Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.109 no.1 Madrid ene. 2017
https://dx.doi.org/10.17235/reed.2016.4705/2016
A survey-based analysis on endoscopic quality indicators compliance among Spanish endoscopists
Ignacio Fernández-Urien1, Federico Argüelles-Arias2, Pedro Alonso3, Javier Júdez4 and Begoña Bermejo5
1Department of Digestive Diseases. Complejo Hospitalario de Navarra. Pamplona, Navarra. Spain.
2Intercenter Digestive Diseases Unit. Hospitales Universitarios Macarena-Rocío. Sevilla, Spain.
3Department of Digestive Diseases. Complejo Hospitalario Universitario A Coruña. A Coruña, Spain.
4Department of Knowledge Management. Sociedad Española de Patología Digestiva.
5Department of Preventive Medicine. Complejo Hospitalario de Navarra. Pamplona, Navarra. Spain
ABSTRACT
Introduction: Endoscopy plays a key role for the diagnosis and management of gastrointestinal disorders. Therefore, quality indicators have been widely proposed in order to optimize endoscopic practice. The aims of this study, promoted by the Spanish Society of Digestive Diseases (SEPD), were to assess the knowledge and compliance to endoscopy quality indicators among Spanish gastroenterologists.
Methods: A 31-questionnaire survey was created based on the endoscopy quality indicators proposed by international guidelines. The survey was distributed among Spanish gastroenterologists who are members of the society. Using only fully completed surveys, a descriptive analysis was performed. Those factors related with a suboptimal quality performance were also investigated.
Results: A total of 1,543 surveys were sent and 281 (18.2%) were received completed. Based on the answers obtained, the management of 14 (70%) out of 20 assessed quality indicators was poor: 5 (83.3%) out of 6 pre-procedure items, 7 (58.3%) out of 12 intra-procedure items and 2 (100%) out of 2 post-procedure items. Young age, public setting, no colorectal cancer (CRC) screening program at the institution and a low volume of procedures/week are factors related to poorer management of the assessed quality indicators.
Conclusions: A significant proportion of Spanish endoscopists do not comply with main endoscopic quality indicators. Factors such as "young" age, public setting, no colorectal cancer screening program and low volume of procedures/week are related to a poorer management of the assessed quality indicators and should be the target for future formative activities.
Key words: Endoscopy. Quality. Indicators. Colonoscopy. Gastroscopy.
Introduction
Nowadays, endoscopy plays a key role for the diagnosis and management of gastrointestinal (GI) disorders. When properly performed, it is generally effective, safe and well tolerated by patients. Esophagogastroduodenoscopy (EGD) and colonoscopy are the most common performed endoscopic procedures in gastroenterology. EGD is widely used for the evaluation of dysphagia, upper GI bleeding and strictures, gastroesophageal reflux, peptic ulcer and celiac diseases and unexplained diarrhea. On the other hand, colonoscopy is generally used for the evaluation of lower GI bleeding and strictures, unexplained diarrhea, inflammatory bowel disease and colorectal cancer (CRC) screening. During EGD and colonoscopy, biopsies as well as hemostasis, dilation, stenting, polypectomy and more therapeutic techniques can be performed.
Despite the appearance of many technical advances in GI endoscopy over the years, the incidence of missed lesions and interval cancers is still higher than desired (1-6). In order to identify these performance gaps and promote better practices to optimize endoscopic procedures among gastroenterologists, the main national and international scientific entities, such as the European and the American Societies of Gastrointestinal Endoscopy (ESGE and ASGE), have put forward a range of quality indicators for these tests (7-11).
The quality of health care can be measured by comparing the performance of an individual or a group of individuals with an ideal or benchmark. The particular parameter that is being used for comparison is termed a quality indicator. Quality indicators may be reported as a ratio between the incidence of correct performance and the opportunity for correct performance, or as the proportion of interventions that achieve a predefined goal (7).
