My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Farmacia Hospitalaria
On-line version ISSN 2171-8695Print version ISSN 1130-6343
Farm Hosp. vol.40 n.6 Toledo Nov./Dec. 2016
https://dx.doi.org/10.7399/fh.2016.40.6.10467
Adaptation of oral medication in people institutionalized in nursing homes for whom medication is crushed: The ADECUA Study
Adecuación de la medicación oral en personas institucionalizadas en residencias de mayores a los que se les tritura la medicación: Estudio ADECUA
Francisco Apolo Carvajal, María González Martínez, Elena Capilla Santamaría, Betsabé Cáliz Hernández, Icíar Cañamares Orbis, Nuria Martínez Casanova, Eugenia Martínez Sánchez, Ainhoa Aranguren Oyarzábal, María José Calvo Alcántara and Encarnación Cruz Martos
General Subdirectorate of Pharmacy and Healthcare Products. SERMAS.
ABSTRACT
Purpose: To evaluate the effect of pharmacist interventions in the adaptation of pharmaceutical forms in elders institutionalized in nursing homes whose medicines are crushed, which may have a clinical relevance.
Methods: Quasi-experimental, multicenter, transversal and prospective study carried out in 10 nursing homes. Subjects for whom drugs were being crushed were identified. Their treatments were reviewed in order to identify drugs that should not be crushed. In these cases, we proposed an alternative to the physician and we evaluated the degree of acceptance, its association to other variables and the pre-post adaptation index (tablets that can be crushed and capsules that can be opened/ total number of tablets and capsules before and after the intervention). Moreover, health professionals received a training course.
Results: Medication was being crushed for 33% (618/1875) of residents (mean: 5 drugs susceptible of crushing). 220 pharmaceutical interventions were performed, mostly in extended release or gastro-resistant drugs (48% were accepted). Switch was the type of intervention more frequent (79%), mainly to immediate release forms or to a different drug. The adaptation index increased from 93% to 95% (p < 0.001). Satisfaction amongst health professionals was high.
Conclusions: The pharmacist intervention improved the correct administration of drugs, thereby increasing safety and efficacy.
Key words: Deglutition; Crush; Oral drug administration; Dosage form.
RESUMEN
Objetivo: Evaluar el efecto de la intervención del farmacéutico en la adecuación de formas farmacéuticas en ancianos institucionalizados en residencias de mayores a los que se les tritura la medicación y cuya alteración pueda tener relevancia clínica.
Método: Estudio cuasiexperimental, multicéntrico, transversal y prospectivo de diseño antes-después en 10 residencias de mayores. Se identificaron los residentes a los que se les trituraba la medicación y se revisó su tratamiento para identificar los medicamentos que no se pueden triturar. Se comunicó al médico responsable la alternativa disponible y se evaluó el grado de aceptación, su asociación con otras variables medidas y el índice de adecuación pre-post, (comprimidos/cápsulas triturables respecto al total de comprimidos/cápsulas antes y después de la intervención). Se impartió además un curso formativo dirigido a los profesionales sanitarios.
Resultados: Se trituraba la medicación al 33% (618/1.875) de los pacientes institucionalizados (media: 5 fármacos susceptibles de trituración). Se realizaron 220 intervenciones, mayoritariamente por tratarse de fármacos de liberación prolongada o gastrorresistentes, de las que se aceptaron el 48%. El tipo de intervención más frecuente fue el cambio (79%), mayoritariamente a formas de liberación inmediata o a otro principio activo. El índice de adecuación pasó del 93% al 95% (p < 0,001). La satisfacción de los profesionales con el curso fue elevada.
Conclusiones: La intervención del farmacéutico ha mejorado la correcta administración de los medicamentos, aumentando con ello su seguridad y eficacia.
Palabras clave: Deglución; Triturar; Administración oral de medicamentos; Forma farmacéutica.
