Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.109 no.2 Madrid feb. 2017
https://dx.doi.org/10.17235/reed.2017.4629/2016
ORIGINAL PAPERS
Validation of SPICE, a method to differenciate small bowel submucosal lesions from innocent bulges on capsule endoscopy
Jaime P. Rodrigues, Rolando Pinho, Adélia Rodrigues, Joana Silva, Ana Ponte, Mafalda Sousa and João Carvalho
Gastroenterology Department. Centro Hospitalar Vila Nova de Gaia/Espinho. Vila Nova de Gaia, Portugal
ABSTRACT
Background and aims: Small bowel submucosal lesions (SBSL) and innocent bulges may have an identical appearance and be difficult to distinguish on small bowel capsule endoscopy (SBCE). Recently, Girelli et al. proposed a score, smooth, protruding lesion index on capsule endoscopy (SPICE), in order to differentiate between the two. We aimed to evaluate and validate SPICE as a differentiation method between innocent bulges and SBSLs.
Methods: We evaluated all SBCEs performed in our department between January 2005 and September 2015, and selected the ones with a smooth, round, protruding lesion in the small bowel. Lesions with suspicious characteristics were excluded. A video clip of the region of interest was created and SPICE was assigned blindly and independently by two endoscopists. We determined the discriminative ability of SPICE using the definitive diagnosis of each patient as the standard criteria.
Results: We included 30 SBCEs corresponding to 12 SBSLs (four gastrointestinal stromal tumors, two neuroendocrine tumors, four lipomas and two polypoid lymphangiectasias) and 18 innocent bulges. SPICE scores ranged from 0 to 4, allowing the distinction between SBSLs and innocent bulges (p < 0.001). SPICE > 2 had a 66.7% sensitivity, 100.0% specificity, 100.0% positive predictive value and 78.3% negative predictive value, and the area under the curve was 0.88 (95% CI, 0.73-1.00; p < 0.001) for the diagnosis of SBSL.
Conclusions: Our data support SPICE, namely a score > 2, as a predictive method of SBSLs. Taking into account its simplicity, it may be very useful in the distinction between SBSLs and innocent bulges on SBCE.
Key words: Capsule endoscopy. SPICE. Submucosal lesions. Intestinal neoplasms. Innocent bulges.
Introduction
Small bowel malignant tumors are some of the rarest neoplasms arising from the gastrointestinal (GI) tract. Taking into account that the small bowel comprises about 70-80% of the length and 90% of the surface area of the alimentary tract, it is interesting to note that they represent only 1-3% of all primary GI tumors (1-3). The overall incidence of small bowel cancer was estimated to be 26.1 per million in men and 17.7 per million in women in a large US population-based study (4), but recent evidence suggests that it is increasing, both in the US (5,6) and Europe (7,8).
Due to nonspecific or absent symptoms and clinical signs, the diagnosis of small bowel tumors (SBTs) is often delayed. Patients commonly present with disseminated disease, and complications such as bleeding, intestinal obstruction or perforation at presentation (9-13). Since prompt diagnosis and treatment may improve the prognosis of patients suffering from this condition, early detection of suspicious lesions is critical to an adequate clinical approach.
In the past few years there has been a lot of development in small bowel endoscopic and radiologic techniques, allowing full visualization of the small intestine. At least part of the increase in small bowel cancer incidence may be explained by this marked improvement in diagnostic methods. Among these, capsule endoscopy (CE), which was introduced into practice in 2001, assumes a more significant role. It is a noninvasive, painless and radiation-free tool, able to provide complete visualization and high-quality images of the small bowel and its mucosa, hence it has rapidly gained acceptance as a standard method for small bowel evaluation. It has a higher diagnostic yield and sensitivity than other radiologic modalities for SBTs, providing earlier diagnosis and treatment (14-17). In the largest database published so far concerning SBTs detected by CE (124 tumors with 5,129 capsule procedures performed), Rondonotti et al. reported a 2.4% prevalence of SBTs (18). In other series, SBTs were found in 8.9% (Cobrin et al. [19]), 6.3% (Bailey et al. [20]), 2.5% (Urbain et al. [21]) and 4.3% (Cheung et al. [22]) of cases. When performed to explore obscure bleeding, the main form of presentation of SBTs (23), the sensitivity for SBT diagnosis is 89-95%, with a specificity of 75-95% (24,25).