Quality indicators for GI endoscopy are now well defined by scientific societies (7-11) and have been proven to deliver better health outcomes, better patient experience and fewer repeated procedures (11). However, the miss rates of pre-malignant and/or malignant lesions, which have been reported in several papers to interval and/or follow-up cancers (1-6), have not decreased over time (5,6). One of the main reasons for that fact may be the huge variation among endoscopists in some crucial quality indicators performance such as colonoscopy completion rates, withdrawal times, colonoscopy surveillance recommendations and adenoma detection rate (ADR) (12-17). Whether it is due to a lack of adequate knowledge and/or information or simply suboptimal performance has been poorly investigated.
The aims of the present study were:
- To evaluate knowledge as well as adherence to endoscopy quality indicators among Spanish gastroenterologists.
- To detect those indicators with a suboptimal management.
- To identify the characteristics of those gastroenterologists with a poorer adherence to quality indicators.
Material and methods
Since the objective of the survey was to assess the endoscopic procedures performance in terms of quality, both ASGE and ESGE recommended quality indicators for GI endoscopic procedures were taken into account (8-10). These recommendations were adapted to the survey design and the endoscopic Spanish practice. Finally, using these recommendations together with the opinion of an expert panel of Spanish endoscopists, a list of pre-procedure, intra-procedure and post-procedure quality indicators to be assessed by the survey was developed (Table I).
Then, in order to design an adequate and attractive survey, several recommended basic rules for surveys development were taken into account:
- The survey was composed of questions that assessed one specific idea (endoscopy performance), which was also explained in a brief introduction.
- Questions and survey were made as short as possible to keep respondents interested and motivated to complete it.
- Similar questions were grouped to keep the survey logical and focused. Page breaks, page titles and instructions were used to help physicians to understand it. Matrix-design questions were limited.
- Everything that could be interpreted in more than one way was spelled out.
- Sensitive questions were not included in order to avoid scare people away. Irrelevant questions were also eliminated.
- Yes/no questions were reduced as much as possible.
- Words, and not numbers, were used when designing answer choices. These answer choices are easier for people to understand.
- Data compilation and management should be easy/possible after survey completion.
Under these basic rules a survey was created by I.F.U., F.A. and P.A. and was programmed by the SEPD (J.J.'s team) using the Survey Monkey software (available at www.surveymonkey.com). The survey, after a dry-run pilot, had a total of 38 questions: 7 questions were related to demographics and the remaining 31 were related to endoscopy performance (Appendix 1). Of these 31 questions, 20 were designed to assess performance quality indicators (6 pre-procedure, 12 intra-procedure and 2 post-procedure items) and 11 were designed to obtain more concrete information. Every assessed quality indicator had a pre-set percentage of positive answers that should be reached in order to be considered as a well-managed item. This value was set using the current recommendations of ASGE and ESGE and when absent, using the consensus of an expert panel of endoscopists (Table I). On the other hand, the relationship between the sub-optimal quality indicators management and some demographic variables of responders such as age, work setting, endoscopies per week and CRC screening program availability was also calculated. The survey was then presented and promoted at the Spanish Digestive Disease Week in June 2015. The SEPD lodged the survey in its web site and invited (from June to August 2015) to all its members (n = 1,543) via e-mail to complete it. Physicians received an e-mail containing a brief introduction of the survey purpose, an invitation signed by the study coordinator (I.F.U.) and a unique link to the online survey. An e-mail reminder was sent to all non-responders at week 4 and 8 and was closed at week 10 after the first e-mail. No incentives were offered for survey completion. Once the deadline was reached, data were compiled and sent for statistical analysis (B.B.). Data management was performed using the 20.0 version of the SPSS software (Chicago, Ill). Qualitative variables are described as simple proportions and quantitative variables, as mean and standard deviation (SD) (normal distribution) or, on the contrary, as median and range (non-normal distribution). For qualitative variables association analysis, lineal Chi-squared tests were performed. A p value under 0.05 was considered to be statistically significant. The Research Ethics Committee of the Complejo Hospitalario de Navarra approved the present study. All participants were informed in writing of the purpose of the survey as well as of possible publications related and future activities based on results.
