My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Revista Española de Enfermedades Digestivas
Print version ISSN 1130-0108
Rev. esp. enferm. dig. vol.109 n.6 Madrid Jun. 2017
https://dx.doi.org/10.17235/reed.2017.4819/2016
CASE REPORT
Fecal microbiota transplantation in recurrent Clostridium difficile infection in a patient with concomitant inflammatory bowel disease
Marta Gravito-Soares1, Elisa Gravito-Soares1, Francisco Portela1,2, Manuela Ferreira1,2 and Carlos Sofia1,2
1Gastroenterology Department. Coimbra University Hospital. Coimbra, Portugal.
2Faculty of Medicine. University of Coimbra. Coimbra, Portugal
Author's contributions: Marta Gravito-Soares and Elisa Gravito-Soares designed the report, collected the patient data, performed the fecal microbiota transplantation and wrote the paper. Manuela Ferreira did the patient follow-up. Francisco Portela and Carlos Sofia analyzed the data and revised the manuscript.
ABSTRACT
The use of fecal microbiota transplantation in recurrent Clostridium difficile infection and coexistent inflammatory bowel disease remains unclear.
A 61-year-old man with ulcerative pancolitis was diagnosed with a third recurrence of Clostridium difficile infection, previously treated with metronidazole, vancomycin and fidaxomicin. Fecal microbiota transplantation of an unrelated healthy donor was performed by the lower route. After a twelve month follow-up, the patient remains asymptomatic without Clostridium difficile infection relapses or inflammatory bowel disease flare-ups.
Fecal microbiota transplantation is relatively simple to perform, well-tolerated, safe and effective in recurrent Clostridium difficile infection with ulcerative pancolitis, as an alternative in case of antibiotic therapy failure.
Key words: Recurrent Clostridium difficile infection. Ulcerative pancolitis. Fecal microbiota transplantation. Inflammatory bowel disease. Infectious colitis.
Introduction
Clostridium difficile infection (CDI) is the leading cause of nosocomial and antibiotic-associated diarrhea (1-3). Recurrent infections are common even after multiple effective antibiotic therapies (1). In Inflammatory bowel disease (IBD), the risk of developing CDI increases due to immunosuppressive therapy, recurrent hospitalizations, pancolitis, nutritional and immunologic status, intestinal dysbiosis and genetic susceptibility. In this condition, the prognosis is worse as it has higher rates of mortality, colectomy and recurrence (3,4). The role of Clostridium difficile in IBD pathogenesis as a cause or consequence is still unknown (2). The standard antibiotic therapy is frequently ineffective in recurrent CDI in IBD patients (5). Fecal microbiota transplantation (FMT) can be an alternative treatment, likely due to the restoration of a disrupted microbiome, thereby breaking the cycle of recurrent CDI (1,3,5). Recent studies showed a high efficacy and safety in recurrent or refractory CDI (5). Nevertheless, very few data about this procedure in recurrent CDI and concomitant IBD patients have been reported (6).
We report a case of a patient with ulcerative pancolitis and three Clostridium difficile recurrences treated with FMT. The response was excellent, resulting in no relapse either of CDI or IBD during 18 months of follow-up.
