Contribution to scientific literature
The relevance of this article lies in the need to understand the level of adherence in order to promote strategies that encourage it, and that will entail an increase in treatment efficacy, and an improvement in patients’ quality of life.
Given the outcomes from this article, we can say that all the multidisciplinary team members must be involved in improving the adherence results, particularly in those patients receiving treatment during long periods of time.
Introduction
Treatment adherence (ADH) is defined as the degree in which a patient acts according to the interval and doses prescribed in the dosing regimen, as a consequence of the commitment by the patient with the recommendations agreed upon by him / her and the healthcare professional1-4.
Patients with cancer are assumed to have particularly good adherence, due to the severity of their disease. However, different studies have demonstrated that this is not always true, and that adherence rates range from 16 to 100%, depending on the medication and measurement method5,6. It has been demonstrated that, after various cycles, there is a tendency to ADH reduction, essentially due to relaxation, lack of motivation, and/or adverse events (AEs)1-5,7.
In recent years, there has been an increase in the use of oral antineoplastic drugs (AOs); this change in therapeutics has an impact on the way to address the disease by patients and healthcare professionals: the patient becomes responsible for treatment management, and adherence becomes an important factor, because it can determine the efficacy of treatment3,6,8,9.
The majority of patients (80%) prefer oral to endovenous treatment, but they often have doubts about its efficacy and importance9. This fact, together with the complexity of treatments and associated AEs, can represent an obstacle at the time of achieving an adequate level of treatment compliance, and therefore efficacy1-5.
Authorized by the EMA in February, 2001, capecitabine is an oral pro-drug of 5FU, used in antineoplastic protocols for breast, colorectal and gastric cancer; the first two conditions have a high incidence. It presents an efficacy similar to parenteral 5FU, but with less associated toxicity1,10 and it can be used in combination with standard chemotherapy or targeted treatment, in this case increasing treatment complexity and the likelihood of AEs5,10,11. Its administration every 12 hours, with resting periods, in cycles and with prolonged duration, requires patient commitment in order to achieve adequate compliance and effective results.
It has been demonstrated that the intervention by the pharmacist providing information about all aspects associated with treatment can increase or encourage AHD2,3,5.
The objective of this article was to analyze ADH in patients on treatment with capecitabine, as well as AEs and the overall health status of patients, and the relationship of these factors with ADH.
Method
An observational prospective study at 7 months (from February to August, 2014), in a cohort of patients on neoadjuvant, adjuvant and metastatic disease treatment with capecitabine (nT), including treatment initiations (ni) as well as treatment continuations (nc), regardless of diagnosis. Those patients who received less than 3 cycles of capecitabine (nE) were excluded, because follow-up was not possible.
Inclusion criteria:
Adult patients with diagnosis of breast cancer, colorectal cancer, gastric cancer, and off-label;
Patients in capecitabine treatment continuation were accepted, as well as those treatment initiations during the 7 months of data collection;
Minimum number of cycles for evaluation: 3 cycles.
We considered one treatment cycle as the administration of capecitabine in:
Adjuvant treatment and metastatic disease:
monotherapy: 1250 mg/m2 / 12h x 14 days / 21 days, oral administration.
combination, according to chemotherapy regimen: 800-1250 mg/m2 / 12h x 14 days / 21 days, oral administration.
Neoadjuvant treatment:
Rectal cancer: 625mg/m2 / 12h continuous oral administration until completing radiotherapy; we considered one cycle as the interval between visits with the Oncologist, every 21-28 days.
Gastric cancer: ECX regimen in neoadjuvant and adjuvant treatment (epirubicin 50 mg/m2 intravenous (iv) on day 1, cisplatin 60 mg/m2 iv on day 1, capecitabine 625 mg/m2 / 12 h orally every 21 days).
(nT total population evaluated; nE excluded patients; n: population evaluated according to inclusion criteria; n = nT-nE; ni: initiation subgroup; nc: continuation subgroup; n = ni + nc)
The schedule for visits to the Pharmacy Unit (V) (8 within 7 months) was conducted according to the day 1 in each cycle, every 21 days, according to the following:
V1 (1st cycle)
Collection of demographical data: age, gender, diagnosis: breast cancer, colorectal cancer, gastric cancer, off-label; and performance status (ECOG PS)12,13.
