My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Medicina Oral, Patología Oral y Cirugía Bucal (Internet)
On-line version ISSN 1698-6946
Med. oral patol. oral cir.bucal (Internet) vol.12 n.5 Sep. 2007
Dyskeratosis congenita. Report of a case with literature review
Ajit Auluck
MDS. Assistant Professor. Oral Medicine and Radiology. Manipal college of Dental Sciences, Mangalore, India
ABSTRACT
Leukoplakic lesion is not uncommon in clinical practice but its occurrence as a component of a syndrome is rare. Dyskeratosis congenita is a rare genodermatosis, which is characterized by triad of skin pigmentation, nail dystrophy and leukoplakic lesion in the oral cavity. It is important for dentists to now about Dyskeratosis Congenita because these leukoplakic lesions can spontaneously undergo malignant transformation. Majority of cases have been reported in dermatology or pediatrics literature whereas only few reports have appeared in dental literature. The purpose of reporting this case with review of recent literature is to create better awareness among dentists about the multisystem manifestations and oro-dental abnormalities of this fatal condition that can aid clinicians in early diagnosis. A case of 26 year old male is reported with details of differential diagnosis, importance of hematological investigations, periodic biopsies and current concepts in management of such patients are briefly reviewed.
Key words: Dyskeratosis congenita, leukoplakia, nail dystrohpy, skin pigmentation, hematological abnormalities.
Introduction
Dyskeratosis congenita (DC) is a rare, hereditary disease, which was first described by Zinsser in 1906(1). Later Engman and Cole et al reported other cases in detail and hence it is also known as Cole-Engman syndrome or Zinsser- Cole-Engman syndrome(2). DC occurs mostly in males and manifests between 5 to 12 years. Classic triad of skin pigmentation, nail dystrophy and oral leukoplakia occur in complete expression of this syndrome. DC is a fatal condition in which majority of the patients develop aplastic anemia and malignant transformation occur in the keratotic white patches(1,2) which is of considerable interest to a dentist.
Case report
A man of 26 years reported for consultation regarding an asymptomatic white patch present on the dorsum of the tongue for last 4 years. He had no habit of chewing or smoking tobacco. No other family member had history of such lesions. Patient did not report of itching, photosensitivity and loss of hair.
The patient was apparently normal till the age of 5 years but started developing symptoms, which are summarized in table-1.
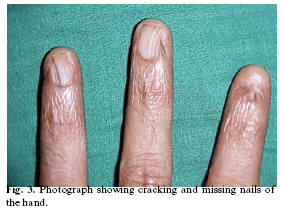
Patient appeared pale but vital signs were within normal limits. Hyperpigmented areas were observed on the forehead (Fig-1), neck and trunk. Nails of both toes and fingers were cracked and atrophied (Fig-2). Left eye was red and patient reported of epiphora (Fig-1).
On intra oral examination a raised white patch of 0.5 cm in diameter was observed on the dorsal surface of the tongue (Fig-3). Lesion was not scrapable with gauze and had no indurated margins. The teeth, periodontium and other oral mucosal surfaces were normal.

Considering the symptoms of skin pigmentation, nail dystrophy and white patch a differential diagnosis of pachyonychia congenita and DC were considered. Hematological investigations, bone aspiration and biopsy of tongue lesion were ordered. Hematological investigations revealed reduced levels of hemoglobin [5.6 mmol/L (normal 8.1-11.2 mmol/L)] and platelets [90X109/L (normal 150 – 450X109/L)] with white blood cells (WBC) count of 4.2X109/L (normal 4X109/L), which is towards the lower limit. On basis of the hematological report a definitive diagnosis of DC was made.
The patient was referred for ophthalmologic consultation.
The Ophthalmologist reported of a growth in the left fundus region with punctal atresia (Fig-1) and a biopsy was recommended.
However, the biopsy was not performed as the patient refused to undergo the procedure.
Histopathological report of the tongue lesion revealed hyperkeratosis with flattened rete ridges. Connective tissue showed fibro-collagenous tissue with proliferating capillaries surrounded by few lymphocytes, adipose tissue and skeletal muscle with areas of hyalinization and atrophy. There was no evidence of malignancy. Bone marrow aspirate showed hypocellular marrow particles suggestive of aplastic anaemia. Trephine biopsy showed bony trabeculae with a markedly hypocellular marrow space in which normal marrow elements including megakaryocytes were not observed.
Discussion
DC is a fatal condition with multisystem manifestations. There are X-linked, autosomal dominant and autosomal recessive forms of DC(3,4). It mainly occurs in the males, inherited as X linked recessive disorder with male:female predilection of 13:1(3). Occurrence in extremely low percentage of the females suggests that there are subsets inherited in an autosomal dominant fashion(3). Autosomal dominant form of dyskeratosis congenita is associated with mutations in the RNA component of telomerase, hTERC, while X linked dyskeratosis congenita is due to mutations in the gene encoding dyskerin, a protein implicated in both telomerase function and RNA processing(5). In DC multiple organ systems are affected due to pleiotropic mutation in the gene(6).
