2036.2003.01562.x. [ Links ]
81. Díez MV, Errecalde MF. Aclaraciones al concepto de genérico. Información terapéutica del Sistema Nacional de Salud 1998;22:68-72. [ Links ]
82. Niv Y. Comparison of proton pump inhibitor-based triple therapy with losec and the generic drug, Omepradex, for efficacy of Helicobacter pylori eradication. Dig Dis Sci 2005;50:623-5. DOI: 10.1007/s10620-005-2546-4. [ Links ]
83. Hatlebakk JG, Katz PO, Kuo B, et al. Nocturnal gastric acidity and acid breakthrough on different regimens of omeprazole 40 mg daily. Aliment Pharmacol Ther 1998;12:1235-40. DOI: 10.1046/j.1365-2036.1998.00426.x. [ Links ]
84. Gisbert JP, Calvet X, Bermejo F, et al. III Conferencia Española de Consenso sobre la infección por Helicobacter pylori. Gastroenterol Hepatol 2013;36:340-74. DOI: 10.1016/j.gastrohep.2013.01.011. [ Links ]
85. Villoria A, Garcia P, Calvet X, et al. Meta-analysis: High-dose proton pump inhibitors vs. standard dose in triple therapy for Helicobacter pylori eradication. Aliment Pharmacol Ther 2008;28:868-77. [ Links ]
86. Vallve M, Vergara M, Gisbert JP, et al. Single vs. double dose of a proton pump inhibitor in triple therapy for Helicobacter pylori eradication: A meta-analysis. Aliment Pharmacol Ther 2002;16:1149-56. DOI: 10.1046/j.1365-2036.2002.01270.x. [ Links ]
87. Lanas A. Inhibidores de la bomba de protones y clopidogrel en el paciente cardiovascular: ¿Riesgo cardiovascular versus riesgo gastrointestinal? Gastroenterol Hepatol 2010;33:1-5. [ Links ]
88. Huang B, Huang Y, Li Y, et al. Adverse cardiovascular effects of concomitant use of proton pump inhibitors and clopidogrel in patients with coronary artery disease: A systematic review and meta-analysis. Arch Med Res 2012;43:212-24. DOI: 10.1016/j.arcmed.2012.04.004. [ Links ]
![]() Correspondence:
Correspondence:
Lara Aguilera Castro.
Department of Gastroenterology and Hepatology.
Hospital Universitario Ramón y Cajal, IRYCIS.
Ctra. Colmenar Viejo, km. 9,100.
28034 Madrid, Spain
e-mail: laraaguilera88@hotmail.com
Received: 16-04-2015
Accepted: 12-06-2015











 texto en
texto en