Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Farmacia Hospitalaria
versión On-line ISSN 2171-8695versión impresa ISSN 1130-6343
Farm Hosp. vol.40 no.6 Toledo nov./dic. 2016
https://dx.doi.org/10.7399/fh.2016.40.6.10424
Effectiveness and safety of pemetrexed for non-small cell lung cancer in the Andalusian Public Health System
Efectividad y seguridad del pemetrexed en el cáncer de pulmón no microcítico en el Sistema Sanitario Público de Andalucía
María Antonia Pérez-Moreno1, Jesús Cotrina-Luque1, Mercedes Galván-Banqueri1, Sandra Flores-Moreno1, Francisco Javier Bautista-Paloma1 and Miguel Ángel Calleja-Hernández2
1 Hospital Pharmacist. Pharmacy Department. Hospital Universitario Virgen del Rocío, Seville.
2 Hospital Pharmacist. Pharmacy Department. Hospital Universitario Virgen de las Nieves, Granada. Spain.
This study was supported by the Health Department of the Spanish Government. (Investigación Clínica Independiente. Ministerio de Sanidad y Política Social).
ABSTRACT
Objective: To evaluate effectiveness and safety profile of pemetrexed in patients with locally advanced or metastatic non-small-cell lung cancer (NSCLC) when it's used on real clinical practice in Andalusia (a Spanish region with 8.5 million inhabitants, 2014 census data).
Methods: An observational multicentre retrospective study was conducted. Adult patients with locally advanced/metastatic NSCLC who received pemetrexed in any hospital in the Andalusian Public Health System during the last term of 2011 were included. We collected patients' baseline characteristics, diagnostic and treatment data, effectiveness variables (response to treatment with pemetrexed and overallsurvival) and main adverse reactions detected.
Results: 172 patients from 17 hospitals were included (77.33% were men), median age 63 years old (between 34 and 83). The predominant histology was adenocarcinoma (84.30%) and 85.20% were diagnosed of lung cancer with IV-stage. 78.49% had been smokers at some point in their lives. Median overall survival from the start of pemetrexed was 9 months (95%CI, 4.1-13.9). Progression of the disease was the most frequent response (33.14%), only one patient had complete response. Stable disease was associated with a higher probability of survival. Main adverse reactions detected were asthenia, haematological reactions, gastrointestinal reactions and dermal o mucous toxicity. No patients discontinued treatment for serious toxicity.
Conclusions: Pemetrexed resulted quite effective in NSCLC when it was used on real clinical practice, with higher survival in non-squamous histology and patients with the best score of Eastern Cooperative Oncology Group scale. The toxicity profile was well tolerated. Prospective studies would be needed to confirm the effect of prognostic factors observed.
Key words: Pemetrexed; Non-small-cell lung cancer; Effectiveness; Safety.
RESUMEN
Objetivo: Evaluar la efectividad y el perfil de seguridad del pemetrexed en pacientes con cáncer de pulmón no microcítico (CPNM) localmente avanzado o metastásico en la práctica clínica real en Andalucía (una región española con 8,5 millones de habitantes según los datos del censo de 2014).
Métodos: Se realizó un estudio retrospectivo multicéntrico observacional, incluyendo aquellos pacientes adultos con CPNM localmente avanzado/metastásico que hubiesen recibido pemetrexed en cualquier hospital del Sistema Sanitario Público de Andalucía durante el último trimestre de 2011. Se revisaron las características basales de los pacientes, los datos relativos al diagnóstico y al tratamiento, las variables de efectividad (en términos de respuesta al tratamiento con pemetrexed y supervivencia global) y las principales reacciones adversas detectadas.
Resultados: Se incluyeron un total de 172 pacientes procedentes de 17 hospitales (77,33% hombres), con una mediana de edad de 63 años (rango: 34 y 83). La histología predominante fue el adenocarcinoma (84,30%) y el 85,20% fueron diagnosticados de cáncer de pulmón en estadio IV. El 78,49% habían sido fumadores en algún momento de sus vidas. La mediana de supervivencia global desde el inicio del pemetrexed fue de 9 meses (IC del 95%, 4,1-13,9). La progresión de la enfermedad fue la respuesta al tratamiento más frecuente (33,14%) y solo un paciente tuvo una respuesta completa. La presencia de enfermedad estable se asoció con una mayor probabilidad de supervivencia. Las principales reacciones adversas detectadas fueron astenia; reacciones hematológicas, gastrointestinales y dermatológicas o toxicidad mucosa. Ninguno de los pacientes interrumpió el tratamiento por toxicidad grave.
