Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista Española de Enfermedades Digestivas
versión impresa ISSN 1130-0108
Rev. esp. enferm. dig. vol.109 no.8 Madrid ago. 2017
https://dx.doi.org/10.17235/reed.2017.4708/2016
Epidemiology of inflammatory bowel disease in Málaga: incidence rate and follow-up of a cohort diagnosed between 2007-2008
Epidemiología de la enfermedad inflamatoria intestinal en Málaga: incidencia y seguimiento a la cohorte diagnosticada en 2007-2008
Ignacio Cueto-Torreblanca, Raquel Camargo-Camero, Raúl Andrade-Bellido, Eduardo Romero-Pérez and Guillermo Alcaín-Martínez
Department of Gastroenterology. Hospital Clínico Universitario Virgen de la Victoria. Málaga, Spain
ABSTRACT
Background and objectives: The incidence of inflammatory bowel disease (IBD) in Spain has been traditionally lower than in Northern European countries. Recent epidemiological studies have found that these differences are diminishing. This study estimates the incidence of IBD in Málaga (Spain), a city in Southern Spain and relates its results to those found in our neighboring countries.
Material and methods: This was a prospective study designed to collect new cases diagnosed during the period from 2007-2008 and follow up these patients. Incidence is expressed as number of patients per 100,000 population per year. The population distribution found in the European Collaborative Study was used to standardize incidence rates.
Results: The gross incidence rate of IBD in Málaga is 9/105, the standardized incidence rate is 12.3/105 (9.7-15.6).
Conclusions: These data are similar to those found in our surroundings, although a higher incidence rate for Crohn's disease (CD) as compared to ulcerative colitis (UC) was found. The clinical characteristics and outcomes of our patients do not differ significantly from those described for other populations.
Key words: Inflammatory bowel disease. Epidemiology.
RESUMEN
Introducción y objetivos: la incidencia de enfermedad inflamatoria intestinal (EII) en España ha sido clásicamente inferior a la incidencia de los países del norte de Europa. Estudios epidemiológicos recientes muestran que la brecha está disminuyendo. Este estudio estima la incidencia de EII en Málaga, una ciudad en el sur de España, y relaciona dichas cifras con los datos de nuestro entorno.
Material y métodos: este es un estudio prospectivo que fue diseñado para recoger los nuevos casos diagnosticados durante los años 2007-2008, y para realizar el seguimiento de estos pacientes. La incidencia se expresa como el número de pacientes por cada 100.000 habitantes por año. La distribución de la población del estudio colaborativo europeo se utilizó para normalizar la tasa de incidencia.
Resultados: la incidencia bruta de la EII en Málaga es 9/105, la incidencia estandarizada es de 12,3/105 (9,7-15,6).
Conclusiones: estos datos son similares a los de nuestro medio ambiente, aunque se observa una mayor incidencia para la enfermedad de Crohn (EC) que para la colitis ulcerosa (CU). Las características clínicas y evolución de nuestro paciente no presentan grandes diferencias con respecto a lo descrito en otras poblaciones.
Palabras clave: Enfermedad inflamatoria intestinal. Epidemiología.
Introduction
The incidence of IBD is increasing in Spain according to studies reported in the past few years (1). Incidence rates have changed since early studies were published and the North-South gradient concept described by the European Collaborative Study (2) no longer applies. A European prospective, multicenter study published in 2014 helped dismiss this notion, which was subsequently replaced by an East-West gradient (3). Spain was classified within the higher-rate band, with a similar incidence to that of traditional high-incidence regions.