Results
A total of 1,543 survey individual invitations were sent by e-mail. Three hundred and thirteen surveys (20.2%) were responded but only 281 (18.2%) were complete. Only complete surveys were considered for the statistical analysis.
Baseline characteristics of responders
Table II shows the baseline characteristics of responders. They were male in 61.9% of the cases, working in public health hospitals in 57.2% of the cases, performing 21-40 endoscopic procedures per week in most of the cases and having an established CRC screening program in their institutions in 63.7% of the cases. The mean age and years in practice of responders was 45.2 ± 10.7 years and 18.8 ± 10.6 years, respectively. Survey responders were from institutions all around Spain and all Spanish regions were represented (Fig. 1).
Survey results
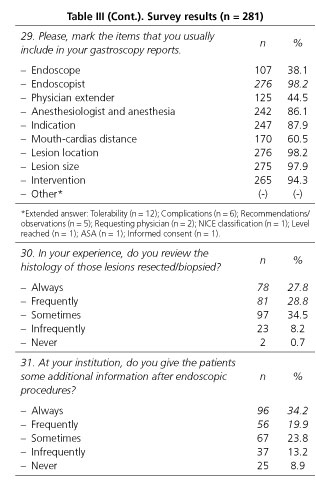
Results obtained from the survey are shown in table III.
Based on the answers obtained, the management of 14 out of the 20 (70%) "quality" items assessed was poor (Table IV): 5 out of 6 pre-procedure items (83.3%), 7 out of 12 intra-procedure items (58.3%) and 2 out of 2 post-procedure items (100%).
The mean difference between ideal (% required answers) and observed scenarios (% observed answers) was greater in post-procedure (40.5%) followed by intra-procedure (30.9%) and pre-procedure quality indicators (15.0%).
On the other hand, "informative" questions (n = 11) and extended answers revealed that:
- The responsible for cleansing regimen information vary significantly between institutions.
- The majority of responders (73.6%) use the Boston cleansing level assessment scale.
- There is consensus regarding colonoscopy repetition due to cleansing level results as well as the best preparation regimen in this scenario.
- There is no consensus regarding the need for better cleansing levels depending on the indication.
- The perception of 82.9% responders is that, in their institutions, experienced endoscopists perform most endoscopic procedures.
- Thirteen point five per cent of responders have an optimal-sized endoscopy room in their institution.
- Seventy-four per cent of responders renew the endoscopic material every seven years or earlier.
- There are almost 40% of responders that are not familiar with obtaining cultures from endoscopes.
- Only 4 out of 14 proposed items were included by more than 90% of responders in their colonoscopy reports. On the other hand, 46 responders proposed the inclusion of nine more items in colonoscopy reports.
- Only 4 out of 9 proposed items were included by more than 90% of responders in their gastroscopy reports. On the other hand, 29 responders include eight more items in their gastroscopy reports.
Relationship between assessed items and participants baseline characteristics (Table V)
Age
The relationship between age and suboptimal management items has been summarized in the first column assessment of table V. Compared to "older" physicians (> 45 years), "younger" physicians (≤ 45 years) perform polypectomy more frequently in patients under anticoagulant/antiplatelet therapy (p = 0.003). Moreover, they estimate that the informed consent form is sometimes incorrectly signed and presented the day of the endoscopic procedure (p = 0.001). Besides, there is a non-significant trend towards "younger" endoscopists regarding the lack of a histologic feedback of resected specimens and towards "older" endoscopists regarding the lack of cleansing scores performance.
Practice setting
The relationship between practice setting and suboptimal management items has been summarized in the second column assessment of table V. Only the frequency of findings photodocumentation was significantly lower in public than in private settings (p = 0.037). However, although the differences were not statistically significant, there was a tendency of a low compliance with the informed consent form in public settings (65.1% vs 81.3% in public and private setting, respectively), use of split dosages in colonoscopy cleansing regimens (55.4% and 68.8% for public and private settings, respectively), complications monitoring (42.2% vs 53.1% for public and private settings, respectively) and histologic feedback of resected specimens (55.4% vs 71.8% for public and private settings, respectively).