Conclusiones: El pemetrexed resultó bastante efectivo en el CPNM cuando fue utilizado en la práctica clínica real, con una mayor supervivencia en histología no escamosa y en los pacientes con mejor puntuación en la escala Eastern Cooperative Oncology Group. El perfil de toxicidad fue bien tolerado. Serían necesarios estudios prospectivos para confirmar el efecto de los factores pronósticos observados.
Palabras clave: Pemetrexed; Cáncer de pulmón no microcítico; Efectividad; Seguridad.
Introduction
Lung cancer (LC) is currently a significant public health problem as it is the most prevalent cancer in the world and the most common cause of death from cancer worldwide. There was estimated to be 1.8 million new cases in 2012 (12.9% of the total) and to be responsible for nearly one in five (1.59 million deaths, 19.4% of the total). The European age-standardised incidence rate of lung cancer in 2012 was 44.9 per 100.000 people1. Developing nations in Asia (e.g., China) and Africa are seeing increased lung cancer rates. In Spain 20.000 adults are newly diagnosed each year2.
Smoking accounts for 80% of the worldwide lung cancer burden in males and at least 50% of the burden in females3. However, new cases for all age groups combined declined 2.6% per year among men and 1.1% per year among women (2005-2009), specially fast in adults aged 35-44, which reflects decrease in smoking rates over the past 30 years4,5..
Globally, survival rates at 5 years after diagnosis are less than 15%6 and approximately 55% of patients who have been newly diagnosed with non-small-cell lung cancer (NSCLC) have distant metastases7.
In spite of the addition of new drugs to the therapeutic arsenal in recent years, disease prognosis continues to be poor, with response rates to first-line chemotherapy (Ch) of 20-40 % and a median survival of 7-12 months8.
Pemetrexed is an anti-metabolite that inhibits several enzymes acting in the folate synthesis, hence disrupting essential metabolic processes necessary for folate-dependent cell replication. At present, three indications are approved by regulatory agencies in locally advanced/ metastatic non-squamous histology: combined with platinum in first-line induction treatment; as monotherapy in first-line maintenance treatment (in patients whose disease has not progressed immediately following a platinum-based chemotherapy regimen) and as second-line treatment in monotherapy. In all three situations patients with predominantly squamous histology are excluded9.
Pemetrexed is currently considered an alternative in first-line and second-line treatment of NSCLC10,11,12, but the number of alternatives13,14,15, the use of pemetrexed in different situations (maintenance or induction) and the possibility of subpopulations that could get an additional benefit to this type of therapies16 makes their use controversial.
Despite its relative recent appearance on the market, there are numerous publications about pemetrexed use, although results are conflicting and not always consistent17. This fact together with the high economic and care impact involved in its use (the cost per patient is six times that of current treatment) 18, makes it necessary to perform an assessment of its effectiveness and safety, so as to identify the patient population that may significantly benefit from this therapy and conditions of use in which they can obtain better results.
For this, the main objective of this study was to evaluate effectiveness and safety profile of pemetrexed in patients with locally advanced or metastatic NSCLC when it's used on real clinical practice in Andalusia (a Spanish region with 8,5 million inhabitants, 2014 census data, where public hospitals provide healthcare for 96% of the population). Secondary objectives were to analyse effectiveness in terms of survival according to relevant characteristics (relating to the patient and to the tumour) and to evaluate the safety depending on patients' age and concomitant use of platinum.
Methods
An observational multicentre retrospective study of the use of pemetrexed was carried out. We collected data of all the patients who received pemetrexed for NSCLC at any Andalusian Public Healthcare Service hospital (Spain) between 1st October 2011 and 31st December 2011.
