Introduction
Drug-on-drug interactions (DDI) are becoming increasingly relevant, due to the growing variety of drugs available and the increased life expectancy of the general population. There is a higher risk of onset of adverse effects caused by DDIs, and of decreases or increases in drug activity caused by interactions, which could compromise or increase the effectiveness of treatment. It is crucial to identify, prevent, and treat DDIs, particularly those considered clinically relevant1,2.
Several studies on DDIs have already been conducted in hospital settings and in primary care. Among other findings, these studies have described DDI onset in approximately 40% of patients3-5, DDIs associated with adverse drug reactions (ADRs4) (14%), hospital admissions for ADRs due to DDIs6 (10%), and fatal events associated with DDIs7 (6%).
The risk of DDIs is particularly high in oncological patients because they receive antineoplastic agents with supportive treatment, in combination with other drugs to treat comorbidities and cancer-related syndromes, such as pain and depression8,9. Moreover, they usually receive drugs with a narrow therapeutic index, and experience organic deterioration due to the underlying pathology and ageing. Such deterioration may affect drug metabolism and renal excretion. In the setting of onco-hematology, many of the effects of DDIs are not recognized as such, either because they are masked by the symptoms of the pathology, or because they overlap with the inherent toxicity of the treatment8.
Studies have investigated the epidemiology of potential DDIs in onco-hematological patients in diverse clinical settings and have obtained disparate results10-15. Riechelmann et al. described DDI rates of 27% to 63%, of which 69% to 88% were moderate or severe. The most common DDIs involved supportive treatment or treatment for comorbidities10-(12. Tavakoli et al.13 reported a lower prevalence of DDIs in cancer patients (31.1%) than in hematologic patients (54.1%), whereas Hadjibabaie et al.15 found a prevalence of 62.9% in the adult and pediatric hematologic population.
Previous studies conducted in Spain show that the prevalence of potential DDIs (32.6%-81.0%) in adult and pediatric onco-hematological inpatients strongly varies according to the population and database used16,17. The drugs most often involved in DDIs included CNS depressants, antiemetics, immunosuppressants, and azole antifungals. The lack of agreement between different databases is an additional challenge when identifying and evaluating possible DDIs18.
Knowledge and the proper management of drug interactions can improve the safety and effectiveness of treatments. The studies conducted in this field have used different methodologies. Therefore, there is a lack of clear definitions of the incidence of interactions, their severity, and risk reduction strategies8. In addition, interaction profiles can vary according to the standard prescribing practices used in different settings. Therefore, effective action plans to minimize DDIs can be developed by identifying the interaction profiles in the work setting. Using a well-known database, the aim of this study was to determine the prevalence of clinically relevant potential DDIs in adult oncology inpatients, and to describe the most common interactions.
Methods
We conducted an observational cross-sectional descriptive study of potential drug interactions detected during the medical treatment of oncological inpatients. The study included all treatments received by adult patients admitted to the Oncology Service in an 860-bed university clinical hospital.
The study period was 4 weeks (February 15-March 14, 2016). All medical treatments were recorded twice a week (Monday and Wednesday), except during holidays, in which case active prescriptions were recorded the next working day. The number of treatments per day was initially estimated to be 25 to 30, comprising a total of 200 to 240 treatments for analysis.
E-prescribing software (Silicon®) and the electronic medical record (Selene®) were used to collect demographic and clinical data: age, sex, patient comorbidities, and tumor location.
The Lexi-Interact®/Lexicomp®(19 ((online)) database was used in this study, as it meets the minimum criteria established by Rodríguez-Terol et al. It is a publicly accessible international database, which is well-known to health professionals and has been cited in different studies20.
Each prescribed medication was recorded according to its active ingredient and subsequently evaluated. If a drug contained 2 or more active ingredients, each active ingredient was separately evaluated. The anatomical-therapeutic-chemical (ATC) classification of drugs was used to classify active ingredients, including the chemical subgroup, into therapeutic groups.
We recorded the total number of drugs prescribed for each treatment, disaggregating the number of antineoplastic and non-antineoplastic drugs. Antineoplastic agents or drugs were defined as those used for the treatment of malignant cancer, regardless of its mechanism of action.