Case report
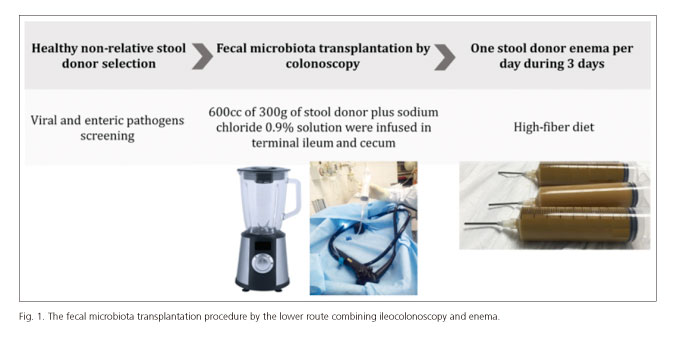
We report the case of a 61-year-old Caucasian male with ulcerative proctitis under topic 5-aminosalicylic acid therapy and a 40 pack-year smoking history and no family history of IBD. The patient had two previous ulcerative colitis flares successfully treated with systemic corticotherapy with oral prednisone at 40 mg/d reduced to 5 mg/week until stopping. During the first flare-up, one year after the diagnosis, the proximal extension of disease was observed with ulcerative pancolitis, and the patient was treated with one daily dose of 3 g oral mesalamine and 1 g enema mesalamine. The second flare-up occurred one year after the first one, and after another cycle of corticoids therapy the patient maintained oral and rectal mesalamine therapy with the same dosage. Two months after the second flare-up, he had three CDI episodes one month apart. He was treated with a ten-day course of oral metronidazole, vancomycin and fidaxomicin, respectively. He had also taken Saccharomyces boulardii during all CDI episodes, with clinical improvement after each one. During the last episode, pseudomembranous colitis was found in the left colonoscopy. All the CDI episodes were diagnosed by clinical and microbiology data and were positive for toxins by the polymerase chain reaction and culture for Clostridium difficile in stools, with compatible histology. He presented at our Emergency Room complaining of a four-day history of abdominal pain and acute diarrhea with six stools per day that did not contained blood, mucus or pus. The physical examination revealed abdominal pain at the deep palpation in the lower abdominal quadrants. Laboratory analysis showed higher inflammatory parameters (10.8*103 of white blood cells and 11.0 of C-reactive protein [< 0.5 mg/dl]). A left colonoscopy was performed, revealing raised yellow and off-white plaques up to 2 cm in diameter over the final 20 cm of the colorectal mucosa. Biopsies confirmed pseudomembranous colitis. The stool test for toxins and culture were positive for C. difficile. A fourth episode or third recurrence of CDI was diagnosed. At that moment, we proposed that the patient undergo FMT in our center. At the patient's request, his wife was the stool donor. His wife had no health problems such as a metabolic syndrome, obesity, food allergy, inflammatory bowel disease (IBD), irritable bowel syndrome, chronic constipation, prior major gastrointestinal procedures or oncologic conditions, and no antibiotic use or infections in the previous three months was reported. The screening was negative for human immunodeficiency virus 1 and 2, syphilis, viral hepatitis A, B and C, Clostridium difficile parasites including Giardia lamblia, as well as the routine stools culture for enteric pathogens, including Shigella, Campylobacter and Salmonella. Initially, a non-selective bowel decontamination therapy was performed to eradicate the Clostridium difficile species. For that purpose, the patient received a 14-day course of oral and rectal vancomycin of 125 mg four times daily, combined with intravenous metronidazole at 500 mg three times daily, followed by a slow tapered vancomycin regimen as follows: 125 mg orally twice daily for seven days, 125 mg orally once daily for seven days and 125 mg orally every two days for seven days. Subsequently, FMT was performed through the lower route combining ileocolonoscopy and enema. A total of 600 cc of 300 g donor stool diluted in sodium chloride 0.9% solution was infused into the terminal ileum and cecum (Fig. 1). At the time of the procedure, the ileum mucosa appeared normal and the transverse/left colon mucosa had endoscopic inactive colitis (Mayo endoscopic score 0) with multiple pseudopolyps. The procedure was well-tolerated. Daily enema administration of 100 ml of fresh donor stool preparation was undertaken over three days post-FMT to complete the treatment. A high-fiber diet was also applied in an attempt to stimulate the proliferation of infused healthy donor intestinal microbiome. The patient improved with no early or late FMT-related complications, maintaining oral mesalamine of 3 g once daily. The outpatient follow-up of 18 months was uneventful, free of symptoms, with one or two stools per day, and with lab and stool analysis every four months revealing no inflammatory parameters or anemia and a sedimentation rate < 20 mm/hr and fecal calprotectin < 50 mg/kg. An ileocolonoscopy with biopsies at 6-months of follow-up was performed, showing scars and pseudopolyps on the transverse and left colon with no inflammatory activity or dysplasia (Mayo endoscopic score 0).
