My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.108 n.11 Madrid Nov. 2016
https://dx.doi.org/10.17235/reed.2016.4323/2016
ORIGINAL PAPERS
A single institution's 21-year experience with surgically resected pancreatic neuroendocrine tumors: an analysis of survival and prognostic factors
Javier A. Cienfuegos1, Fernando Rotellar1, Joseba Salguero1,7, Miguel Ruiz-Canela2, Jorge M. Núñez-Córdoba2,3,4, Iosu Sola5, Alberto Benito6, Pablo Martí-Cruchaga1, Gabriel Zozaya1, Fernando Pardo1 and José Luis Hernández-Lizoáin1
1Department of General Surgery. Clínica Universidad de Navarra. Facultad de Medicina, Universidad de Navarra. Pamplona, Navarra. Spain.
2Department of Preventive Medicine and Public Health. Facultad de Medicina. Universidad de Navarra. Pamplona, Navarra. Spain.
3Division of Biostatistics, Research Support Service. Central Clinical Trials Unit. Clínica Universidad de Navarra. Facultad de Medicina. Universidad de Navarra. Pamplona, Navarra. Spain.
4Epidemiology and Public Health Area. Navarra Institute for Health Research (IdiSNA). Pamplona, Navarra. Spain.
5Department of Pathology. Hospital San Pedro. Logroño, La Rioja. Spain.
6Department of Radiology. Clínica Universidad de Navarra. Facultad de Medicina, Universidad de Navarra. Pamplona, Navarra. Spain. 7Department of Urology. Hospital Universitario Reina Sofia. Córdoba
ABSTRACT
Background: Pancreatic neuroendocrine tumors (pNETs) comprise a heterogeneous group of tumors with a varied biological behavior. In the present study, we analyzed the experience of 79 pNETs resected between 1993 and 2015. The pathologic prognostic factors (European Neuroendocrine Tumor Society, ENETS; and AJCC) classification, vascular invasion (VI), proliferation index (ki-67) and the presence of necrosis were retrospectively reviewed.
Methods: The clinical data of 79 patients with pNETs who underwent surgery were retrospectively analyzed. Mortality rates and Kaplan-Meier estimates were used to evaluate survival over time for pathologic stages, tumor functionality, and vascular invasion. Cox proportional hazards models were used to calculate the hazard ratio regarding ENETS, AJCC staging, sex, tumor functionality and vascular invasion.
Results: The male:female ratio was 40:39. Twenty-one patients (26%) had functional tumors and 58 (73.4%) had non-functional tumors, of which 35 (44.3%) were diagnosed incidentally. Seventeen Whipple procedures, 46 distal pancreatectomies (including 26 laparoscopic and 20 open procedures), 8 laparoscopic central pancreatectomies, 1 laparoscopic resection of the uncinated process and 7 enucleations (one laparoscopic) were performed. Vascular invasion and necrosis were observed in 29 of 75 cases (38.6%) and in 16 cases (29%), respectively. The comparison between survivor functions of ENETS staging categories showed statistically significant differences (p = 0.042). Mortality rate was higher in patients with non-functioning tumors compared with hormonally functioning tumors (p = 0.052) and in those with vascular invasion (p = 0.186).
Conclusions: In spite of the heterogeneity of pNETs, the ENETS TNM classification efficiently predicts long-term prognosis. The non-functioning tumors and the presence of vascular invasion are associated with poor prognosis.
Key words: Neuroendocrine tumor pancreas. Prognostic factors. Parenchyma-sparing laparoscopic pancreatectomy.
Introduction
Pancreatic neuroendocrine tumors (pNETs) account for between 2% and 4% of all pancreatic tumors, with an incidence between 2-5 per million people, and include a heterogeneous group of neoplasms with variable clinical manifestations and prognosis (1,2). In recent years, the prevalence of such tumors has increased due to the use of more sensitive imaging techniques (multi-slice computer tomography (CT), magnetic resonance imaging (MRI), and octreotide scintiscan (111 In-DTPA). This is particularly true for tumors diagnosed incidentally, which represent up to 40% of all cases (1-3).
Given their low prevalence and the confusing nomenclature used over decades such as "insular tumors" and "neuroendocrine carcinomas", and the lack of homogeneous clinical criteria, our understanding of the prognostic factors and the natural history of the disorder has remained imprecise (4).