Oral and written information about treatment to each patient, as well as about management of the most common AEs.
V2,V5 and V6 (2nd, 5th and 6th cycle)
Follow-up visits: Review of evolution reported by the Oncologist in the clinical record of the patient; solution of patient doubts regarding administration, storage, and management of AEs derived of treatment; surplus tablet count.
V3 and V7 (3rd and 7th cycle)
Quality of life assessment: Each patient was handed the SF-12 test (Figure 1)1,14 that had to be completed and returned at their next visit.
AE assessment: Each patient was handed the AE test (Figure 2)7,15 that should be completed and returned at their next visit.
V4 and V8 (4th and 8th cycle)
Review of toxicities described by the Oncologist in the electronic clinical record (IANUS®) from V1 to V4 and from V5 to V8, according to the NCICTCAE Classification v4.016. Assessment of AE test results handed to patients at V3 and V7.
Quality of life assessment: Review of SF-12 test results.
ASH assessment: The adherence for each patient was calculated through the Morisky Test2,17,18, Sackett’s self-reported compliance test (Figure 2)2,17,18 and the count of surplus tablets. All patients were dispensed the exact medication for each cycle; the frequency of dispensing was obtained from the Outpatient Dispensing Program (Silicon®).
We considered that a patient had good adherence when the results of the Morisky and Sackett tests were correct; thay is to say, if the patient answered adequately to the questions in the tests (Morisky: NO/ YES/NO/NO; Sackett: NO/YES/NO), and the surplus medication count was null.
Those data obtained were entered in an EXCEL® data-base, as well as the pharmaceutical interventions conducted. For data processing, patients were classified into 2 subgroups: treatment initiation (ni) and continuation (nc).
Statistical analysis was conducted through the SPSS® program, version 15, using the Chi-square test and Fisher Exact Test.
Results
From a cohort of 130 patients (nT), 111 were evaluated (n: 57 men, 54 women), with a mean age of 66.7 years (range: from 32 to 87 years). Initiation subgroup: ni = 71 and continuation subgroup nc = 40. Nineteen (19) patients were excluded (nE) due to treatment duration < 3 cycles (only one cycle dispensed). Patient distribution based on diagnosis and treatment regimen is shown in Figure 3.

Figure 3 Sample distribution base don diagnosis and therapeutic regimen (CAPIRI-BEVACIZUMAB: bevacizumab 7.5 mg/kg IV at day 1 + irinotecan 250mg/m2 IV at day 1 + capecitabine 850-1000mg/m2/12hx14d oral, every/21d; CAPOX-BEVACIZUMAB: bevacizumab 7.5 mg/kg IV at day 1 + oxaliplatin 130mg/m2 IV D1 + capecitabine 850-1000mg/m2/12hx14d oral, every/21d; CDDP-capecitabine: cisplatin 75mg/m2 IV, at day 1 + capecitabine 850-1000 mg/m2/12hx14d oral, every/21d; CAPECITABINE-LAPATINIB: capecitabine 1250mg/m2/12hx14d/21d, oral + lapatinib 1250 mg/24h, continuous oral administration; CAPECITABINA-RDT: capecitabine 625mg/m2/12h oral + radiotherapy; ECX: epirubicin 50mg/m2 IV, at day 1 + cisplatin 60mg/m2 IV, at day 1 + capecitabine 626 mg/m2/12hx21d oral, every/21d; CAPOX: oxaliplatin 130mg/m2 IV at day 1 + capecitabine 850-1000mg/m2/12hx14d oral, every/21d; CAPECITABINE: capecitabine 1250mg/m2/12hx14d/21d, oral.
Level of adherence
At V4, the percentage of patients with good adherence was 78.4%, superior in the initiation subgroup (ni81.7% vs. nc72.5%). At V8, only 19 patients were still on treatment. The adherence rate of these patients was 89.5%, superior in the initiation subgroup (ni91.6% vs. nc85.7%) (Figure 4).

