Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.109 no.2 Madrid feb. 2017
https://dx.doi.org/10.17235/reed.2016.4600/2016
ORIGINAL PAPERS
Clinical status, quality of life, and work productivity in Crohn's disease patients after one year of treatment with adalimumab
Cristina Saro1, Daniel Ceballos2, Fernando Muñoz3, Cristóbal de-la-Coba1, María Dolores Aguilar4, Pablo Lázaro4, Valle García-Sánchez5, Mariola Hernández6, Jesús Barrio7, Ruth de-Francisco8, Luis I. Fernández9 and Manuel Barreiro-de-Acosta10
1Gastroenterology Service. Hospital Cabueñes. Gijón, Spain.
2Gastroenterology Service. Hospital Dr. Negrín. Las Palmas, Spain.
3Gastroenterology Service. Hospital de Salamanca. Salamanca, Spain.
4Research Division. Advanced Techniques in Health Services Research. Madrid, Spain.
5Digestive System Service. Hospital Universitario Reina Sofía. Instituto Maimónides de Investigación Biomédica de Córdoba (IMIBIC). Universidad de Córdoba. Córdoba, Spain.
6Gastroenterology Service. Hospital Virgen del Puerto. Plasencia, Spain.
7Gastroenterology Service. Hospital Universitario Río Hortega. Valladolid, Spain.
8Gastroenterology Service. Hospital Universitario Central de Asturias. Oviedo, Spain.
9Gastroenterology Service. Hospital Clínico Universitario de Valladolid. Valladolid, Spain.
10Gastroenterology Service. Hospital Clínico Universitario de Santiago. Santiago de Compostela, Spain
On behalf of the EFICADEC researchers group: Patricia Romero, Hospital General Universitario Santa María del Rosell, Cartagena, Murcia (Spain); Víctor Orive, Hospital Universitario de Basurto, Bilbao, Vizcaya (Spain); José Luis Cabriada, Hospital de Galdakao-Usansolo, Galdakao, Vizcaya (Spain); Alfredo Lucendo, Hospital General de Tomelloso, Tomelloso, Ciudad Real (Spain); Víctor Amo, Hospital Regional Universitario Carlos Haya, Málaga (Spain); Daniel Carpio, Hospital Montecelo, Pontevedra (Spain); Andrés Sansó, Hospital de Manacor, Palma de Mallorca (Spain); Miguel Pato, Complejo Hospitalario Universitario de Orense, Orense (Spain); Juan Ramón Pineda, Hospital del Meixoeiro, Vigo (Spain); Elena Gento, Hospital Universitario de Burgos, Burgos (Spain); Daniel Ginard, Hospital Universitario Son Espases, Palma de Mallorca (Spain); Juan Antonio Borrallo, Hospital Don Benito-Villanueva de la Serena, Don Benito, Badajoz (Spain); Santos Pereira, Hospital Xeral Cíes, Vigo (Spain); Juan Jesús Puente, Hospital Alto Guadalquivir, Andújar, Jaén (Spain); Erwin Quevedo, Hospital de Mérida, Mérida, Badajoz (Spain); Francisco Gallego, Hospital de Poniente, El Egido, Almería (Spain); Javier Castro, Hospital Arquitecto Marcide, Ferrol, A Coruña (Spain); Benito González, Complejo Hospitalario Universitario A Coruña, A Coruña (Spain); Teresa Castro, Hospital de Jerez, Jerez de la Frontera, Cádiz (Spain); Antonio Rodríguez, Hospital Universitario de Salamanca, Salamanca (Spain); Leopoldo Rosés, Complejo Hospitalario Xeral-Calde, Lugo (Spain); Olga Merino, Hospital Universitario de Cruces, Baracaldo, Vizcaya (Spain); Ana Trapero, Complejo Hospitalario de Jaén, Jaén (Spain); Eduardo Leo, Hospital Universitario Virgen del Rocío, Sevilla (Spain); and Ma José Alcalá, Hospital Obispo Polanco, Teruel (Spain).
This work was supported by a grant from the Spanish Working Group on Crohn's Disease and Ulcerative Colitis (Grupo Español de Trabajo en Enfermedad de Crohn y Colitis Ulcerosa [GETECCU]).
ABSTRACT
Objective: Clinical trials have shown the efficacy of adalimumab in Crohn's disease, but the outcome in regular practice remains unknown. The aim of the study was to examine clinical status, quality of life, and work productivity of Crohn's disease patients receiving adalimumab for one year in the context of usual clinical practice.