In 2010, the World Health Organization (WHO), the American Joint Cancer Commission (AJCC), the European Neuroendocrine Society (ENETS) and the North American Neuroendocrine Tumor Society (NAETS) established specific criteria for degree of proliferation (the ki67 index) and staging (TNM) (5).
Since then, several series and multicenter studies have been published which have shed light on the biological behavior of these tumors. While the AJCC extrapolates the TNM categories from adenocarcinoma of the pancreas (6), the classification proposed by ENETS includes the experience of a multicenter series of neuroendocrine tumors of the pancreas. Fortunately, the discrepancies between the two classifications have little impact on prognosis or therapy (7,8).
In this study, the clinical, pathologic and surgical results from 79 pNETs treated in our center are described, and the associated prognostic factors and the new developments in surgical techniques are analyzed.
Patients and methods
The medical records of patients with pNETs treated surgically at the Clínica Universidad de Navarra from 1993 to 2015 were retrospectively reviewed. Approval from the Institutional Review Board was obtained.
Demographic data included gender, age at surgery, signs and symptoms present at the time of diagnosis, body mass index (BMI), and location of the tumor. Patients presenting with symptoms and an excess of serum hormone markers were classified as having "functional" tumors. Nonfunctional tumors were classified as "symptomatic" when patients presented with symptoms related to tumor mass prompting abdominal CT or MRI. Patients who presented as a result of unrelated conditions were classified as having "incidental" tumors or "incidentalomas" (9).
The preoperative clinical staging was performed by CT and MRI, and endoscopic ultrasound (EUS) with fine-needle aspiration (FNA) with an on-site cytopathologist, whenever possible. The clinical diagnosis of multiple endocrine neoplasia type 1 (MEN1) was based on previously published accepted criteria (1,2).
Surgical indications were discussed by a multidisciplinary committee. The surgical strategy was based on the anatomical location of the tumor as well as on oncological requirements. After 2004 most of the procedures were carried-out by laparoscopy. Intra-operative ultrasonography was used to assess tumor morphology and its relationship with the pancreatic duct as well as with the splenic vessels before and following pancreatic resection. Enucleation was defined as the excision of the pancreatic lesion with a minimal margin.
Surgical procedures were grouped as open or laparoscopic, which included pancreaticoduodenectomy (PD), central pancreatectomy (CP), uncinate process resection, distal pancreatectomy (DP) or enucleation (EN). In DP, the spleen was preserved wherever possible with splenic artery and vein preservation (SV-PDP). The techniques of laparoscopic central pancreatectomy (LCP) and laparoscopic resection of the uncinate process have been previously described (10,11). Our technique for laparoscopic distal pancreatectomy with splenic vessel preservation-distal pancreatectomy (SVP-DP) has been previously reported (12). Resection type was graded according to the R categories of the International Union against Cancer (UICC; R0: no residual tumor, R1: microscopic residual tumor, R2: grossly residual tumor).
Overall morbidity was defined as any complication, and these were graded according to the Dindo-Clavien system. Definitions of pancreatic fistula, delayed gastric emptying and postoperative bleeding were based on the International Study Group of Pancreatic Surgery (13-15). The onset of postoperative pancreatogenic diabetes (type IIIc) was based on the criteria of the American Diabetes Association (16).
Perioperative mortality was defined as death within 30 days of surgery or the same hospital admission. Follow-up time was calculated from the date of surgery to the last date of contact.
The histological grade, as well as the nomenclature and staging of all specimens, was based on the TNM classification of ENETS (2010) for pancreatic neuroendocrine neoplasms (5,17).
Tumor size was recorded from the pathology report. The examination of the resection margins and the posterior margin or mesopancreas was carried out according to pathology guidelines. Histological examination included regular hematoxylin and eosin staining, and additional staining with immunohistochemical markers (synaptophysin, chromogranin A). The proliferative index was expressed as the percentage of Ki-67 (using the MIB-1 antibody) in the areas of strongest immunostaining. G1 NETs were defined as having a ki-67 index of > 20%, according to the updated 2010 WHO (17) classification. Lymph node involvement, lymphovascular invasion, and necrosis were recorded whenever possible.