Inclusion criteria were: patients with age ≥ 18 years old with locally advanced (Illb) or metastatic (IV) NSCLC treated with pemetrexed in any Andalusian Health System hospital in the last term of 2011.
We recorded the following variables:
• Anthropometric data (patients' age, gender and body surface area).
• Renal function and smoking habits.
• Diagnostic and treatment data: tumour histology, metastatic location, disease stage and Eastern Cooperative Oncology Group stage (ECOG) perform status at diagnosis and at the beginning of pemetrexed treatment, indication of pemetrexed and the reasons for which treatment with pemetrexed had been finalized (scheduled end of treatment, voluntary abandonment, severe toxicity, disease progression, death, loss to follow up or deterioration of general condition).
• Effectiveness variables: The response to treatment was considered as complete response (CR), partial response (PR), stable disease (SD), disease progression (DP) or unknown. Furthermore, survival data were measured: Overall survival (OS, months) calculated as the time between initiation of chemotherapy and date of death and Progression-free survival (PFS, months), calculated as the time between the start of the chemotherapy and date of progression or end date tracking.
• Safety variables: Grade 3-4 adverse reactions included in the clinical history. The measures that were taken at the presence of toxicity were also reviewed.
Effectiveness variables were studied according to relevant characteristics of treatment and diagnosis.
We analysed if the combination with platinum and age influenced about the frequency of adverse reactions.
The treatment information was obtained from the oncology pharmacy database in each hospital. All patient demographic data were taken from digital clinical histories. In order to collect the data, all oncologist pharmacists for each center were contacted. They identified the patients who received pemetrexed from the chemotherapy pharmacy systems at their hospitals. Following this, a researcher visited each hospital to collect the data. At least one visit was made to each center. Visits were planned when required in order to finish collecting information.
A descriptive analysis of the data was carried out (mean, median, range and interquartile range). The description of the qualitative variables was conducted using frequency distribution and percentages. Quantitative variables were described by using measures of central tendency (mean or median) accompanied by a measure of dispersion (standard deviation or interquartile range). All the calculations were performed using a two-tailed test and confidence intervals of 95%, with the statistics program SPSS® 16.0. The statistical significance threshold was established at a P <0.05.
To compare means the nonparametric Mann-Whitney U test was applied and for the comparison of proportions Fischer's exact test was applied. The probability of survival was estimated with the Kaplan-Meier and to calculate the statistical mean of the different groups caption the Mantel-Cox test was used.
The approval of the Research Ethics Committee coordinating centers and the Spanish Drug and Devices Agency was obtained before starting the study. It was classified as a post-authorization study. According to this classification it was not necessary to obtain informed consent. It conforms to the Organic Protection of Data Law 15/1999, of December 13 (OPDL), and in order to protect patients' confidential data, they were identified by a corresponding numerical code preceded by the initials of the hospital, for example: HVR (Hospital Virgen del Rocio).
Results
Patients, disease and treatment characteristics
172 patients from a total of 17 hospitals were included in 2011. The median age was 63 years old (between 34 and 83 years old) and 42.4% older or equal to 65 years. 77.33 % were men. All patients had adequate renal function, 85.20% were diagnosed for lung cancer with metastatic-stage tumour (IV) and 78.49% had been smokers at some point in their lives (only 7% had stopped smoking at the time of diagnosis).
The predominant histology was adenocarcinoma (84.30%). At the beginning of the treatment with pemetrexed, 91.28% of the patients had stage IV tumour and 60.46% of them ECOG 1. Patients' baseline characteristics are shown in table 1.
At the end of the study 95 patients had died.
37.2% of patients with NSCLC received pemetrexed in combination with platinum for first-line treatment without maintenance therapy and 27.9% for first-line treatment in combination with platinum followed by maintenance pemetrexed.
Effectiveness results
The median overall survival from the start of pemetrexed was 9 months (95% CI 4.08 to 13.92). Figure 1 shows the Kaplan-Meier curve for OS.
In table 2 survival analyses results according to the possible prognostic factors studied has been collected.