Contribution to literature
Dysphagia in elderly people may be caused by a diversity of alterations, both structural and functional. The physiopathology is determined by different factors, including ageing itself. The prevalence of dysphagia varies depending on the setting where the study is conducted. Ekberg et al. found a prevalence ranging from 3 to 50% in the European population1. It affects between 56% and 78% of institutionalized elderly people, depending on the level of severity considered2. In this setting, other studies have verified a prevalence of dysphagia between 50 and 60%3,4. In studies conducted in other countries, it has been confirmed that many medicinal products are incorrectly manipulated, and can cause toxicity problems and lack of effectiveness5,6. In Spain, some deaths were reported consequence of manipulating extended release tablets that changed its bioavailability by crushing. Therefore, it is necessary to detect these medication errors and suggest alternatives for these patients. To our knowledge, there have been no studies so far in nursing homes to assess the role of the pharmacist in this area.
There is evidence of a high prevalence of crushing medicines in nursing homes in Spain, and there is a frequent inadequate crushing7. Many healthcare professionals are not aware of the consequences of crushing. Our study demonstrates that there are pharmacological alternatives for most of medicines that can be administered without any previous alteration, and that the pharmacist plays an essential role in the training of these professionals and adapting the correct pharmaceutical forms.
Introduction
Dysphagia or difficulty swallowing, is a term that indicates the existence of a subjective sensation of difficulty when the alimentary bolus passes during swallowing8. It can include a wide range of sensations, from the existence of problems in order to initiate swallowing, to the sensation that food has become stuck in the oesophagus. Dysphagia is a frequent health issue in the institutionalized elderly population, as a consequence of physiological and pathological changes (particularly neurological and degenerative conditions) associated with the ageing process. It is estimated that the prevalence of dysphagia in this population group varies between 56 and 78%4,9-12. Moreover, in some cases these swallowing problems will require the use of feeding tube for the administration of nutrients.
Elderly people institutionalized in nursing homes with swallowing problems refer difficulties for taking whole capsules and tablets13, which can lead to medication errors due to lack of adherence or wrong manipulation of pharmaceutical forms14.
In this situation, healthcare professionals are forced to use techniques to manipulate pharmaceutical forms, such as crushing tablets or opening capsules in order to facilitate their administration. The lack of knowledge by healthcare professionals of the consequences caused by the alteration of the pharmaceutical form on the effectiveness and safety of the medication6,15, as well as the lack of instructions indicating how to carry out this technique, will lead to an increased risk of iatrogenia and medication errors. A study published in 2012 showed that these practices were carried out for 32% of residents institutionalized in geriatric units, with swallowing and behavioural alterations as the most frequent causes for crushing; and that this practice was not allowed in 42% of medications analyzed5.
Tablet crushing practice shown in the hospital setting can also be reproduced in the Primary Care setting, and particularly in the case of nursing homes.
The primary aim is to evaluate the intervention carried out by the pharmacist in the adaptation of pharmaceutical forms in institutionalized people in nursing homes whose medicines are usually crushed and this form alteration could have potential clinical relevance. The secondary aims are to estimate the prevalence of crushing solid pharmaceutical forms for institutionalized persons, to evaluate the influence of age and shift of administration in the prevalence of crushing and to identify the medicines most frequently crushed with potentially negative consequences.
Methods
A quasi-experimental, multicentre, transversal and prospective study, before-and-after design, conducted in ten nursing homes with a pharmacist in their multidisciplinary team. At the same time, an observational transversal study was conducted to estimate the prevalence of the crushing practice in these centres.
The study included all institutionalized residents whose treatment was prepared in dosette boxes and had at least one medicine crushed in any of the shifts. Residents who self-administered their medicines were excluded and those who died or were transferred to other centres during the course of the study were considered as follow-up losses.
The duration of the study was five months, from May to September, 2015. The study protocol was reviewed and approved by the Regional Clinical Research Ethics Committee.
