Introduction
Congenital coagulopathies (CCs) are a series of conditions characterized by bleeding disorders resulting from blood clotting alterations. They are complex difficult-to-manage chronic conditions, the most significant of whichin terms of both prevalence and severity are hemophilia A and B and von Willebrand's disease1.
At present, there are around 400,000 people with hemophilia worldwide, of which nearly 3,000 live in Spain2. Approximately 80-85% of cases are type A hemophilia, which is associated to a deficiency in coagulation factor VIII; patients with type B hemophilia, where the deficient clotting factor is factor IX, account for the remaining 15-20%3. Both types of hemophilia are hereditary x-linked recessive conditions, where men suffer from the disease while women act as carriers of the affected gene4. The main symptom of these conditions is potentially life-threatening recurrent internal or external bleeds, which may be spontaneous or induced by slight trauma. Fifty percent of patients suffer from severe hemophilia A1. The most formidable complication caused by bleeding is hemarthrosis, a hemophilic arthropathy that may cause severe mobility restrictions. Moreover, patients may present with muscle hematomas as well as neurologic disorders, compartment syndrome, shortness of breath, hypovolemia and intracranial hemorrhage1.
Standard treatment of hemophilia is based mainly on replacement therapy, i.e., intravenous infusions of the deficient coagulation factor aimed at treating or preventing bleeds. Such intravenous infusions may be administered on demand or prophylactically and, in Spain, they can be selfadministered by patients if they are 8 years of age or older. The amount of deficient coagulation factor to be infused and the frequency of administration depend on the type of hemophilia and the severity, type and location of the bleeds5. The most significant complication associated with replacement therapy is the development of inhibitors, which typically result in an increase in bleeding risk, reducing the patients' quality of life and increasing their mortality2.
As mentioned above, another example of a CC is von Willebrand's disease, a hereditary autosomal recessive condition where a disruption in the gene coding for Von Willebrand factor (vWF) produces qualitative and/or quantitative alterations in the vWF. The prevalence of this condition is 1-3%1, and its most characteristic symptom is the appearance of mucocutaneous bleeds of variable intensity6. As with hemophilia, three levels of severity can be distinguished in von Willebrand's disease, depending on the plasma concentration of vWF. The most severe phenotype is the most unusual one7.
Treatment of von Willebrand's disease currently consists in the administration of antifibrinolytics and desmopressin, which trigger the release of the vWF that could have built up in endothelial cells. This treatment is not indicated in all patients, with recourse to exogenous replacement factor or other alternatives being necessary in cases where it cannot be administered1,8.
There are other CCs that result in a deficiency or alteration of other factors or proteins involved in hemostasis. It is not easy to fathom the real incidence of these CCs and replacement treatment of the deficient or altered factors is not always available1.
As CCs are hereditary and, for the time being, incurable chronic conditions, the people who suffer from them are forced to live with situations that negatively impact their quality of life. These situations are highly diverse and are usually related to three kinds of factors: biological factors (pain, complications derived from the treatments, comorbidities, multiple surgical procedures; prolonged hospital stays, etc.); psychological factors (pain, stress, feelings of guilt, depression, low perceived social support, poor adherence to treatment, etc.); and social factors (negative reactions by family members, lack of support networks, financial instability, difficulties in managing pediatric patients, difficulty to travel, etc.)9.
The main contribution of hospital pharmacists to patients, society and the health system consists in promoting the improvement of patients' health outcomes by providing a pharmacological therapy adapted to their individual characteristics; optimizing the effectiveness, efficiency and safety of the treatments; making available all the information they need; and promoting clinical continuity across all levels of care10.
The purpose of this study was to identify and promote initiatives geared towards improving the way hospital pharmacy departments manage patients with CCs, thus contributing to obtaining the best health outcomes for these patients in a changing healthcare environment.
Methods
The authors came together in a coordinating committee charged with identifying the most significant milestones along the journey travelled by patients with CCs, particularly hemophilia, where hospital pharmacists (HPs) have a role to play (Figure 1). The Committee was also responsible for identifying the key actions performed by HPs as well as the main needs patients may have at each stage along their journey.