The main limitation of small bowel capsule endoscopy (SBCE) is the impracticality of the technique, namely the inability to take biopsies, to perform therapeutic interventions, and to handle the capsule during the examination. Another drawback of SBCE is represented by the difficulty in distinguishing a submucosal lesion from an innocent bulge, a smooth protrusion of normal mucosa resulting from loop angulation and/or the impression of an adjacent loop. Up to half of the most common histological types of small bowel cancer, namely adenocarcinomas, neuroendocrine tumors, lymphomas and sarcomas (9,23), can appear endoscopically as smooth, round, protruding lesions (18). To determine the nature of small bowel protrusions on CE, Shyung et al. described a scoring system composed of five suspicious characteristics: bleeding, mucosal disruption, irregular surface, color, and white villi (26). However, when these signs are not present, innocent bulges and submucosal lesions may look very similar and the later can be missed when their overlying mucosa is intact. In fact, evidence is growing that CE can miss a significant number of tumors in the small bowel (15,27-30). Moreover, Sato et al. showed that recent chromoendoscopy techniques, namely flexible spectral imaging color enhancement (FICE), are not particularly useful for improving the detection of tumors on CE (31).
Taking all this into consideration, the importance of finding new tools to improve the diagnosis of small bowel lesions is fundamental. In a previous retrospective and observational study, Girelli et al. identified four morphologic and temporal criteria that characterized innocent bulging: a mass with ill-defined boundaries, diameter larger than its height, nonvisible lumen in the frames in which it appears and mass image lasting less than ten minutes (32). Based on these criteria the same authors developed a scoring system called SPICE (smooth, protruding lesions index on capsule endoscopy), aimed at discriminating submucosal masses from innocent bulges (33) (Table I). SPICE proved to be accurate in distinguishing both of them when suspicious characteristics are lacking; a score greater than two showed a sensitivity of 83.3% and a specificity of 89.4% and was predictive of small bowel malignancy. Recently, the European Society of Gastrointestinal Endoscopy cited the score in their clinical guidelines, highlighting that further validation and validation studies are needed (34).
In the present study we aimed to evaluate and validate this recent scoring system as a method of to differenciate submucosal lesions of the small bowel from innocent bulges.
Material and methods
SPICE classification
As previously mentioned, an innocent bulge is associated with the following characteristics: an ill-defined boundary with the surrounding mucosa, a diameter larger than its height, nonvisible lumen in the frames in which it appears, and an image lasting less than ten minutes (32). SPICE classification was accomplished using the same principles as in the original article by Girelli et al.: the three morphological criteria were measured in the frame(s) allowing a better characterization of the variable of interest, while the temporal criterion was measured from the first to the last frame in which the protrusion appeared; the lumen was considered as visible when at least three consecutive small bowel folds were seen; lesion diameter and height were roughly estimated and, in doubtful cases, a ruler was applied on the screen to perform an exact measurement. SPICE was calculated by summing the number of unfulfilled criteria for innocent bulges (Table I), then we tested whether or not this scoring system was able to distinguish submucosal masses from innocent bulges.
Study design
All consecutive CEs performed in our department, between January 2005 and September 2015, were evaluated, and the ones with a smooth, round protruding lesion of any size in the small bowel, namely between the pylorus and ileocecal valve, independently of the duration of the image of the protrusion, were selected. Only patients who had been studied with upper endoscopy and colonoscopy prior to CE were included. Patients with no follow-up were excluded, follow-up was considered as having at least one appointment and another diagnostic study of the small bowel, endoscopic (enteroscopy) or radiologic (computed tomography enterography, magnetic resonance enterography and/or barium study of small bowel) after the CE. Other exclusion criteria included: the presence of suspicious characteristics (such as erythema, bleeding, mucosal disruption in the form of erosion or ulcer, irregular surface or exudates) or other signs normally reported in submucosal lesions (such as converging folds and abnormal vascularity) on the protrusion, technical capsule failures, poor small bowel visualization and staging of polyposis syndromes.