Endoscopies per week
The relationship between number of endoscopic procedures performed per week and suboptimal management items has been summarized in the third column assessment of table V. It has been found that the use of cleansing scales and ADR knowledge is significantly lower among physicians with a low volume of endoscopic procedures per week (p = 0.026 and p = 0.001, respectively). On the other hand, there was a tendency among those endoscopists performing more procedures per week to use a better timing for cleansing regimens, correct complete colonoscopy definition, lower frequency of "risky" polypectomies and better performance of tattooing.
CRC screening program
The relationship between availability of CRC cancer screening programs and suboptimal management items has been summarized in the fourth column assessment of table V. Compliance with informed consent forms was significantly better in those institutions with CRC screening program (72.6% vs 60.0%; p = 0.006). On the other hand, the use of split dosages in cleansing regimens (58.6% vs 52.0%; p = 0.001), the knowledge of the ADR (19.0% vs 14.0%; p = 0.003), "risky" polypectomies (45.2% vs 41.0%; p = 0.006) and complications monitoring (50.8% vs 30.0%; p = 0.002) was significantly more frequent in those institutions with CRC screening program.
Discussion
Colonoscopy is currently considered as the gold standard procedure for examination of the colon. Properly performed, it is usually safe, accurate and well tolerated by patients. However, despite being the gold standard, colonoscopy is not always perfect. From back-to-back colonoscopy studies, it has been estimated that up to 22-28% of polyps and 20-24% of adenomas are missed, and mostly in the right colon (3,5). In fact, the preventive effect of colonoscopy is more evident for distal CRCs compared to proximal lesions (2,4,18-21). On the other hand, up to 8% of CRCs occur within three years after a previous colonoscopy and they are thought to be missed lesions (1-4,22,23). Despite important technical advances and increased theoretical professional awareness, the polyp/cancer miss rate has not decreased significantly over time (5,6). One of the reasons that could explain this fact is that the effectiveness of optical colonoscopy varies among individuals and it is well known that it depends on several factors such as colon cleansing level, withdrawal times, adenoma detection rate, expertise or cecal intubation rate (12-17). These factors are currently known as quality indicators for colonoscopy and the majority of scientific societies recommend endoscopists to comply with them in their daily practice. The establishment of a minimum quality standard is now, in the CRC screening era, essential to define and shape a quality colonoscopy service, and this should be extended to all endoscopic procedures.
CRC remains being the third most common non-cutaneous cancer diagnosed in both men and women and the third leading cause of cancer-related death (24). These figures have resulted in the initiation of different CRC screening strategies all over the world. Optical colonoscopy is being commonly used as the first-line technique for CRC screening in the United States (25) while in Europe colonoscopy is usually performed after a positive fecal occult blood test (26). CRC screening programs are based on the fact that screening for CRC can affect mortality from the disease in two ways: by detecting cancers at an early and curable stage, and by detecting and removing adenomas (18,19,27-31). Consequently, the number of colonoscopies performed all around the world has increased dramatically. For the success of these mass-screening programs, it is of outmost importance to perform high quality colonoscopies according to the latest state of knowledge. However, there are still some unanswered questions regarding quality indicators: a) are quality indicators well known by all endoscopists?; b) how often do endoscopists receive a feedback of their procedures; and c) what is the endoscopist adherence to quality indicators? In fact, there is a lack of information in the literature regarding these issues. The rationale for the present study was to evaluate the perception of Spanish endoscopists regarding the quality of their endoscopic procedures in order to identify items with a poor management and the most deficient endoscopist profile. This could serve for the development of future strategies focused on the improvement of endoscopic practice.