Figure 4 Distribution of the ADH % recorded at V4 and V8, in the total sample and by sub-groups. Correlation of ADH test data and surplus medication count.
The remaining 92 patients were not adequate for assessment at V8 due to treatment completion, progression, and/or change of therapy.
Regarding medication count after a cycle, only 2 patients had surplus medication due to specific doses missed.
One study limitation was the reduced number of patients who returned the Morisky and Sackett tests at V8; and even though the level of adherence was 89.5% (91.6% ni vs. 85.7% nc), the sample was not representative regarding the initial study population (17.11% from n = 111). We would need a study with longer duration, in order to draw the conclusion that the pharmaceutical care provided encouraged adherence.
Adverse Events (AEs)
Clinical assessment by the Oncologist: clinical record review.
The AEs found match those described in the product specifications10. A 33% of patients presented skin toxicity, 14% presented abdominal pain, 26% asthenia, 25% diarrhoea, and there was neurological toxicity in 25% of patients, particularly in those on the CAPOX regimen (oxaliplatin 130mg/m2 iv at day 1 + capecitabine 1000mg/ m2 /12h x 14 days orally, every 21 days), requiring reduction or temporary discontinuation of oxaliplatin.
In the initiation subgroup, the most common (> 10%) Grade 1 AEs were: skin toxicity, diarrhoea and neurotoxicity; for Grade 2 (> 7%), skin toxicity and asthenia; and for Grade 3 G3 (> 3%) gastrointestinal pain. There was no Grade 4 toxicity.
In the continuation subgroup, the most common (> 9%) G1 event was skin toxicity; for G2, gastrointestinal pain (4.5%); for G3, skin toxicity and neurotoxicity (1.8%); and G4 neutropenia (0.9%).
Figure 5 shows the ADH level at V4 and V8, in patients with G2/G3 toxicities.

Figure 5 Adverse events. Clinical assessment by the Oncologist in the total population. ADH results vs. patient assessment for moderate intensity and duration ≥ 7 days
All AEs associated with capecitabine were reversible and did not require treatment discontinuation.
b. Subjective assessment by the patient
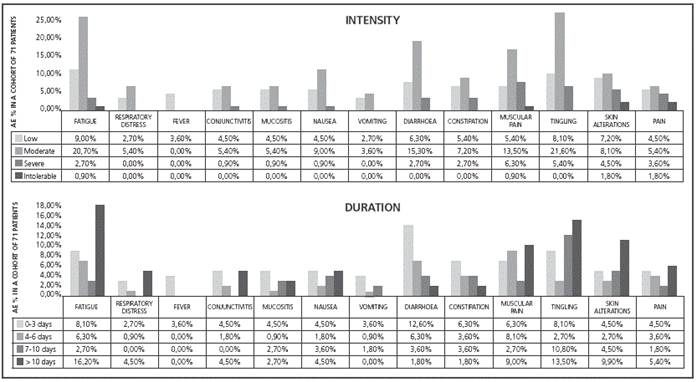
A 74.7% of patients returned the AE test (Figure 2), and 63.96% of them stated some AE in duration-intensity (Figure 6); the most relevant in terms of duration were: fatigue, tingling, diarrhoea, muscular pain. Figure 5 shows the ADH level at V4 and V8 in patients who stated AEs with duration ≥ 7 days and “moderate” intensity.
Quality of Life
Seventy-one (71) patients (63.96%) returned the SF-12 test at V4, and 15 (13.51%) at V8. Figure 7 shows the distribution of patients by ECOG15,16 and the results of the 1st question in the SF-12 test vs. ADH level at V4 and V8.

Figure 7 Distribution of population according to ECOG PS. ADH results vs. Question 1 in the SF-12 (quality of life).
In terms of the result of the 5th question, Were you limited in the kind of work or daily activities?: At V4, 34.30% of patients in the initiation subgroup needed to modify their daily activities vs. 21.4% in the continuation subgroup. At V8, 42.90% of patients in the initiation subgroup needed to modify their habits vs. 0% in the continuation subgroup. The difference was statistically significant (p = 0.016), applying Fisher statistical test.