A video clip comprising all the protrusion time intervals was created and anonymized. SPICE was assigned blindly and independently by two experienced endoscopists; any disagreement on the final score was resolved by consensus after video re-evaluation.
Relevant clinical information was retrospectively collected from medical records, including demographic characteristics, other findings detected on CE besides protrusion lesions, and reports from other diagnostic studies subsequently performed. The final diagnosis of each lesion was the standard criterion that was subsequently matched with the total SPICE scores of each video.
Diagnostic definition
Protrusions with a final histologic diagnosis or pathognomonic endoscopic features were considered as small bowel submucosal lesions. Apparent lesions detected on the initial CE, not confirmed on the subsequent study and with no specific symptoms or clinical signs on the subsequent appointment(s) were considered as innocent bulges.
Capsule endoscopy
The Given® Video Capsule system and the MiroCam® Video Capsule system were used in the study. Written informed consent was obtained from all patients. CE studies were carried out according to our department protocol, which includes an overnight fast, suspension of iron supplements eight days before the procedure and a liquid diet in the last meal. After capsule ingestion, patients were allowed to eat a light snack four hours later. Patients were evaluated with real-time view 30 minutes after capsule ingestion, and a prokinetic agent (metoclopramide) was administered when the capsule was found in the stomach.
Statistical analysis
Statistical analysis was performed using the IBM® SPSS® Statistics software (SPSS Inc., Chicago), version 23.0.
Categorical variables are presented as frequencies and percentages while continuous variables are presented as means and standard deviations (SD) for variables with normal distributions, or medians and interquartile ranges (IQR) for variables with skewed distributions. Normal distribution of the data was tested with Kolmogorov-Smirnov and Shapiro-Wilk tests or via analysis of skewness and kurtosis (maximum tolerable values of skewness and kurtosis of 1). Categorical variables were compared using the Chi-squared test or Fisher's exact test, as appropriate, while continuous variables were compared using the Student's t-test for variables with normal distributions or non-parametric tests (Mann-Whitney) for variables with skewed distributions. Odds ratios (OR) with a 95% confidence interval (CI) were also calculated.
All reported p values are two-tailed, with a p value of 0.05 indicating statistical significance.
Sensitivity, specificity, negative and positive predictive values (NPV and PPV, respectively) and the area under the receiver operating characteristic curve (AUC) were calculated for a SPICE cut-off value of 2. The 95% confidence intervals (CIs) were included for sensitivity, specificity, PPV, NPV and AUC.
Results
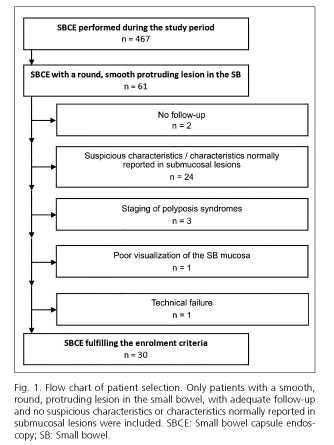
During the study period our department performed 467 SBCEs, of which 61 (13.1%) presented with a round, smooth protruding lesion. Thirty-one capsule endoscopies were excluded from subsequent analysis due to the following reasons: absence of follow-up (n = 2); presence of suspicious characteristics or characteristics normally reported in submucosal lesions (n = 24); poor visualization of the small-bowel (n = 1); technical failure (n = 1); and staging of polyposis syndromes (n = 3). Ultimately, 30 videos (6.4%), corresponding to 28 patients, fulfilled the enrolment criteria (Fig. 1). Multiple round, smooth protruding lesions were not found during the same procedure. The capsule reached the colon in all 30 (100%) examinations.