This is a survey-based study, and this design has some pros and cons. The main advantage of the present study is that surveys can be easily distributed (e.g., on-line, resulting in a high number of completed answers). In fact, a total of 1,543 surveys were sent and 281 (18.2%) were received completed, which is an adequate response rate, if not excellent. Based on the opinion of a survey-dedicated web site (www.surveymonkey.com), the response rates of on-line surveys vary depending on factors such as relationship with responders, length, complexity and interest of the survey, but a response rate of 20-30% should be considered as a success. Although, this study reached an 18.2% rate of completed surveys, we believe that 281 answered surveys are more than enough to obtain solid conclusions. In a recent study with a similar design, the completion rate was 12.6% (17). Other surveys into gastroenterology practice revealed 10-30% completion rates (32-34). On the other hand, on-line survey answers could sometimes be inaccurate. It is well known that the tendency in surveys is to overestimate our knowledge and minimize our mistakes or weaknesses. In fact, the ideal situation would be assessing the adherence to quality indicators directly in the endoscopy room. Anyway, our results could serve as a preliminary contact with quality indicators compliance in our country.
The professional average profile of those endoscopists who participated in the survey was one of a 44 years old man, working mainly in public hospitals having a CRC screening program, with an endoscopic experience of 18 years and with a volume of endoscopic procedures per week between 21 and 40, which is equivalent to 3-4 days per week of endoscopic practice. Geographically, endoscopists from all around Spain participated in the survey. These characteristics, together with the adequate number of surveys completed, allow for the extrapolation of our results to the general population since there were not extreme values (i.e., outliers) in the distribution of endoscopists' demographic data.
Our first significant finding is that the management of 70% of the quality indicators assessed was suboptimal. This was significantly more evident in post-procedure quality items followed by intra-procedure and pre-procedure quality items. It called our attention that many of the survey responders were not aware of their ADR, which is probably the most important colonoscopy quality indicator. In fact, ADR reflects the technical skills and care to achieve a good examination of the entire colon. Consequently, a high ADR reduces the probability of interval cancers (13,14,16). Thus, the ASGE recommends colonoscopists to know their ADR and keep it higher than 30% in man and 20% in woman (population aged ≥ 50 years undergoing screening colonoscopy) (8). In contrast, most survey participants admitted colonoscope withdrawal times longer than six minutes. As it has been widely demonstrated, the time spent on colonoscope withdrawal is an important quality indicator that has been well correlated with the ADR (13). Based on this fact, one may argue that probably the ADR of our survey responders would be adequate but unknown or just unmeasured. Future efforts focusing on ADR knowledge and measurement among Spanish endoscopists should be done. Equally, the feedback from lesions previously biopsied or resected (i.e., histology), as well as the patients' outcome (i.e., complications), should be also recorded. In fact, colonoscopy procedures should not finish after colonoscope withdrawal. Regarding the quality of bowel preparation, it is usually defined as the ability to visualize the mucosa after stool and fluid have been suctioned. Bowel preparation is a well-known quality indicator and thus, it should be documented in each colonoscopy report. If bowel cleansing is inadequate to identify polyps > 5 mm in size, the ASGE recommends repeating the procedure within one year (8). In fact, a poor bowel preparation reduces detection of both small and large polyps resulting in an increase incidence of interval cancers (35). In order to optimize the colon cleansing level as well as its evaluation, most guidelines recommend the use of objective scales such as the Boston Preparation Scale or the Otawa Bowel Preparation Scale (9). On the other hand, the use of split dosages adapting it to the time of the colonoscopy have demonstrated an improvement of the bowel cleansing level as well as an increase of the ADR (36,37). Despite the large number of papers demonstrating these facts, our results confirm that they were not optimally managed among our study population.
According to our results, endoscopists "at risk" for a suboptimal quality indicators management may be identified since they have a specific profile. In fact, "younger" endoscopists, public practice setting, low volume of procedures per week and no CRC screening program are items related to more deficiencies. As a recent study has concluded, endoscopic experience, which is commonly reflected by age, volume of procedures per week and participation in a CRC screening program, affects the ADR (38). Our results prove these factors affect ADR but also many of the proposed quality indicators, such as polypectomy under antiplatelets and/or oral anticoagulants therapy, histology and complications feedback, photodocumentation of normal and abnormal findings, use of cleansing scales, complete colonoscopy definition, and use of split dosages.