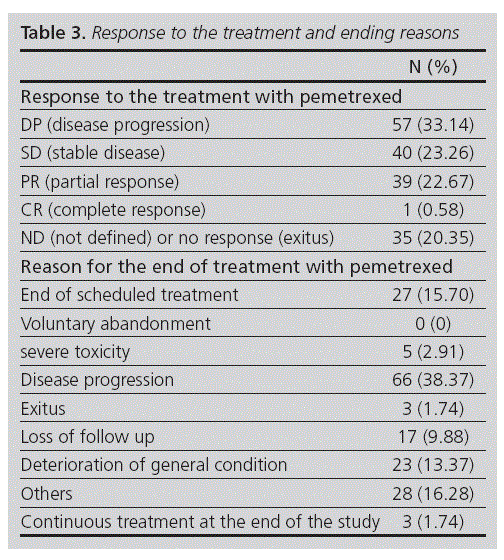
The response to treatment with pemetrexed and the main reasons for withdrawal or suspension of the treatment are shown in Table 3. Progression of the disease (PD) has been the most common treatment response to pemetrexed (33.14%) and after treatment with pemetrexed one patient had complete response (CR). The presence of stable disease was associated with a higher probability of survival.
The analyses of OS for each of histologic groups demonstrated improved OS in patients with adenocarcinoma and large-cell carcinoma. It was also shown improved survival in patients with the best score of ECOG scale (especially ECOG 0) and when the drug was prescribed as first-line treatment in combination with platinum followed by maintenance treatment with pemetrexed or when it was used in second line monotherapy treatment. Kaplan-Meier curves for OS according to these variables are shown in Figure 2.
It seems there was trend, but not significant, to longer OS in women, in patients younger than 65 years and when the stage at the start of treatment with pemetrexed was IV.
Safety results
Main grade 3-4 adverse reactions detected were asthenia, haematological reactions, gastrointestinal reactions and dermal o mucous toxicity. The values of the safety variables have been detailed in Table 4.
It was detected higher frequency of adverse reactions when pemetrexed was used in combination with platinum than when it was used in monotherapy, but there were no statistically significant differences. Safety results in terms of age were comparable between patients under 65 years and those with 65 years or older.
No patients discontinued treatment for serious toxicity associated with it.
The measures taken to the toxicity associated with pemetrexed were: end of the treatment (in 5 patients, due to asthenia, anemia, neutropenia and renal toxicity), doses reduction 20% (in 7 patients), delay of the next cycle of chemotherapy (for 4 patients), two patients required hospitalization (because of severe thrombocytopenia and rash) and for 10 patients other measures.
Discussion
This study (that is part of an ambitious research project supported by Spanish Government) has evaluated the effectiveness and safety of the treatment with pemetrexed in non-small-cell lung cancer when it's used on real clinical practice, specifically in a homogeneous region with more than 8 million inhabitants and it is the first study to target full medical histories instead of databases.
The majority of patients treated with pemetrexed were male, which is consistent with the fact that LC continues to be more prevalent in this sex1. In addition, a high percentage of patients had been smokers at some point in their lives, proving that this habit represents a very important risk factor for the development of this disease19.
There are published previous studies which assessed the use of pemetrexed in NSCLC, but with a population of less patients20,21. Also, unlike the previous ones, the present study evaluates effectiveness of pemetrexed in all lines prescribed for treatment pemetrexed.
As for the effectiveness results in terms of response to treatment, disease progression was the most common response. However, the rate of treatment response (complete response or partial response) was quite high, significantly higher than previous studies published as Villanueva et al.21 or Picaza et al.20, although the percentage of patients with stage IV was also very high. Further, compared with the pivotal clinical trials for the approval of three indications of pemetrexed in the present study we found a greater response to treatment with pemetrexed. In combination with platinum for first-line induction treatment, Scagliotti et al.22 detected a response rate of 30.6%, Hanna et al.23 in second line treatment found 9.1%, and Paz-Ares et al.8 described 3% of response for first-line maintenance treatment.
