Introduction
The management of complex chronic patients poses a significant challenge. As the needs of such patients change over time, a continuous reassessment and a close monitoring of their condition are essential. To make things even more challenging, these patients are usually required to resort to different levels in the healthcare system1. The term complex chronic patient is frequently related to the image of an elderly patient with multiple long-standing conditions. However, other kinds of patients also fall into the complex category. Patients who undergo allogeneic haematopoietic stem cell transplantation (allo HSCT), for example, comprise a group of highly complex haematologic patients given the large amount of drugs included in their therapeutic regimen, the need to continuously monitor their evolution; and their predisposition to complications such as graft-versus-host disease (GvHD), infection, and failure to recover normal peripheral circulation levels (graft failure), which may require hospitaliszation.
Although pharmacists have for many years now been part of the multidisciplinary team involved in haematopoietic stem cell transplantation (HSCT), their training and specific role, as well as their responsibilities within the team, were only defined in 2016, when the Pharmacy Special Interest Group of the American Society for Blood and Marrow Transplantation (ASBMT) drew up the HCT Clinical Pharmacist Role Description Statement in order to specify the duties of the specialist pharmacist in the multidisciplinary care of allo HSCT patients2. More recently, the European Society for Blood and Marrow Transplantation (EBMT) published the first European recommendations3. Both documents discuss the positive influence that pharmacists can exert on therapeutic adherence.
There are hardly any studies in the literature that evaluate the positive influence of pharmacists on the therapeutic adherence of HSCT patients4,5. In such patients, lack of adherence or incorrect administration of medication may triggerlead to serious complications and adverse events that could even result in hospital admission (on some occasions in the intensive care unit)6.
Data available from Europe are too scarce to serve as a foundation for a pharmaceutical care program for HSCT patients. The present study is based on the hypothesis that therapeutic adherence in allo HSCT patients may be compromised by the complexity of their condition and could influence the clinical outcomes of these patients as adherence tends to be related to the onset of complications and the incidence of readmissions. Thus, the main purpose of this study is to measure the adherence to prophylaxis against graft faillure, to prophylaxis against GvHD and to prophylaxis against infections seen in patients undergoing allo HSCT. The study also analyses the incidence of acute complications and the readmission rates based on adherence.
Methods
This is a retrospective observational study conducted in a third-level hospital. The data were obtained from the patients' electronic clinical records and from the Pharmacy Department's medication management system. All adult patients in the Haematology Department undergo to allo HSCT between May 2017 and May 2018 were included in the study. The study variables were evaluated between days 0 and +100 (considered to be the acute post-engraftment phase) where monitoring the occurence of early complications is crucial.
Data were collected on the patients' age, sex, baseline condition, source of stem cells (bone marrow or peripheral blood), conditioning regimen intensity (myeloablative or reduced intensity), major histocompatibility complex (human leukocyte antigen (HLA)) compatibility, donor's sex, and on the prophylactic drug therapy administered. An assessment was made of adherence to cyclosporine, tacrolimus and sirolimus prophylaxis against graft failure; cyclosporine, tacrolimus and sirolimus (associated or not to mycophenolate mofetil (MMF)) prophylaxis against GvHD; and, lastly, prophylaxis against infections (posaconazole or voriconazole prophylaxis for fungal infections and valganciclovir prophylaxis for viral infections).
Whenever possible, adherence was evaluated using the Pharmacy Department's dispensation records. For cyclosporine, adherence was determined by a direct measurement of the serum concentration of the drug, which is typically dispensed to allo HSCT patients at the Community Pharmacy although the diagnosis of HSCT-related conditions is typically made in the hospital setting.
As they were prescribed off-label, MMF, sirolimus and tacrolimus were dispensed by the hospital Pharmacy Department. Most of the patients on sirolimus or tacrolimus collected the drug at their referral hospital, hence their adherence was evaluated (as was also the case with cyclosporine) from the drugs' serum levels.