Diagnosis
Twelve of the 30 selected protrusions (40.0%) were considered to have a final diagnosis of SBSL; this included four gastrointestinal stromal tumors (GIST), two neuroendocrine tumors (NET), four lipomas and two polypoid lymphangiectasias. All SBTs, namely GISTs and NETs, accounting for 20.0% of the selected protrusions, were diagnosed after resective surgery and subsequent histological analysis. The diagnosis of lipoma and lymphangiectasia was considered after visualization of characteristic findings with single-balloon enteroscopy. The final diagnosis of the protrusions classified as innocent bulges (n = 18, 60.0%) was considered after a variable combination of several diagnostic studies, including barium studies of the small-bowel, computed tomography enterography, magnetic resonance enterography, and/or single-balloon enteroscopy associated with an uneventful follow-up.
Patients
The mean age of patients was 54.5 ± 18.3 years and 50.0% (n = 15) were female (Table II). The age of the patients was significantly higher in the SBSL group (64.5 ± 11.4 years) than in the innocent bulge group (47.8 ± 19.2 years); p = 0.011. There was a significant association between female gender and the diagnosis of a small bowel lesion; p = 0.025 (OR = 6.0; 95% CI, 1.2-30.7).
Indication
The indications for SBCE in the selected patients were: unexplained iron deficiency anemia (n = 13, 43.3%), obscure GI bleeding (n = 8, 26.7%), suspected Crohn's disease (n = 4, 13.3%), abdominal pain (n = 3, 10.0%) and primary neoplasia screening (n = 2, 6.7%). There was a significant statistical association between the group of patients who underwent the procedure due to anemia or obscure GI bleeding (100.0% in the SBSL group vs 50.0% in the bulge group) and the diagnosis of a lesion; p = 0.004.
Follow-up
The overall median follow-up time was 45.4 ± 44.3 months and 54.3 ± 48.9 if we consider only the innocent bulge group.
Localization
Considering the localization of the protrusions through the small bowel, we found one (3.3%) localized in the duodenum, 13 (43.3%) in the jejunum and 16 (53.3%) in the ileum. There was no significant difference between the final diagnosis of the groups regarding the localization of the protrusions; p > 0.05.
SPICE evaluation
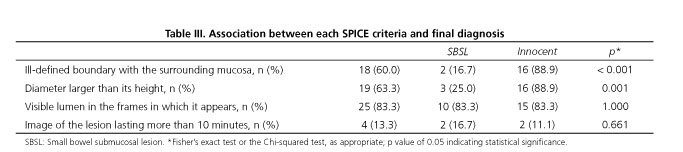
Analyzing the four criteria that constitute SPICE (Table III), there were significantly more protrusions in the innocent bulge group (n = 16, 88.9%) compared to the lesion group (n = 2, 16.7%) they had ill-defined boundaries; p < 0.001 (OR = 40.0; 95% CI, 4.8-331.0). With regard to the shape, 88.9% (n = 16) of the protrusions in the innocent bulge group had a diameter larger than its height vs 25.0% (n = 3) in the SBSL group, thus there was also a significant association between the second SPICE criteria (protrusion diameter larger than its height) and the diagnosis of an innocent bulge; p = 0.001 (OR = 24.0; 95% CI, 3.4-171.5). On the other hand, no differences between the absolute diameter of the protrusions on the SBSL group (10.4 ± 4.7 millimeters) and those of the innocent bulge group (11.6 ± 2.2 millimeters) was found; p = 0.449. There was no difference between the bulge and lesion groups regarding visualization of the lumen in the same frames of the protrusion (n = 3, 83.3% vs n = 2, 83.3%); p = 1.000. Likewise, no association between the final criteria of SPICE, protrusion time interval higher than ten minutes, and the final diagnosis (n = 2, 11.1% in the bulge group vs n = 2, 16.7% in the lesion group) was found; p = 0.661. Furthermore, the absolute protrusion time interval did not differ significantly between the SBSL (median = 74.0; IQR = 10.0-209.0 seconds) and the innocent bulge groups (median = 136.5; IQR = 36.5-350.0 seconds); p = 0.553.