Patients with extrapancreatic disease at the time of diagnosis and those who underwent a R2 resection were seen by an oncologist to decide on the need for neoadjuvant or adjuvant treatment based on cytotoxic agents (streptozocin, temozolomide), cytostatic agents (everolimus) or hepatic-directed therapies (chemoembolization, radiofrequency ablation and administration of 90-Yttrium labeled microspheres) (18).
Follow-up evaluations were carried out every 3-6 months for the first three years and then annually at the surgeon's discretion. The diagnosis of recurrence was defined as the earliest imaging of a new neoplasm after complete resection and disease progression was defined as new neoplasm growth following incomplete resection.
Statistical analysis
Descriptive statistics included the mean, standard deviation (SD), median and range for continuous variables and frequencies for categorical variables. In line with other authors (19), the ENETS staging classification was simplified to four categories: I, II, III, and IV.
Patients were followed up from the date of their surgery until death, date of the last contact, or December 31st 2015. Mortality rates (per 1,000 person-years) were calculated by dividing the total number of deaths by the total number of person-years of follow-up. Mortality rates were stratified by ENETS staging categories (I, II, III and IV), tumor functionality (functional tumor vs non-functional tumor) and vascular invasion (no vs yes). Analyses were restricted to patients with non-missing information. Kaplan-Meier estimates were used to evaluate survival over time, with differences assessed using the log-rank test for equality of survivor functions for different pathologic stages, tumor functionality, and vascular invasion. Cox proportional hazards models were used to calculate the hazard ratio (HR) and 95% confidence interval (CI) of pNET-related death and ENETS stages (I to IIb vs IIIa to IV), AJCC state (IA and IB vs IIA to IV), age (using the median as a cut-off, ≤ 55 y vs > 55 y), sex, tumor functionality (yes vs no), and vascular invasion (yes vs no) were used to predict tumor mortality. P-values were two-sided, and the statistical significance level was defined as p < 0.05. The analysis was performed using Stata 14 software (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX: StataCorp LP).
Results
From December 2003 to December 2015, 84 patients underwent surgical resection of pNETs. Of the original 84 patients, 5 cases were excluded: one patient with MEN-1 and simultaneous gastric adenocarcinoma, two patients with pancreatic fibrosis, one patient with peripancreatic paraganglioma and one focal nesidioblastosis. Thus, 79 patients were finally included in the study.
Clinical characteristics
The demographic data of the 79 patients are shown in table I. Three cases were hereditary (MEN type 1) and 76 were sporadic. The mean patient age was 54 years (SD: 13; range 22-78). The male:female ratio was 40:39. Twenty-one patients (26%) had functional tumors: 10 insulinomas, 6 gastrinomas, 2 glucagonomas, 1 vipoma. One tumor produced the peptide related to the parathyroid hormone (PTH-rP), with hypercalcemia and a carcinoid tumor. Of the 79 patients, 58 (73.4%) had nonfunctional tumors, 35 of which (44.3%) were diagnosed incidentally ("incidentalomas"). In 41 cases (51.8%), the diagnosis was previously confirmed with cytology. Twenty-four tumors (30.3%) were located in the head of the pancreas, 16 (20.2%) in the body and 39 (49.3%) in the tail of the pancreas (Table I). Seventeen Whipple procedures (PD), 46 DP (26 laparoscopic and 20 open procedures), eight laparoscopic CP, one laparoscopic resection of the uncinate process and seven enucleations (one of which was carried out laparoscopically) were performed.
Of the 79 patients, 21 had synchronous liver metastases (stage IV), 10 underwent subsequent liver resections, and 6 had "curative" resections (R0).
Forty-three out of the 79 patients (54.4%) experienced some postoperative complications (Clavien grade I/II: 47%; Clavien grade III/IV: 53%) (Table I). Eight pancreatic fistulas (10%) were recorded, 6 of them grade B and 2 grade C (sepsis, repeat operation), all of which were in the open surgery group. Seven patients (8.8%) experienced intra-abdominal hemorrhage, 3 in the laparoscopic group and 4 in the open surgery group, and one patient had moderate intraluminal hemorrhage (Table I). Three patients (3.7%) undergoing PD experienced delay in gastric emptying: 2 grade A and 1 grade B. Thirteen patients developed new-onset post-pancreatectomy diabetes: 2 patients undergoing PD, 3 PC, and 8 DP. One patient (1.7%) died due to abdominal bleeding, portal thrombosis and sepsis 30 days after undergoing an open distal pancreatectomy with resection of the celiac trunk (Appleby procedure).