This study was developed in three phases:
Phase 1. Before the intervention
Those residents whose medicines were usually crushed were identified with a personal interview by the healthcare professionals responsible for administration, both in the morning and in the afternoon shift.
Afterwards, the pharmacist reviewed the treatment of these residents, checking if crushing was adequate for each medicine according to the summary of product characteristics and two guidelines on correct administration of medicines16,17. In case of lack of information and/or discrepancy in bibliography, information was asked to the manufacturing pharmaceutical company. If there were no stability studies, it was considered adequate, at the judgment of the researcher, to crush those tablets coated with the aim to mask an unpleasant taste or smell, or to protect from light or humidity, or chewable tablets. The formulation of orodispersible and effervescent tablets allows their administration in people with swallowing problems. In case of someone crushed them, it was considered clinically irrelevant. Regarding capsules, it was considered adequate to open capsules with powder or pellets inside, with bibliography supporting that these could be opened, emptied and dispersed in water, though the drug had an unpleasant taste or smell. During data collection, it was considered inadequate to crush acetylsalicylic acid, although its summary of product characteristic state that it can be crushed, due to its gastric damaging effects, with the risk of ulcer for acetylsalicylic acid enteric coated of 6% vs. 23% in other formulations without it18. Demographical data were collected (age and gender), as well as the reason for crushing medicines. Regarding treatment, the following data were collected:
- Total number of lines of prescription, including medicines (there can be different lines with the same drug), oxygen, dietary supplements, thickening agents, and healthcare products. Both, chronic and acute treatments were recorded.
- Number of capsules and tablets that can be modified.
- Number of tablets that can be crushed and capsules that can be opened according to their summary of product characteristics.
- Number of tablets that can be crushed and capsules that can be opened according to bibliography.
- Number of tablets that can be crushed and capsules that can be opened according to the clinical judgment of the researcher.
- Number of tablets defined as not adequate to be crushed and capsules that cannot be opened.
- Reason for not crushing (extended release, gastro-resistant, soft gelatine capsule, clinical trial medicines, teratogenic or cytotoxic drugs).
The prevalence of crushing practice was calculated, defined as the ratio between the number of residents whose medication was crushed and the total number of residents included in the study. The adaptation index was calculated and defined as the ratio of medicines adequately crushed and the number of medicines that may be crushed.
Phase 2. Intervention
Two types of interventions were conducted in this phase.
Firstly, a 4-hour educational programme was designed and certified about the rational use of medicines in nursing homes. This programme included recommendations to strengthen safe strategies in the different stages of medication process in the centres. The administration section represented half of the programme's content, and it consisted in an explanation of how pharmaceutical forms should be administered, including a specific subsection for medication crushing, highlighting specially those types of tablets that should not be crushed, and instructions for drug administration. Secondly, all incidences detected in administration, as well as the pharmacological alternative or pharmacist recommendation, were communicated to the physician through written and oral report. No alternatives were considered for those medications that could be crushed, because the switch to other pharmaceutical forms can cause more medication errors in nursing homes19.
The number of meetings conducted, number of attendees, knowledge acquired, and level of satisfaction regarding educational program as well as the number and type of interventions suggested (switching to another drug, addition of another medicine, interruption, or instructions for drug administration) were all recorded. The addition of another medication was considered for those cases where there was no alternative and the consequences of crushing could be alleviated by adding on another medication. Switching to a different pharmaceutical form, to another drug, or from fixed-dose combination to single drugs was specified as intervention subtype.
Phase 3. After the intervention.
In this phase, the pharmacist assessed the effect of the actions.
To this aim, the pharmacist evaluated the intervention on prescription within the 30 days after it was reported. The number of interventions accepted was recorded, and the reason for not accepting (treatment about to be completed, medicines by special authorization, a patient without clinical criteria for crushing, prescription by specialist, discrepancy with the medical criteria, lack of time, and unknown).