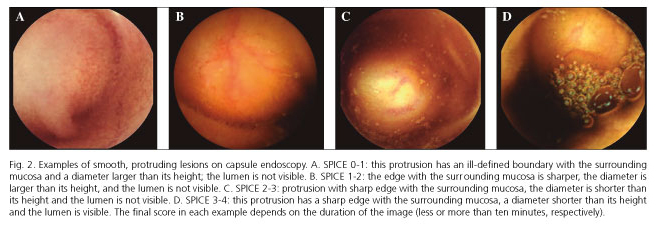
SPICE scores ranged from 0 to 4 (Fig. 2) and were significantly higher in the SBSL group (2.58 ± 1.00) than in the innocent bulge group (1.17 ± 0.71); p < 0.001. Adopting the suggested cut-off value > 2, SPICE had a sensitivity of 66.7% (95% CI, 34.9-90.1%); specificity of 100.0% (95% CI, 81.5-100.0%); positive predictive value of 100.0% (95% CI, 63.1-100.0%) and negative predictive value of 81.8% (95% CI, 59.7-94.8%). The area under the receiver operating characteristic curve was 0.88 (95% CI, 0.73-1.00), p < 0.001 (Fig. 3).
Characteristics of SBSL and innocent bulge groups are summarized in table II.
Discussion
Small bowel research has been considered to be the final frontier for endoscopists. Since the release of CE about 15 years ago, the diagnosis of multiple small bowel diseases was revolutionized. However, equivocal findings are not uncommon and diagnostic dilemmas persist. The endoscopist has to make a diagnosis commonly based on a short video segment, lesions cannot be touched and biopsy specimens cannot be obtained. Additionally, the sensitivity of SCBE is not perfect as it can miss different types of lesions. In the last few years, several reports proved that CE can miss a significant number of tumors in the small bowel, with a miss rate of approximately 20% (15,27-30). There are many potential reasons for this disturbing false-negative rate of SBTs. These include: a limited field of vision, a non-continuous image capture, collapsed bowel or folds and loop angulations that hide lesions, poor bowel cleansing, and incomplete studies. In addition, tumors are typically unifocal, hence more likely to be missed than diffuse lesions; finally, mass lesions located in the proximal small bowel may be missed on the basis of capsule velocity or tumble. These findings suggest that, despite the major advances in small bowel examination by CE, the detection of small bowel mass lesions by this modality remains a challenge and the differential diagnosis between masses and bulges is not straightforward.
A consensus group in 2007 proposed a classification of mass lesions of the small bowel that could define the probability of a tumor (high, intermediate or low) depending on major signs (bleeding, mucosal disruption, irregular surface, polypoid appearance and color) and minor signs (delayed passage, white villi and invagination) (35). This classification would subsequently guide the management of patients with suspected SBT. When these endoscopic features are absent, bulges and masses may look very similar, and based on this assumption, Girelli et al. identified four criteria (three morphologic, one temporal) that permitted the distinction of these two entities (32). Combining these criteria, they created a score, SPICE (smooth, protruding lesions index on capsule endoscopy) that proved to be accurate in distinguishing submucosal masses from innocent bulges; to better understand its real clinical importance, further validation studies are needed (33).
In the present study the prevalence of selected smooth, round, protruding lesions was 6.4%, identical to previous reports. Of these, 40% were considered as a small bowel submucosal lesion and 20% as a submucosal tumor, a figure also similar to previous data. The histologic type of diagnosed tumors, GI stromal and neuroendocrine tumors, was also in some way expected considering that these are two of the most common small bowel tumors and that they arise from the subepithelial layers of the bowel wall (9). As previously reported in the literature, increasing age was a predictive factor for the diagnosis of a small bowel lesion. Also, our data support the current understanding of the most common form of presentations of SBTs, such as iron deficiency anemia and obscure GI bleeding, which were also predictors of the diagnosis of lesions.
Considering each criteria of SPICE, the characteristics of the boundaries with the surrounding mucosa and the diameter of the protrusion comparatively to its height presented discriminating capability between a lesion and an innocent bulge, and may be the main focus of attention of the capsule reader. On the other hand, neither the visibility of the lumen or the duration of the image lesion allowed the distinction between the two entities.