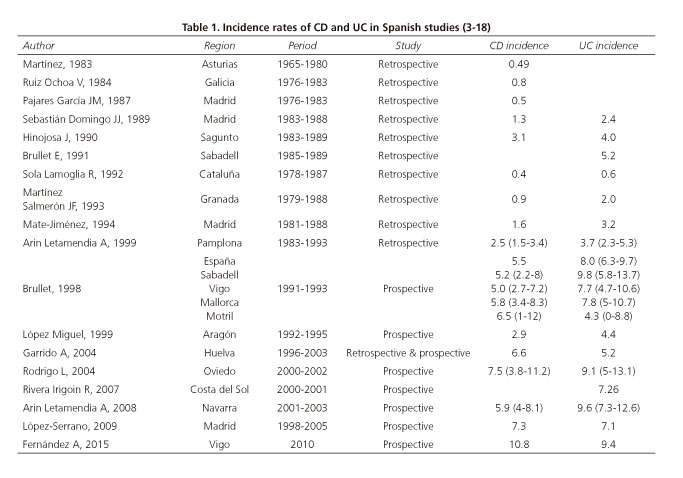
In Spain, IBD incidence data from epidemiological studies have gradually approached those obtained in Northern Europe (4-19,24) (Table 1). The reasons for such convergence include improved design in Spanish studies, improved diagnosis in clinical daily practice and an actual increase in patient numbers. No prior epidemiological studies were available in Málaga although, studies performed in other cities of Southern Spain do exist.
Early epidemiological studies found that IBD was more predominant in males than females within Spain (5). The slight predominance of UC amongst males was consistent with the findings obtained in other European countries although this predominance was not seen for CD. More recent studies have ironed out this difference to a certain extent (18). No differences have been reported in terms of age distribution between Spanish studies as compared to other European countries. Except for the latest research which shows that the incidence does not follow a classical bimodal distribution but plateaus over the second, third and fourth decades of life (19).
Follow-up data have been reported for several European and American cohorts. However, few Spanish studies include a long-term follow-up of a patient cohort with IBD.
The goal of this study was to determine the gross and standardized incidence rate of IBD in a population from Southern Spain, as well as the phenotypes and therapy needs by long-term follow-up of this cohort's.
Materials and methods
We designed a prospective study to determine the incidence of IBD in our setting. Newly diagnosed patients with IBD from January 1st 2007 to December 31st 2008 in the healthcare area served by the Hospital Clínico Universitario Virgen de la Victoria (Málaga, Spain) were included in the study. This area had 424,101 patients at the onset of the study and 457,258 at the end of the study. The percentage of women in the cohort was 50.9%. Health care in our area is predominantly public but private centers were also contacted in order to maintain undetected cases to a minimum. The Hospital Clínico Universitario Virgen de la Victoria does not serve the pediatric population which represents 15.9% of the total.
A network was set up to identify newly diagnosed patients at the various health care levels (gastroenterology, surgery, radiology and pathology units). All patients included in the study met the Lennard-Jones criteria (20). Clinical phenotypes were categorized according to the Montreal classification. Additionally, data were recorded on disease severity, therapy with immunomodulators or anti-TNFs and both surgical and non-surgical complications. The mean follow-up was 70.6 months from December 2013. A total of 62 (78.48%) patients completed the study.
Information was recorded using the Microsoft Excel database included in Microsoft Office Professional 2010 package. Statistical analysis was performed using the SPSS and Epidat 4.0 packages. Incidence is expressed as the number of patients per 100,000 population per year. The population distribution in the European Collaborative Study was used for incidence rate standardization. Pearson's chi-squared or Fisher's exact test were used for epidemiological data, Montreal classification characteristics, and clinical outcome.
Results
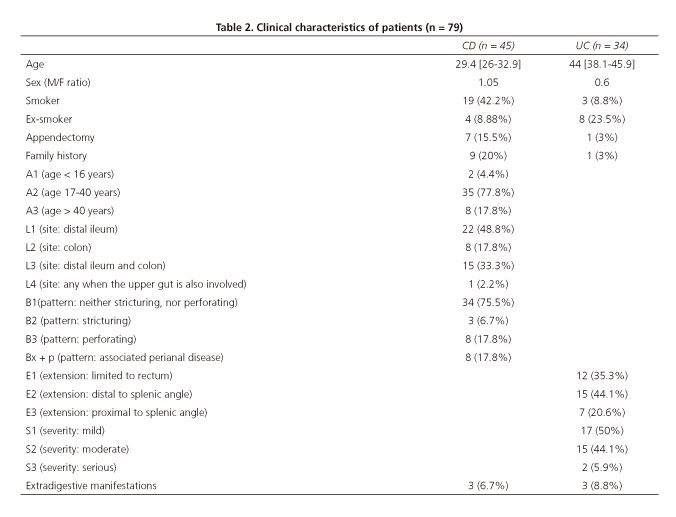
During the period from 2007-2008 a total of 79 patients were diagnosed with IBD, 34 patients with UC and 45 with CD. No patients diagnosed with indeterminate colitis were identified (Table 2).