Besides, it was evaluated if the level of acceptance was associated with any of the variables collected (centre, physician, medication and type of intervention), and the monthly economic impact derived was calculated. This impact was obtained based on the prices of medications at recomended retail price including value add taxes (RRP including VAT) that appear in the nomenclator of February, 2015. Finally, the post-intervention adequacy index was determined, and it was compared with the value before the intervention, in order to assess the primary objective.
Statistical analysis
The sample size calculated to find statistically significant differences with 80% potency was of 202 nursing home residents, assuming an increase in the adequacy index of 80% to 90%.
A statistical analysis was conducted of the data collected, evaluating the association between variables through the χ2 test for qualitative variables, using the Bonferroni Correction for comparing multiple proportions, and the Student t-test (parametric) or Mann-Whitney U Test (non-parametric) for quantitative variables. McNemar test was used for the calculation of the difference in proportions for paired samples. The IBM® SPSS® Statistics 22.0 program for statistic analysis was used.
Results
There were 1875 residents identified, within a range of 56-to-107-years of age, whose medicines were prepared in dosette boxes. From these, 618 (33.0%) residents had their oral medication usually crushed, and this was more frequent in the women subgroup (see table 1), and in the older residents (p = 0.012). It was also observed that there was more medicine crushing in the morning shift than in the afternoon (p < 0.05). The main reason for crushing was lack of cooperation by the patient due to cognitive impairment, followed by difficulty swallowing (see table 2).
The educational programme was held in 41 instances at different times that included all working shifts. The number of attendees was 436 healthcare professionals. A significant increase was observed in knowledge about safety in crushing medicines, going from 9.8% in the previous questionnaire to 85.2% in the questionnaire after the course (OR = 53.2; CI95%: 32.5-87.5). Healthcare professionals rated their satisfaction as sufficient or complete, in 97.9% of cases.
From the 618 residents included in Phase 2, only the treatment of 579 was reviewed, due to follow-up losses for death or transfer. In total, 4682 lines of prescription were reviewed (mean: 8.1 lines of treatment/ patient). From these, 3031 (64.7%) were for medicines that can be crushed or altered (mean: 5.2 medications/residents); 1752 (57.8%) could be manipulated according to the summary of product characteristics, and this number increased to 2643 (87.2%) after the review by the pharmacist of the bibliography available (see table 3). After consulting with the manufacturer regarding discrepancies and lack of information, and the subsequent clinical evaluation by the pharmacist, 7.3% (n = 220) could not be crushed, and therefore the adequacy index before the intervention was 92.7%. The main reasons the tablet could not be crushed or the pharmaceutical form could not be altered were: extended-released medication (62.8%) and having a gastroresistant coating (31.4%), where manipulation affected its safety or effectiveness.
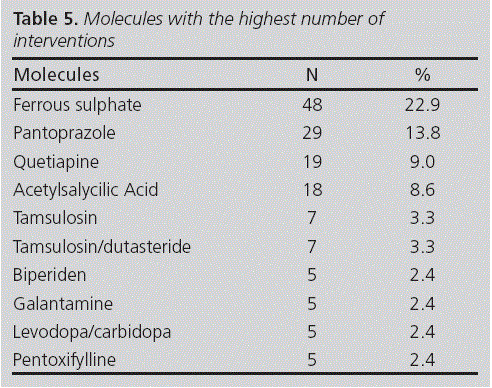
After the review, there were ten follow-up losses, and therefore 210 alternative recommendations were suggested by the pharmacist. Table 4 shows the type and subtype of intervention conducted. There were interventions on 78 different medicines, corresponding to 42 drugs. A 70.5% of interventions were conducted upon just ten drugs (see table 5). All recommendations for adding on another drug were conducted for residents at high risk of gastrointestinal bleeding and on treatment with acetylsalicylic acid without any gastric protection with a proton pump inhibitor; in these cases, it was suggested to add on omeprazole.
