Contribution to scientific literature
Drug Information Centres were created in 1962 in the United States, and since then they have provided information considered useful, timely, and with good quality. The use of new medicines and new ways of administration creates doubts in healthcare professionals, particularly in the hospital setting, and these can be solved by the DICs. Clinical practice is indirectly benefitted by DIC’s service whit the improvement of drug therapy.
Introduction
The term “drug information” may have different meanings, depending on the context where it is used. One of these meanings is the information on medicines provided by a professional with specific skills and functions, that can be printed or verbal1,2,3.
The International Pharmaceutical Federation (FIP) and the American Society of Health-System Pharmacists have stated that the pharmacist plays an essential role in the provision of reliable and genuine information to patients1,4. Taking into account the increasing access to on-line information by patients, the pharmacist can act as a guide and interpreter. The professional should be involved in public and private organizations that prepare and spread objective and genuine information on medicines for patients and healthcare professionals3. Governments should develop policies where pharmacists play a key role in the provision of information on medicines for patients, and encourage people to look for advice from their pharmacists regarding medicines and their use3,4.
Drug Information Centres (DICs) are defined as operational units that provide scientific-technical information on medicines in an objective and timely manner, representing an optimal strategy to address specific needs for information. For this aim, they have adequate sources and specifically qualified professionals, who will generate independent and adequate information for the questions presented or the needs identified1.
Rational Use of Medicines (RUM) can be defined as encouraging patients to receive the medicine adequate for their clinical needs, in the doses needed for their own individual requirements, during an adequate period of time, and at the lowest cost for them and their community.
Most of the information that can be accessed by prescribers comes from the pharmaceutical industry, which can be biased to economic interests; therefore, as a strategy to promote RUM, the World Health Organization advocates for the importance of evidence-based information without conflicts of interests being provided by Drug Information Centres and by independent drug bulletins5.
The Brazilian Drug Information Centre (Cebrim/FCP) was created through a proposal by the Federal Council of Pharmacy (FCP) for the implementation of a DIC in the national setting in 1989, with the objective of changing the course of pharmacists professionals and use of medicines in Brazil. The creation of this centre was made official on March 22nd 1996, through the publication of the FCP Resolution No. 285, which approved the new administration and staff structure of the Federal Council of Pharmacy, including the Cebrim/FCP as a information section6. The task of Cebrim/FCP is to provide information on medicines, based on the best evidence, to healthcare professionals, as well as to promote safe and effective therapeutical practices, with the best cost-benefit for the community6.
This study intends to define the activities by the Cebrim/FCP Drug Information Centre, to describe passive information when dealing with the demand by healthcare professionals, and the satisfaction by users regarding the service provided.
Material and methods
Reactive Information
The centre has a computerized system for recording and storing those questions and their answers: the Sistema de Informação Farmacoterapêutica (SIFAR) (Pharmacotherapeutical Information System). This system generates management reports, which were collected during the period from January 1st, 2010 to December 31st, 2015, and were analyzed in this study. For this aim, it was necessary to describe the information activities by Cebrim/FCP, to quantify of questions received, to identify the profiles of users and professionals within the SIFAR, to determine the subjects most frequently requested by users, the time until the response, the most widely used information sources, and also to describe user satisfaction regarding the answers provided by Cebrim/FCP.
To calculate the mean of questions received per year in the centre, weekends and holidays were not considered, because the centre has no work hours for those periods. Thus, 252 working days per year were considered for 2010, 2011 and 2012, 255 working days in total for 2013 and 2014, and 251 working days in total for 2015. The profile and profession of the users are accounted through the initial record by the user when they first access the on-line system. The computerized system also allows records of patients, health organizations, and other DICs. These data generate reports for analysis of the type and frequency of the different professionals with the highest number of consultations, as well as the percentage of patients who are users of the centre.
The topics requested refer to the main subject of the question received. In cases with more than one subject, both will be selected and recorded in the computerized system; these data will generate management information for the analysis and determination of the most frequent topics of consultation.
The system registers the time of response by the centre, considered from the time and date when the user sent the question until the time and date when the centre sent the answer. These data generate the calculation of the total time used for preparing the answer, that is presented in five cathegories: <59 minutes, from 1 to 5 hours, from 6 to 23 hours, and more than 3 days. The system considers and includes weekends.
The system records and enters the information sources consulted for the preparation of each answer provided. A report is generated that allows to classify information sources into primary, secondary and tertiary, as well as to determine the frequency of use for each source.
Satisfaction by the user
In 2010, the assessment of satisfaction was conducted through a pilot study where a questionnaire was applied to a random sample of users; this was sent and answered by e-mail. These results were not considered for this analysis, due to the different methodologies used.
From 2011 onwards, the evaluation of answers by the users has been conducted through an on-line computerized system for immediate and voluntary scoring of the answer provided. The user assesses the answer from Cebrim/FCP using a 5-score Likert-type scale, where 1 is the worst score and 5 is the best score. This annual analysis included only those information requests (IRs) that included patients, and out of these, the ones that were assessed by the users, per year.