Overall survival data were similar or higher relative to the published literature, except for first-line treatment combined with platinum, in which survival rates were lower than the study Scagliotti et al.22 Similarly to published clinical trials differences in survival was observed according tumour histology, being significantly higher in non-squamous histology (adenocarcinoma and large cell), which confirms the fact that there are subpopulations of patients who may derive more benefit from treatment with pemetrexed. This finding differs from those obtained from previous retrospective studies such as Villanueva et al.21, in which no differences were found in survival according to histology, but in this study the majority was squamous histology.
Pemetrexed has been shown to be significantly safer than other therapies for lung cancer treatment in different lines. In our study, in 27 patients treated with pemetrexed grade 3-4 adverse reactions were detected, which seems a low percentage of the total patients (15,70%), with low rates of haematological and gastrointestinal toxicity, even lower than published data, especially in case of neutropenia. Although most frequent were when pemetrexed was associated with platinum, no statistically significant differences were found. Nor does the development of adverse reactions associated with age. This perhaps could be due to the low frequency of detected events.
This study is not without limitations. It refers to the Andalusian population, so it is a local study. Results could not be representative of the rest of areas. It is also a retrospective study without control group, so there are a lot of censored data because some of the clinical histories did not have all the required information of the data collection notebook, so the results shown may be somewhat bias. However, it provides valuable information about the effectiveness and safety of the drug in healthcare daily practice.
Conclusions
Pemetrexed resulted quite effective when it was used on real clinical practice, with higher survival in non-squamous histology (adenocarcinoma and large cell) and patients with the best score of ECOG scale. The toxicity profile was well tolerated.
The results should be carefully interpreted because of the small sample size and retrospective design of the study. Prospective studies would be needed to confirm the effect of prognostic factors observed.
Acknowledgments and affiliations
The authors wish to acknowledge their cooperation to other components of the Pharmacy Department of Virgen del Rocio Hospital and of other hospitals who have collaborated with data collection: Ma Teresa Garrido Martínez (Juan Ramón Jiménez Hospital), Nuria Muñoz Muñoz (Virgen Macarena Hospital), Silvia Artacho Criado (Virgen de Valme Hospital), Ana Isabel Gago Sánchez (Reina Sofía Hospital), Juan José Fernández Avila (Torrecárdenas Hospital), Inmaculada Vallejo Rodriguez (Virgen de las Nieves Hospital), Miguel Damas Fernández Fijares (San Cecilio Hospital), Francisco Nieto Cobo (Baza Hospital), Marcos Camacho Romera (Santa Ana Hospital), Marga Garrido (Costa del Sol Hospital), Begoña Muros de Fuentes (Virgen de la Victoria Hospital), Beatriz Mora Rodriguez (Regional de Málaga Hospital), Rocío Gavira Moreno (Jerez de la Frontera Hospital), José Carlos Roldán (La Línea Hospital), Ma José Martínez Bautista (Puerta del Mar Hospital), Carmen Martínez Díaz (Puerto Real Hospital), Pablo Villanueva Jimenez (Punta de Europa Hospital). Also, to Postgraduate Department of the Pharmacy Faculty from the University of Granada.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
No ethical approval was necessary as this is an observational and retrospective study.
