Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.108 no.10 Madrid oct. 2016
https://dx.doi.org/10.17235/reed.2016.3671/2015
ORIGINAL PAPERS
Management of refractory esophageal stenosis in the pediatric age
Manejo de las estenosis esofágicas refractarias en edad pediátrica
Fernando Alberca-de-las-Parras1, María Navalón-Rubio2 and Juan Egea-Valenzuela1
1Department of Digestive Diseases. Unit of Gastrointestinal Endoscopy.
2Department of Pediatrics. Unit of Pediatric Gastroenterology. Hospital Clínico Universitario Virgen de la Arrixaca. Murcia, Spain.
ABSTRACT
Introduction: Refractory esophageal stenosis (RES) is a major health problem in the pediatric population. Several techniques such as stent placement or C-mitomycin (CM) have been described as alternative treatments. We present our experience with both techniques, in our case with biodegradable stents (BS) and sometimes the association with stents and CM.
Material and methods: Six patients have been included: 2 post-operative fistulas in patients with type I esophageal atresia; 1 operated atresia without fistula; and 3 caustic strictures. 5 BS were placed in 4 children: 3 of them in cases of atresia (2 prosthesis in one case) and the other one in a case of stricture. CM was used in 5 cases: in 2 of them from the beginning, and in the other 3 cases after failure of the stent.
Results: When placed in fistulas, BS were fully covered. One of them successfully treated the fistula, but the other one was not effective. One stenosis was successfully treated with SB (in the case of persistent fistula), but recurrence was observed in the other 2 cases. One of these was solved with CM, and the other one needed a second stent. In the remaining 2 cases (one atresia and one caustic stricture) CM was effective after 1 and 2 sessions respectively. Overall, 5 out of 6 stenosis have been successfully treated (83.3%), and 1 out of 2 fistulas (50%).
Conclusions: Association of BS and CM has been effective in the management of RES in children.
Key words: Esophageal stenosis. Pediatrics. Biodegradable stent. Mitomycin.
RESUMEN
Introducción: las estenosis esofágicas refractarias (EER) a las dilataciones suponen un importante problema de salud en la pediatría. Se han descrito técnicas alternativas como la colocación de prótesis y el uso de mitomicina C (MC). Presentamos nuestra experiencia en una serie consecutiva con ambas técnicas, en nuestro caso con prótesis biodegradables (PB) y a veces ambas técnicas asociadas.
Material y métodos: se incluyeron 6 pacientes, dos con fístula postquirúrgica en atresias esofágicas tipo 1 operadas, otro con atresia operada sin fístula y otros tres con estenosis caústicas. Se colocaron 5 PB en 4 niños, 3 en atresias operadas (en un paciente 2 prótesis) y 1 en estenosis. Dos de los niños con atresia tenían fístula asociada.<>Se utilizó MC en 5 casos, en dos de forma primaria y en tres tras recidiva al fracasar la prótesis.
Resultados: las PB colocadas en las fístulas fueron recubiertas y en un caso solucionaron la fístula y en otro no. En las estenosis fueron efectivas en solo una (con la fístula no resuelta), recidivando en 2. La MC solucionó uno de estos casos, debiendo ponerse una segunda prótesis en el otro paciente. En los otros dos (una atresia y otra por caústicos), la MC fue efectiva, solucionando las estenosis tras 1 y 2 sesiones respectivamente. En el cómputo total hemos solucionado las estenosis en 5 de 6 estenosis refractarias (83,3%) y en 1 de 2 fístulas (50%).
Conclusiones: la asociación de prótesis biodegradables y mitomicina ha sido efectiva en el rescate de algunos casos de estenosis esofágicas refractarias en niños, teniendo un papel secundario en la resolución de fístulas.
Palabras clave: Estenosis esofágica. Pediatría. Prótesis biodegradables. Mitomicina.
Introduction
Dilation is the reference technique in the management of esophageal stenosis in the pediatric age. There are moderately sized randomized studies analyzing therapeutic alternatives and the different techniques used in clinical practice are based on published cases or descriptive studies of case series, mainly in adult populations. Moreover there are certain ethical problems when developing new therapies and these patients are usually managed conservatively.
The concept of stricture refractory to dilation has not been absolutely defined, but a number of 3 non-effective dilations are usually used (1). The differences between caustic and postoperative stenosis remain unclear as well.
Metallic stents have been used (steel and nitinol) (2), as well as biliary (3) and tracheobronchial (4) prosthesis and silicone (5), plastic (Polyflex) (1), polytetrafluoroethylene (6) or biodegradable (polydioxanone) (7) stents in cases of post-operative stenosis and caustic strictures in children, with variable results and high rates of stent migration.
The advantage of BS in the pediatric population is that there is no need for their removal, although in a study including 8 children with metallic stents, no difficulties for their removal were reported (2).