Discussion
The FMT procedure involves an infusion of a liquid suspension of a healthy donor stool into a recipient bowel in order to cure a specific pathology related to gastrointestinal dysbiosis (3,8). It can be considered as a form of organ transplantation of more than 1,000 microbial species, but it is simpler to perform than other organ transplants, as no immunologic donor/recipient matching or post-procedure immunosuppression is required (1-3).
Since the first report of FMT use in recurrent CDI in 1958, a review of case series and a single-randomized control trial have shown its effectiveness compared to standard antibiotic treatment, with a cure rate of nearly 90% (1,3,4). Increasing research with regards to the microbiome in CDI revealed that FMT can resolve the dysbiosis by restoring phylogenetic diversity of gut microbiota and breaking the cycle of recurrent CDI (1,5,8).
To date, no standardized protocol for microbiota transplantation exists (1,5). Several routes of FMT instillation were suggested, although the best strategy remains unclear (3,5), and from our perspective it should be determined on a case by case basis. The lower proximal gastrointestinal FMT seems to be more advantageous regarding CDI treatment, because it is able to reach the terminal ileum or cecum (compared to only the left colon by enema), and it has the possibility to determine the presence and extension of pathologic mucosa and exclude coexistent conditions (3). Additionally, this administration route permits a larger volume suspension infusion, is more anatomically practical and seems to have a higher success rate (5). The bowel lavage before FMT may contribute to a significant reduction of pathogenic microorganisms allowing the colonization of donor fecal microbiota (3,5). In this case, we chose the lower route, after bowel lavage, combining ileocolonoscopy to the proximal colon and enema to the distal colon, since it was a pancolitis and CDI with pseudomembranous colitis in the left colon and rectum.
Other concerns regarding the FMT procedure are under investigation, such as the volume of infusion to administer, the type and duration of antibiotic therapy before transplantation, and the minimum number of infusions necessary to achieve a cure. The ideal characteristics of donors and effects of FMT on the resident flora need further research (1,5). Nonetheless, recent literature did not show significant differences between related and unrelated donors (1,4,6). However, it has been shown that the use of more than 50g of stool donor, administration of more than 500mL of infusion and performing FMT after effective antibiotic therapy are associated with higher rate of infection resolution (1,6). We used an unrelated donor, reducing the infection transmission probability, and 300 g of stool donor, totaling a volume of 600 ml of infusion FMT. The FMT was performed after antibiotic therapy.
Another area of FMT use is the treatment of CDI coinfection in immunocompromised patients (5). This group of patients, including IBD patients, has an increased risk of CDI and associated complications, and this condition should be ruled out in all IBD hospitalized patients with a disease flare-up (2,4,5,8). Regarding colonic IBD, the higher incidence of CDI was found in patients with left sided and extensive disease compared to distal disease (2). For patients with CDI and IBD, the classic antibiotic therapies are recognized as ineffective and there are no data for fidaxomicin. FMT has emerged as a potential therapy (2,7), but effectiveness, safety and potential adverse related-FMT events are not well understood (5,9). The very few case series about FMT use for superimposed CDI in IBD have shown the efficacy of FMT in CDI resolution and relapse prevention, with few adverse effects and without infection complications (6). Although some reports postulate that the coexistence with IBD may require repeated FMT to ensure successful CDI eradication (9), FMT seems to be an untargeted approach to modify the underlying dysbiosis present in IBD (1,7).
In relation to IBD treatment, the first cases of FMT transient success date back to 1989 (5). Recent randomized controlled trials and a meta-analysis demonstrated a variable efficacy of FMT in IBD, especially for ulcerative colitis (7,9,10). The risk of IBD reactivation is still another worry, since a case of transient flare-up of ulcerative colitis after FMT has been reported (11).