Discussion
Even though an ADH close to 100% is assumed in
Oncology, data published in bibliography show that it is similar to that in other conditions, due the complexity and duration of treatments.1-4 In those studies published about adherence to capecitabine, there is a range from 75 to 91%; this is caused, among other things, by the effect of toxicities on patient attitude at the time of taking their medication1-5. Besides, a reduction has been observed over time, and it can go from 99% to 64% at 3 months2.
There are discrepancies about the minimum percentage of adherence to treatment for obtaining therapeutic efficacy. For Gordis et al (1969), the minimum was 75%; Ebrahim et al (1998) and Mayer et al (2009) considered 80% as “acceptable”. But for Patterson et al (2000) and Timmers et al (2014), 95% must be the minimum adherence value in order to obtain an optimal level of efficacy2,4. Low adherence has a direct impact on treatment efficacy, and is a critical factor at the time of achieving a favourable clinical response1,2,4,6.
In the studies published about ADH for oral antineoplastic agents, the ADH rates ranged between 40 and 100%, and 85% was considered acceptable2.
In our patient cohort, there was a 78.4% ADH rate, lower than the minimum established by Timmers et al.2,4, but similar to those collected in other studies1,3,5,7; it was superior in the initiation vs. the continuation subgroup. This difference can be due to the fact that patients initiating treatment are more motivated by their recent diagnosis and by the anxiety caused by lack of treatment, and appear very receptive to the oral and written information provided.
It is considered that even though cyclic dosing regimens, such as the one for capecitabine, are more complex than those with continuous dosing, more attention by the patient is required and ADH will be higher; however, this has not occurred in our study.
When treatment is prolonged over time, toxicities, disease evolution or treatment modifications can have a negative impact on motivation, and lead to ADH reduction2,3,4,6. This appears in the data obtained for the continuation subgroup in our study, where ADH was lower.
The majority of toxicities described by patients had reduced duration and intensity (except for tingling, fatigue, muscular pain and diarrhoea); these were the most frequent, and had an impact on quality of life. Even though there is a perception of good tolerability to treatment, the level of adherence in the initiation subgroup was always higher, possibly due to the cumulative toxicity of the treatment observed in the continuation subgroup and its impact on quality of life. On the other hand, even though G2-3 toxicities were more frequent in the initiation subgroup, ADH in these patients was always higher than in the continuation subgroup, probably due to closer monitoring at treatment initiation. It is important to monitor AEs adequately during treatment, due to their influence in the quality of life of patients and ADH.
Regarding quality of life, the majority of patients conducted their normal activities, and showed a favourable attitude towards treatment; fatigue was the most limiting factor, and the one with the highest impact on their mood.
Results are not very different between both subgroups in the emotional aspect, though they differ in the physical aspect. The percentage of patients who considered having a good health status was higher in the initiation subgroup; however, these patients stated a higher feeling of physical difficulty for daily life, possibly due to the limitations derived from therapy initiation, such as a regular administration schedule for capecitabine, hydration and diet requirements, and skin care.
A correlation was observed between quality of life and adherence. In patients with a positive assessment of their quality of life, ADH was superior, and it was higher in the initiation subgroup. Therefore, we could say that a quality of life perceived as good by our patients will encourage treatment adherence.
Patients in the continuation subgroup require a higher control in order to maintain an adequate adherence. Lack of treatment compliance can be due to disease evolution or deterioration in quality of life, which are parameters with impact on motivation11,12,13.
In conclusion, oral antineoplastic drugs present advantages for the oncological patient, but the development of toxicities and the alteration of quality of life can lead to a reduction in adherence. Even though the adherence to capecitabine is relatively high, there are an important number of patients with lack of adherence in the continuation subgroup; therefore, individualized follow-up is required, as well as implementing new strategies to encourage adherence, particularly in patients on treatments prolonged over time.











 texto en
texto en