When using stents, an important fact is that these can be customized (5) and compared to dilation they provide maintained radial expansive force with time, so stenosis can be treated more easily or decrease the frequency of dilations (8).
Only one case of BS in the pediatric age has been published (7).
Several authors have reported the effectiveness of CM injected (9) or extended (10) over the stenotic area in pediatric cases of RES. CM is an anthracycline with antineoplastic and anti-proliferative properties due to its capacity to inhibit the activity of fibroblasts, suppressing the formation of fibrosis and scares due to apoptosis. It has been used for the treatment of stenosis in ophthalmology and otolaryngology (9,11,12).
We present 6 cases of RES treated with BS and/or CM which may provide a new approach in this population so often treated with interventional procedures.
Methods
Six patients have been included (February 2010-December 2013). All the procedures took place in an operating room under general anesthesia, and with endoscopic support. We normally used pediatric endoscopes (Pentax EG-1580K and 5.9 mm Olympus GIF-XP160), but in children over 15 kg we used larger diameter endoscopes (9.2 mm Olympus GIF-165) as this makes the technique easier (there is no need for guide wire exchange) and this also shortens the anesthesia times.
In 4 children, an endoscopic gastrostomy was also placed. In one of them, this was used as a lower access point for stenosis canulation and introduction of a BS.
Stent placement procedure
In 3 cases, customized BS ("SX ELLA-BD" by ELLA-CS, Hradec Kralove, Czech Republic) were placed after measuring the stenosis length, the portion of healthy esophagus over and above the stricture and the diameter of the esophagus, especially at the proximal end. We ordered BS according to these measurements, with a smaller introducer system in an 8 month-old child, as the standards (28F) were too big for this patient. In this case we used a straight stent with no cups because there was not enough room in the introducer. In the other 2 cases, as the esophagus was significantly dilated, standard BS were used.
In all the cases soft and low radial force dilation was performed initially (HERCULES balloon by COOK-WINSTON, Salem, USA), so the introducer system could be placed. All the patients were under proton-pump inhibitors for the treatment of gastroesophageal reflux and anti-reflux surgery had been previously done in 2 patients.
C-mitomycin procedure (Fig. 1)
We prefer the use of endoscopes with a working channel greater than 2.8 mm as the procedure can be done in just one step. The stenotic area needs to be dilated (10-12 mm balloons) (1a, 2a) prior to CM administration. The technique will be different depending on the stenosis length (Fig. 1): in short strictures (< 1 cm) 0.4 ml of CM (0.4 mg/ml) is injected in every quadrant (1b) (13); in long strictures (> 1 cm) we perform an adaptation of the technique described by Broto (14):
- After placement of the guide wire the endoscope is removed.
- As a technical innovation a 15mm biliary extraction balloon is placed distal to the stenosis (this prevents CM migration and damage of healthy tissue) (2b).
- Using a double lumen biliary brush, CM (0.4 mg/ml) is carefully distributed over the stenosis (2c). The total amount of CM depends on the child's weight: in children < 12.5 kg, 2.5 ml of CM are used; in patients > 12.5 kg, 5 ml are administered.
- Then a 8 mm balloon is inflated for 5 minutes in order to provide a closer adherence of CM to the esophageal wall (2d).
- Finally the esophagus is cleaned up with water and all the liquid is removed using the endoscope (2e).
In children younger than 2 years old (or under 10 kg) it is not possible to use a biliary brush if the stenosis is long. In this situation we only apply CM over the stenosis and extend it with a smaller brush.
The decision about the initial use of BS or CM was based on the previous evolution of the stenosis and its length. In short, for stenosis with no previous complications we started with CM. In patients with an associated fistula, previous complications or long stenosis, we decided to use BS first.
Results
We present the descriptive analysis of a prospective case series of RES in children who have been treated with alternative techniques such as BS and CM.
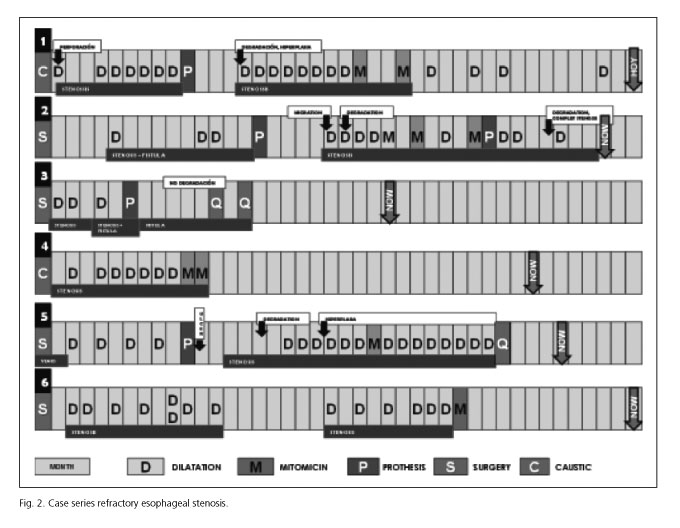
Case 1
A 6 year-old girl with severe esophagitis and gastritis after caustic ingestion. After one month the patient underwent dilation due to esophageal stricture, presenting a mild perforation which was treated conservatively. After 4 months the patient was sent again for esophageal and antral dilation. The gastric stricture was resolved (6 dilations), but the esophageal remained (9 dilations). After 10 months a "ELLA" BS (16 x 60 mm - customized) was placed.