In DC both the hard and the soft tissues in the oral cavity are affected. Oral mucosal changes manifests in the form of a white keratotic patch. Earlier cases reported of vesicles and ulcerations preceding development of leukoplakia(7). But these may represent herpetic infections which may occur due to immunocompromised status of these patients secondary to bone marrowsuppression(8). In our case the patient had no vesicles or ulcerations or any other associated oro-mucosal abnormality.
Intraorally, the white patch may affect the buccal mucosa, tongue or palate, with tongue most commonly affected(1) as seen in our patient. Usually, white patch associated with dyskeratosis congenita is leukoplakia but lichen planus or lichenoid lesions instead of leukoplakia are also reported(5). Lichenoid lesions may be a component of chronic graft versus host disease. Electron microscopy studies revealed that cells in dyskeratosis congenita have an embryonic immature nucleus, which have higher chances to undergo malignant transformation(9). In addition, barrier zone of epithelium is less effective in dyskeratosis congenita than the normal epithelium causing increased permeability of noxious substances and carcinogens to the germinal layers(10). Hence increased malignant transformation rate is seen in leukoplakic areas associated with dyskeratosis congenita(9). However, in the case reported here the biopsy report was not suggestive of any malignant changes.
In DC severe periodontal destruction occur due to anomalies in ectodermally derived structures and diminished host response caused by neutropenia(10). Patients have gingival inflammation, bleeding, recession and bone loss that simulates juvenile periodontitis(10). In addition there may be defects in the structure of the enamel like hypocalcification(7) that may cause dental caries. However, our patient had a healthy periodontium and teeth were normal.
Dermatologic manifestations occur in form of reticular hyperpigmentation of skin. These pigmented areas are associated with atrophy of epidermis, capillary hyperplasia and melanin pigment deposited near the blood vessels(3,6). Such pigmentation was observed in our patient over the skin of the face (Fig-1) and chest. Dystrophy of nails is associated with the onset of skin pigmentation(2). Nail abnormalities can result in longitudinal splitting and furrowing. Nails are brittle or completely lost(3). In our patient skin and nail changes started simultaneously (Table-2) and nails were atrophied with absence of nails in some digits (Fig-2).
In DC eye involvement can lead to epiphora, growth in fundus, blepharitis and loss of eyelashes(3). Our patient had a growth in the left fundus of the eye but biopsy could not be performed to ascertain the nature of the lesion.
Majority of patients develop a haematopoietic disorder resembling Fanconis anaemia because initially the bone marrow is normal but gradually fat cells and fibrotic tissue replace the haematopoietic cells(7). Haematopoietic disorder manifests as marrow hypoplasia, anaemia or thrombocytopenia(6). With development of pancytopenia there occurs secondary infections that is the cause of death in majority of these patients(6,10). In addition to marrow failure and malignant changes in white patches, pulmonary complications are also serious complications in patients with DC(11). Our patient was pale and had anemia. Moreover, marrow was hypocellular and patient had thrombocytopenia.
Immunologic abnormalities occur affecting both the humoral and cell mediated immunity causing high incidence of opportunistic infections(6,10). Abnormalities in cell mediated immunity is evident by delayed hypersensitivity reaction to skin test (antigen inoculation) and no sensitization to dinitrochlorobenzene(6). Abnormalities in immune systems like lymphoid depletion, absence of primary germinal follicles and fibrosis of lymph nodes can also occur(6). No opportunistic infections were seen in this patient. Tests to confirm these immunological abnormalities were also not performed.
There may be hyperpigmentation of the buccal mucosa that may be reactive post inflammatory hypermelanosis(8). Other abnormalities like alopecia, taurodontism, mental retardation, small genitals premature aging, nutmeg liver, horseshoe kidneys, amyloidosis and development of Hodgkins disease and adenocarcinoma are also reported(3,6,8) but not observed in this patient.
Differential diagnosis
The differential diagnosis includes fanconis anaemia, pachyonychia congenital, white spongy nevus and Graft versus host disease (GVHD).
DC is considered as a variant of Fanconis anaemia because aplastic anaemia and cutaneous pigmentation occur in both but the two conditions differ from each other. Fanconis anaemia may have a possible association with oral squamous cell carcinoma(12). In contrast to Dyskeratosis congenita, Fanconis anaemia is an autosomal dominant condition with concomitant skeletal and renal abnormalities. But there is no nail dystrophy or oral leukoplakia seen in Fanconis anaemia as reported in this case.