The mean hospital stay was 16.7 days (SD: 17.0; median: 12; range: 5-102) in open pancreatectomies and 7.3 days (SD: 5.7; median: 5.5; range: 3-32) in laparoscopic procedures.
The mean tumor size was 3.2 cm (SD: 3.5; median: 2; range: 0.3-23). In 8 (10.13%) cases there were multiple tumors and endocrine cystic tumors were found in 5 cases (6.3%). In 59 cases (74.6%) the surgery was curative (R0), in 4 cases (5%) there was involvement of the retro pancreatic resection margin following PD (R1), and in the remaining 21 cases (26.5%) patients had synchronous liver metastases that were already noted.
Vascular invasion was observed in 29 (38.6%) of the 75 cases in which this parameter was measured, and no vascular invasion was found in 46 (61.3%) cases. In 16 cases (29%) tumor necrosis was observed.
Pathologic staging
Table II summarizes staging using the ENETS and AJCC classification. Based on these criteria, 29 cases were stage I, 13 cases IIa, 3 patients IIb, and 6 cases IIIa. Seven patients had lymph node involvement (N1), corresponding to stage IIIb, and 21 were stage IV according to ENETS. Thirty-nine patients (49%) were considered to have low-grade tumors, 19 patients (24%) had intermediate-grade tumors and one patient (1%) had a high-grade tumor. There was insufficient documentation to assign a pathologic grade in 20 patients.
Prognostic factors
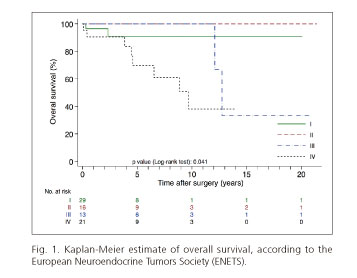
We observed 12 deaths out of 431.4 person-years of follow-up, giving an overall mortality rate close to 28 per 1,000 person-years. Mortality rates according to ENETS staging categories were 17 deaths per 1,000 person-years for stage I, 24 deaths per 1,000 person-years for stage III and 66 deaths per 1,000 person-years for stage IV. No deaths occurred in the stage II group during the follow-up period. The comparison between survivor functions of ENETS staging categories showed statistically significant differences (p = 0.042) (Fig. 1).
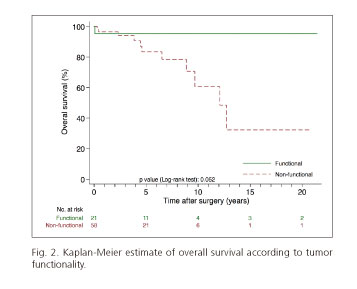
Mortality rate was higher in patients with non-functioning tumors compared with patients with hormonally functioning tumors (39 deaths per 1,000 person-years vs seven deaths per 1,000 person-years, respectively; p = 0.052) (Fig. 2). Differences in mortality rates in patients with vascular invasion (mortality rate: 48 deaths per 1,000 person-years) were apparent when compared with those patients without vascular invasion (mortality rate: 17 deaths per 1,000 person-years), although these differences were not statistically significant at the 5-percent level (p = 0.186) (Fig. 3).
Recurrence of the tumor occurred in 13 patients (16.4%): 7 in the liver, 2 in local lymph nodes, 1 in the pancreas, 1 in bone and 2 in the pancreas and liver.
Table III shows the association between clinical characteristics and pathology findings with the risk of tumor-related deaths. The hazard ratio for death among patients with ENETS stages IIIa to IV compared with states I to IIb was 4.7 (CI 95% 1.0-21.8). A similar association was found following the AJCC states. A higher risk was found among patients with non-functional tumors and vascular invasion although these associations were not statistically significant (Table III).
Discussion
pNETs represent a heterogeneous group of neoplasms with a very variable clinical progression (1,20). Not only do pancreatic tumors have a low incidence (1-2%), but until 2010 no homogeneous pathologic staging criteria existed and the nomenclature used (insular tumors, endocrine carcinoma) was confusing. In 2010 the AJCC, ENETS, NAETS and the WHO established TNM classification criteria (4,5).