In Phase 3, the recommendations were accepted in 48.5% of cases at 30 days. The causes for rejection appear in table 6; the first cause is unknown justification, followed by discrepancy with the clinical criterion. The level of acceptance for recommendations was associated with the type of intervention (see Table 4); the most frequently accepted interventions were instructions for administration (OR = 0.01; CI95%: 0.00-0.83) and treatment discontinuation (OR = 0.20; CI95%: 0.06-0.62) vs. switching to another drug (p < 0.05). Statistically significant differences were found between some centres, but no relationship was established with the reason for lack of acceptance, medications, or physicians in charge. The interventions recommended would have represented monthly economic savings of 898€, if completely accepted. However, the benefit obtained in money terms by the recommendations accepted was of 187€ per month.
The adequacy index after the intervention was 95.5%, and benefit by the pharmaceutical intervention was obtained per each 27 crushed medications (21 to 40; p < 0.001).
Discussion
The prevalence of crushing obtained in our study, as well as the reasons for medication crushing, are similar to the literature for the same population group5,20. However, the percentage of drugs that should not be crushed described here is much lower than the one found by Caussin et al.5; therefore, the degree of adaptation found before the intervention is very high. This is due to the fact that only those preparations that can have an important clinical relevance if altered have been selected. Another reason is that the clinical criteria of the researcher have been included, based on the information provided by the pharmaceutical companies. If we consider drugs that can be crushed according to their summary of product characteristics, the percentage would coincide with the one described in the French study by Caussin et al.5.
On the other hand, female gender and age have been associated with medication crushing, we cannot draw clear conclusions; in the first case, the reason for this association can be the higher percentage of women included in our sample, that can be explained because they have a longer life expectancy vs. men21 and, in the case of age, the magnitude of the effect has low importance and clinical relevance, and outcomes could be influenced by other confounders, such as the clinical situation of residents. Regarding the administration shifts, it is very likely that the outcomes are due to the fact that the morning shift includes breakfast and lunch, where most of the medication is administered to residents in these centres. The afternoon shift includes the medication administered at tea-time and dinner; the drugs most frequently administered at this time are hypnotics, anxiolytics, statins, and acenocoumarol. These medications are usually smaller, and many of them are orodispersible. However, this result was not expected, because the status of the residents gets worse over the day, and this will generally have a negative impact on their swallowing reflex.
Our outcomes by type of intervention indicate there is an alternative for practically all medicines that cannot be crushed; only 2.4% of the total required instructions for administration. These data refer to an oral anticoagulant that could not be replaced, and three medications that were part of a clinical trial. Moreover, most medications present another pharmaceutical form with the same drug, or an equivalent therapeutic alternative from the same therapeutic group. It is surprising that the intervention switching prescription has been associated with a lower level of acceptance than treatment discontinuation. It is possible that the subsequent time of evaluation has not been adequate or that reminders must be sent to physicians in order to increase the percentage of acceptance in this group, because the reason for not following the recommendations in almost two-thirds of cases was lack of time or unknown.
Though the previous adequacy index was very high, and superior to the one assumed when calculating the sample size, the potency of the study allowed to establish a statistically significant relationship between the pharmacist intervention and an improvement in the adequacy index. One out of five residents reviewed benefited from the pharmacist intervention.
The objective of this study was to increase safety and not to reduce costs; however, there were modest economic savings with the interventions accepted. These savings included only the direct costs of medicines, because we have not found studies that assess the number of visits to emergency department, hospitalizations, or adverse reactions prevented through this type of intervention, and their associated cost. If these indirect costs were included, it can be assumed that the savings obtained would be much higher, not only in economic terms but also regarding an improvement in the management of resources and patient safety.
On the other hand, healthcare professionals had a positive assessment of the educational programme, which increased their knowledge on administration and drug manipulation. However, there was no assessment of its impact on the number of medication errors associated with the manipulation of pharmaceutical forms, though it has been described in literature that the development by a pharmacist of an educational programme on this matter and in the same setting will lead to a reduction in this type of errors22.