The score and the proposed cut-off proved globally to be an excellent discriminative instrument between SBSL and innocent bulges. A SPICE > 2 presented a sensitivity of 67% and a specificity of 100%, corresponding to four false-negative and zero false-positive findings. Regarding the false-negative rate, only one lesion with malignant potential, namely a GIST, was missed by the score; the remaining were benign lesions, without the need of subsequent therapy or prognostic significance. The misidentified GIST was histologically diagnosed after resective surgery and was found to have extra-luminal growth that is extrinsic to the small bowel wall. This characteristic together with a fast protrusion time interval and its relatively small size hampered a higher SPICE score and a correct classification. Even with these features, the clinical suspicion of this lesion was high and the patient underwent enteroscopy and subsequently surgery that allowed an accurate diagnosis. SPICE may help the endoscopist to estimate the probability of a submucosal lesion with an excellent degree of confidence, especially for ruling out the presence of a lesion and identifying innocent bulges, and may also be a major tool to aid the tricky interpretation of small bowel protrusions with overlying normal mucosa.
The present study has a few limitations. Primarily, it is a retrospective study of a referral population and part of the initial evaluation of some patients, including endoscopy and colonoscopy, was also performed at other centers. The number of enrolled patients is relatively small, the rarity of small bowel tumors and the strict exclusion criteria such as the exclusion of protrusions with suspicious criteria are the main reasons for this. Although all patients with a submucosal tumor had a histological diagnosis, this was not available in all patients of the innocent bulge group. However, normal findings during the subsequent studies performed as well as the long, uneventful follow-up increase our confidence in the accuracy of the final diagnosis of these patients.
This work also has important merits. The protrusions were classified by two independent endoscopists via detailed analysis of the whole video segment where they appear, enabling a rigorous stratification of each one of them. The strict exclusion criteria enabled us to select only protrusions that create doubts in clinical practice, making interpretation of the results more valuable. The median follow-up of the patients was prolonged, mainly of the innocent bulge group. Although relatively small, the number of protrusions included was higher than in the study by Girelli et al., confirming their results and allowing a more realistic and precise estimation of the utility of the score.
In conclusion, attempts to distinguish SBSLs from innocent bulges rely mainly on the presence of suspicious characteristics. SPICE is a recently developed scoring system based on simple morphologic and temporal criteria, easy to classify and useful for distinguishing both of them when these suspicious characteristics are absent. Our results are generally in agreement with those by Girelli et al. proving the accuracy of the score and its potential clinical benefits. Taking into account the lower incidence of SBTs, its use may avoid unnecessary small bowel studies and even inappropriate surgery. On the other hand, it may allow the diagnosis of tumors at an earlier stage, possibly improving the prognosis of patients. Prospective studies with larger numbers of patients may be required to further analyze the diagnostic and therapeutic impact of SPICE and confirm these data.