The fact that adherence was determined based on the dispensation records followed the premise that patients cannot take medication that is not dispensed to them, but also patients take the medication they have been dispensed in an appropriate manner. Dispensation records and reports on the amounts of medication dispensed at different dates during the study period were processed using the Paciwin® outpatient management module, which was used to calculate the subjects' adherence rate.
Mean adherence during the dispensation period (MDP) was defined as the proportion of days a patient had enough medication to take 100% of the doses prescribed. It was calculated with the equation shown in Figure 1.
The dispensation period was composed of the amount of days on which the medication was dispensed, from the first to the previous to the last dispensation day. The number of days between dispensations is the total number of days elapsed between the first and the last dispensation. For a patient to be defined as adherent they had to reach a total adherence rate ≥ 95%7,8.
To evaluate adherence to cyclosporine, tacrolimus and sirolimus based on serum concentrations, a series of threshold levels were established. Patients had to stay above those levels in order to be classified as adherent, unless there was some reason (according to the Haematology Department) that justified (i.e., drug-to-drug interactions or impaired absorption). For cyclosporine, for which optimal concentration levels following allo HSCT ranged between 150 a 250 ng/mL, the threshold was set at 100 ng/mL, considering the drug's short half-life (6.3-20.4 hours) and that a drop of 50 ng/mL below the target level, in the absence of clinical justification, may be considered an alarming signal of poor adherence. The threshold was set at 5 ng/mL for tacrolimus and at 6 ng/mL for sirolimus9. The incidence of acute complications resulting from allo HSCT was studied in three different contexts. Firstly, secondary graft failure, defined as the loss of at least two cell lines in patients who had achieved haematopoietic engraftment (in the absence of other complications that might have been responsible for such a loss). Secondly, acute GvHD (aGvHD) which, according to the criteria defined by the International Bone Marrow Transplantation Registry (IBMTR), refers to the appearance of GvHD between days 0 and
+100 post-engraftment10. Lastly, infection, defined as fever above 38 °C or evidence of infection confirmed by a positive culture or histology from one or more sterile sites, including the bloodstream.
The readmission rate due to such complications was calculated for days 0 to +100 (Figure 2). The readmission rate was defined as the percentage of admissions occurring during the study period after discharge of the engrafted patients. Any unexpected admission following a previous discharge from the same hospital was considered a readmission. The numerator of the readmission rate was the number of discharges among patients readmitted within ≤ 100 days of their previous discharge. The denominator was the number of patients discharged over the study period, excluding patients who passed away during that time.
A comparison was made between the incidence of complications and the readmission rates of adherent and non-adherent patients.
To quantify the association between adherence and the incidence of complications, an estimation was made using the odds ratio (OR) with a confidence interval of 95% (CI95%). The same calculation was used to quantify the association between adherence and readmission rates. The SPSS Statistics® (v21) package was used for statistical analysis .
The study was evaluated and approved by the Research Ethics' Committee of our hospital in June 2018.
Results
A total of 46 patients were included in the study, 69.6% (n = 32) of whom were male. Mean age was 47.7 ± 12.1 years. The demographic and clinical variables analised are detailed in Table 1, along with all the different drug combinations used as prophylaxis against graft failure and against GvHD, depending on the conditioning protocols defined for each patient.