Based on these new criteria, the most experienced research groups updated their results (7,19). In our study, we followed the ENETS norms designed specifically for pNETs and contrasted our data with those from the series published after 2010. Although some authors have reported discrepancies between the AJCC and the ENETS TNM classifications, such variations (generally in stage III) have little effect on prognosis (21).
In our study, the demographic pattern, clinical presentation, and histological data are very similar to those of recent series (6,19). Of note, we had a greater number of nonfunctional tumors (74.6%) and especially of "incidental" tumors (44%) due to the use of more sensitive diagnostic techniques such as CT, MRI, and EUS. The phenotype described corresponds to that reported by the majority of authors (6,22).
In our study, it is also worth highlighting the increased use since 2004 of pancreatic parenchyma-sparing laparoscopic resections such as enucleation, central pancreatectomy (CP) and SVP-DP, and of these techniques, enucleation is less frequently used (23,24) (Table I). This has led to reductions in hospital stay and operative morbidity to levels similar to those reported by other authors in several recent meta-analyses and systematic reviews, as these are the only source of data as no prospective randomized studies comparing open surgery with laparoscopy exist (25,26). It is worth pointing out that, in contrast to other studies, in this group there was no conversion to open surgery, and there were no pancreatic fistulas. The absence of postoperative pancreatic fistula (POPF) in the laparoscopy group may be due to several reasons. Firstly, the number of enucleations was lower (1/36 cases) and the greater incidence of POPF in enucleations in comparison with standard resections is well established. Secondly, we routinely administered octreotide and avoided the use of drains in the "pancreatic bed", as has been suggested in recent studies by Hüttnet et al. (27). A recent meta-analysis reported a lower incidence of POPF in standard resections as compared to enucleations (OR = 2.09, p > 0.001; I2-16%). The same group reported an overall incidence of POPF of 41% in 166 enucleations performed using open surgery.
In those patients with no evidence of liver metastases (15/26), the splenic vessels were spared, and we observed no cases of thrombosis or obliteration of the splenic vein, in contrast to the 27% reported by Tien et al. (28).
Several technical reasons could explain these results, such as avoidance of the use of the ultrasonic dissector in the dissections and sealing of the small branches of the splenic vein. It has been reported that the temperature of the dissector blades can reach 150 oC thus damaging the endothelium of the splenic vein, which is more sensitive to thermal trauma as it lacks a muscle layer and elastic fibers (29).
In our series, 13 patients (16.5%) developed new-onset pancreatogenic diabetes (NOPD) between one month and nine years after surgery, a figure similar to that reported by other authors and in a recent meta-analysis (14%) (30). Eight patients had undergone DP, 3 CP, and 2 Whipple procedures. It is well established that, in contrast to what occurs in rodents, the proliferation of β cells following pancreatic resection is minimal in humans. Some authors have linked the development of NOPD to the volume of the pancreas resected.
Survival analysis using the ENETS TNM classification distinguished between two subgroups of patients: those with a good prognosis (a 5-year OS of 90.5% and 100%) corresponding to stages I and II, and another group with a poorer prognosis (a 5-year OS of 100% and 69.6%, respectively) corresponding to stages III and IV (Fig. 1). These data are in line with the majority of published series (7,19). The patterns of recurrence were also consistent with those reported by other authors who show the liver to be the location where recurrence most often occurs.
The data on tumor size, vascular invasion (38.67%), the presence of necrosis (16%) and lymph node involvement (24.1%) were similar to the figures reported in recent series (6,8).
Apart from the presence of distant metastases (stage IV), vascular invasion was predictive of poorer prognosis whereas no significant differences for lymph node involvement were observed.
Twenty-one patients (26%) had functional tumors, and 58 (73.4%) had nonfunctional tumors, an incidence similar to that reported in the most recent series although quite different from studies published 20 years ago in which a predominance of tumors associated with hormonal syndromes was shown. We found no significant differences in survival between both groups although the difference almost reached statistical significance (p = 0.052) as has been reported by other authors (8,21,31).