The main limitation of this study is that no health outcomes were evaluated. The reason for this is that a long-term follow-up would be required for assessing the safety and efficacy parameters, with definition of all the subrogated variables of all treatments that are incorrectly crushed previously to initiating the study, and with the likely existence of many factors of confusion. We believe that it would be necessary to conduct other studies to evaluate the health outcomes associated with an incorrect manipulation of medicines by clinical condition.
Another limitation in our study is its quasi-experimental design without a control group. However, it was not considered ethical to deprive a group of residents of an optimization in their pharmacological treatment, particularly when this could have negative consequences on their health.
The outcomes obtained in this study lead to the conclusion that the incorporation of a pharmacist to nursing homes rovides added value, increasing the safety and effectiveness of pharmacological treatments.
Conflict of interests
No conflict of interests.
Bibliography
1. Ekberg O, Hamdy S, Woisard V, Wuttge-Hannig A, Ortega P. Social and psychological burden of dysphagia: its impact on diagnosis and treatment. Dysphagia. 2002;17(2):139-46. [ Links ]
2. Clavé P, Arreola V, Velasco M, Quer M, Castellví JM, Almirall J, et al. Diagnóstico y tratamiento de la disfagia orofaríngea funcional. Aspectos de interés para el cirujano digestivo. Cir Esp. 2007 Aug;82(2):62-76. [ Links ]
3. Lin L-C, Wu S-C, Chen HS, Wang T-G, Chen M-Y. Prevalence of impaired swallowing in institutionalized older people in taiwan. J Am Geriatr Soc. 2002 Jun;50(6):1118-23. [ Links ]
4. Ferrero López M, García Gollarte J, Botella Trelis J, Juan Vidal O. Detección de disfagia en mayores institucionalizados. Rev Esp Geriatría Gerontol. 2012;47(4):143-7. [ Links ]
5. Caussin M, Mourier W, Philippe S, Capet C, Adam M, Reynero N, et al. L'écrasement des médicaments en geriatrie: une pratique <<artisanale>> avec de fréquentes erreurs qui nécessitait des recommandations. Rev Médecine Interne Fondée Par Société Natl Francaise Médecine Interne. 2012 Oct;33(10):546-51. [ Links ]
6. Ecraser un comprimé ou ouvrir une gélule: beaucoup d'incertitudes, quelques dangers avérés. Rev Prescrire. 2014;34(366): 267-73. [ Links ]
7. Estudio EARCAS: Eventos Adversos en Residencias y Centros Asistenciales Sociosanitarios (Internet). Madrid: Ministerio de Sanidad, de Sanidad, Política Social e Igualdad; 2011. Available from: http://www.msssi.gob.es/organizacion/sns/planCalidadSNS/docs/EARCAS.pdf. [ Links ]
8. Madrid (Comunidad Autónoma), Dirección General de Farmacia y Productos Sanitarios. Guía farmacogeriátrica. Madrid: Dirección General de Farmacia y Productos Sanitarios; 2004. [ Links ]
9. Hernández Martín J, Correa Ballester M, Vial Escolano R, Forcano García M, Gómez Navarro R, González García P. Adaptación de la guía farmacoterapéutica de un hospital sociosanitario a pacientes con disfagia. Farm Hosp. 2013;37(3):198-208. [ Links ]
10. Damián J, Valderrama-Gama E, Rodríguez-Artalejo F, Martín-Moreno JM. Estado de salud y capacidad funcional de la población que vive en residencias de mayores en Madrid. Gac Sanit. 2004;18(4):268-274. [ Links ]