The M/F ratio for IBD was 0.8 (44 male and 35 female). The mean age of patients diagnosed with IBD was 35.7 years of age (32.1-39.2), 44 (38-49.9) years of age for UC and 29.4 (26-32.9) years of age for CD.
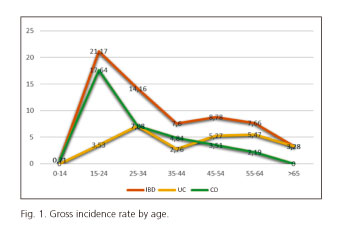
The annual gross incidence rate of IBD in our cohort was 9/105, 10.2/105 for males and 7.8/105 for females. Gross incidence for patients with UC was 3.9/10 5, 5/105 for males and 2.7/105 for females. Gross incidence for patients with CD was 5.1/105, 5/105 for males and 5.1/105 for females (Table 3, Fig. 1).
Incidence excluding patients younger than 14 years of age was 10.5/105, 12.1/105 for males and 9/105 for females. This incidence rate was 4.6/105 for patients with UC and 5.9/105 for patients with CD.
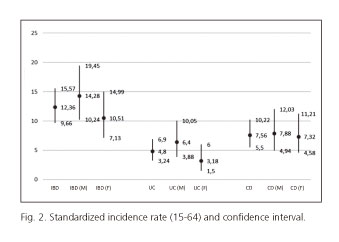
The standardized incidence rate for IBD within the age range of 15-64 years was 12.3/105 (9.7-15.6), 14.3/105 (10.2-19.5) for males and 10.5/105 (7.1-15) for females. The standardized incidence rate within the age range of 15-64 years was 4.8/105 (3.2-6.9) for UC and 7.6/105 (5.5-10.2) for CD (Fig. 2).
The most common presentation for UC patients was distal involvement beyond the splenic flexure (E2, 44.1%) and mild severity (S1, 50%). With regard to the CD phenotype, most were in the A2 category (77.8%) according to age at diagnosis, in the L1 category (48.9%) for location and in the B1 category (75.6%) for behavior. No relevant changes were seen with regard to phenotype shift during the course of disease, only 2.94% of B1 cases evolved to B2 and 5.88% of these evolved further to B3.
The mean time from symptom onset was 4.73 months for patients with UC and 5.03 months for CD. The median time from symptom onset was 3 months for UC and 2 months for CD. No significant differences were identified with regard to phenotype.
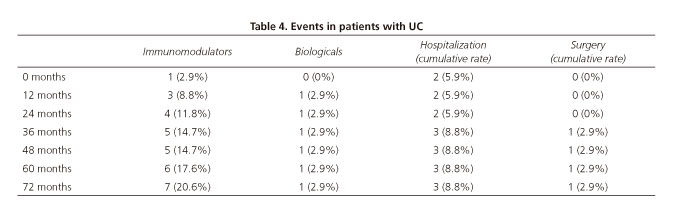
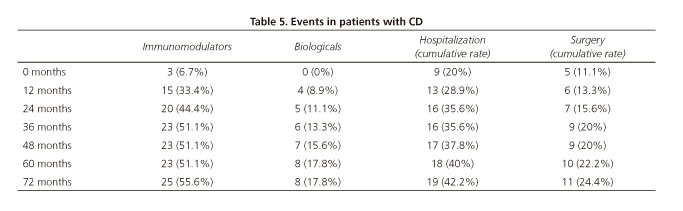
During the first year, only 3 (8.8%) patients with UC and 15 (33.3%) patients with CD required immunomodulators. At the end of the follow-up period, 7 (20.6%) patients with UC and 25 (55.6%) patients with CD had received immunomodulators. With regards to biologic therapy, only 1 (2.9%) patient with UC required this form of treatment, whereas 8 (17.8%) patients with CD needed it throughout follow-up. Immunomodulators or biologics were initiated during the first year for 50% of the patients who required them (Tables 4 and 5).