Material and methods: This was a prospective, observational study with a one-year follow-up. After baseline, Crohn's disease patients were evaluated at 1, 3, 6, 9, and 12 months after starting treatment with adalimumab. Outcome variables included: clinical status (measured with CDAI), quality of life (measured with EuroQoL-5D and IBDQ), and work productivity (measured with WPAI questionnaire). These outcome variables were compared using the Student's t test or Wilcoxon test for paired comparison data according to the data distribution. Statistical significance was set at two-sided p < 0.05.
Results: The sample was composed of 126 patients (age [mean] 39.1 ± [standard deviation] 13.8 years; 51% male). Significant changes were observed during the follow-up period: CDAI decreased from [median] 194 ([25-75 percentiles] 121-269) to 48.2 (10.1-122.0) (p < 0.05); the EuroQoL-5D increased from 0.735 (0.633-0.790) to 0.797 (0.726-1.000) (p < 0.05); the EuroQoL-5D visual analogue scale increased from 50.0 (40-70) to 80.0 (60-90); (p < 0.05) and the IBDQ increased from 56.7 (51.6-61.5) to 67.5 (60.1-73.6) (p < 0.05). The total work productivity impact decreased from 53% to 24% (p < 0.05).
Conclusions: In regular practice, adalimumab is clinically effective in the treatment of Crohn's disease patients and results in a significant improvement in quality of life and work productivity.
Key words: Adalimumab. Crohn's disease. IBD. Quality of life. Work productivity.
Introduction
Crohn's disease (CD) is a chronic inflammatory illness characterized by periods of remission and exacerbation which may involve any area of the gastrointestinal tract. Past studies have estimated CD incidence rate in Europe between 2.5 and 7.5 cases/100,000 inhabitants/year (1-3); however, the rate has been increasing in recent years, with the latest estimates being around 9 cases/100,000 inhabitants/year (4-7). CD is usually diagnosed during young adulthood, and the mortality rate, although higher than that of the overall population, is not very high. Thus, patients are affected mostly by CD-related morbidity and treatment for most of their lives, with the corresponding impact on their health-related quality of life (HRQoL) and work productivity.
There is clear evidence of the negative impact of CD on HRQoL. Cohen, in a recent meta-analysis of 22 articles, concluded that HRQoL is worse in CD patients than in healthy controls and patients with ulcerative colitis, and similar to that of other chronic diseases. HRQoL levels also worsen with higher disease activity (8). Studies of CD in Spain report evidence of a high impact of the disease on overall wellbeing, social function and vitality; a moderate impact on emotional function, psychological function, and physical pain; and a mild impact on physical function (9).
In addition to generic measurement tools such as the EuroQoL-5D or the SF-36, there are specific instruments to assess quality of life in CD patients such as the commonly used Inflammatory Bowel Disease Questionnaire (IBDQ), with a 36-item version (10) and a 9-item version (11).
There are also studies that show the negative impact of CD on work productivity and activity impairment (12). A version of the assessment tool Work Productivity and Activity Impairment questionnaire (WPAI) has been adapted specifically for CD (13-15) and validated in Spain (16,17).
With regard to treatment, new therapies have emerged, such as those referred to as biological treatments. These treatments include elements (usually monoclonal antibodies) designed to block certain molecules involved in the inflammatory response or the corresponding receptors in cell membranes. Biological compounds have revolutionized the therapeutic approach to CD, especially for those patients with severe or refractory disease. A recent meta-analysis of clinical trials concluded that biological treatments are effective for the induction of remission of active CD and the prevention of flare-ups (18). Clinical trials have also reported improvements in work productivity (19,20) and quality of life (21-23).
The objective of this study was to examine these reported associations in regular clinical practice, i.e., outside the controlled environment of a clinical trial. Thus, our aim was to measure the improvement in clinical health status, quality of life, and work productivity in CD patients who are naïve to biological therapy for one year after starting treatment with adalimumab in the context of regular practice.
Materials and methods
This is an observational cohort study with a prospective (one year follow-up) and retrospective design. This work is part of the larger study named Efficacy of Adalimumab in Crohn's Disease (EFICADEC is the acronym in Spanish).
Baseline data, collected during the first visit (V0), included socio-demographic information, CD history, clinical status and quality of life at V0, and information on work activity during the previous week and previous year. During the five following visits at months 1, 3, 6 ,9, and 12 from baseline (V1, V2, V3, V4, and V5, respectively), data collected included clinical data, quality of life, and work and non-work activity information for the week prior to the visit. We included CD patients who are naïve to any biological treatment for whom adalimumab treatment was prescribed as part of regular clinical practice. Exclusion criteria included incomplete clinical records for the 12 months prior to the start of treatment with adalimumab and any anti-TNF treatment prior to the start of adalimumab therapy.