Thirty-five cases (44%) were diagnosed incidentally, which is in line with other series where this figure has ranged from 35% to 45% and even reached 80-82% in some series. Although the resection of < 2 cm tumors has been controversial, most authors indicate resection in centers with experience and pancreatic parenchyma-sparing techniques, ideally performed using laparoscopy (17). Fischer, Cherenfant and others (32,33) have reported an incidence of carcinoma and systemic and lymph node metastases of between 13% and 25%, respectively, in incidental tumors of ≤ 2 cm. In small tumors with a ki67 index over 3% (grades II and III), these authors reported a 5-year survival rate of 77%, similar to that of high-grade tumors, which serves to reinforce an interventionist approach (7).
In our series, as was noted above, disease-free survival in these tumors was almost 95% at five years. These results must be contrasted with good clinical judgment in patients who are older or who have multiple disorders where clinical monitoring may be an option.
In 21 patients (26.5%) with liver metastases (stage IV), resection of the primary tumor was carried out, and subsequent resection of the liver was performed in 10 patients, which was curative in 6 cases. Although the management of stage IV tumors is controversial, some studies have reported a better response to systemic treatment and an increase in the length of disease-free survival in patients where the primary tumor is resected and even after the cytoreduction in advanced nonfunctional tumors (34).
We are aware of the limitations of the study as it is retrospective in design and was carried out over a period from 1993 to 2015, which includes the publication in 2010 of the classification systems and nomenclature for NETs (5,17). This limitation was inevitable as shown in other retrospective single and multi-center series. Two of the authors (JAC and JS) exhaustively and independently reviewed the pathology reports and the TNM staging established by ENETS and AJCC, and high levels of agreement were obtained. For this study, the ENETS classification was chosen as it was more specific to neuroendocrine tumors.
Similarly, during the study period in 2004, laparoscopic resections were introduced, requiring great technical skill. As we have already pointed out, there are no prospective randomized studies comparing open surgery with laparoscopy or the different types of resection: enucleation, CP or DP with sparing of the splenic vessels. However, several meta-analyses and systematic reviews have been published which confirm the benefit of laparoscopic techniques regarding morbidity, hospital stay and postoperative pain. We followed the best clinical evidence available from meta-analyses and consensus guidelines published after 2010 (24,26).
Due to the strict monitoring, the limited number of cases (n = 79) and the heterogeneous nature of the tumors, we were not able to perform a multivariate analysis of prognostic factors. A further possible limitation might be the impact on survival of the introduction of new agents used as neoadjuvant or adjuvant therapies over the past two decades. Given that few changes have been reported, we believe that the retrospective nature could imply some degree of variation in collecting relevant prognostic data, especially ki67 staining interpretation, vascular invasion, lymph node invasion or presence of necrosis. As previously mentioned, there were no clear pathological guidelines until 2010 (17).
Conclusions
In spite of the heterogeneity in the biological behavior of pNETs (functional vs nonfunctional tumors, incidental tumors vs normally diagnosed tumors) and the multiple parameters described (vascular invasion, necrosis, Ki-67 proliferation grade, lymph node involvement and perineural invasion), the ENETS TNM classification efficiently predicts long-term prognosis in patients.
Surgical resection is the treatment of choice, even for incidental tumors. Laparoscopic resections associated with pancreatic parenchyma-sparing techniques are the procedures of choice in centers with experience in pancreatic surgery.
In advanced stages (IV), pancreatic resection should be combined with curative resection (R0) of the metastases whenever feasible against a background of multimodal treatments such as chemoembolization and chemotherapy.
Acknowledgments
All authors have read and approved the manuscript, and it is not under consideration elsewhere. The authors are not aware of any affiliations, memberships, funding, or financial holdings that might perceive as affecting the objectivity of the manuscript.