After an intensive review of the informative questions, we may conclude that there is no consensus in some important endoscopic issues such as the optimal size of the endoscopy rooms, material renewal or endoscopes disinfection. Moreover, the items included in the upper and lower endoscopy reports reflected a high variability among endoscopists. In fact, only 4 out of the 9 (44.4%) items recommended to be included in the upper GI endoscopy reports were included by more than 90% of responders. On the other hand, only 4 out of the 14 (28.5%) items recommended to be included on the lower GI endoscopy were included by more than 90% of the responders. This reflects the need for homogeneous report forms among endoscopists.
This study has some limitations. Firstly, this is a survey-based study, with autodeclared answers. Responses may not reflect daily practice. Answers may show "the best picture" possible. However, with a high number of answers obtained, counting with general honest replies, this effect may be minimized. On the other hand, we have detected that the questions used in the survey regarding the use of oral anticoagulants and antiplatelet drugs may be imprecise (questions 5 and 17) since Nonsteroidal anti-inflammatory drugs (NSAIDs) and low dosages of ASA (100 mg) do not usually interfere with the endoscopic practice, and this should have been specified. Future uses of a similar survey to explore tendencies or post-intervention changes should edit these questions.
In summary, this survey-based study reveals that a significant proportion of Spanish endoscopists do not comply with the main quality indicators proposed by the international societies of endoscopy. "Young" endoscopists, public practice setting, no CRC screening program and a low volume of procedures per week should be the main targets for future formative interventions.
References
1. Hosokawa O, Shirasaki S, Kaizaki Y, et al. Invasive colorectal cancer detected up to 3 years after a colonoscopy negative for cancer. Endosc 2003;35:506-10. DOI: 10.1055/s-2003-39665. [ Links ]
2. Bressler B, Paszat LF, Vinden C, et al. Colonoscopic miss rates for right-sided colon cancer: A population-based analysis. Gastroenterol 2004;127:452-6. DOI: 10.1053/j.gastro.2004.05.032. [ Links ]
3. Van Rijn JC, Reitsma JB, Stoker J, et al. Polyp miss rate determined by tandem colonoscopy: A systematic review. Am J Gastroenterol 2006;101:343-50. DOI: 10.1111/j.1572-0241.2006.00390.x. [ Links ]
4. Bressler B, Paszat LF, Chen Z, et al. Rates of new or missed colorectal cancers after colonoscopy and their risk factors: A population-based analysis. Gastroenterol 2007;132:96-102. DOI: 10.1053/j.gastro.2006.10.027. [ Links ]
5. Leufkens AM, Van Oijen MG, Vleggaar FP, et al. Factors influencing the miss rate of polyps in a back-to-back colonoscopy study. Endoscopy 2012;44:470-5. DOI: 10.1055/s-0031-1291666. [ Links ]
6. Samadder NJ, Curtin K, Tuohy TM, et al. Characteristics of missed or interval colorectal cancer and patient survival: A population-based study. Gastroenterol 2014;146(4):950-60. DOI: 10.1053/j.gastro.2014.01.013. [ Links ]
7. Rizk MK, Sawhney MS, Cohen J, et al. Quality indicators common to all GI endoscopic procedures. Gastrointest Endosc 2015;81(1):3-16. DOI: 10.1016/j.gie.2014.07.055. [ Links ]
8. Rex DK, Schoenfeld PS, Cohen J, et al. Quality indicators for colonoscopy. Gastrointest Endosc 2015;81(1):31-53. DOI: 10.1016/j.gie.2014.07.058. [ Links ]