Only one patient (2.9%) with UC required surgery during follow-up. During the first year, 5 (11.1%) patients with CD needed surgery. At the end of follow-up, 11 (24.4%) patients had been operated on. Three (8.8%) patients with UC and 19 (42.2%) with CD needed hospitalization during the follow-up. Figures for the initial year included 2 (5.9%) patients with UC and 13 (28.9%) with CD (Tables 4 and 5).
The statistical analysis showed that patients with CD had poorer outcomes than patients with UC, which was reflected by an increased immunomodulator usage (p = 0.036), higher rate of hospital admissions (p = 0.015) and a greater number of surgeries (p = 0.021). Having a B2 or B3 behavior at diagnosis was associated with an increased need for immunomodulators (p = 0.009), hospital admission (p = 0.049) and surgical procedures (p = 0.018) during the first three years. This association is not sustained over time as none of these factors were associated with behavior at the end of the follow-up period. Furthermore, a statistically significant association was observed between younger age at diagnosis with CD and the use of biologics (p = 0.022). No statistically significant associations were identified for patients with UC, likely due to the low number of events collected.
Discussion
The present study is the first to provide data from an IBD cohort in our setting that includes gross and standardized incidence rates in order to relate IBD incidence in our population with that in Spain and Europe. When the study was developed, every effort was made to minimize missing newly-diagnosed patients by expanding the recruitment to multiple sites, both public and private healthcare centers. The gross incidence informs about population specific figures. Epidemiological differences preclude comparisons of gross incidence rates between different populations. Standardized incidence allows comparisons of incidence rates between different populations by weighting gross incidence rates with respect to a reference population.
The European Collaborative Study (2) showed a North-South gradient for IBD and placed Spain within the lower range of incidence, 8.7/105 for UC and 3.9/105 for CD. A similarly designed multicenter epidemiological study in Spain confirmed these data (11). Over time, other studies have been reported in Oviedo (16), Madrid (17) and Navarre (18), which showed a higher IBD incidence in Spain that approaches that of northern European countries. The data most recently reported data are from the Vigo cohort (19) and provide the highest incidence rates ever found in Spain.
Overall incidence in our cohort (Table 3) is higher than the incidence found by earlier Spanish studies in a hospital setting. In Madrid in 1989, the estimated incidence rates were 2.4/105 for UC and 1.3/105 for CD (6). Our incidence rate is similar to that obtained in recent studies. For example, in Oviedo in 2009, the estimated incidence rates were 7.1/105 for UC and 7.3/105 for CD (16). In our cohort that excluded the pediatric population, the incidence was slightly lower than that described for the Vigo cohort, 10.8/105 for CD and 9.4/105 for UC (19).
The Spanish multicenter study (13) was the first study that provided standardized incidence rates that were higher for UC (8/105) than for CD (5.5/105). However, both our gross and standardized estimates recorded a higher incidence for CD as compared to UC (Table 3). This higher incidence for CD versus UC, both gross and standardized, has been reported in other southern Spanish cohorts (13) (15) (Table 1).
Some European studies provide similar estimates. During 2004-06, the incidence rates recorded in Oberpfalz (Germany) were 3.9/105 for UC and 6.6/105 for CD (21), during 2000-04, the incidence rates estimated for the Kvarner Bay region (Croatia) were 4.3/105 for UC and 7/105 for CD (22).
The M/F ratio of our IBD cohort was 0.8. UC slightly predominates in males with a ratio of 0.55, whereas this ratio is 1.05 for CD. A review of incidence rates worldwide (23) excluded the notion that sex plays a role in the incidence of IBD. Our estimates fall within the range described in recent reports. In our cohort, the mean age at diagnosis is delayed for over 5 years for UC as compared to CD. CD incidence peaks at 15-24 years of age (17.6/105), UC incidence peaks at 25-34 years of age (7.1/105). These values are consistent with those in the reported series.
Clinical phenotypes and patient outcomes in our cohort are similar to those described in other European countries (25- 27). However, both the Epicom study on a European scale and the recently reported Vigo cohort (19) showed slightly lower percentages of 66-68% for the non-stricturing, non-fistulizing pattern. The proportion of patients in this study with L4 (2.2%) is lower than that recorded by other studies (10-15%), which is accounted for by the fact that upper gut tests are not routinely carried out in our newly diagnosed patients.