References
1. Halperin DM, Kulke MH, Yao JC. A tale of two tumors: Treating pancreatic and extrapancreatic neuroendocrine tumors. Annu Rev Med 2015;66:1-16. DOI: 10.1146/annurev-med-061813-012908. [ Links ]
2. Kondo NI, Ikeda Y. Practical management and treatment of pancreatic neuroendocrine tumors. Gland Surg 2014;3:276-83. [ Links ]
3. Rindi G, Wiedenmann B. Neuroendocrine neoplasms of the gut and pancreas: New insights. Nat Rev Endocrinol 2011;8:54-64. DOI: 10.1038/nrendo.2011.120. [ Links ]
4. Klimstra DS, Modlin IR, Adsay NV, et al. Pathology reporting of neuroendocrine tumors: Application of the Delphic consensus process to the development of a minimum pathology data set. Am J Surg Pathol 2010;34:300-13. DOI: 10.1097/PAS.0b013e3181ce1447. [ Links ]
5. Kloppel G, Rindi G, Perren A, et al. The ENETS and AJCC/UICC TNM classifications of the neuroendocrine tumors of the gastrointestinal tract and the pancreas: A statement. Virchows Arch 2010;456:595-7. DOI: 10.1007/s00428-010-0924-6. [ Links ]
6. Rindi G, Falconi M, Klersy C, et al. TNM staging of neoplasms of the endocrine pancreas: Results from a large international cohort study. J Natl Cancer Inst 2012;104:764-77. DOI: 10.1093/jnci/djs208. [ Links ]
7. Ellison TA, Wolfgang CL, Shi C, et al. A single institution's 26-year experience with nonfunctional pancreatic neuroendocrine tumors: A validation of current staging systems and a new prognostic nomogram. Ann Surg 2014;259:204-12. DOI: 10.1097/SLA.0b013e31828f3174. [ Links ]
8. Strosberg JR, Cheema A, Weber J, et al. Prognostic validity of a novel american joint committee on cancer staging classification for pancreatic neuroendocrine tumors. J Clin Oncol 2011;29:3044-9. DOI: 10.1200/JCO.2011.35.1817. [ Links ]
9. Cherenfant J, Stocker SJ, Gage MK, et al. Predicting aggressive behavior in nonfunctioning pancreatic neuroendocrine tumors. Surgery 2013;154:785-91;discussion 791-3. DOI: 10.1016/j.surg.2013.07.004. [ Links ]
10. Rotellar F, Pardo F, Montiel C, et al. Totally laparoscopic roux-en-Y duct-to-mucosa pancreaticojejunostomy after middle pancreatectomy: A consecutive nine-case series at a single institution. Ann Surg 2008;247:938-44. DOI: 10.1097/SLA.0b013e3181724e4a. [ Links ]
11. Rotellar F, Pardo F, Benito A, et al. Laparoscopic resection of the uncinate process of the pancreas: The inframesocolic approach and hanging maneuver of the mesenteric root. Surg Endosc 2011;25:3426-7. DOI: 10.1007/s00464-011-1740-z. [ Links ]
12. Rotellar F, Pardo F, Cervera M, et al. Laparoscopic distal pancreatectomy with or without splenectomy. Surgical technique. Surg Endosc 2006;20(Suppl 1):S245. [ Links ]
13. Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: An international study group (ISGPF) definition. Surgery 2005;138:8-13. DOI: 10.1016/j.surg.2005.05.001. [ Links ]
14. Wente MN, Veit JA, Bassi C, et al. Postpancreatectomy hemorrhage (PPH): An international study group of pancreatic surgery (ISGPS) definition. Surgery 2007;142:20-5. DOI: 10.1016/j.surg.2007.02.001. [ Links ]
15. Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: A suggested definition by the international study group of pancreatic surgery (ISGPS). Surgery 2007;142:761-8. DOI: 10.1016/j.surg.2007.05.005. [ Links ]
16. American Diabetes Association. (2) Classification and diagnosis of diabetes. Diabetes Care 2015;38(Suppl):S8-S16. DOI: 10.2337/dc15-S005. [ Links ]
17. Klimstra D, Arnold R, Capella C, et al. Neuroendocrine neoplasms of the pancreas. In: Bosman FT, Carneiro F, Hruban R, Theise ND, eds. WHO classification of tumours of the digestive system. 4th ed. Lyon: International Agency for Research on Cancer; 2010. p. 322-6. [ Links ]
18. Eads JR, Meropol NJ. A new era for the systemic therapy of neuroendocrine tumors. Oncologist 2012;17:326-38. DOI: 10.1634/theoncologist.2011-0356. [ Links ]