Results
Service description
Cebrim/FCP is a technical area of the Brazilian Federal Council of Pharmacy, acting independently and answering IR from all over the country, essentially by pharmacists. The working hours of the centre are from 8 a.m. to 7 p.m., from Monday to Friday. There are three pharmacists working full-time, as well as interns from the Pharmacy Course. The centre has an adequate physical setting, equipped with computers and updated sources of information, according to the criteria by FIP for Drug Information Centres7 and the recommendations by Silva and Saavedra (2014) regarding the skills and competence required for informing professionals 8. All information requests from Cebrim/FCP is peer reviewed (100%); that is to say, one pharmacist prepare and another pharmacist review the answer.
Reactive information
The number of IRs answered per day during the six years analyzed increased from 2010 to 2013, from 1.7 to 3.0 questions in 2013 per working day. Meanwhile, in the following years, there was a reduction in the mean number of requests/ working day, to 2.7 and 2.3 respectively. The mean of IRs during the period of the study was of 639 questions (Table 1).
Table 1 Description of the passive reactive information service by CEBRIM/FCP in the period from 2010 to 2015

The analysis of the profile of CEBRIM/FCP users highlighted pharmacists and undergraduate Pharmacy students, with >91% of the consultations conducted during this period; and some of the most frequent subjects in the requests to CEBRIM/FCP included IRs on administration and way of use, indication, drug interactions, and legislation; these were presented at high rates. The most recurrent subjects are presented in Table 1.
The results regarding the time spent to prepare the answer and send it to the users was <24 hours in all years for approximately 50% of IRs ; the proportions per year were: 52.4% in 2011, 78.9% in 2012, 76.6% in 2013, 62.2% in 2014 and 55.9% in 2015. There was an exception in 2010, where only 42.5% of IRs were answered in <24 hours.
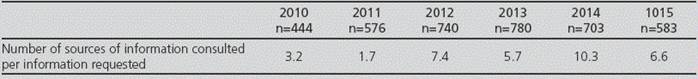
Most of the IRs received by CEBRIM/FCP were answered with tertiary sources (53.5%). For approximately 30% of the answers, the secondary sources available were consulted; and for 1.5% to 6% of the IRs, primary sources were required, due to question complexity (Table 1). The relation between the total number of bibliographic sources used for answer preparation and the total number of questions answered per year shows that in 2010 and 2011, the mean for sources used was 2.45, and from 2012 onwards, the mean for sources used increased up to 7.5 (Table 2).
User Satisfaction
From 2011 to 2015, the answers were assessed by a mean 44% of the total number of users of the service. Overall, from those who assessed the answers, almost 80% were satisfied or very satisfied with the service (Table 3).
Discussion
Cebrim/FCP presented a variable number of IRs per year (annual mean =639) and their main users were pharmacists and undergraduate students. The most recurrent questions were about administration and way to use the medicines.
In this study, the results showed an increase in the annual mean number of IRs in 2010 and 2013; this can be explained by the promotion of the service conducted through presentations, and the distribution of disclosure materials, conducted by the team of pharmacists for healthcare professionals. A similar outcome was reported by Oliveira et al. , who showed the evolution of the number of questions for the Unipar Centre, starting with 48 IRs/year in 1996 and going up to 959 IRs/year in 2009; this is considered the outcome of a serious and responsible task9. The outcomes by Sessa et al. showed a variable number of IRs, initially 645 in 1999 up to a constant 300 IRs per year, which continued until 2010 without any alterations10. A community information centre analyzed five years of production of IRs, and their findings were a mean of 816 IRs per year11.
In a different way, a hospital centre received an average 1,160 questions per year from those healthcare professionals working in the hospital12. And an information centre acting in the Regional Health Ministry of the Bahia State in Brazil assessed their production of IRs during a period of 12 years of action; their outcomes were different, with an approximate mean of 300 IRs per year13. The different results from these two centres could be explained through the different institutional nature of each of them, a hospital and a public institution, both with action particularly targeted to their internal population.
Regarding the profile of users, these results were expected, because these professionals are workers in the public system object of the actions by the Federal Council of Pharmacy, an organization including CEBRIM/FCP. Pharmacy students are also recurrent users of this type of service; thus, a positive outcome is achieved regarding one of the objectives of the centre, which is disclosure and closeness with universities (Table 1). Veber et al. reached similar outcomes in a study conducted at a University DIC; their analysis of the profile of IRs revealed that most consultations were conducted by pharmacists from their own institution and working in the town cared for by the DIC14. In a DIC linked to a hospital, it was frequent to observe a high number of requests made by nurses (30 to 40%) and physicians (11 to 17%), besides pharmacists12. Results achieved by Santos et al. (2011) confirmed the importance of pharmacist action for providing information on medicines in clinical practice in the hospital setting.