We calculated that a sample size of 100 patients was required to detect differences in the CDAI mean of 100 points, assuming a standard deviation of 250 (α = 0.05; β = 0.2). To achieve this final sample size, it was necessary to recruit 132 patients into the study based on an attrition rate between 25% and 36% observed in the CHARM study (24).
The outcome variables under study included: clinical variables, quality of life and work productivity measures at the different clinical visits. The following instruments collected the relevant data: Crohn's Disease Activity Index (CDAI) (25) assessed clinical status (proportion of patients with CDAI < 150, in remission, and global score); Perianal Disease Activity Index (PDAI), or Irvine PDAI (26) (remission is defined as a baseline value > 0 and a value = 0 for items 1, 2, 3, and 5, at the end of the one year treatment). Other outcome variables of interest included: corticoid tapering rate and, at the end of the study period, changes in quality of life as measured by EuroQoL-5D (the descriptive system index ranging from 0 to 1 and the visual analog scale [VAS] from 0 to 100) (27), and by IBDQ-9 (normalized scale from 0 to 100) (11). Finally, impact on work productivity and activity impairment was measured with the WPAI adapted to CD (17), which evaluates four different aspects of CD impact on work productivity during the previous seven days: percentage of lost work hours (absenteeism), percentage of impact on hours worked (presenteeism), percentage of total work productivity impact (TWPI: absenteeism and presenteeism combined), and percentage of impact on activity outside work.
Missing values in outcome variables (CDAI, PDAI, IBDQ and EuroQoL-5D) were replaced via an imputation method. We imputed the predicted values based on the slope of the regression line drawn between the value of the previous visit and the value of the subsequent visit, adjusted for time. For those cases where there was no visit following that with the missing data, the missing value was replaced with the value of the previous visit.
In the descriptive analysis, qualitative or categorical variables were expressed in percentages. Quantitative variables were tested for normality; if the variable was normally distributed, we described it by its mean and standard deviation (mean ± SD). Hypothesis testing for these variables was performed with the Student's t test for paired data. For variables not normally distributed we reported the median and the interquartile range (median [P25-P75]). Hypotheses contrasts involving these variables were tested using the Wilcoxon tests for paired data. For bivariate analyses Pearson's Chi-squared statistic or Fisher's exact test when there were five or fewer observations in a category were used. Statistical significance was set at two-sided p < 0.05.
Results
A total of 132 patients were recruited from 33 centers located in 11 geographical regions. Of these 132 records, six were eliminated due to incomplete data. For the total of five visits for each of the remaining 126 patients, eight missing values had to be imputed in CDAI, ten values in IBDQ, and seven in EuroQoL-5D. Of the 33 patients with perianal disease, 25 PDAI missing values were imputed. Forty-three patients presented a CDAI < 150 at study entry.
Socio-demographic characteristics
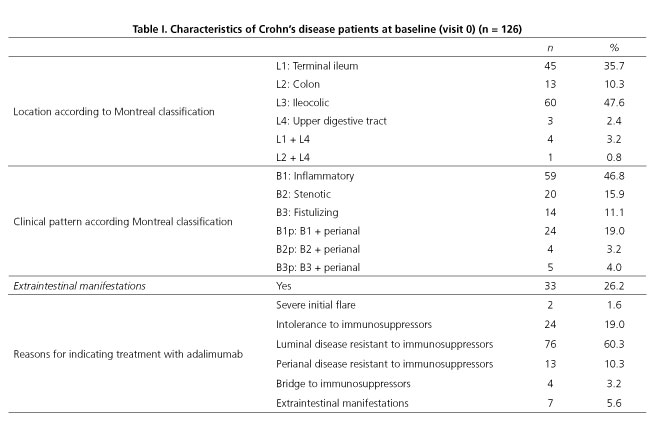
Women made up almost half the sample population (49.2%), the mean age was 39.1 ± 13.8 years, and patients had been diagnosed with CD over a median of seven years (RIC 3-12). At diagnosis, 25 patients (19.8%) presented with extraintestinal manifestations and another 25 had perianal involvement. At baseline (V0), 76 patients (60.3%) were in the labor force, of which 51.3% had been on sick leave in the previous year. Among those on sick leave in the previous year due to CD, the median number of days off work was 53 (13.5-120).