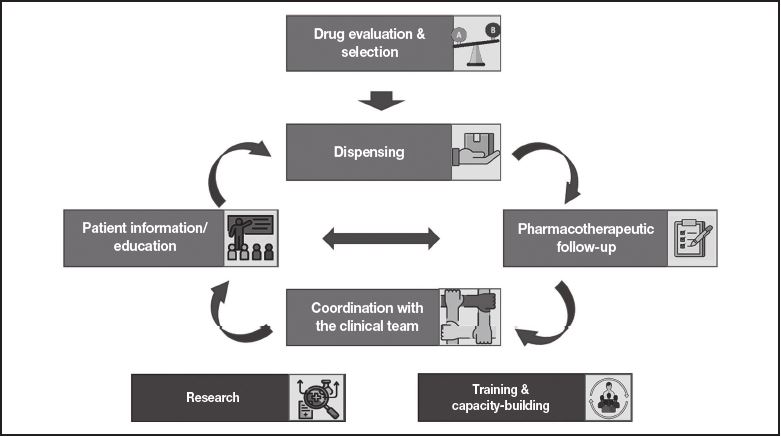
Figure 1. Main stages in the management of patients with congenital coagulopathies that involve the intervention of hospital pharmacists
Subsequently, a group of 16 HPs was created, all of them experienced in the management and treatment of patients with CCs, to identify and undertake initiatives aimed at improving the standard of care provided to these patients. To this effect, a structured pre-read was sent to each HP to promote individual reflection. At a later stage, a debate was held on the key attributes of HPs and the challenges they faced, and on the current needs of patients with CCs. Particular emphasis was laid on the areas where HPs could add the greatest value to the management of these patients and on the importance of ensuring proper coordination between the work of HPs and that of the other members of the clinical team. Taking these challenges and opportunities into consideration, a series of initiatives were identified that should be implemented to improve the standard of care provided by hospital pharmacy departments to these patients (Table 1).
Table 1 (cont.).

*FEDHEMO: Spanish Hemophilia Federation; HP: hospital pharmacist; SEFH: Spanish Society of Hospital Pharmacists
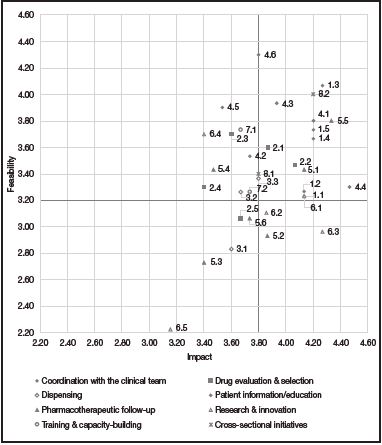
These consensus-based initiatives were prioritized by the 16 HPs individually according to their impact and feasibility. The initiatives' impact, i.e., their potential to ensure that the HP's intervention would result in an improvement in the patients' status, was scored on a scale from 1 (initiatives with the lowest impact) to 5 (those with the highest impact). Feasibility scores were the mean of the scores for two items. On the one hand, the resources needed, i.e., the financial or spatial resources required to implement the different initiatives identified (scores for this item were assigned on a scale from 1 to 5, with higher scores corresponding to higher levels of feasibility); and, on the other hand, decision-making, i.e., the complexity or difficulty inherent in making the decisions required for the implementation of each initiative (scores were assigned on a scale from 1 to 5, with higher scores corresponding to higher levels of feasibility).
The scores given by each HP were collected and aggregated for the analysis. The mean score assigned by each individual for both impact and feasibility was also recorded (Figure 2).
Once all the initiatives were prioritized, the key action areas to be addressed were identified, all of them including top priority initiatives in terms of their impact and feasibility.
Results
First of all, the coordinating committee identified the areas where the role played by hospital pharmacists was believed to be key to the management of patients with CCs. As shown inFigure 1, these areas are as follows: coordination with the other members of the clinical team in charge of taking care of these patients (hematologists, nursing staff, emergency staff, trauma surgeons, rehab physicians, etc.); drug evaluation and selection; dispensing; patient information and education; pharmacotherapeutic follow-up; research and innovation in the field of CCs; and capacity-building and training of the hospital pharmacy staff.
Within each of these areas, participating HPs discussed first of all the key actions and specific functions they performed vis-a vis patients with CCs to ensure that the best health outcomes could be achieved. That was a starting point that provided a reflection framework to identify the initiatives required to further improve the standard of care provided. This process led to identification of 34 initiatives, which were grouped under 8 domains (Table 1).
A prioritization matrix was put together (Figure 2) to represent the different initiatives based on their impact and feasibility. The matrix therefore categorized initiatives into top priority initiatives (high impact and high feasibility), intermediate priority initiatives (high impact and low feasibility, or vice versa) and low priority initiatives (low impact and low feasibility).
Of the 34 initiatives proposed, 33 obtained a score equal to or higher than 2.5 in terms of impact and feasibility. A total of 15 actions were defined as top priority, i.e., those whose score was equal to or higher than the mean overall score (3.8 for impact and 3.2 for feasibility).