11. García Aparicio J, Herrero Herrero JI, Moreno Gómez AM, Martínez Sotelo J, González del Valle E, Fernández de la Fuente MA. Pilotaje de un protocolo para la adecuación de la forma farmacéutica de la medicación oral al grado de disfagia, de los pacientes ingresados en un servicio de medicina interna. Nutr Hosp. 2011 Oct;26(5):933-9. [ Links ]
12. Khan A, Carmona R, Traube M. Dysphagia in the elderly. Clin Geriatr Med. 2014 Feb;30(1):43-53. [ Links ]
13. Aguilar Salmeron R. Administración de medicamentos a personas con dificultades para tragar (Internet). Cedimcat; (cited 2014 Dec 12). Available from: http://www.cedimcat.info/index.php?lang=ca. [ Links ]
14. Barber ND, Alldred DP, Raynor DK, Dickinson R, Garfield S, Jesson B, et al. Care Homes' Use of Medicines Study: prevalence, causes and potential harm of medication errors in care homes for older people. Qual Saf Health Care. 2009 Oct;18(5):341-6. [ Links ]
15. Royal Pharmaceutical Society. Pharmaceutical Issues when Crushing, OpeneningorSplitting OralDosage Forms. (Internet). 2011 (cited 2014 May 3). Available from: http://www.medicinesmanagementstoke.nhs.uk/documents/RPS_Pharmaceutical_Issues_when_Crushing_Opening_Splitting_sdosageforms_june_2011.pdf. [ Links ]
16. Arenaza Peña A, Arias Fernández L, Benítez Giménez M, Bilbao Gómez-Martino C, Borrego Hernando MI, Fernández Ruiz-Morón A. Guía de administración de medicamentos por sondas de alimentación enteral (Internet). Madrid: Hospital Clínico San Carlos; 2012 (cited 2014 Dec 12). Available from: http://www.madrid.org/cs/Satellite?blobcol=urldata&blobheader=applica tion/pdf&blobheadername1=Content-disposition&blobheadername2=cadena&blobheadervalue1=filename%3DGuia+de+administraci %C3%B3n+de+medicamentos+por+sondas+de+alimentaci%C3%B3n+enteral.pdf&blobheadervalue2=language%3Des%26site%3DHospital ClinicoSanCarlos&blobkey=id&blobtable=MungoBlobs&blobwhere=1352812967550&ssbinary=true. [ Links ]
17. Stockport Clinical Commisioning Group. Medication administration in patients with swallowing difficulties or dysphagia (Internet). (cited 2014 Dec 12). Available from: http://stockportccg.org/wp-content/uploads/2012/01/34838_Med_Admin_Dysphagia.pdf. [ Links ]
18. Silvoso GR, Ivey KJ, Butt JH, Lockard OO, Holt SD, Sisk C, et al. Incidence of gastric lesions in patients with rheumatic disease on chronic aspirin therapy. Ann Intern Med. 1979 Oct;91(4):517-20. [ Links ]
19. Alldred DP, Standage C, Fletcher O, Savage I, Carpenter J, Barber N, et al. The influence of formulation and medicine delivery system on medication administration errors in care homes for older people. BMJ Qual Saf. 2011 May;20(5):397-401. [ Links ]
20. Paradiso L, Roughead E, Gilbert A, Cosh D, Nation R, Barnes L, et al. Crushing or altering medications: what's happening in residential aged-care facilities? Australas J Ageing. 2002;21:123-7. [ Links ]
21. Instituto Nacional de Estadística. Esperanza de vida (Internet). 2015 (cited 2015 Sep 30). Available from: file:///C:/Users/Mar%C3%ADa/Downloads/4_1_Esperanza_de_vid.pdf. [ Links ]
22. Verrue CL, Mehuys E, Somers A, Van Maele G, Remon JP, Petrovic M. Medication administration in nursing homes: pharmacists' contribution to error prevention. J Am Med Dir Assoc. 2010 May;11(4):275-83. [ Links ]
![]() Correspondence:
Correspondence:
Correo electrónico: francisco.apolo@salud.madrid.org
(Francisco Apolo Carvajal).
Recibido el 19 de febrero de 2016;
aceptado el 14 de septiembre de 2016.











 text in
text in