References
1. O'Riordan BG, Vilor M, Herrera L. Small bowel tumors: An overview. Dig Dis 1996;14:245-57. DOI: 10.1159/000171556. [ Links ]
2. Neugut AI, Jacobson JS, Suh S, et al. The epidemiology of cancer of the small bowel. Cancer Epidemiol Biomarkers Prev 1998;7:243-51. [ Links ]
3. Pan SY, Morrison H. Epidemiology of cancer of the small intestine. World J Gastrointest Oncol 2011;3:33-42. [ Links ]
4. Goodman MT, Matsuno RK, Shvetsov YB. Racial and ethnic variation in the incidence of small-bowel cancer subtypes in the United States, 1995-2008. Dis Colon Rectum 2013;56:441-8. DOI: 10.1097/DCR.0b013e31826b9d0a. [ Links ]
5. Haselkorn T, Whittemore AS, Lilienfeld DE. Incidence of small bowel cancer in the United States and worldwide: Geographic, temporal, and racial differences. Cancer Causes Control 2005;16:781-7. DOI: 10.1007/s10552-005-3635-6. [ Links ]
6. Bilimoria KY, Bentrem DJ, Wayne JD, et al. Small bowel cancer in the United States: Changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg 2009;249:63-71. DOI: 10.1097/SLA.0b013e31818e4641. [ Links ]
7. Lu Y, Frobom R, Lagergren J. Incidence patterns of small bowel cancer in a population-based study in Sweden: Increase in duodenal adenocarcinoma. Cancer Epidemiol 2012;36:e158-63. DOI: 10.1016/j.canep.2012.01.008. [ Links ]
8. Bojesen RD, Andersson M, Riis LB, et al. Incidence of, phenotypes of and survival from small bowel cancer in Denmark, 1994-2010: A population-based study. J Gastroenterol 2016. DOI: 10.1007/s00535-016-1171-7: 1-9. [ Links ]
9. Reynolds I, Healy P, McNamara DA. Malignant tumours of the small intestine. Surgeon 2014;12:263-70. DOI: 10.1016/j.surge.2014.02.003. [ Links ]
10. Sailer J, Zacherl J, Schima W. MDCT of small bowel tumours. Cancer Imaging 2007;7:224-33. DOI: 10.1102/1470-7330.2007.0032. [ Links ]
11. Aparicio T, Zaanan A, Svrcek M, et al. Small bowel adenocarcinoma: Epidemiology, risk factors, diagnosis and treatment. Dig Liver Dis 2014;46:97-104. DOI: 10.1016/j.dld.2013.04.013. [ Links ]
12. Landerholm K, Falkmer S, Jarhult J. Epidemiology of small bowel carcinoids in a defined population. World J Surg 2010;34:1500-5. DOI: 10.1007/s00268-010-0519-z. [ Links ]
13. Pennazio M, Rondonotti E, De Franchis R. Capsule endoscopy in neoplastic diseases. World J Gastroenterol 2008;14:5245-53. DOI: 10.3748/wjg.14.5245. [ Links ]
14. Honda W, Ohmiya N, Hirooka Y, et al. Enteroscopic and radiologic diagnoses, treatment, and prognoses of small-bowel tumors. Gastrointest Endosc 2012;76:344-54. DOI: 10.1016/j.gie.2012.04.443. [ Links ]
15. Han JW, Hong SN, Jang HJ, et al. Clinical efficacy of various diagnostic tests for small bowel tumors and clinical features of tumors missed by capsule endoscopy. Gastroenterol Res Pract 2015;2015:6. DOI: 10.1155/2015/623208. [ Links ]
16. Triester SL, Leighton JA, Leontiadis GI, et al. A meta-analysis of the yield of capsule endoscopy compared to other diagnostic modalities in patients with obscure gastrointestinal bleeding. Am J Gastroenterol 2005;100:2407-18. DOI: 10.1111/j.1572-0241.2005.00274.x. [ Links ]
17. De Mascarenhas-Saraiva MN, Da Silva Araujo Lopes LM. Small-bowel tumors diagnosed by wireless capsule endoscopy: Report of five cases. Endoscopy 2003;35:865-8. DOI: 10.1055/s-2003-42625. [ Links ]
18. Rondonotti E, Pennazio M, Toth E, et al. Small-bowel neoplasms in patients undergoing video capsule endoscopy: A multicenter European study. Endoscopy 2008;40:488-95. DOI: 10.1055/s-2007-995783. [ Links ]
19. Cobrin GM, Pittman RH, Lewis BS. Increased diagnostic yield of small bowel tumors with capsule endoscopy. Cancer 2006;107:22-7. DOI: 10.1002/cncr.21975. [ Links ]