The child reported intermittent thoracic pain for 4 months, with a good response to medical treatment. Moreover, she presented stenosis due to proximal hyperplasia (resolved after 3 dilations). Distal stenosis recurred after 4 months (as long as the stent was absorbed) with the need for more dilations. After 9 months we decided to use CM injection in this patient (2 sessions) with a good response. Eighteen months later this child still has a good oral intake.
Case 2
A 3 year-old boy presented with a VACTERL syndrome (esophageal and anorectal atresia) requiring multiple surgical reconstructions and a gastrostomy.
There was a need for dilations due to stenosis of the esophageal-ileo-coloplasty, but these were discontinued as he presented with a cervicothoracic abscess. After one year, as dilations were unsatisfactory and the fistula remained, a "ELLA" BS (fully covered stent, 18 x 60 mm - customized) was placed. We faced great technical difficulties as the anatomy of proximal esophagus was severely altered, so we used the gastrostomy orifice to pull out the wire-guide. During follow-up the patient presented with a new cervical abscess (because the stent moved distally) and this was treated with replacement of the BS and antibiotics. After 8 months he presented with re-stenosis and CM was then used, with the placement of a second stent (uncovered). Finally, after absorption of this stent the fistula was resolved, but a complete esophageal stenosis occurred.
The time for complete stent absorption was 5 and 4 months respectively.
Case 3
An 8 month-old girl with type I long-gap esophageal atresia who required surgery when she was 2 months old. She presented with anastomotic stenosis and came for dilation on 3 occasions (every 4 weeks). During the third dilation, the wire passed to the mediastinum and the child remained in the intensive care unit as she developed mediastinitis.
At 7 months the patient was suffering recurrent aspiration pneumonia and a broncho-esophageal fistula was observed. After discussion in a multidisciplinary committee, at 8 months a partially covered "ELLA" BS (60 x 150 x 40 mm) was placed. A straight stent, with no cups, was customized so an appropriate introducer could be used as the patient was a small child of 5 kg.
Oral intake was restarted, but she went onto have respiratory complications again after 7 months. At this time the stent was totally absorbed and the stenosis was resolved, but a fistula was observed again, with the need for surgical repair: cervical esophagostomy and further esofagogastroplasty. After surgery she had a fair evolution from her respiratory and gastrointestinal symptoms.
Case 4
A 2 year-old girl with caustic stenosis required dilations every 4 weeks as she presented frequent restenosis. The stricture was 1 cm long and the lumen only 2 mm. After 7 dilations we decided to initiate CM. She received 2 sessions (the second one 40 days after the first) of 1.6 ml of CM injection followed by progressive dilations, complete resolution was observed. Caustic ingestion occurred 2.5 years ago, and the patient has not required any more dilations in the last 19 months.
Case 5
A 13 year-old boy with an operated type 1 atresia (Sharli esophagogastroplasty) presenting with chronic dysphagia due to fibrotic proximal esophagus and stricture of the anastomosis. After 2 unsatisfactory dilations (2 months between dilations), a standard uncovered "ELLA" BS (23 x 8 mm) was placed just under Kilian's area as the proximal esophagus was very tortuous. Two weeks later he presented with severe throat pain, and an ulcer was observed under the proximal end of the stent. Perforation was discarded in a computed tomography and medical treatment with pain killers and enteral nutrition were started with a good evolution and wound healing after 2 weeks. The patient needed more dilations, with a higher frequency than previously, and after 6 of them CM was associated with no response. Finally definitive surgical treatment was decided one year after the placement of the BS.
In this patient reabsorption of the stent was observed at 5 months, with presence of a great amount of hyperplastic residual tissue and a need for a higher number of dilations.
Case 6
A 2 year-old girl was operated on when she was 2 months old due to a type 1 esophageal long-gap atresia. Two months later she presented with stenosis of the anastomosis and required dilations (14 sessions every 4-8 weeks as restenosis was observed). Moreover this patient was born with a severe tracheomalacia and this caused significant problems with nasotracheal intubation and extubation difficulties during all the dilation procedures.
The use of CM was decided after 14 dilations and the patient has shown a great response after only one session as no new stenosis and no symptoms have been observed after 19 months.