Clinical status at baseline
Table I shows the clinical status of CD patients at baseline (V0). Based on the Montreal classification by location of the disease, 35.7% of CD was in L1; 10.3%, in L2; 47.6%, in L3; 2.4%, in L4; 3.2%, in L1 + L4; and 0.8%, in L2 + L4. In terms of disease behavior, 46.8% of patients presented an inflammatory pattern, 15.9% presented a stenotic pattern, and 11.1% presented a fistulizing pattern. At baseline, there were 33 patients (26.2%) with perianal involvement, and 33 patients presented extraintestinal manifestations as well.
Adalimumab regimen
As a first induction dose, 85.7% of patients received 160 mg of adalimumab and another 13.5% received 80 mg. The second induction dose consisted of 80 mg for 86.5% of patients and 40 mg for 13.5%. Throughout the study, a maintenance dose of 40 mg was administered every 15 days in 92% of cases.
Response to treatment
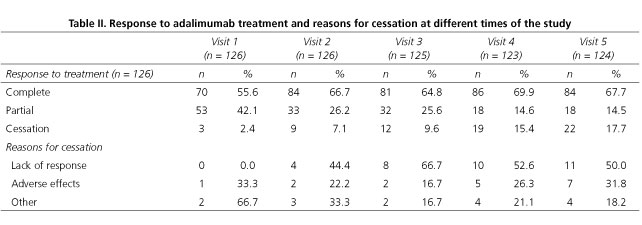
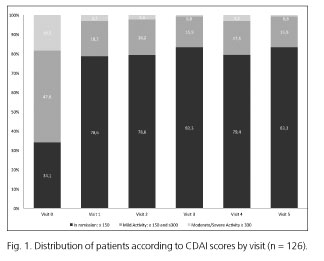
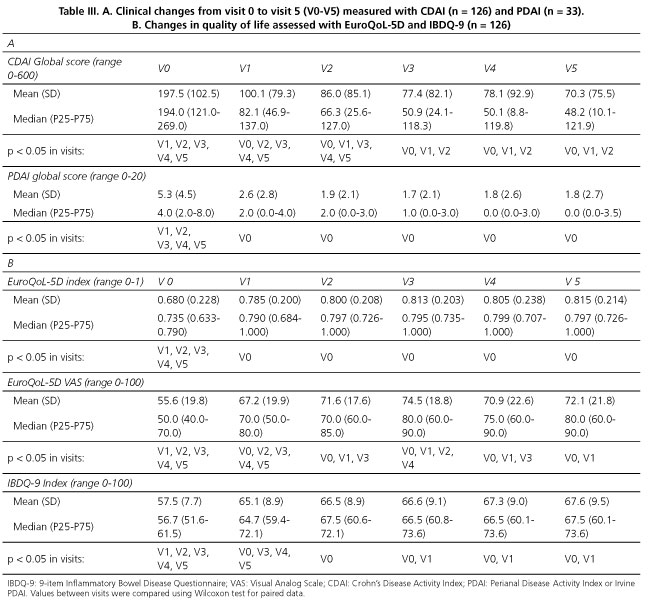
Table II shows response to adalimumab treatment data according to clinical criteria. At V1 (one month into treatment) complete response (CR) was observed in 55.6% of cases, partial response (PR) was seen in 42.1%, and treatment had to be discontinued in 2.4% of patients. By the end of the 12 months of treatment (V5), CR was 67.7%, PR was 14.5%, and treatment was discontinued for 17.7% of patients. Approximately one third of treatment cessations, both at V1 as well as at V5, were due to adverse effects. By the end of the follow-up, half of the treatment interruptions were caused by a lack of response to adalimumab. Corticoid tapering rate during follow-up was 87.4%, although in two cases where the patients had not taken corticoids in the previous year treatment with corticoids had to be reintroduced. CDAI changes throughout the study period are shown in figure 1 and table III. The proportion of patients in remission increased significantly from V0 to V1 (34.1% vs 78.6%) (p < 0.001), with a moderate increase until reaching 83.3% by the end of follow-up (Fig. 1). CDAI global score decreased significantly (p < 0.05) in each consecutive visit from V0 to V3, after which changes were no longer statistically significant. During the entire period, the median score decreased from 194 (121-269) to 48.2 (10.1-122) (Table III). At V1, 57.9% of patients had improved 70 or more points compared to their V0 score, and 45.2% had improved 100 or more points. At six months of treatment (V3) these percentages were 65.1% and 57.9%, respectively, and at 12 months of treatment (V5) these proportions held at 69.8% and 58.7%, respectively.