In line with the methodology previously described by Smithburger et al.21 and adapted by Fernández de Palencia et al.16,17, all patients in the census were considered to be new patients for each day of the study. All prescription lines were checked in the Lexicomp® database and all drug interactions detected were recorded in pairs, indicating the degree of severity and level of evidence assigned by the database, the mechanism of interaction, the description of the potential effect, and whether the interacting drug pair were both antineoplastic agents, an antineoplastic with other medication, or both were general medications. Any active ingredients not included in the database were excluded from the descriptive analysis.
All potential interactions classified by the database with a level of risk C, D, or X were considered clinically relevant, regardless of their degree of severity and level of evidence or documentation (Table 1)19. The degree of severity and level of evidence or documentation refers to each pair of interacting drugs studied individually.
A descriptive analysis was conducted of the demographic and clinical data, the drugs most commonly prescribed in the analyzed treatments, and the 20 most prevalent drug interactions detected by the database. The analysis was conducted using the SPSS 20.0 software package for Windows®. Continuous variables and variables that showed a normal distribution after applying the Kolmogorov-Smirnov test are expressed as mean and standard deviation. Variables that did not show a normal distribution are expressed as median and interquartile range (25th percentile-75th percentile). Qualitative variables are expressed as absolute frequency and relative frequency in percentages.
The epidemiological characteristics of drug interactions were analyzed by determining the prevalence of treatments that involved some type of interaction (total and by level of risk):
Results
We analyzed 218 medical treatments, each of which was considered as a patient, whose main characteristics were: 61.5% males, mean age of 63.4 ± 14.6 years; Most of the patients were pluripatológicos, being the most prevalent comorbidity hypertension, presenting in a 35.3% of the cases, followed by diabetes mellitus type II in 26.1% and dyslipidemia in 20.2%. The most frequent tumour location was lung (21.1%), followed by colorectal (14.6%) and breast (9.2%).
The median number of drugs tested per treatment was 9 (6-12). A total of 2069 drugs were prescribed, of which 27 were antineoplastics. In total, 84.9% of the treatments were composed of 5 or more drugs. Table 2 shows the 20 most commonly prescribed drugs (71.2% of the total).
The most commonly used antineoplastic drugs were fluorouracil (n = 5) and etoposide (n = 4), followed by carboplatin (n = 3) and cisplatin (n = 2); other antineoplastic agents were prescribed once each during the study period: docetaxel, paclitaxel, gemcitabine, methotrexate, bevacizumab, cetuximab, and exemestane.
Of the 218 medical treatments analyzed, only 11 (5.0%) did not involve DDIs. Of the remaining 207 treatments, there were 1850 potential DDIs, which were grouped into 378 drug pairs. The median number of DDIs per treatment relative to the total was 6 (3-12). Of all DDIs detected, 1675 were classified as level of risk C (90.5%), 95 as level D (5.1%) and 80 as level X (4.3%).
Table 3 shows the 20 pairs of DDIs most commonly detected in relation to the total (37.2%). They are shown in descending order of onset frequency, percentage, level of risk, degree of severity, level of evidence, the mechanism of action, and the description of the potential effect.
Table 3 The most commonly detected pharmacological interactions. Risk, severity, and reliability rating. Description and mechanism.

Severity Rating: 1: Mild; 2: Moderate; 3: Severe
Reliability Rating: 1: Poor; 2: Fair; 3: Good; 4: Excellent
Mechanism of action: 0: Unknown; 1: Pharmacokinetic; 2: Pharmacodynamic; 3: Pharmacokinetic and Pharmacodynamic.
The drugs most commonly involved in DDIs were opioid analgesics, antipsychotics (especially butyrophenones), benzodiazepines, pyrazolones, followed by glucocorticoids and heparins.
Of the 1850 DDIs detected, only 10 DDIs involved antineoplastics, and only 1 DDI involved a pair of antineoplastics (paclitaxel and carboplatin) (see Table 4). Paclitaxel was the drug most commonly associated with interactions with non-antineoplastic drugs, specifically with nebivolol, atorvastatin, and doxazosin.