Series analysis
We present the results of 5 stent placements in 4 patients (2 stents were needed in one case). The stenosis was solved in 2 cases, we observed partial or temporary response in 3 cases, and there was clinical worsening in one patient (secondary to esophageal ulceration). Three stents were customized and the other 2 were the standard commercialized ones (with the smallest lumen: 23 x 8 x 23 mm). In 3 cases, uncovered stents were used, fully covered in one patient, and partially covered in the other one. In those patients with a fistula (2 cases), fully covered stents were effective but not the partially covered.
The observed complications with BS were: chest pain (2/5, 40%), severe hyperplasia (2/5, 40%), mild hyperplasia with no need for dilation (2/5, 40%), migration with further replacement of the stent (1/5, 20%) and ulcer (1/5, 20%). All these complications were managed conservatively.
In the case of CM, 5 therapies were applied to 6 patients (2 sessions in 4 patients, and 1 session in the other one). Stenosis was permanently resolved in 3 cases (60%) and CM injection was not effective in the other 2 cases (40%). No adverse effects were registered. The average time spent during CM sessions was 28.7 minutes (16-75 minutes).
Globally, and considering both techniques, 4 out of 6 stenosis were resolved (66.6%), as well as 1 out of 2 fistula (50%). The fistula in which our technique was unsatisfactory required surgical treatment. The 2 cases of stenosis with no response were also sent for surgery. One of them presented complete esophageal closure and reparative surgery was necessary.
The follow-up of the patients after BS or CM was between 8 months and 2 years.
Discussion
The concept of refractoriness has not been well defined because there are doubts over whether the limit to define it should be established at three to five dilations (1,15). A recurrent or refractory stenosis is defined as an anatomic restriction secondary to luminal scarring or fibrosis which conditions dysphagia with no endoscopic evidence of inflammatory process. This may happen if a diameter of 14 mm is not achieved in 5 sessions every 2 weeks (refractory) or if a satisfactory diameter is not maintained in 4 weeks after 14 mm were achieved (recurrent) (16). These criteria are not easy to apply in children so clinical, endoscopic and radiologic criteria should be considered in this population as we have done.
Dilations are aggressive procedures performed under deep sedation, with a high risk of complications (micro breaks of the esophageal wall that lead to scarring and fibrosis) and also with high rate of recurrences.
One additional problem in our population was that two of the children had perforations, one of them after dilation, and the other one due to postoperative leaks which made any further management complicated as inner and outer fibrosis was produced. The covered stent resolved the fistula, even after a short migration time, but the stenosis remained, and this patient was also not a good candidate for reparative surgery. In case number 3, there was not enough contact between the dilated esophagus and the stent because this had no cups (the introducer was extremely small) and food was deposited between them. This could be the reason why the fistula was not resolved. We consider the presence of cups in the stents essential (especially if the esophagus is dilated due to long-time evolution stenosis) so these will remain placed and the risk of migration will be lower. In case number 5 there was not a good response probably because we used a standard and longer stent which produced an excavated and painful ulcer on the proximal esophagus.
BS have shown to be as safe and effective as in benign esophageal stenosis in adults (17). The main advantage of BS is that there is no need for removal. In adults, fully or partially covered stents are usually used in benign diseases, but these need to be removed after some time and in some circumstances more stents are needed after removal (18). Severe complications have been described when removing esophageal stents, such as perforations, bleeding, and problems related to sedation and the need for further endoscopic procedures. This is why we have worked with BS as our aim was to avoid the removal of the stents. Higher migration rate have not been reported with BS compared with other types of prosthesis. In one of our cases, the BS migrated but it was successfully replaced. In a German case series including 12 children the rate of migration was higher when using plastic stents than with silicone or nitinol stents (19).
One disadvantage of BS is early degradation promoted by acid. Moreover this degradation is more evident at high pressure points such as stenosis. We have observed degradations from 4 to 7 months, with no relation with the presence of anti-reflux surgery (the case of 7 months degradation had anti-reflux associated with anti-secretory agents).
One hundred and thirty-two cases of BS have been reported in esophageal benign and malignant disease (Table I), but there is only one case in a pediatric patient and this stent was ineffective because it caused excessive reflux and presented distal migration (7). There are two more cases of BS placed in children in Minsk, but these have not been published (information obtained from the manufacturer and also referred by Vandenplas) (7). In a Belgian series (abstract) including 24 BS in 20 patients, with no data with regard to esophageal stents or the ages of the patients, the authors report that after 6 months, in 52% of the cases there is a need for further procedures and 60% of the patients reported pain (31).
Higher rates of hyperplasia, including severe cases, have been reported when using these stents (24,32). We have observed 2 cases of severe hyperplasia with a need for further dilations, which was previously reported in the literature (33). In one of the cases, the patient had anti-reflux surgery (a very competent esophageal-ileo-colo-gastroplasty). In our series, hyperplasia seems to be more important than in the adult series (only 1 case in 21 BS) (17). Our hypothesis is that the higher reparative tissue capacity in children can be the reason for this phenomenon.