Regarding the 33 patients with perianal disease at study entry, the last follow-up PDAI was available for 29, of which ten (34.4%) experienced remission of the perianal disease. Table IIIA shows that the global score for the index decreased from 4 (2-8) to 0 (0-3.5) (p < 0.05). It is worth noting that each of the intermediate differences in scores, i.e., between baseline and each of the following visits, was also statistically significant.
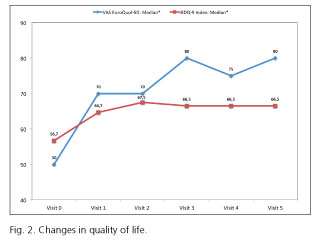
Quality of life and work and social impact
Table IIIB shows the results regarding quality of life. Here we observed that the median for EuroQoL-5D increased from a baseline value of 0.735 (0.633-0.790) to 0.797 (0.726-1.000) (p < 0.05) after 12 months on adalimumab. Again, the changes between V0 and each of the following visits were also statistically significant. The corresponding VAS increased from 50.0 (40-70) at V0 to 80.0 (60-90) at V5 (p < 0.05). The global score for the IBDQ increased significantly from 56.7 (51.6-61.5) at V0 to 64.7 (59.4-72.1) at V1, and continued to improve until V2 (67.5; 60.6-72.1), maintaining these gains by the end of the 12 months (67.5; 60.1-73.6) (p < 0.05). Figure 2 shows that the evolution of the EuroQoL-5D VAS was more gradual and substantive than that for IBDQ. As part of the WPAI test results we observed that CD-caused absenteeism decreased significantly from V1 until the end of follow-up, almost one third of patients reported a percentage > 50% at V0 but, by their last visit (V5), under one tenth of patients reported percentages > 50%. A similar evolution was detected for presenteeism and, consequently, for TWPI (absenteeism and presenteeism combined). TWPI diminished significantly from V1 until the end of the study; at V0 over half of all patients reported a TWPI of 50% but by V5 only one fifth of them were as high. Percentage of impact of CD on regular activity outside work exhibited a very similar pattern to that of the global work productivity pattern.
Discussion
Our results indicate that adalimumab, as administered in regular clinical practice in a scheduled regimen, is highly effective on CD from the first month of treatment, and this effect persists for at least an entire year.
Regarding response to treatment, our results show that a CR peak is reached at about three months into treatment (V2) and this level remains more or less constant for the rest of the follow-up. In case of maintained partial response, clinicians opted for treatment suspension. Treatment was stopped in 1 of every 5 to 6 patients. Over half of the time the decision to stop was due to a lack of response, and in one third of the cases treatment cessation was due to adverse effects.
All outcome variables (CDAI, PDAI, EuroQoL-5D, IBDQ, WPAI) experience a statistically significant improvement after the first month of treatment with adalimumab. Improvement tends to continue after this first spurt but at a slower pace (and not always with statistically significant changes) up to visit 2 or 3 (at three or six months, respectively). After this, gains are maintained, and became stable through to the end of the follow-up period. In a previous study with infliximab in patients with active CD, a similar pattern was found with significant improvement at four weeks in quality of life and the CDAI. This improvement remained at week 8 (28).
The percentage of patients in total remission according to CDAI increased by 49% at six months, mirroring data reported in the CHARM study (22). Specifically, this study reported a 40% increase in patients receiving a regimen of 40 mg of adalimumab every two weeks and a 47% increase among those on a weekly regimen. It is worth mentioning that the percentage of patients improving by ≥ 100 and ≥ 70 in the first six months from V0 (65.1% and 57.9%, respectively) was slightly greater than the numbers reported in CHARM (51.7% and 54.1% on bimonthly regimen, respectively, and 52.2% and 56.1% on a weekly regimen).