References
1. Globocan 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. International Agent for Resarch on cancer. Available from: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx (accesed July 15th 2015). [ Links ]
2. Global Resourse for Advancing Cancer Education. Available from: http://cancergrace.org/lung/tag/spanish-lung-cancer-group/ (accesed July 15th 2015). [ Links ]
3. Siegel R, Naishadham D and Jemal A. Cancer statistics, 2013. CA: a cancer journal for clinicians. 2013; 63(1):11-30. [ Links ]
4. Society AC. Lung cancer key statistics. Available http://www.cancer.org/cancer/lungcancer-nonsmallcell/detailedguide/non-small-celllung-cancer-key-statistics (accessed 2014 November 29). [ Links ]
5. Henley SJ, Richards TB, Underwood JM, et al. Lung cancer incidence trends among men and women-united states, 2005-2009. MMWR. Morbidity and mortality weekly report. 2014; 63(1):1-5. [ Links ]
6. DiPiro JT, Talbert RL, Yee GC, et al. Pharmacotherapy a pathophysiologic approach. Vol. 132. Eighth Edition. Lenexa: ACCP; 2011; p. 2157-73. [ Links ]
7. Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11-30. [ Links ]
8. Paz-Ares LG, Altug S, Vaury AT, et al. Treatment rationale and study design for a phase III double-blind, placebo-controlled study of maintenance pemetrexed plus best supportive care versus best supportive care immediately following induction treatment with pemetrexed plus cisplatin for advanced nonsquamous non-small cell lung cancer. BMC Cancer. 2010;10:85. [ Links ]
9. Pemetrexed product information (AlimtaH). 2013. Available from: http://www.ema.europa.eu/docs/es_ES/document_library/EPAR_Product_Information/human/000564/WC500025611.pdf (Accesed July 15th 2015). [ Links ]
10. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology - Small Cell Lung Cancer, 2010. Available from: www.nccn.org/professionals/physician_gls/default. asp (Accesed July 15th 2015). [ Links ]
11. NSCLC Spanish Society of Medical Oncology Guide. Available from: http://www.seom.org/publicaciones/guias-clinicas/102720-guia-seom-de-cancer-pulmon-no-microcitico#esp (Accesed July 15th 2015). [ Links ]
12. Non squamous - NSCLC guide. Andalusian hospital pharmacotherapy guide. Informe Genesis 3.0 version, published on July 2010. Available from: http://gruposdetrabajo.sefh.es/ genesis/genesis/Enlaces/InformesHosp_abc.htm#P (Accesed July 15th 2015). [ Links ]
13. Hong J, Kyung SY, Lee SP, et al. Pemetrexed versus gefitinib versus erlotinib in previously treated patients with non-small cell lung cancer. Korean J Intern Med. 2010;25:294-300. [ Links ]
14. Pilkington G, Boland A, Brown T, et al. A systematic review of the clinical effectiveness of first-line chemotherapy for adult patients with locally advanced or metastatic non-small cell lung cancer. Thorax. 2015 ;70:359-67. [ Links ]
15. Madureira P, de Mello RA, de Vasconcelos A, et al. Immunotherapy for lung cancer: for whom the bell tolls? Tumour Biol. 2015;36:1411-22. [ Links ]
16. Yang CH, Simms L, Park K, et al. Efficacy and safety of cisplatin/ pemetrexed versus cisplatin/gemcitabine as first-line treatment in East Asian patients with advanced non-small cell lung cancer: results of an exploratory subgroup analysis of a phase III trial. J Thorac Oncol. 2010;5:688-95. [ Links ]
17. Pérez-Moreno MA, Galván-Banqueri M, Flores-Moreno S, et al. Systematic review of efficacy and safety of pemetrexed in non-small cell lung cancer. Int Journal of Clinical Pharmacy 2014;36:476-487. [ Links ]
18. Isla D, González-Rojas N, Nieves D, et al. Treatment patterns, use of resources, and costs of advanced non-small-cell lung cancer patients in Spain: results from a Delphi panel. Clin Transl Oncol. 2011;13:460-71. [ Links ]
19. Yu Y, Liu H, Zheng S, et al. Gender susceptibility for cigarette smoking-attributable lung cancer: a systematic review and meta-analysis. Lung Cancer. 2014;85:351-60. [ Links ]
20. Picaza E, Agustin Ma J, Varela I, et al. Estudio retrospectivo de la efectividad y seguridad de pemetrexed en mesotelioma pleural maligno y cáncer de pulmón no microcítico. Atención Farmacéutica. 2008; 10: 2: 73-78. [ Links ]
21. Villanueva-Herraiz S, Ortega-García MP, Camps-Herrero C, et al. Estudio de utilización de pemetrexed en el cáncer de pulmón no microcítico. Farm Hosp. 2010;34:194-203. [ Links ]
22. Scagliotti GV, Parikh P, von Pawel J, et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol. 2008;26:3543-51. [ Links ]
23. Hanna N, Shepherd FA, Fossella FV, et al. Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy. J Clin Oncol. 2004;22:1589-97. [ Links ]
![]() Correspondence:
Correspondence:
Correo electrónico: sg.pm.mary@gmail.com
(María Antonia Pérez-Moreno).
Recibido el 4 de diciembre de 2015;
aceptado el 25 de julio de 2016.