In our case, we performed a FMT to treat the third CDI recurrence and prevent new relapses in an IBD patient through a lower route combining colonoscopy and enema. We considered a prior long-period of antibiotic therapy with metronidazole and vancomycin together, and then a tapered vancomycin regime in order to obtain an effective Clostridium difficile eradication. Although there are controversial results in the literature, to date, after 18 months of follow-up, our patient is well with no CDI relapses or IBD flares and no adverse FMT-related events.
Fecal microbiota transplantation is relatively simple to perform, well-tolerated, safe and effective in recurrent CDI with ulcerative pancolitis, as an alternative in cases of prior antibiotic treatment failure. Our results suggest that a good donor selection, an initial extended combined antibiotic therapy, and a combined lower route to reach the proximal and distal colon could be effective and safe to treat and mainly to prevent recurrent CDI in IBD patients with sustained improvements for both diseases. More case reports are necessary to conclude if FMT is effective for patients with ulcerative pancolitis and recurrent CDI.
References
1. Terrier M, Simonet M, Bichard P, et al. Recurrent Clostridium difficile infections: The importance of the intestinal microbiota. World J Gastroenterol 2014;20:7416-23. DOI: 10.3748/wjg.v20.i23.7416. [ Links ]
2. Niltzan O, Elisa M, Chazan B, et al. Clostridium difficile and inflammatory bowel disease: Role in pathogenesis and implications in treatment. World J Gastroenterol 2013;19:7577-85. DOI: 10.3748/wjg.v19.i43.7577. [ Links ]
3. Ponte A, Pinho R, Mota M, et al. Initial experience with fecal microbiota transplantation in Clostridium difficile infection - Transplant protocol and preliminar results. Rev Esp Enferm Dig 2015;107:402-7. DOI: 10.17235/reed.2015.3767/2015. [ Links ]
4. Trifan A, Stanciu C, Girieanu I, et al. Impact of Clostridium difficile infection on inflammatory bowel disease outcome: A review. World J Gastroenterol 2014;20:11736-42. DOI: 10.3748/wjg.v20.i33.11736. [ Links ]
5. Kelly C, Kahn S, Kashyap P, et al. Update on fecal microbiota transplantation 2015. Indications, methodologies, mechanisms, and outlook. Gastroenterol 2015;149:223-37. DOI: 10.1053/j.gastro.2015.05.008. [ Links ]
6. Kelly C, Ihunnah C, Fischer M, et al. Fecal microbiota transplant for treatment of Clostridium difficile infection in immunocompromised patients. Am J Gastroenterol 2014;109:1065-71. DOI: 10.1038/ajg.2014.133. [ Links ]
7. Xu Q, Cao H-L, Wang W-Q, et al. Fecal microbiota transplantation broadening its application beyond intestinal disorders. World J Gastroenterol 2015;21:102-11. DOI: 10.3748/wjg.v21.i1.102. [ Links ]
8. Surawicz C, Brandt L, Binion D, et al. Guidelines to diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol 2013;108:478-98. DOI: 10.1038/ajg.2013.4. [ Links ]
9. Colman R, Rubin D. Fecal microbiota transplantation as therapy for inflammatory bowel disease: A systematic review and meta-analysis. J Crohns Colitis 2014;8:1569-81. DOI: 10.1016/j.crohns.2014.08.006. [ Links ]
10. Moayyedi P, Surette M, Libertucci J, et al. Fecal microbiota transplantation induces remission in patients with active ulcerative colitis in a randomized controlled trial. Gastroenterol 2015;149:102-9. DOI: 10.1053/j.gastro.2015.04.001. [ Links ]
11. De Leon L, Watson J, Kelly C. Transient flare of ulcerative colitis after fecal microbiota transplantation for recurrent Clostridium difficile infection. Clin Gastroenterol Hepatol 2013;11:1036-8. DOI: 10.1016/j.cgh.2013.04.045. [ Links ]
![]() Correspondence:
Correspondence:
Marta Gravito-Soares.
Gastroenterology Department.
Centro Hospitalar e Universitário de Coimbra.
Praceta Professor Mota Pinto.
3000-075 Coimbra, Portugal
e-mail: ms18498@gmail.com
Received: 03-01-2017
Accepted: 27-01-2017