Table 4 The most common pharmacological interactions involving an antineoplastic agent. Risk, severity, and reliability ratings. Description and mechanism.
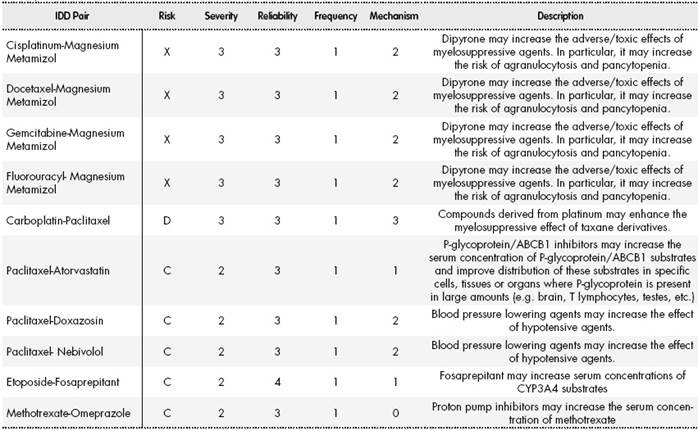
Severity Rating: 1: Mild; 2: Moderate; 3: Severe
Reliability Rating: 1: Poor; 2: Fair; 3: Good; 4: Excellent
Mechanism of action: 0: Unknown, 1: Pharmacokinetic; 2: Pharmacodynamic; 3: Pharmacokinetic and Pharmacodynamic.
In addition to the DDIs shown in Table 3 and Table 4, other relevant DDIs include: Level X: Haloperidol combined with tiotropium bromide (risk of anticholinergic symptoms) or domperidone (QT interval prolongation); ipratropium with butylscopolamine or dexchlorpheniramine (anticholinergic syndrome); linezolid with morphine (increased morphine toxicity) or metamizole (increased risk of myelosuppression); metoclopramide with quetiapine (increased antipsychotic toxicity); and clopidogrel with omeprazole (decreased effect of clopidogrel).
Level D (among others): Metoclopramide combined with desvenlafaxine, paroxetine, sertraline, or amitriptyline; haloperidol with levofloxacin, ondansetron, paroxetine, or zolpidem; metamizole with aspirin, furosemide, torasemide, or paroxetine.
The main mechanisms of interaction in the 20 most prevalent DDIs were pharmacodynamic factors (70.0%), followed by pharmacokinetic factors (15.0%).
Treatments with at least 1 clinically relevant DDI had a prevalence of 95%; of these, 94.5% were level C, 26.1% were level D, and 26.1% were level X.
Discussion
This observational study adds to the body of knowledge on the epidemiology and potential severity of DDIs in oncology inpatients. The prevalence of DDIs was very high (95% of all interactions, of which 26.1% were level X interactions).
It is difficult to compare the results with those of other studies, because of the different methodologies used and different settings analyzed. The scientific literature contains studies on DDIs in adult oncology patients in hospital and outpatient settings. However, the clinical settings are not comparable and the studies address specific situations: hospitalized patients who do not receive chemotherapy12, or who do receive chemotherapy13, or who only receive supportive palliative treatment10; and outpatients treated with standard chemotherapy11,14 or treated with oral antineoplastics22,23. In contrast to these studies, the present study addresses the typical situation of oncological inpatients, regardless of the reason for their admission to hospital and the treatment administered.
In addition, different authors have used different methodologies. In the present study, each new treatment was considered as a different patient. This methodology was based on that previously described by Smithburger et al.21 and Fernández de Palencia et al.16,17,24. The majority of other studies on oncology patients used different methodologies, which analyzed overall treatment and provided DDI data relative to the patients evaluated.
The database used also had a relevant influence on the results. Fernández de Palencia et al. studied the onset of potential interactions in the same setting and used the same methodology as that of the present study; however, they used the Micromedex® (MMX) and Drug Interaction Facts® (DIF) databases17. These authors found significant differences between these databases in the prevalence of DDIs during treatment: the MMX database showed a prevalence of 81%, whereas the DIF database showed a prevalence of only 32.6%17. It should be noted that, for the same population and setting, the Lexicomp® database showed a prevalence of 95%, which was higher than that previously described. This prevalence was also higher than that observed in adult hematological patients (74.1% with MMX; 56.8% with DIF);24 the difference was even more marked in relation to the pediatric onco-hematological population (44.7% with MMX; 51.3% with DIF)16.