Table 1. Descriptive analysis of the demographic and clinical variables of the sample
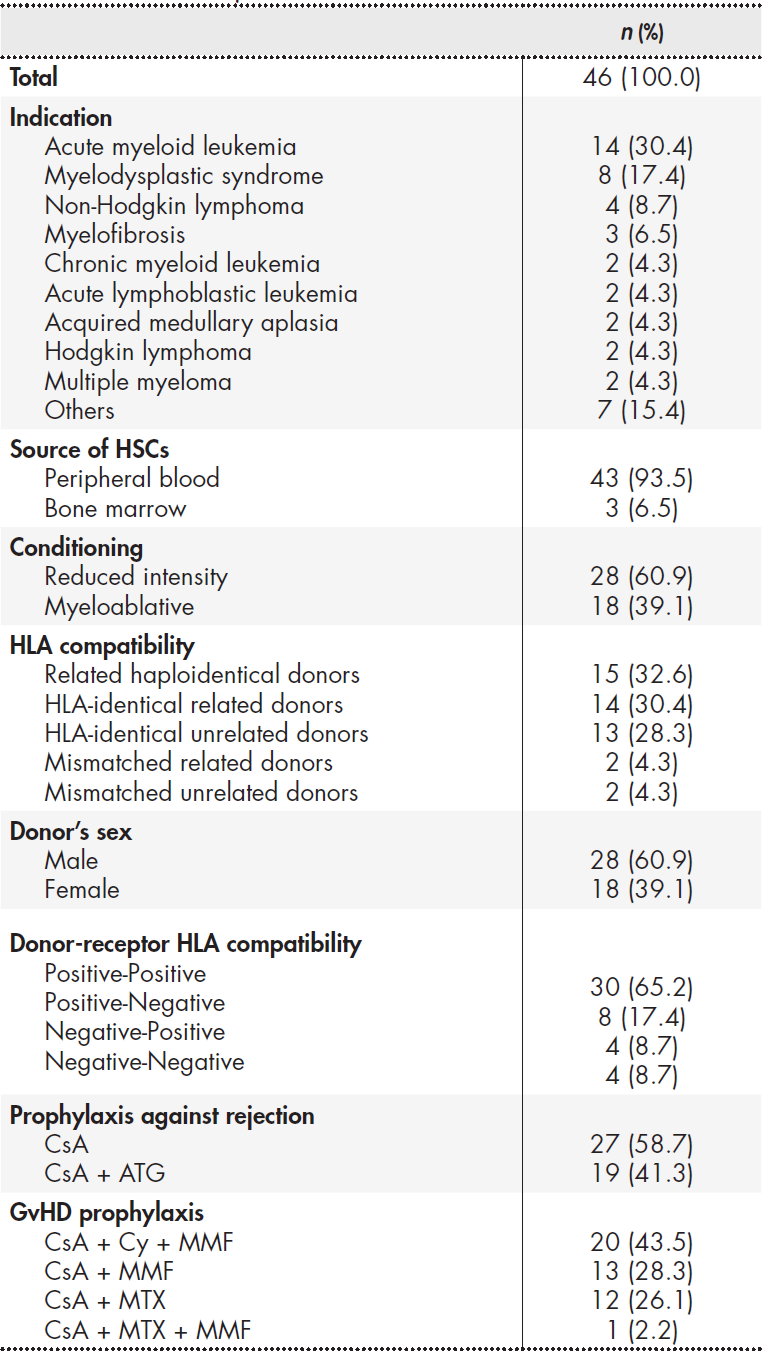
ATG: antithymocite globulin; CsA: cyclosporine A; Cy: cyclophosphamide; GvHD: graft-versus-host disease; HLA: human leukocyte antigen; HSCs: haematopoietic stem cells; MMF: mycophenolate mofetil; MTX: metothrexate. Drugs were administered orally wherever possible, except for ATG, Cy and MTX, which were administered intravenously.
All the patients were started on cyclosporine for prevention of graft failure and GvHD, although four of them (8.7%) were switched to a different immunosuppressant due to toxicity issues: three (6.5%) were switched to sirolimus and one (2.2%) to tacrolimus. A total of 73.9% (n = 34) of patients also received MMF as prophylaxis against GvHD, of whom 41.3% (n = 19) were still on the drug at discharge.
Initial anti-infection prophylaxis was administered in all cases following a typical post-engraftment protocol based on fluconazole, acyclovir and trimethoprim/ sulfamethoxazole. On discharge, 41.3% (n = 19) of patients received at least one of the studied drugs as secondary prophylaxis. A total of 28.3% of patients (n = 13) were administered antifungal prophylaxis on discharge (posaconazole in 61.5% of patients, voriconazole in 30.8% and both drugs in 7.7%). Ten patients (21.7%) received antiviral prophylaxis with valganciclovir.