9. Rembacken B, Hassan C, Riemann JF, et al. Quality in screening colonoscopy: Position statement of the European Society of Gastrointestinal Endoscopy (ESGE). Endosc 2012;44(10):957-68. DOI: 10.1055/s-0032-1325686. [ Links ]
10. Park WG, Shaheen NJ, Cohen J, et al. Quality indicators for EGD. Gastrointest Endosc 2015;81(1):17-30. DOI: 10.1016/j.gie.2014.07.057. [ Links ]
11. Rutter MD, Rees CJ. Quality in gastrointestinal endoscopy. Endoscopy 2014;46(6):526-8. DOI: 10.1055/s-0034-1365738. [ Links ]
12. Mysliwiec PA, Brown ML, Klabunde CN, et al. Are physicians doing too much colonoscopy? A national survey of colorectal surveillance after polypectomy. Ann Int Med 2004;141(4):264-71. DOI: 10.7326/0003-4819-141-4-200408170-00006. [ Links ]
13. Barclay RL, Vicari JJ, Doughty AS, et al. Colonoscopic withdrawal times and adenoma detection during screening colonoscopy. New Engl J Med 2006;355(24):2533-41. DOI: 10.1056/NEJMoa055498. [ Links ]
14. Simmons DT, Harewood GC, Baron TH, et al. Impact of endoscopist withdrawal speed on polyp yield: Implications for optimal colonoscopy withdrawal time. Alim Pharm Therap 2006;24(6):965-71. DOI: 10.1111/j.1365-2036.2006.03080.x. [ Links ]
15. Saini SD, Nayak RS, Kuhn L, et al. Why don't gastroenterologists follow colon polyp surveillance guidelines? Results of a national survey. J Clin Gastroenterol 2009;43(6):554-8. DOI: 10.1097/MCG.0b013e31818242ad. [ Links ]
16. Imperiale TF, Glowinski EA, Juliar BE, et al. Variation in polyp detection rates at screening colonoscopy. Gastrointest Endosc 2009;69(7):1288-95. DOI: 10.1016/j.gie.2007.11.043. [ Links ]
17. Gellad ZF, Voils CI, Lin L, et al. Physician perceptions on colonoscopy quality: Results of a national survey of gastroenterologists. Gastroenterol Res Pract 2014;2014:510494. DOI: 10.1155/2014/510494. [ Links ]
18. Nishihara R, Wu K, Lochhead P, et al. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med 2013;369:1095-105. DOI: 10.1056/NEJMoa1301969. [ Links ]
19. Baxter NN, Goldwasser MA, Paszat LF, et al. Association of colonoscopy and death from colorectal cancer. Ann Intern Med 2009;150:1-8. DOI: 10.7326/0003-4819-150-1-200901060-00306. [ Links ]
20. Lakoff J, Paszat LF, Saskin R, et al. Risk of developing proximal versus distal colorectal cancer after a negative colonoscopy: A population-based study. Clin Gastroenterol Hepatol 2008;6:1117-21. DOI: 10.1016/j.cgh.2008.05.016. [ Links ]
21. Singh H, Nugent Z, Demers AA, et al. The reduction in colorectal cancer mortality after colonoscopy varies by site of the cancer. Gastroenterol 2010;139:1128-37. DOI: 10.1053/j.gastro.2010.06.052. [ Links ]
22. Carballo F, Muñoz-Navas M. Prevention or cure in times of crisis: The case of screening for colorectal cancer. Rev Esp Enferm Dig 2012;104(10):537-45. DOI: 10.4321/S1130-01082012001000006. [ Links ]
23. Zauber AG, Winawer SJ, O'Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med 2012;366:687-96. DOI: 10.1056/NEJMoa1100370. [ Links ]
24. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013;63:11-30. DOI: 10.3322/caac.21166. [ Links ]
25. Bibbins-Domingo K, Grossman DC, Curry SJ, et al. Screening for colorectal cancer: US Preventive Services Task Force Recommendation Statement. JAMA 2016;315(23):2564-75. DOI: 10.1001/jama.2016.5989. [ Links ]