Most of the detected DDIs involved supportive drugs and drugs targeting comorbidities, with little involvement of antineoplastic agents (<1%). This result was influenced by the study setting, which involved the low use of antineoplastic agents during patient admission and a population mainly composed of patients hospitalized for complications derived from the disease or its treatment, or of patients in the terminal phase. Nonetheless, a similar trend has been previously described, even in active treatment settings11,14. For example, Riechelmann et al.11 found that approximately 13% of DDIs involved antineoplastic agents vs 87% with non-antineoplastic agents.
The prescriptions profile matched that previously described for the same population, with similarities between the most commonly prescribed drugs and between the number of drugs per treatment (9 in the present study, 11 in the study by Fernández de Palencia17). However, interaction profiles vary considerably according to the database used: for example, there was no match whatsoever between the MMX and DIF databases18.
A comparison of the DDIs detected in this study using the Lexicomp® database and the studies by Fernández de Palencia using the MMX and DIF databases17 shows that:
- Regarding the most prevalent DDIs, the MMX database had the highest level of agreement with the Lexicomp® database, both of which describe benzodiazepine interactions with opioids. However, at the time of this analysis, Lexicomp® classified these DDIs as level C (moderate), whereas the MMX database classified them as severe. Both databases consider the association between haloperidol and metoclopramide to be a DDI, and thus this association is contraindicated in both databases. The MMX database showed a prevalence of 81%, which is similar to that observed with Lexicomp®. Both databases identified pharmacodynamic factors as the main mechanism of action underlying the detected DDIs (63%-70%).
- In contrast, marked differences were found between these databases and the DIF database in relation to prevalence (32.6%), and there was little agreement on the most commonly detected DDIs. The DIF database identified pharmacokinetic factors (48.3%) as the predominant mechanism of action underlying the DDIs.
- A striking difference between databases was the identification of several DDIs commonly involving metamizole. Neither the MMX nor the DIF databases include this drug, which is not marketed in the US. Its inclusion is relevant because it is widely used in Spain and could have contributed to the higher prevalence of DDIs detected in the present study. We draw attention to the DDIs between metamizole and the antineoplastic agents used during the study. This DDI is classified as level of risk X because of the increased risk of spinal aplasia and agranulocytosis; therefore, alternative analgesics should be considered during active treatment.
The following potential DDIs between CNS depressants should be noted, because of their synergistic effect: the increased risk of anticholinergic syndrome when using ipratropium or tiotropium with haloperidol, butylscopolamine or dexchlorpheniramine; the increased risk of toxicity when using antipsychotics and antidepressants with metoclopramide; and multiple interactions with metamizole.
Differences were found between the results of the present study and those of previous studies on the hematological and pediatric population (pediatric onco-hematology). Differences were found between the profiles of the most commonly described DDIs, and between the profiles of the most commonly prescribed drugs; in particular, there was greater use of immuno-suppressive agents and azole antifungal agents, which were the drugs most commonly involved in potential interactions16,24.
The results of the study further corroborate the difficulty of assessing DDIs in clinical practice, in terms of their potential severity and their effects on therapy. The results also confirm the need to use and compare different databases for decision making.
There was a very high prevalence of treatments with clinically relevant DDIs. Those with level of risk X (26%) are of particular concern.
Due to their frequency of onset and potential severity, we highlight the risk of respiratory depression with the concomitant use of CNS depressants, the risk of onset of anticholinergic symptoms when combining morphine or haloperidol with butylscopolamine, ipratropium bromide or dexchlorpheniramine, and multiple interactions involving metamizole.
Contribution to the scientific literatura
The present study adds to the body of knowledge on the epidemiology and potential severity of drug-drug interactions in oncology inpatients. It further corroborates the difficulty of assessing interactions in clinical practice regarding their potential severity and effects on therapy. The study also confirms the need to use and compare different databases in order to improve decision making











 texto en
texto en 