19. Strosberg JR, Weber JM, Feldman M, et al. Prognostic validity of the American joint committee on cancer staging classification for midgut neuroendocrine tumors. J Clin Oncol 2013;31:420-5. DOI: 10.1200/JCO.2012.44.5924. [ Links ]
20. Birnbaum DJ, Turrini O, Ewald J, et al. Pancreatic neuroendocrine tumor: A multivariate analysis of factors influencing survival. Eur J Surg Oncol 2014;40:1564-71. DOI: 10.1016/j.ejso.2014.06.004. [ Links ]
21. Han X, Xu X, Jin D, et al. Clinicopathological characteristics and prognosis-related factors of resectable pancreatic neuroendocrine tumors: A retrospective study of 104 cases in a single Chinese center. Pancreas 2014;43:526-31. DOI: 10.1097/MPA.0000000000000065. [ Links ]
22. Varas M, Gornals J, Ponseti JM, et al. Pancreatic endocrine tumors or apudomas. Rev Esp Enferm Dig 2011;103:184-90. DOI: 10.4321/S1130-01082011000400003. [ Links ]
23. Abu Hilal M, Takhar AS. Laparoscopic left pancreatectomy: Current concepts. Pancreatology 2013;13:443-8. DOI: 10.1016/j.pan.2013.04.196. [ Links ]
24. Venkat R, Edil BH, Schulick RD, et al. Laparoscopic distal pancreatectomy is associated with significantly less overall morbidity compared to the open technique: A systematic review and meta-analysis. Ann Surg 2012;255:1048-59. DOI: 10.1097/SLA.0b013e318251ee09. [ Links ]
25. Iacono C, Verlato G, Ruzzenente A, et al. Systematic review of central pancreatectomy and meta-analysis of central versus distal pancreatectomy. Br J Surg 2013;100:873-85. DOI: 10.1002/bjs.9136. [ Links ]
26. Pericleous S, Middleton N, McKay SC, et al. Systematic review and meta-analysis of case-matched studies comparing open and laparoscopic distal pancreatectomy: Is it a safe procedure? Pancreas 2012;41:993-1000. [ Links ]
27. Huttner FJ, Koessler-Ebs J, Hackert T, et al. Meta-analysis of surgical outcome after enucleation versus standard resection for pancreatic neoplasms. Br J Surg 2015;102:1026-36. DOI: 10.1002/bjs.9819. [ Links ]
28. Tien YW, Liu KL, Hu RH, et al. Risk of varices bleeding after spleen-preserving distal pancreatectomy with excision of splenic artery and vein. Ann Surg Oncol 2010;17:2193-8. DOI: 10.1245/s10434-010-0983-6. [ Links ]
29. Yoon YS, Lee KH, Han HS, et al. Patency of splenic vessels after laparoscopic spleen and splenic vessel-preserving distal pancreatectomy. Br J Surg 2009;96:633-40. DOI: 10.1002/bjs.6609. [ Links ]
30. De Bruijn KM, Van Eijck CH. New-onset diabetes after distal pancreatectomy: A systematic review. Ann Surg 2015;261:854-61. DOI: 10.1097/SLA.0000000000000819. [ Links ]
31. Birnbaum DJ, Gaujoux S, Cherif R, et al. Sporadic nonfunctioning pancreatic neuroendocrine tumors: Prognostic significance of incidental diagnosis. Surgery 2014;155:13-21. DOI: 10.1016/j.surg.2013.08.007. [ Links ]
32. Fischer L, Bergmann F, Schimmack S, et al. Outcome of surgery for pancreatic neuroendocrine neoplasms. Br J Surg 2014. DOI: 10.1002/bjs.9603. [ Links ]
33. Cherenfant J, Talamonti MS, Hall CR, et al. Comparison of tumor markers for predicting outcomes after resection of nonfunctioning pancreatic neuroendocrine tumors. Surgery 2014;156:1504-10;discussion 1510-1. DOI: 10.1016/j.surg.2014.08.043. [ Links ]
34. Cusati D, Zhang L, Harmsen WS, et al. Metastatic nonfunctioning pancreatic neuroendocrine carcinoma to liver: Surgical treatment and outcomes. J Am Coll Surg 2012;215:117-24;discussion 124-5. DOI: 10.1016/j.jamcollsurg.2012.05.002. [ Links ]
![]() Correspondence:
Correspondence:
Javier A.Cienfuegos.
Department of General Surgery.
Clínica Universidad de Navarra.
Universidad de Navarra.
Av. Pío XII, 36.
31008 Pamplona, Navarra. Spain
e-mail: fjacien@unav.es
Received: 18-03-2016
Accepted: 04-08-2016