Regarding our results, it is important to analyze our failures and to detect the causes: in case number 2, the patient had been operated on several times before and presented with an anfractuous esophagus, which was almost impassable. Previous perforations caused a lack of elasticity which avoided the normal liberation of the stents. The covered stent solved the fistula despite of the fact that it migrated, but up to now it has been impossible to solve the stenosis, and this patient is not a good candidate for a new surgery. In case number 3, the stent was cup-free (due to the small size of the introducer) and this caused a poor contact between the dilated esophagus and the stent. Food and secretions accumulated in this area could be the reason for non-response of the fistula. This is why we think that cups are essential in the stents, especially in dilated esophagus, secondary to long term stenosis. Cups could also avoid the migration. In case number 5, the reasons for failure were similar to those in case number 2 as this was a rotated stenosis after atresia corrective surgery. Moreover, the placement of a bigger stent just under Killian's area produced a painful and deep ulcer.
Failure of the treatment has been related to patients previously operated on for atresia, previous complications and fibrotic esophagus. In these cases we think that the therapeutic strategies should be reconsidered and new protocols using stents with a greater radial force should be defined, although in the latest published data there are no differences when comparing the stents available nowadays (34). One possible solution could be based on the use of fully covered metallic stents for longer periods of time. On the other hand a case series including 59 BS in 21 adults presenting with benign esophageal stenosis has been recently published, with a low response rate (25%), so authors conclude that these stents could be useful as a bridge therapy, with the aim of increasing the period between dilations, but not with a curative aim (35).
With regard to the fistula, we observed a healing of the process in one case, but there was recurrence in the other one, who was an infant with a significantly dilated esophagus and a huge fistula, so the stent did not expand and make the correct contact with the esophageal walls. Treatment and resolution of the fistula associated to atresia using covered plastic stents has been previously reported (36). We have contributed one more case but unfortunately the stenosis has n healed in our patient. In a case series including 5 patients with anastomotic leaks, the use of BS was effective in 4 cases, but one patient needed three stents and the authors report 3 stent migrations (28).
CM has been useful as adjuvant treatment after stent placement in a case of caustic stenosis. In one other case, also in a stenosis after caustic ingestion, the early use of CM has solved the stenosis after only 2 sessions. In case number 2 we observed some kind of tachyphylaxis as there was increasing inter-dilation periods in the first 3 doses of CM but, after the 4th dose the therapeutic effect disappeared, with the need for new dilations in the same way as before the treatment. This phenomenon has not been reported previously.
With regard to CM, it has been shown that the therapeutic effect is higher when using higher concentrations and a longer contact of the drug with the tissue. It is important to use the correct dose and maintain CM on the mucosa for at least 5 minutes (37,38). Several techniques have been proposed for application of CM, such as soaked pads, mucosal injection, the use of spray catheters and stents that progressively release the drug. Rigid endoscopes and fibro-endoscopes have been used and application of CM has also been described using a catheter through a gastrostomy stoma (39).
Although it has not been well defined, it seems that 5 CM sessions could be a good limit for maintained use (14), but some authors have reached 6 sessions with no complications (40). CM is safe when applied topically and, although it is cytotoxic and may damage normal mucosa (41), this has not been described in a case series including gastroenterology patients, in which no significant complications are seen.
Forty-two pediatric patients with RES treated with CM have been reported, with only two case series (Table II): in a multicenter study including 16 patients, complete and partial response are seen in 62.5% and 19% respectively (5 years follow-up) (41); in a prospective series including 10 patients, 100% of response is reported (40). The other ones are isolated selected cases with therapeutic success.
It is very important to set the size of the stents correctly. We should use a slightly bigger size than the diameter of the upper esophagus, even when this strategy can produce chest pain or produce decubitus ulcers.
Although placement of BS is an expensive procedure, with higher costs when using customized stents, we may be saving additional costs related to hospitalization, anesthetic procedures, the use of operating rooms, and recuperation of the children and job absences of the parents if we can increase the periods between dilations. Moreover, these children usually undergo multiple and very frequent invasive procedures and we can offer them some kind of benefit with this strategy. Cost-effectiveness studies are necessary to assess the real economic impact.
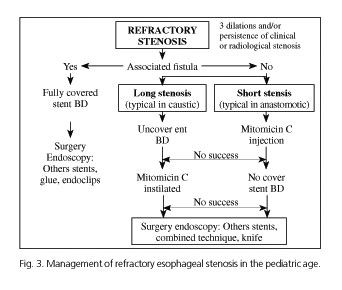
Analyzing this review, and with our experience, we propose an algorithm for the management of RES, with the need for the collaboration of pediatric surgeons as surgery is the standard treatment (Fig. 3). Once RES diagnosis is established we may run endoscopic treatments depending on the existence or not of associated fistula, which is mainly a surgical pathology. If there is no fistula, decisions will depend on the length of the stenosis: in long (< 1 cm) and fibrotic stenosis (as caustic) the use of covered stents will be the procedure of choice; in short stenosis the option will be CM sessions (up to 5 sessions). If there is no response, the alternative technique should be considered. Failure of both techniques means need for surgery, different stents, stricturotomies, etc. One more possibility is association of several techniques as stenting could help to maintain the effects of CM.