Because CD is usually diagnosed during young adulthood (30.1 ± 13.6 year-olds), this disease has a substantial impact on the patient work life by requiring a considerable amount of sick leave. Adalimumab reduced the number of lost work hours and work productivity due to CD considerably as early as one month after the start of treatment. Our results differ slightly from those presented in other studies on the effects of adalimumab on work productivity. One meta-analysis (29) concluded that after one year of treatment the weighted average of improvement for absenteeism was 8.7% (range: 7.2%-10.3%), 22.2% for presenteeism (range: 18.2%-26.1%), and 24.9% for TWPI (range: 20.0%-29.7%) versus our weighted averages of 21.7%, 16.8% and 29.1%, respectively. In the CHOICE (30) study, carried out in CD patients treated with adalimumab after failure of infliximab, 8% of patients improved with regard to absenteeism, as well as 15% and 18% in presenteeism and TWPI, respectively. These results in the multicenter study CARE (20) are 9.6%, 17.3% and 18.4% respectively. The most striking finding of our results, in relation to these previous studies, is the value of absenteeism at baseline (32%) versus 15% in CHOICE (30) and 23% in CARE (20). These variations may be explained away by the different work environments in the different countries where the studies were carried out, as well as by differences in patient profiles. In our study, absenteeism is greater but presenteeism is lower, which translates into a smaller difference in the total measure of work productivity, TWPI. Regarding the impact on activity outside work score in the CHOICE (30) study, an improvement of around 20% was obtained at four weeks after starting adalimumab treatment, remaining stable for the rest of the year. However, in our study, the period of improvement of this score is longer (until week 12), reaching a greater improvement (27%) and then remaining stable.
Our results regarding quality of life (EuroQoL index) are also slightly different from those reported by Yu et al. (31) based on patients with moderate-severe CD. In this study, Yu et al. compared the efficacy of adalimumab and infliximab and found that the quality of life after 12 months of treatment with adalimumab (measured with the EuroQoL-5D index) was somewhat higher than our results (0.865 and 0.815, respectively). However, they failed to report the improvement recorded during the first 12 months of treatment, which reached 0.135 in our sample. Other studies, using different instruments for measuring quality of life, obtained findings consistent with ours in terms of quality of life improvement (30,32).
Our results should be interpreted in the context of the study limitations. The main limitation of the EFICADEC study in general is the inherent difficulty of extracting retrospective data from patient clinical history of the year prior to baseline. However, all analyses for this work were based exclusively on prospective data collected via validated questionnaires filled out during the subsequent visits, thus we expect these data to be highly reliable. Another limitation could derive from imputing missing data on the global scores for CDAI, EuroQoL-5D, IBDQ, and PDAI. Despite this, the number of data imputed was very low (1.2% [8/630] in CDAI; 1.1% [7/630] in EuroQoL-5D; 1.6% [10/630] in IBDQ, and 6.0% [25/165] in PDAI). These low percentages combined with the method used to calculate the imputed value (regression imputation based on the previous and following observations to the missing data; in the absence of the following value, the missing value was replaced with that of the previous visit) minimize the possibility of biasing the results.
Based on our results, we draw the following conclusions:
1. Patients on adalimumab showed improvements in clinical measures (CDAI, PDAI) similar to those reported in clinical trials. These improvements are noticeable from the first month of treatment and progressive up to three or six months of treatment, when it reaches its peak and stabilizes until the end of the study period of one year.
2. After the one-year treatment, over 50% of patients stop taking corticoids.
3. The impact of adalimumab on work productivity is significant and substantial. How presenteeism and absenteeism evolve in our study is slightly different than those in previous studies, though the behavior of TWPAI is similar to that found in these studies.
4. HRQoL, measured with EuroQoL-5D and IBDQ, improves substantially from the onset of adalimumab treatment and gains remain stable throughout the follow-up period.
Conflict of interest and funding
Cristina Saro has served as a speaker for Abbvie and MSD and advisory board member for Abbvie and MSD. Fernando Muñoz has served as a speaker for Abbvie and MSD and advisory board member for Abbvie and MSD. Daniel Ceballos has served as a speaker for Abbvie, MSD, Otsuka, Fresenius-Kabi, Faes and Shire, consultant for Abbvie and has received research funding from Abbvie, MSD and Otsuka. Cristóbal de la Coba has served as a speaker for Abbvie. Pablo Lázaro is an employee of Advanced Techniques in Health Services Research and has not served as a speaker or consultant for any organization. María Dolores Aguilar is an employee of Advanced Techniques in Health Services Research and has not served as a speaker or consultant for any organization. Valle García-Sánchez has served as a speaker, consultant and advisory board member for Abbvie, MSD and Ferring, and has not received research funding from them. Luis I. Fernández has served as a speaker, consultant and advisory board member for Abbvie, MSD and Shire, and has received research funding from Abbvie and MSD. Jesús Barrio has served as a speaker, consultant and advisory board member for Abbvie, MSD and Shire, and has received research funding from Geteccu. Manuel Barreiro-de Acosta has served as a speaker, consultant and advisory board member for Abbie and MSD, and has not received research funding from any. For the remaining authors none were declared.