The most frequent subjects found in this study were also the most common reported by Sessa et al. , with IRs about indication in 20.5% of the questions received per year, identification in 13.9%/year of aspects regarding stability and storage in 12.7%/year of questions10. Sartori et al. reported that the most requested subjects in a university centre were: indications of use, identification, stability, and legislation15.
Even though the records of DIC users cannot be considered representative of overall healthcare professionals, a periodical analysis of frequent subjects can point out at any latent problems and the information needs by the community in the healthcare area 16.
CEBRIM/FCP is located in the Federal Capital of the country, very close to health institutions and the regulatory agency; for this reason, there are frequent questions about legislation (from 6 to 8% of the IRs per year), which are observed in the outcomes found. During the years of the study, there were many changes in the Brazilian Pharmaceutical Legislation, which could justify the increase in questions on this matter. For example, the approval of the Collegiate Board Resolution RDC 20/2011 by Anvisa on antibiotics, the RDC 13/2013 published by Anvisa, which determines the Good Practices for Manufacturing (GPM) of traditional phytotherapy products, and the Resolutions RES No. 585/2013 and 586/2013 by the Federal Council of Pharmacy on clinical responsibilities of pharmacists.
There were variations in the time of response to IRs. The reduction in the demand for reactive information can be explained by an increase in the time of response by the centre. Besides, CEBRIM/FCP underwent a reorganization process, with a reduction in the staff of pharmacists, and only one pharmacist with full-time commitment to reactive information. According to Reppe et al. (2014), the number of sources consulted was one of the main factors that take time during the preparation of answers17, which could confirm the outcomes reached in this study (Table 1).
Similar results were reported by Santos et al. (2011) for a study in a hospital DIC, where 76% of users considered that their question had been answered in due time12. An external committee for assessment of American DICs considered in their study the criteria of timeliness and punctuality as the time of response offered in <24 hours, or less than the interval specified by the user18. The quality of service can be measured by recording the time of answer used by the centre and the level of satisfaction by the user, which includes the perception by the user regarding the answer being sent in due time19.
The frequent use of tertiary bibliographic sources, even though considered reliable, can present outdated information, which suggests the low complexity of the questions asked in Cebrim/FCP. Similar outcomes were reached by Santos et al. (2011), with a great use of tertiary sources for answering the questions, with 1.3 to 0.4 tertiary sources used per IRs; the frequency of use of secondary sources was modified from 0.08 to 0.05 in the same period, and regarding the use of primary sources of information, the authors reported from 0.03 to 0.01 sources per answer prepared12.
According to the guidelines reported by Vacca et al. (2012), good-quality information is representative when it presents >2 references20. This standard was maintained in CEBRIM/FCP during the years studied. However, 2011 was an atypical year in CEBRIM/FCP due to the exit of pharmacists who provided the service; and this fact was reflected in the service provided, and therefore that year presented results below what was established by the Vacca guidelines and the performance in the other years of the study; the answers provided by CEBRIM/FCP presented a median number of sources consulted according to the international standard.
Regarding user satisfaction, the outcomes by CEBRIM/FCP were different to those by Santos et al. (2011), which showed that 46% of users considered that the work conducted was optimal12. Fischer et al. (2012) consulted their users about the answers received, and found that 94.5% of users declared that they were satisfied with the ease of access, 88.1% with the quality of the information, and all subjects declared that they would consult the centre again21.
Programs for monitoring the activities by information centres allow to inform, among other aspects, about the impact of the information provided, such as therapeutical changes, changes in prescription patterns, and user satisfaction19. Besides, satisfaction surveys conducted among the users of Drug Information Centres are recommended by government health institutions and pharmaceutical societies, which look for and recommend 100% of user satisfaction 22.
It is observed that the level of satisfaction by the users of CEBRIM/FCP did not experience any alterations during the years when the system of assessment was implemented, which suggests a continued quality of the service provided by the centre; however, it shows the need for actions of constant improvement in order to reach the international standards determined. The on-line assessment of satisfaction by the user allows having a larger sample of usersensures the willingness and impartiality of the evaluation, and therefore, a real overview of the opinion of the user about the service provided.
Conclusions
CEBRIM/FCP addresses a stable number of information request, mostly by pharmacists, about pharmaceutical data, including subjects such as administration and way of use of medicines, indication, and drug interactions, among others. Regarding the time of response to IRs, between 41% and 78% of questions were answered within less than 24 hours. Most of the information sources consulted in order to prepare the answers were tertiary, and secondary to a lower extent. Almost 80% of the users who evaluated the answers provided declared that they were satisfied or very satisfied with the service. The Drug Information Centre by the Federal Council of Pharmacy has contributed directly and indirectly to a better pharmacotherapy and rational use of medicines.











 text in
text in