20. Bailey AA, Debinski HS, Appleyard MN, et al. Diagnosis and outcome of small bowel tumors found by capsule endoscopy: A three-center Australian experience. Am J Gastroenterol 2006;101:2237-43. DOI: 10.1111/j.1572-0241.2006.00749.x. [ Links ]
21. Urbain D, De Looze D, Demedts I, et al. Video capsule endoscopy in small-bowel malignancy: A multicenter Belgian study. Endoscopy 2006;38:408-11. DOI: 10.1055/s-2005-921203. [ Links ]
22. Cheung DY, Lee IS, Chang DK, et al. Capsule endoscopy in small bowel tumors: A multicenter Korean study. J Gastroenterol Hepatol 2010;25:1079-86. DOI: 10.1111/j.1440-1746.2010.06292.x. [ Links ]
23. Cardoso H, Rodrigues JT, Marques M, et al. Malignant small bowel tumors: Diagnosis, management and prognosis. Acta Med Port 2015;28:448-56. DOI: 10.20344/amp.6479. [ Links ]
24. Pennazio M, Santucci R, Rondonotti E, et al. Outcome of patients with obscure gastrointestinal bleeding after capsule endoscopy: Report of 100 consecutive cases. Gastroenterol 2004;126:643-53. DOI: 10.1053/j.gastro.2003.11.057. [ Links ]
25. Hartmann D, Schmidt H, Bolz G, et al. A prospective two-center study comparing wireless capsule endoscopy with intraoperative enteroscopy in patients with obscure GI bleeding. Gastrointest Endosc 2005;61:826-32. DOI: 10.1016/S0016-5107(05)00372-X. [ Links ]
26. Shyung LR, Lin Sc Fau-Shih SC, Shih Sc Fau-Chang WH, et al. Proposed scoring system to determine small bowel mass lesions using capsule endoscopy. [ Links ]
27. Ross A, Mehdizadeh S, Tokar J, et al. Double balloon enteroscopy detects small bowel mass lesions missed by capsule endoscopy. Dig Dis Sci 2008;53:2140-3. DOI: 10.1007/s10620-007-0110-0. [ Links ]
28. Zagorowicz ES, Pietrzak AM, Wronska E, et al. Small bowel tumors detected and missed during capsule endoscopy: Single center experience. World J Gastroenterol 2013;19:9043-8. DOI: 10.3748/wjg.v19.i47.9043. [ Links ]
29. Chong AK, Chin BW, Meredith CG. Clinically significant small-bowel pathology identified by double-balloon enteroscopy but missed by capsule endoscopy. Gastrointest Endosc 2006;64:445-9. DOI: 10.1016/j.gie.2006.04.007. [ Links ]
30. Baichi M, Arifuddin RM, Mantry PS. Small bowel masses found and missed on capsule endoscopy for obscure bleeding. Gastrointest Endosc 2007;42(9):1127-32. [ Links ]
31. Sato Y, Sagawa T, Hirakawa M, et al. Clinical utility of capsule endoscopy with flexible spectral imaging color enhancement for diagnosis of small bowel lesions. Endosc Int Open 2014;2:E80-7. DOI: 10.1055/s-0034-1365526. [ Links ]
32. Girelli CM, Porta P. Bulge or mass? A diagnostic dilemma of capsule endoscopy. Endoscopy 2008;40:703-4. DOI: 10.1055/s-2008-1077483. [ Links ]
33. Girelli CM, Porta P, Colombo E, et al. Development of a novel index to discriminate bulge from mass on small-bowel capsule endoscopy. Gastrointest Endosc 2011;74:1067-74;quiz1115e1061-5. DOI: 10.1016/j.gie.2011.07.022. [ Links ]
34. Pennazio M, Spada C, Eliakim R, et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 2015;47:352-76. DOI: 10.1055/s-0034-1391855. [ Links ]
35. Mergener K, Ponchon T, Gralnek I, et al. Literature review and recommendations for clinical application of small-bowel capsule endoscopy, based on a panel discussion by international experts. Consensus statements for small-bowel capsule endoscopy, 2006/2007. Endoscopy 2007;39:895-909. DOI: 10.1055/s-2007-966930. [ Links ]
![]() Correspondence:
Correspondence:
Jaime Pereira Rodrigues.
Gastroenterology Department.
Centro Hospitalar Vila Nova de Gaia/Espinho.
Rua Dr. Francisco Sá Carneiro.
4434-502 Vila Nova de Gaia, Portugal
e-mail: jaimepereirarodrigues@gmail.com
Received: 03-10-2016
Accepted: 29-10-2016