The use of immunomodulators and biologics peaked during the first year in our cohort, this is likely due to the attempts to rapidly control inflammation. Our first-year estimates are similar to those of the western European ECCO-Epicom Inception cohort (28) with regard to CD. Our cohort significantly differs with regards to patients with UC, with a lower usage of immunomodulators and biologics and lower hospitalization and surgical treatment rates.
In summary, our cohort exhibits clinical and epidemiological characteristics similar to those reported by other studies. The clinical course is consistent with that of other cohorts with poorer outcomes for CD. With regard to IBD incidence, a less apparent North-South gradient was confirmed. Finally, we highlight the greater incidence rate of CD versus UC in our population, a fact also reported by other recent studies which may suggest a trend.
References
1. Pajares JM, Gisbert JP. Epidemiología de la enfermedad inflamatoria intestinal en España. Una revisión sistemática. Rev Esp Enferm Dig 2001;93:9-14. [ Links ]
2. Shivananda S, Lennard-Jones J, Logan R, et al. Incidence of inflammatory bowel disease across Europe: is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease (EC-IBD). Gut 1996;39:690-7. [ Links ]
3. Burisch J, Pedersen N, Cukovic-Cavka S, et al; EpiCom-group. East-West gradient in the incidence of inflammatory bowel disease in Europe: the ECCO-EpiCom inception cohort. Gut 2014;63:588-97. DOI: 10.1136/gutjnl-2013-304636. [ Links ]
4. Martinez G, Fernandez Y, Rodrigo Saez L, et al. Estudio epidemiológico de la Enfermedad de Crohn en la región de Asturias. Rev Esp Enferm Apar Dig 1983;63:534-41. [ Links ]
5. Ruiz Ochoa V. Estudio epidemiológico de la enfermedad de Crohn en Galicia en el período de 1976 a 1983. Rev Esp Enferm Apar Dig 1984;66:273-9. [ Links ]
6. Sebastian Domingo JJ, Banares Canizares R, Velo Bellver JL, et al. Los aspectos epidemiológicos de la Enfermedad inflamatoria intestinal crónica en un área de la Comunidad Autónoma de Madrid. An Med Interna 1989;6:519-22. [ Links ]
7. Hinojosa J, Primo J, Lledo S, et al. Incidencia de enfermedad inflamatoria intestinal en Sagunto. Rev Esp Enferm Dig 1990;78:283-7. [ Links ]
8. Brullet E, Rue M, Montserrat A, et al. Un estudio epidemiológico descriptivo de Colitis ulcerosa en una población hospitalaria (1985-1989). Med Clin 1991;97:45-9. [ Links ]
9. Sola Lamoglia R, Garcia-Puges AM, Mones Xiol J, et al. Enfermedad inflamatoria intestinal en Cataluña (Barcelona y Gerona). Rev Esp Enferm Dig 1992;81:7-14. [ Links ]
10. Martinez-Salmeron JF, Rodrigo M, de Teresa J, et al. Epidemiología de Enfermedad inflamatoria intestinal en la provincial de Granada, España: un estudio prospectivo de 1979 a 1988. Gut 1993;34:1207-9. DOI: 10.1136/gut.34.9.1207. [ Links ]
11. Mate-Jimenez J, Munoz S, Vicent D, et al. Incidence and prevalence of ulcerative colitis and Crohn's disease in urban and rural areas of Spain from 1981 to 1988. J Clin Gastroenterol 1994;18:27-31. DOI: 10.1097/00004836-199401000-00008. [ Links ]
12. Arin Letamendia A, Burusco Paternain MJ, Borda Celaya F, et al. Epidemiological aspects of inflammatory bowel disease in the Pamplona area. Rev Esp Enferm Dig 1999;91:769-76. [ Links ]
13. Brullet E, Bonfill X, Urrútia G, et al. Estudio epidemiológico sobre la incidencia de enfermedad inflamatoria intestinal en cuatro áreas españolas. Med Clin 1998;110:651-6. [ Links ]