26. European Colorectal Cancer Screening Guidelines Working Group, Von Karsa L, Patnick J, et al. European guidelines for quality assurance in colorectal cancer screening and diagnosis: Overview and introduction to the full supplement publication. Endosc 2013;45(1):51-9. DOI: 10.1055/s-0032-1325997. [ Links ]
27. Winawer SJ, Zauber AG, Ho MN, et al. Prevention of colorectal cancer by colonoscopic polypectomy. N Engl J Med 1993;329:1977-81. DOI: 10.1056/NEJM199312303292701. [ Links ]
28. Levin B, Lieberman DA, McFarland B, et al. Screening and surveillance for the early detection of colorectal cancer and adenomatous polyps, 2008: A joint guideline from the American Cancer Society, the US Multi-Society Task Force on Colorectal Cancer, and the American College of Radiology. Gastroenterol 2008;134:1570-95. DOI: 10.1053/j.gastro.2008.02.002. [ Links ]
29. Hardcastle JD, Chamberlain JO, Robinson MH, et al. Randomised controlled trial of faecal-occult-blood screening for colorectal cancer. Lancet 1996;348:1472-7. DOI: 10.1016/S0140-6736(96)03386-7. [ Links ]
30. Kronborg O, Fenger C, Olsen J, et al. Randomised study of screening for colorectal cancer with faecal-occult-blood test. Lancet 1996;348:1467-71. DOI: 10.1016/S0140-6736(96)03430-7. [ Links ]
31. Mandel JS, Bond JH, Church TR, et al. Reducing mortality from colorectal cancer by screening for fecal occult blood. N Engl J Med 1993;328:1365-71. DOI: 10.1056/NEJM199305133281901. [ Links ]
32. Hillyer GC, Basch CH, Basch CE, et al. Gastroenterologists' perceived barriers to optimal pre-colonoscopy bowel preparation: Results of a national survey. J Cancer Educ 2012;27:526-32. DOI: 10.1007/s13187-012-0364-x. [ Links ]
33. Tinsley A, Naymagon S, Trindade AJ, et al. A survey of current practice of venous thromboembolism prophylaxis in hospitalized inflammatory bowel disease patients in the United States. J Clin Gastroenterol 2013;47(1):e1-e6. DOI: 10.1097/MCG.0b013e31824c0dea. [ Links ]
34. Whitson MJ, Bodian CA, Aisenberg J, et al. Is production pressure jeopardizing the quality of colonoscopy? A survey of U.S. endoscopists' practices and perceptions. Gastrointest Endosc 2012;75(3):641-8. DOI: 10.1016/j.gie.2011.10.032. [ Links ]
35. Lieberman DA, Rex DK, Winawer SJ, et al. Guidelines for colonoscopy surveillance after screening and polypectomy: A consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterol 2012;143:844-57. DOI: 10.1053/j.gastro.2012.06.001. [ Links ]
36. Enestvedt BK, Tofani C, Laine LA, et al. 4-liter split-dose polyethylene glycol is superior to other bowel preparations, based on systematic review and meta-analysis. Clin Gastroenterol Hepatol 2012;10:1225-31. DOI: 10.1016/j.cgh.2012.08.029. [ Links ]
37. Kilgore TW, Abdinoor AA, Szary NM, et al. Bowel preparation with split-dose polyethylene glycol before colonoscopy: A meta-analysis of randomized controlled trials. Gastrointest Endosc 2011;73:1240-5. DOI: 10.1016/j.gie.2011.02.007. [ Links ]
38. Jover R, Zapater P, Bujanda L, et al. COLONPREV Study Investigators. Endoscopist characteristics that influence the quality of colonoscopy. Endosc 2016;48(3):241-7. DOI: 10.1055/s-0042-100185. [ Links ]
![]() Correspondence:
Correspondence:
Ignacio Fernández-Urien.
Department of Digestive Diseases.
Complejo Hospitalario de Navarra.
C. Irunlarrea, 3.
31008 Pamplona, Navarra. Spain
e-mail: ifurien@yahoo.es
Received: 02-11-2016
Accepted: 19-12-2016