Conclusion
We think that the possibly of developing customized BS allows the design of appropriate stents for each individual case. BS do not need to be removed but side effects can be significant and a limiting factor. These stents have been effective when treating fistula, but this effectiveness has been more limited to the management of esophageal stenosis, which have presented recurrences but on the other hand have also needed dilations for longer time periods. Anti-reflux treatment must be intensified as this leads to longer stent maintenance and lower rates of side effects.
CM is an alternative for stenosis with no response to dilations and its use is associated with low rates of side effects.
References
1. Broto J, Asensio M, Vernet JM. Results of a new technique in the threatment of severe esophageal stenosis in children: poliflex stents. J Pediatr Gastroenterol Nutr 2003;37(2):203-6. DOI: 10.1097/00005176-200308000-00024. [ Links ]
2. Zhang C, Yu JM, Fan GP, et al. The use of a retrievable self-expanding stent in treating childhood benign esophageal strictures. J Pediatr Surg 2005r;40(3):501-4. DOI: 10.1016/j.jpedsurg.2004.11.041. [ Links ]
3. Ruthmann O, Richter S, Fischer A. Biliary stenting of an iatrogenic esophageal perforation following corrosive esophagitis in a 5-year-old child. Endoscopy 2009;41(Suppl 2):E325-6. DOI: 10.1055/s-0029-1214939. [ Links ]
4. Best C, Sudel B, Foker JE, et al. Esophageal stenting in children: indications, application, effectiveness and complications. Gastrointest Endosc 2009;70(6):1248-53. DOI: 10.1016/j.gie.2009.07.022. [ Links ]
5. Foschia F, De Angelis P, Torroni F, et al. Custom dynamic stent for esophageal strictures in children. J Pediatr Surg 2011;46(5):848-53. DOI: 10.1016/j.jpedsurg.2011.02.014. [ Links ]
6. Atabek C, Surer I, Demirbag S. Increasing tendency in caustic esophageal burns and long-term polytetrafluorethylene stenting in severe cases: 10 years experience. J Pediatr Surg 2007;42(4):626-40 DOI: 10.1016/j.jpedsurg.2006.12.012. [ Links ]
7. Vandenplas Y, Hauser B, Devreker T. A biodegradable esophageal stent in the treatment of a corrosive esophageal stenosis in a child. J Pediatr Gastroenterol Nutr 2009;49(2):254-7. DOI: 10.1097/MPG.0b013e31819de871. [ Links ]
8. Kramer RE, Quiros JA. Esophageal stents for severe strictures in young children: experience, benefits and risk. Curr Gastroenterol Rep 2010;12(3):203-10. DOI: 10.1007/s11894-010-0105-4. [ Links ]
9. Rahbar R, Jones DT, Nuss RC, et al. The role of mitomycin in the prevention and treatment of scar formation in the pediatric aerodigestive tract: friend or foe? Arch Otolaryngol Head Neck Surg 2002;128:401-6. [ Links ]
10. Afzal NA, Albert D, Thomas AL. A child with oesophageal strictures. Lancet 2002;359(9311):1032. DOI: 10.1016/S0140-6736(02)08095-9. [ Links ]
11. Rahbar R, Shapshay SM, Healy GB. Mitomycin: effects on laryngeal and tracheal stenosis, benefits, and complications. Ann Otol Rhinol Laryngol 2001;110:1-6. DOI: 10.1177/000348940111000101. [ Links ]
12. Kao SCS, Lia CL, Tseng JH, et al. Dacrocystorhinostomy with intraoperative mitomycin-C. Ophthalmology 1997;104:86-91. DOI: 10.1016/S0161-6420(97)30357-1. [ Links ]
13. Spier BJ Sawma VA, Gopal DV. Intralesional mitomycin C: successful treatment for benign recalcitrant esophageal stricture. Gastrointest Endosc 2009;69(1):152-3. DOI: 10.1016/j.gie.2008.05.060. [ Links ]
14. Broto J, Gil-Vernet JM, Royo G. Use of mitomycin C in esophageal stenosis. Cir Pediatr 2007;20:57-8. [ Links ]
15. Antoniou D, Soutis M, Christopoulos-Geroulanos G. Anastomotic strictures following esophageal atresia repair: a 20-year experience with endoscopic ballon dilatation. J Pediatr Gastroenterol Nutr 2010;51(4):464-7. DOI: 10.1097/MPG.0b013e3181d682ac. [ Links ]