The mean hospital stay for performing the allo HSCT procedure was 33 ± 19.4 days.
Adherence
Globally, 82.6% (n = 38) of patients were adherent to prophylaxis against graft failure, of which 84.8% were adherent to cyclosporine prophylaxis, 100.0% to sirolimus prophylaxis whereas 0% to tacrolimus prophylaxis. Thirty-seven patients (80.4%) were considered adherent to prophylaxis against GvHD (84.8% to cyclosporine prophylaxis, 100% to sirolimus prophylaxis, 0% to tacrolimus prophylaxis and 84.2% to MMF prophylaxis). It should be mentioned that the only patient on tacrolimus, as well as two of the patients on sirolimus, collected their drug from their referral hospital, other than ours. The only patient on sirolimus whose dispensation records were available was found to be adherent both on the basis of serum concentrations and the frequency of drug collection from the Pharmacy Department. All patients complied with their anti-infection prophylaxis regimen.
Complications
No graft failure was observed during the study period.
Overall incidence of aGvHD was 47.8% (n = 22). Table 2 shows the type and severity of documented aGvHD reactions. The incidence of aGvHD in patients who were not adherent to prophylactic treatment at discharge was 55.6% (n = 5), compared with 45.9% (n = 17) for adherent patients. This difference, however, was not statistically significant (OR = 0.68; CI95% = 0.157-2.943; p = 0.718). The readmission rate due to aGvHD was 18.8%. All aGvHD-related readmissions occurred among adherent patients.
As all patients experienced at least one episode of febrile neutropenia or documented infection while they were admitted for their HSCT, only episodes occurring after discharge were considered. A total of 67.4% (n = 31) of transplanted patients presented with fever or infection. In the subgroup of patients receiving posaconazole, voriconazole or valganciclovir, the incidence of fever or documented infection reached 68.4% (n = 13). It was not possible to compare the incidence of both events in adherent vs. non-adherent patients as all patients were classified as adherent. The readmission rate due to fever or infection was 75%.
Discussion
This is the first Spanish study to evaluate adherence in patients under-going allo HSCT. The results obtained suggest high adherence to prophylactic treatment, ranging between 80.4% and 100%. Such results are consistent with those published in the literature11. Nevertheless, the fact that 17.4% and 19.6% of patients were found to be non-adherent to prophylaxis against graft failure or against GvHD, respectively, reflects the complexities involved in treating this patient population.
In chronic patients, an adherence rate equal to or higher than 80% is considered acceptable, although this figure should not be extrapolated to other situations. It would seem reasonable to think that patients with complex conditions would exhibit adherence-to-medication rates nearing 100% in order to optimize their clinical outcomes. Haematologic cancer patients are particularly vulnerable, and lack of adherence in these individuals could have a significant impact on their survival and survival11.
In the context of HSCT, Morrison et al. reviewed the adherence data published until 2017. Only five studies (two of them on pediatric patients) evaluated adherence to oral medication during the acute phase of HSCT, with adherence rates ranging between 33% and 94.7%5. In a study published in 2018, Lehrer et al. used an 8-item questionnaire to evaluate adherence between 60 and 180 days post-engraftment and found poor adherence levels in 50% of patients12.
It has been suggested that a lack of adherence to immunosuppressants and other oral drugs could increase the risk of developing GvHD and infection, or suffering a relapse5. Nonetheless, no reports exist in the literature that propose an adherence threshold above which the appearance of the above mentioned complications becomes more likely. Gresch et al. carried out a secondary analysis of the data reported by the PROVIVO trial, which looks into long-term survival following allo HSCT13. These authors investigated the relationship between lack of adherence and therapeutic regimen, as well as the relationship between lack of adherence and development of GvHD. That study is the first one to find a positive association between lack of adherence and the appearance of GvHD. Adherence was evaluated by means of administering a questionnaire and measuring immunosuppressant serum concentrations14.