As regards actions considered a priority, most of them pertained to the clinical domain (29.4%), followed by the patient information and education domain (23.5%). For their part, actions under the drug evaluation and selection, pharmacotherapeutic follow-up and cross-sectional initiative domains accounted for 11.8% of initiatives considered a priority, respectively. Lastly, the initiatives under the dispensing and the research and innovation domains accounted for 5.9% of top priority actions, respectively.
During the second phase, three overall priority action areas were defined (Table 2), which encompassed the highest priority initiatives. These priority action areas were: 1) Preparation of a guide aimed at improving clinical coordination (intra- and inter-hospital communication and coordination); 2) design of an expert patient program; and 3) personalization of the therapeutic strategy.
Table 2. Action areas proposed to improve management of patients with congenital coagulopathies by hospital pharmacists

CCs: congenital coagulopathies; CSUR: Centers, Services and Reference Units of the National Health System; HP: hospital pharmacist; PREM: patient-reported experience; PROM: patient-reported outcome measure
Discussion
CCs are complex conditions that have a significant impact on patients not only from a phsyiopathological point of view throughout all the different stages of their lives, but also from a psychological and social standpoint. For that reason, it is vital to identify the areas within the management of these chronic conditions where healthcare providers can add value to their patients, ensuring at all times that they cater for their needs.
HPs are playing an increasingly significant role in the management of patients, not least those with CCs. Although the approach to patients with CCs was traditionally limited to third-level hospitals, a growing number of less specialized facilities are now treating those patients, which means that the preparation, coordination and continuous improvement of the HPs from different hospitals is key to successfully approach their condition and optimize their health outcomes and quality of life. The prioritization carried out as part of this study together with the action areas proposed have been influenced by a changing reality.
In this respect, the three overall action areas resulting from our análisis were identified and described with a view to aligning and grouping together the most impactful and feasible initiatives in a practical easy-toimplement manner, making it possible for HPs to cater for the patients' needs. The proposed action areas are in line with and have been designed to confirm to the CMO pharmaceutical care model, aimed at providing pharmaceutical care with consideration to individual patient needs (capacity), aligning each patient's short, medium and long-term goals in collaboration with other healthcare providers (motivation), and responding to the patients' needs in real time through the new information technologies (opportunity)11.
The development of a guide intended to improve clinical coordination would help convey the experiences of hospitals with large numbers of patients with CCs to smaller facilities, fostering and formalizing intra- and inter-hospital communication and coordination and establishing communication channels between HPs, hematologists and other healthcare providers. The guide would also benefit patients as it would improve their experience of the clinical process, aid in homogenizing the information and the messages that they receive from the different professionals involved and improve pharmacotherapeutic follow-up. It would also improve communication between healthcare providers, increase the visibility of HPs and make it easier to understand their role in the multidisciplinary team. Moreover, a global view of the patient would be adopted and there would be an increase in knowledge and education. As regards the health system, the benefits would consist in an optimization of resources, which would increase efficacy, lead to better health outcomes and greater patient satisfaction, and enhance the prestige of the public health system.
Expert patient programs have been successfully implemented for other conditions, where they have proved their usefulness. Given the complex nature of CCs and the strict treatment regimens patients must follow, initiatives involving information and education activities are of the essence. According to the experts, the benefits of these programs include an increased commitment of patients with their pharmacotherapeutic goals; more informed decision-making through empowerment and capacity-building; an increase in adherence; treatment optimization and an ensuing reduction in the number of bleeds; and an increase in safety and quality of life.
Lastly, in order to improve safety, effectiveness and adherence, a suggestion mas made to further personalize therapeutic strategies by combining the different drug evaluation and selection, dispensing, patient information and education, pharmacotherapeutic follow-up and cross-sectional initiatives identified. It was thus concluded that developing a guide or model that made it easier for HPs to establish the most appropriate therapeutic strategy for each patient with a CC could be extremely beneficial as it would improve overall health outcomes and significantly increase the patients' quality of life.
Preparation of a guide or a model that makes it easier for HPs to personalize the therapeutic strategy and the care of patients would go a long way towards ensuring that patients receive a treatment adapted to their clinical situation, lifestyle and expectations that allows them to live their lives to the full thanks to a therapy tailored to their needs. HPs would also be better off as they would be provided with an overall view of each patient, which would help them make more informed decisions and increase their effectiveness at preventing complications, thus enhancing patient satisfaction. Furthermore, making the guide generally available would facilitate the sharing of the information provided with other professionals.
This analysis has identified and prioritized a series of actions into three global priority action areas which, if addressed, could contribute to improving the way patients with CCs, particularly those with hemophilia, are currently approached. The actions proposed, all of them amenable to implementation in the Spanish health system, could make it possible for HPs to exert a real impact on the lives of their patients.











 texto en
texto en