16. Kochman ML, McClave, SA, Boyce HW. The refractory and the recurrent esophageal stricture: a definition. Gastrointestinal Endoscopy 2005;62(3):474-5. DOI: 10.1016/j.gie.2005.04.050. [ Links ]
17. Repici A, Vieggaar FP, Hassan C. Efficacy and sefaty of biodegradable stents for refractory benign esophageal strictures: the BEST (Biodegradable Esophageal Stent) study. Gastrointestinal Endoscopy 2010;72(5):927-34. DOI: 10.1016/j.gie.2010.07.031. [ Links ]
18. Hirdes MM, Siersema PD, Houben MH, et al. Stent-in-Stent Technique for Removal of Embedded Esophageal Self-Expanding Metal Stents. Am J Gastroenterol 2011;106:286-93. DOI: 10.1038/ajg.2010.394. [ Links ]
19. Reinshagen K, Kähler G, Manegold BC. Evaluation of stents in treating childhood benign esophageal strictures. Klin Padiatr 2009;221(1):25-30. DOI: 10.1055/s-2007-984377. [ Links ]
20. Goldin E, Fiorini A. Ratan Y. A new biodegradable and self-expandable stent for benign esophageal strictures. Gastrointestinal Endoscopy 1996;43(4):294SW. DOI: 10.1016/S0016-5107(96)80017-4. [ Links ]
21. Fry SW, Fleischer DE. Management of a refractory benign esophageal stricture with a new biodegradable stent. Gastrointest Endosc 1997;45(2):179-82. DOI: 10.1016/S0016-5107(97)70244-X. [ Links ]
22. Tanaka T, Takahashi M, Nitta N. Newly developed biodegradable stents for benign gastrointestinal tract stenoses: a preliminary clinical trial. Digestion 2006;74:199-205 DOI: 10.1159/000100504. [ Links ]
23. Saito Y, Tanaka T, Andoh A. Usefulness of biodegradable stents constructed of poly-l-lactic acid monofilaments in patients with benign esophageal stenosis. World J Gastroenterol 2007;13(29):3977-80. DOI: 10.3748/wjg.v13.i29.3977. [ Links ]
24. Dhar A, Topping JH, Johns E. Biodegradable stents in refractory benign oesophageal strictures. First report of 4 patients from UK. Gastrointest Endosc 2009;M1487. DOI: 10.1016/j.gie.2009.03.655. [ Links ]
25. Orive-Calzada A, Alvarez-Rubio M, Romero-Izquierdo S, et al. Severe epithelial hyperplasia as a complication of a novel biodegradable stent. Endoscopy 2009;41(Suppl 2):E137-E138. DOI: 10.1055/s-0029-1214634. [ Links ]
26. Stivaros SM, Williams LR, Senger C. Woven polydioxanone biodegradable stents: a new treatment option for benign and malignant oesophageal strictures. Eur Radiol 2010;20:1069-72. DOI: 10.1007/s00330-009-1662-5. [ Links ]
27. van Boekel PG, Vieggar FP, Siersema PD. A comparison of temporary self-expanding plastic and biodegradable stents for refactory benign esophagel strictures. Clin Gastroenterol hepatol 2011;9(8):653-9. DOI: 10.1016/j.cgh.2011.04.006. [ Links ]
28. Cerná M, Köcher M, Válek V. Covered biodegradable stent: new therapeutic option for the management of esophageal perforation or anastomotic leak. Cardiovasc Interv Radiol 2011;34:1267-71. DOI: 10.1007/s00270-010-0059-9. [ Links ]
29. van Hooft JE, van Berge Henegouwen MI. Endoscopic treatment of benign anastomotic esophagogastric strictures with a biodegradable stent. Gastrointest Endosc 2011;73:1043-7. DOI: 10.1016/j.gie.2011.01.001. [ Links ]
30. Griffiths EA, Gregory CJ, Pursnani KG. The use of biodegradable (SX-ELLA) oesophageal stents to treat dysphagia due to benign and malignant oesophageal disease. Surg Endosc 2012;26(8):2367-75. DOI: 10.1007/s00464-012-2192-9 . [ Links ]
31. Ibrahim M, Vandermeeren A, Van Maele V. Belgian multicenter experience with the biodegradable ella stent in benign strictures of the digestive tract. Endoscopy 2010;42 (Suppl 1):A259. [ Links ]
32. Güitrón-Cantú A, Adalid-Martínez R, Gutiérrez-Bermúdez JA. Reacción a cuerpo extraño de una prótesis esofágica biodegradable. Informe de un caso Revista de Gastroenterología de México 2010;2:203-7. [ Links ]
33. Hair CS, Devonshire DA. Severe hyperplastic tissue stenosis of a novel biodegradable esophageal stent and subsequent successful management with high-pressure balloon dilation. Endoscopy 2010;42(Suppl 2):E132-E133. DOI: 10.1055/s-0029-1244011. [ Links ]