References
1. Shivananda S, Lennard-Jones J, Logan R, et al. Incidence of inflammatory bowel disease across Europe: Is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease (EC-IBD). Gut 1996;39:690-7. DOI: 10.1136/gut.39.5.690. [ Links ]
2. Saro C, Lacort M, Argüelles G, et al. Incidencia y prevalencia de la enfermedad inflamatoria intestinal en Gijón, Asturias, España. Gastroenterol Hepatol 2000;23:322-7. [ Links ]
3. Burisch J, Pedersen N, Čuković-Čavka S, et al. East-West gradient in the incidence of inflammatory bowel disease in Europe: The ECCO-EpiCom inception cohort. Gut 2013;0:1-10. DOI: 10.1136/gutjnl-2013-304636. [ Links ]
4. Nerich V, Monnet E, Etienne A, et al. Geographical variations of inflammatory bowel disease in France: A study based on national health insurance data. Inflamm Bowel Dis 2006;12:218-26. DOI: 10.1097/01.MIB.0000206540.38834.8c. [ Links ]
5. Sincic BM, Vucelic B, Persic M, et al. Incidence of inflammatory bowel disease in Primorsko-Goranska County, Croatia, 2000-2004: A prospective population-based study. Scand J Gastroenterol 2006;41:437-44. DOI: 10.1080/00365520500320094. [ Links ]
6. Vind I, Riis L, Jess T, et al. Increasing incidences of inflammatory bowel disease and decreasing surgery rates in Copenhagen City and County, 2003-2005: A population-based study from the Danish Crohn colitis database. Am J Gastroenterol 2006;101:1274-82. DOI: 10.1111/j.1572-0241.2006.00552.x. [ Links ]
7. Saro C, De la Coba C, Lacort M, et al. Changes in incidence of inflammatory bowel disease during the last 15 years: An epidemiological prospective population-based study from Gijón (Spain) (1992-2006). Gut 2007;56(Suppl 3):A129. [ Links ]
8. Cohen RD. The quality of life in patients with Crohn's disease. Aliment Pharmacol Ther 2002;16:1603-9. DOI: 10.1046/j.1365-2036.2002.01323.x. [ Links ]
9. Juan J, Estiarte R, Colomé E, et al. Burden of illness of Crohn's disease in Spain. Dig Liver Dis 2003;35:853-61. DOI: 10.1016/j.dld.2003.07.002. [ Links ]
10. López-Vivancos J, Casellas F, Badía X, et al. Validation of the Spanish version of the Inflammatory Bowel Disease Questionnaire in ulcerative colitis and Crohn's disease. Digest 1999;60:274-80. DOI: 10.1159/000007670. [ Links ]
11. Alcalá MJ, Casellas F, Fontanet G, et al. Shortened questionnaire on quality of life for inflammatory bowel disease. Inflamm Bowel Dis 2004;10:383-91. DOI: 10.1097/00054725-200407000-00009. [ Links ]
12. Wyke RJ, Edwards FC, Allan RN. Employment problems and prospects for patients with inflammatory bowel disease. Gut 1988;29:1229-35. DOI: 10.1136/gut.29.9.1229. [ Links ]
13. Reilly MC, Gerlier L, Brown M. Discriminant validity of the work productivity and activity impairment questionnaire in patients with Crohn's disease. Value Health 2006;9:A246-7. DOI: 10.1016/S1098-3015(10)63342-6. [ Links ]
14. Feagan B, Schreiber S, Selke B, et al. Moderate to severe Crohn's disease induces high productivity losses: Work Productivity and Activity Impairment (WPAI) baseline data from the PRECiSE program. Gastroenterol 2006;130:A483. [ Links ]
15. Feagan B, Brown M, Gerlier L, et al. The effects of certolizumab pegol on work productivity and daily activities of Crohn's disease patients: Data from PRECISE 2. Gut 2007;55(Suppl V):A139. DOI: 10.1016/S1873-9954(07)70023-X. [ Links ]
16. Vergara M, Montserrat A, Casellas F, et al. Validation of the Spanish Work Productivity and Activity Impairment Questionnaire: Crohn's disease version. Eur J Gastroenterol Hepatol 2009;21:809-15. DOI: 10.1097/MEG.0b013e32830f4c9e. [ Links ]
17. Vergara M, Montserrat A, Casellas F, et al. A new validation of the Spanish Work Productivity and Activity Impairment Questionnaire-Crohn's disease version. Value Health 2011;14:859-61. DOI: 10.1016/j.jval.2011.02.1179. [ Links ]