The present study, like those mentioned above, shows that a significantly high percentage of allo HSCT patients present with adherence difficulties. However, adherence seems to be a modifiable factor 15 that may justify the delivery of specific interventions. Chieng et al. demonstrated that making weekly consultations with a specialist pharmacist available to ambulatory patients improves self-reported adherence16. Similarly, Corrêa et al. found that pharmaceutical care integrated into the multidisciplinary HSCT team contribute to a greater success in attaining the patients' therapeutic targets with regard to the use of immunosuppressant17. This positive effect of pharmaceutical interventions has also been observed in other populations, such as that of renal transplant patients18.
None of our patients experienced secondary graft failure. Nonethe-less, nearly half of them presented with at least one aGvHD episode. The incidence of aGvHD in our study was similar to that reported by other authors19-21. Although we were unable to demonstrate an association between lack of adherence and the risk of complications, we did observe a trend toward a higher incidence of aGvHD (OR = 0.68) among patients who were non-adherent to their immunosuppressive treatment. This finding ought to be confirmed by future prospective studies.
The incidence of infection in allo HSCT patients is highly variable, ranging from 1 to 65% in different studies on patients with aspergillosis and cytomegalovirus reactivation22-25. In our patients, the incidence of infection following discharge reached 67.4%, which is slightly higher than the rates reported in the above mentioned studies.
In 2015, McKenna et al. analised the most frequent causes of readmission during the acute post-implantation period in patients who underwent autologous or allogeneic transplantation. Nearly 40% of allo HSCT patients were readmitted for reasons other than a relapse of their condition. The most frequent reason was fever (58%) followed by GvHD (13.9%)6. Despite the recommendation for anti-infection prophylaxis, wide variability was found in the use, dosage and time of administration of antibiotics. Our infection-related readmission rate was 75%, while our aGvHD-related readmission rate was 18.8%. These figures are consistent with those reported in the previously mentioned study.
The retrospective nature of our study is one of its main limitations. Another one is the fact that adherence was evaluated only in quantitative terms and using one single measurement method. Conceiving adherence as “the amount of medication a patient actually takes as compared with the amount prescribed by their physician" does not capture the whole notion of adherence. The World Health Organization defines adherence as the degree to which the behavior of a patient, as regards taking medication, following a diet or changing their lifestyle, is consistent with the recommendations made by their healthcare provider. This definition comprises both quantitative and qualitative adherence, i.e. taking the right amount of medication at the right time, without missing a dose and observing any relevant dietary restrictions. Accordingly, some patients in the study could have been classified as adherent when in fact they were not. The ideal thing is to combine two measurement methods, either two indirect ones (a registry and self-completed questionnaires) or an indirect method combined with a direct one (for example serum levels of the medication). Conducting a prospective study would help overcome this problem allowing a more accurate determination of adherence in our target population.
One of the main strengths of the study is that it presents the first adherence data available for Spanish patients undergoing allo HSCT, with an acceptably-sized sample considering that the typical sample size in the published studies ranges between 6 and 138 patients5. Moreover, the present study was designed and conducted jointly by the Pharmacy and Haematology Departments with a view to establishing a starting point for the development of a comprehensive pharmaceutical care program for allo HSCT patients. Such a program would involve not only the integration of the pharmacist into the multidisciplinary HSCT team, a reality in our hospital since November 2017, but also the reconciliation of medication on admission and at discharge; the monitoring of adherence; and the promotion of healthcare education, as specified in the latest EBMT recommendations3. This initiative will help improve adherence and clinical outcomes.
To conclude, although adherence to prophylaxis against acute complications in patients undergoing allo HSCT is higher than 80%, there is still a considerable proportion of patients who do not take their medication correctly. A trend was observed toward a higher incidence of aGvHD among patients who were non-adherent to their prophylactic regimen. The full integration of a pharmacist into the allo HSCT team would improve the patients' knowledge about their treatment, optimize therapeutic adherence, and enhance clinical outcomes in this patient population.











 text in
text in 