34. Hirdes MM, Vleggaar FP, de Beule, M. In vitro evaluation of the radial and axial force of self-expanding esophageal stents. Endoscopy 2013;45(12):997-1005. DOI: 10.1055/s-0033-1344985. [ Links ]
35. Hirdes MM, Siersema PD, van Boeckel PG. Single and sequential biodegradable stent placement for refractory benign esophageal strictures: a prospective follow-up study. Endoscopy 2012;44(7):649-54. DOI: 10.1055/s-0032-1309818. [ Links ]
36. Adler DG, Pleskow DK. Closure of a benign tracheoesophageal fistula by using a coated, self-expanding plastic stent in a patient with a history of esophageal atresia. Gastrointest Endosc 2005;61(6):765-8. DOI: 10.1016/S0016-5107(05)00083-0. [ Links ]
37. Khaw PT, Doyle JW, Sherwood MB, et al. Prolonged localized tissue effects from 5-minute exposure to fluorouracil and mitomycin. Arch Ophthalmol 1993;111:263-7. DOI: 10.1001/archopht.1993.01090020117035. [ Links ]
38. Lee DA, Lee TC, Cortes AE. Effects of mithramycin, mitomycin, daunorubicin and bleomycin on human fibroblast attachment and proliferation. Invest Ophthalmol Vis Sci 1990;31:2136-44. [ Links ]
39. Heran MK, Baird R, Blair GK. Topical mitomycin-C for recalcitrant esophageal strictures: a novel endoscopic/fluoroscopic technique for safe endoluminal delivery. J Pediatr Surg 2008;43(5):815-8. DOI: 10.1016/j.jpedsurg.2007.12.017. [ Links ]
40. Kumar A, Bhatnagar V. Topical application of mitomycin-C in corrsive esophageal strictures. J Indian Assoc Pediatr Surg 2005;10:25-7. DOI: 10.4103/0971-9261.16071. [ Links ]
41. Rosseneu S, Afzal N, Yerushalmi B. Topical application of mitomycin-C in oesophageal strictures. J Pediatr Gastroenterol Nutr 2007;44(3):336-41. DOI: 10.1097/MPG.0b013e31802c6e45. [ Links ]
42. Uhlen S, Fayoux P, Vachin F, et al. Mitomycin C: an alternative conservative treatment for refractory esophageal stricture in children? Endoscopy 2006;38(4):404-7. DOI: 10.1055/s-2006-925054. [ Links ]
43. Olutoye OO, Shulman RJ, Cotton RT. Mitomycin C in the management of pediatric caustic esophageal strictures: a case report. J Pediatr Surg 2006;41(5):e1-3. DOI: 10.1016/j.jpedsurg.2005.12.051. [ Links ]
44. Gillespie MB, Day TA, Sharma AK. Role of mitomycin in upper digestive tract stricture. Head Neck 2007;29(1):12-7. DOI: 10.1002/hed.20476. [ Links ]
45. Zur KB, Putnam PE, Rutter MJ. Combined retrograde and anterograde hypopharyngeal puncture and dilatation in a child with complete hypopharyngeal stenosis. Int J Pediatr Otorhinolaryngol 2007;71(1):153-7. DOI: 10.1016/j.ijporl.2006.07.023. [ Links ]
46. Fröhlich T, Greess H, Köhler H. Topical mitomycin C treatment of a benign oesophageal stricture following caustic ingestion in a four-year-old boy. Z Gastroenterol 2007;45:255-8. DOI: 10.1055/s-2006-927153. [ Links ]
47. Daher P, Riachy E, Georges B. Topical application of mitomycin C in the treatment of esophageal and tracheobronchial stricture: a report of 2 cases. J Pediatr Surg 2007;42(9):E9-11. DOI: 10.1016/j.jpedsurg.2007.06.007. [ Links ]
48. Chung J, Connolly B, Langer J. Fluoroscopy-guided topical application of mitomycin-C in a case of refractory esophageal stricture. J Vasc Interv Radiol 2010;21(1):152-5. DOI: 10.1016/j.jvir.2009.09.016. [ Links ]
49. Machida H Tominaga K., Minamino H. Locoregional MMC for refractory esophageal stricture after ESD. Endoscopy 2012;44:622-5. [ Links ]
![]() Correspondence:
Correspondence:
Fernando Alberca-de-las-Parras.
Department of Digestive Diseases. Unit of Gastrointestinal Endoscopy.
Hospital Clínico Universitario Virgen de la Arrixaca.
Ctra. Madrid-Cartagena, s/n.
30120 El Palmar, Murcia. Spain.
e-mail: alberca.fernando@gmail.com
Received: 14-01-2016
Accepted: 11-06-2016











 texto en
texto en