18. Ford AC, Sandborn WJ, Khan KJ, et al. Efficacy of biological therapies in inflammatory bowel disease: Systematic review and meta-analysis. Am J Gastroenterol 2011;106:644-59. DOI: 10.1038/ajg.2011.73. [ Links ]
19. Ford AC, Sandborn WJ, Khan KJ, et al. Clinical trial: The effects of certolizumab pegol therapy on work productivity in patients with moderate-to-severe Crohn's disease in the PRECiSE 2 study. Aliment Pharmacol Ther 2010;31:1276-85. DOI: 10.1111/j.1365-2036.2010.04303.x. [ Links ]
20. Louis E, Löfberg R, Reinisch W, et al. Adalimumab improves patient-reported outcomes and reduces indirect costs in patients with moderate to severe Crohn's disease: Results from the CARE trial. J Crohns Colitis 2013;7:34-43. DOI: 10.1016/j.crohns.2012.02.017. [ Links ]
21. Feagan BG, Yan S, Bala M, et al. The effects of infliximab maintenance therapy on health-related quality of life. AJG 2003;10(98):2232-8. DOI: 10.1111/j.1572-0241.2003.07674.x. [ Links ]
22. Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn's disease: The CHARM trial. Gastroenterol 2007;132:52-65. DOI: 10.1053/j.gastro.2006.11.041. [ Links ]
23. Koelewijn C, Schrijvers A, Oldenburg B. Infliximab use in patients with Crohn's disease: Quality of life, costs and resource use. Neth J Med 2006;7(64):212-8. [ Links ]
24. Feagan BG, Panaccione R, Sandborn WJ, et al. Effects of adalimumab therapy on incidence of hospitalization and surgery in Crohn's disease: Results from the CHARM study. Gastroenterol 2008;135:1493-9. DOI: 10.1053/j.gastro.2008.07.069. [ Links ]
25. Best WR, Becktel JM, Singleton JW, et al. Development of a Crohn's disease activity index. Gastroenterol 1976;70:439-44. [ Links ]
26. Irvine EJ. Usual therapy improves perianal Crohn's disease as measured by a new disease activity index. McMaster IBD Study Group. J Clin Gastroenterol 1995;20:27-32. [ Links ]
27. Badía X, Fernández E, Segura A. Influence of socio-demographic and health status variables on evaluation of health status in a Spanish population. Eur J Public Health 1995;5:87-93. DOI: 10.1093/eurpub/5.2.87. [ Links ]
28. Van Balkom BP, Schoon EJ, Stockbrugger RW, et al. Effects of anti-tumour necrosis factor-a therapy on the quality of life in Crohn's disease. Aliment Pharmacol Ther 2002;16:1101-7. DOI: 10.1046/j.1365-2036.2002.01268.x. [ Links ]
29. Binion DG, Louis E, Oldenburg B, et al. Effect of adalimumab on work productivity and indirect costs in moderate to severe Crohn's disease: A meta-analysis. Can J Gastroenterol 2011;25:492-6. DOI: 10.1155/2011/938194. [ Links ]
30. Lichtiger S, Binion DG, Wolf DC, et al. The CHOICE trial: Adalimumab demonstrates safety, fistula healing, improved quality of life and increased work productivity in patients with Crohn's disease who failed prior infliximab therapy. Aliment Pharmacol Ther 2010;32:1228-39. DOI: 10.1111/j.1365-2036.2010.04466.x. [ Links ]
31. Yu AP, Johnson S, Wang ST, et al. Cost utility of adalimumab versus infliximab maintenance therapies in the Unites States for moderately to severely active Crohn's disease. Pharmaecon 2009;27:609-21. DOI: 10.2165/11312710-000000000-00000. [ Links ]
32. Casellas F, Robles V, Borruel N, et al. Restoration of quality of life of patients with inflammatory bowel disease after one year with antiTNFalpha treatment. J Crohn's Colitis 2012;6:881-6. DOI: 10.1016/j.crohns.2012.01.019. [ Links ]
![]() Correspondence:
Correspondence:
Manuel Barreiro-de-Acosta.
Gastroenterology Service.
Hospital Clínico Universitario de Santiago.
Travesía de Choupana, s/n.
15706 Santiago de Compostela.
A Coruña, Spain
e-mail: manubarreiro@hotmail.com
Received: 08-09-2016
Accepted: 22-10-2016