My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.108 n.10 Madrid Oct. 2016
https://dx.doi.org/10.17235/reed.2016.4340/2016
LETTERS TO THE EDITOR
Clinical, laboratory, serological, and histological profile of sprue-like enteropathy associated with olmesartan use
Perfil clínico-analítico, serológico e histológico de la enteropatía sprue-like asociada con la toma de olmesartán
Key words: Enteropathy. Diarrhea. Olmesartan. Sprue.
Palabras clave: Enteropatía. Diarrea. Olmesartán. Esprue.
Dear Editor,
Olmesartan is one of the various angiotensin II receptor blockers (ARBs) used for the management of high blood pressure. A clinico-pathological condition that mimicked celiac disease in association with the use of olmesartan was described in 2012 and designated olmesartan-associated sprue-like enteropathy (OSLE). It was first described by Rubio-Tapia et al. (1) in a group of patients with diarrhea, weight loss, negative anti-transglutaminase antibodies, evidence of enteropathy, and no clinical response to gluten-free diet who had other enteropathy causes ruled out and finally responded both clinically and histologically to olmesartan discontinuation. A systematic review and a French study on OSLE have been recently reported, which support an association between olmesartan use and the development of the condition (2,3). The pathogenic mechanisms of the disease remain unclear as of today.
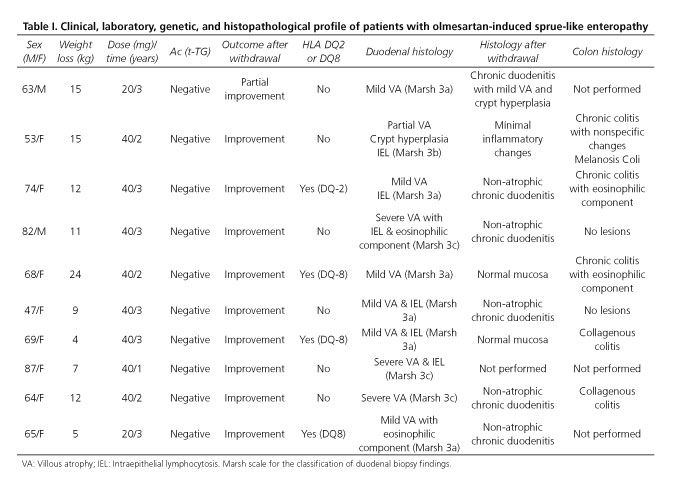
In our institution an observational, descriptive study was performed in 12 patients who met the clinical, histopathological, and evolutionary criteria of OSLE from May 2013 to December 2015. Mean age was 67 years (range: 47-87), and the series included 9 women and 3 men. Mean treatment duration was 32 months (range: 12-60). All patients had watery diarrhea, weight loss, and negative celiac serology. They were admitted with severe clinical illness including prerenal kidney failure, metabolic acidosis, water-electrolyte imbalance, and malnutrition parameters. Most common laboratory abnormalities included anemia and hypoalbuminemia. Duodenal biopsy histology revealed villous atrophy in all 12 patients. In the genetic study five patients had a DQ2/DQ8 haplotype, which represented 41.6% of the total sample. All patients responded well to drug discontinuation, and 100% of patients with a follow-up biopsy showed histological recovery (Table I).
While the causing mechanism remains unknown, the long latency seen from drug exposure to symptom onset, the finding of lymphocytes and collagenous colitis, and the condition's high association rate with HLA-DQ2/8 all suggest a cell-mediated delayed hypersensitivity reaction (2,4). This would speak against a class effect of ARBs. However, an alternate hypothesis suggests that the mechanism of action would involve the inhibition of the transforming growth factor beta (TGF-β), an important mediator of intestinal immune homeostasis, as seen with all ARBs (3). Since this enteropathy has been strongly linked to olmesartan use alone, the delayed hypersensitivity reaction hypothesis seems more plausible. Two cases of sprue-like enteropathy potentially associated with ARBs other than olmesartan were reported last year (3,5). The first case was included in a French study and was related to irbesartan use, whereas the second case involves a patient with severe enteropathy associated with valsartan. As of today, however, evidence is insufficient to confirm this enteropathy type in association with ARBs other than olmesartan. Therefore, considering the data reported by our team on these 12 patients with OSLE and the reviewed literature, we may conclude that olmesartan must be considered as a cause of severe diarrhea in order to facilitate an early identification of OSLE patients, particularly of those with severe diarrhea, duodenal villous atrophy, and negative celiac serology. Drug discontinuation is vital for suspected olmesartan-induced sprue-like enteropathy as it results in rapid clinical improvement and slower histological recovery.
Esteban Sáez-González, Francia Carolina Díaz-Jaime and
Adolfo del-Val-Antoñana
Gastroenterology Unit. Department of Digestive Medicine.
Hospital Universitari i Politècnic La Fe.
Valencia, Spain.
References
1. Rubio-Tapia A, Herman ML, Ludvigsson JF, et al. Severe sprue-like enteropathy associated with olmesartan. Mayo Clin Proc 2012;87:732-38. DOI: 10.1016/j.mayocp.2012.06.003. [ Links ]
2. Laniro G, Bibbò S, Montalto M, et al. Systematic review: Sprue-like enteropathy associated with olmesartan. Aliment Pharmacol Ther 2014;40:16-23. DOI: 10.1111/apt.12780. [ Links ]
3. Marthey L, Cadiot G, Seksik P, et al. Olmesartan-associated enteropathy: Results of a national survey. Aliment Pharmacol Ther 2014;40:1103-09. DOI: 10.1111/apt.12937. [ Links ]
4. Rubio-Tapia A. Editorial: Sprue-like enteropathy associated with olmesartan - Broadening the differential diagnosis of enteropathy. Aliment Pharmacol Ther 2014;40:1359-63. DOI: 10.1111/apt.13000. [ Links ]
5. Margot L, Herman MD, Rubio-Tapia A, et al. A case of severe sprue-like enteropathy associated with valsartan. ACG Case Rep J 2015;2:92-94. [ Links ]











 text in
text in