14. Lopez Miguel C, Sicilia B, Sierra E, et al. Incidence of inflammatory bowel disease in Aragon: outcome of a prospective population-based study. Gastroenterol Hepatol 1999;22:323-8. [ Links ]
15. Garrido A, Martinez MJ, Ortega JA, et al. Epidemiology of chronic inflammatory bowel disease in the Northern area of Huelva. Rev Esp Enferm Dig 2004;96:687-94. [ Links ]
16. Rodrigo L, Riestra S, Nino P, et al. A population-based study on the incidence of inflammatory bowel disease in Oviedo (Northern Spain). Rev Esp Enferm Dig 2004;96:296-305. DOI: 10.4321/S1130-01082004000500002. [ Links ]
17. Lopez-Serrano P, Perez-Calle JL, Carrera-Alonso E, et al. Epidemiologic study on the current incidence of inflammatory bowel disease in Madrid. Rev Esp Enferm Dig 2009;101:768-72. DOI: 10.4321/S1130-01082009001100004. [ Links ]
18. Arin Letamendia A, Borda Celaya F, Burusco Paternain MJ, et al. High incidence rates of inflammatory bowel disease in Navarra (Spain). Results of a prospective, population-based study. Gastroenterol Hepatol 2008;31:111-6. [ Links ]
19. Fernández A, Hernández V, Martínez-Ares D, et al; EpiCom Group. Incidence and phenotype at diagnosis of inflammatory bowel disease. Results in Spain of the EpiCom study. Gastroenterol Hepatol 2015;38(9):534-40. DOI: 10.1016/j.gastrohep.2015.03.001. [ Links ]
20. Lennard-Jones JE. Classification of inflammatory bowel disease. Scand J Gastroenterol 1989;24(Suppl 170):2-6. DOI: 10.3109/00365528909091339. [ Links ]
21. Ott C, Obermeier F, Thieler S, et al. The incidence of inflammatory bowel disease in a rural region of Southern Germany: a prospective population-based study. Eur J Gastroenterol Hepatol 2008;20(9):917-23. DOI: 10.1097/MEG.0b013e3282f97b33. [ Links ]
22. Sinci BM, Vuceli B, Persi M, et al. Incidence of inflammatory bowel disease in Primorsko-goranska County, Croatia, 2000-2004: A prospective population-based study. Scand J Gastroenterol 2006;41:437-44. DOI: 10.1080/00365520500320094. [ Links ]
23. Molodecky NA, Soon IS, Rabi DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012;142:46-54. DOI: 10.1053/j.gastro.2011.10.001. [ Links ]
24. Jess T, Riis L, Vind I, et al. Changes in clinical characteristics, course, and prognosis of inflammatory bowel disease during the last 5 decades: a population-based study from Copenhagen, Denmark. Inflamm Bowel Dis 2007;13:481-9. DOI: 10.1002/ibd.20036. [ Links ]
25. Louis E, Collard A, Oger AF, et al. Behaviour of Crohn's disease according to the Vienna classification: changing pattern over the course of the disease. Gut 2001;49:777-82. DOI: 10.1136/gut.49.6.777. [ Links ]
26. Romberg-Camps MJ, Dagnelie PC, Kester AD, et al. Influence of phenotype at diagnosis and of other potential prognostic factors on the course of inflammatory bowel disease. Am J Gastroenterol 2009;104:371-83. DOI: 10.1038/ajg.2008.38. [ Links ]
27. Burisch J, Pedersen N, Cukovic-Cavka S, et al. Initial disease course and treatment in an inflammatory bowel disease inception cohort in Europe: The ECCO-EpiCom Cohort. Inflamm Bowel Dis 2014;20:36-46. DOI: 10.1097/01.MIB.0000436277.13917.c4. [ Links ]
![]() Correspondence:
Correspondence:
Ignacio Cueto-Torreblanca.
Department of Gastroenterology.
Hospital Clínico Universitario Virgen de la Victoria.
Campus de Teatinos, s/n.
29010 Málaga, Spain
e-mail: nachoct@gmail.com
Received: 18-11-2016
Accepted: 21-01-2017











 texto en
texto en