Pachyonychia congenita is an autosomal dominant condition characterized by nail abnormalities, hyperkeratosis or hyperhidrosis of the palms and the sole and mucosal leukoplakia. In Pachyonychia congenita the nails become thick and shed at an early age and the mucosal leukoplakias do not undergo malignant transformation. Hematological abnormalities do not occur in pachyonychia congenita.
White spongy nevus is a congenital leukokeratosis that presents with diffuse milky-white plaques on oral mucous membrane. The keratotic plaques are extensive and persist through out life, without a tendency for malignant transformation. It is not associated with skin, nail or hematological abnormalities as seen in DC.
In patients with DC who have undergone bone marrow transplantation, graft versus host disease can be considered as a differential diagnosis(8) because they have several features in common(13), including skin and nail changes. DC should be suspected in cases where the chronological pattern is not consistent with chronic graft versus host disease. In DC leukoplakias rather than lichenoid lesions occur. If white lesion shows lichenoid lesion like characteristics GVHD should be suspected.
Treatment
There is no effective and curative treatment for DC. Some interceptive and preventive measures can be adopted for which an early diagnosis is essential. Patient should be kept under observation and recalled for periodic follow up (14). In follow-up visits biopsy and complete blood picture should be advised to detect any malignant changes in white patches or developing haematopoietic disorder.
Chemotherapy with bleomycin or cyclophosphamide is effective for treating leukoplakia(1) associated with DC. Tomita and Ishii(15) reported that a vitamin A derivative, etretinate is effective for treating leukoplakia in DC. However side effects like exfoliative cheilitis, angular cheilitis and teratogenicity can occur. Excisional biopsy can also be indicated to eliminate the possibility of development of carcinoma in leukoplakic areas. For extensive lesions in which surgery is not possible administration of steroids and testosterone can be used(1,4). In future gene therapy can provide an alternative therapy for management of this fatal condition.
Conclusion
In a young patient, if a white keratotic patch is observed without history of tobacco consumption with concomitant muco-cutaneous and systemic disorders a clinician should suspect Dyskeratosis Congenita. Dentist should be able to recognize this fatal condition in its early stages and advice appropriate hematological investigations. Biopsy of the white patch should be performed and patient recalled for regular follow up. Dentists may be first to see and diagnose Dyskeratosis Congenita and have an important role in monitoring the oral malignant changes in the mucosa.
References
1. Tanaka A, Kumagai S, Nakagawa K, Yamamoto E. Cole-Engman syndrome associated with leukoplakia of the tongue: a case report. J Oral Maxillofac Surg 1999;57:1138-41. [ Links ]
2. Boek KD, Degreef H, Verwilghen R, Corbel L, Daele MCV. Thrombocytopenia: first symptom of dyskeratosis congenita. Pediatrics 1981;67:898-902. [ Links ]
3. Ogden GR, Connor E, Chisholm. Dyskeratois congenita: report of a case and review of literature. Oral Surg Oral Med Oral Pathol 1988;65:586-91. [ Links ]
4. Marrone A, Mason PJ. Dyskeratosis Congenita. Cell Mol Life Sci 2003;60:507-17. [ Links ]
5. Bessler M, Wilson DB, Mason PJ. Dyskeratosis congenital and telomerase. Curr Opin Pediatr 2004;16:23-8. [ Links ]
6. Womer R, Clark JE, Wood P, Sabio H, Kelly TE. Dyskeratosis congenita: two examples of this multisystem disorder. Pediatrics 1983;71:603-9. [ Links ]
7. Loh HS, Koh ML, Giam YC. Dyskeratosis congenita in two male cousins. Br J Oral Maxillofac Surg 1987;25:492-9. [ Links ]
8. Triester N, Lehmann LE, Cherrick I. Dyskeratosis congenita Vs chronic graft versus host disease: report of a case and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2004;98:566-71. [ Links ]
9. Mckay GS, Ogden GR, Chisholm DM. Lingual hyperkeratosis in dyskeratosis congenita: ultrastructural findings. J Oral Pathol Med 1991;20:196-9. [ Links ]
10. Yavuzyilmaz Ezel, Yamanik Nermin, Yetgin Sevgi, Kansu Ozgen. Oral-Dental findings in Dyskeratosis Congenita. J Oral Pathol Med 1992;21:280-4. [ Links ]
11. Sabesan T, Baheerathan NN, Hankovan V. Dyskeratosis congenital: Its connections with oral and maxillofacial surgery. Br J Oral Maxillofac Surg 2005, July 14 (epub ahead of print). [ Links ]
12. Dean A, Alamillos FJ, Velez A, Velasco F, Rodas J, Garcia A. Squamous cell carcinoma of the oral cavity and Fanconi s anemia. An association to bear in mind. Med Oral 1999;4: 410-415. [ Links ]
13. de la Rosa Garcia E, Bologna Molina R, Vega Gonzalez Tde J. Graft-versus-host disease, an eight case report and literature review. Med Oral Patol Oral Cir Bucal 2006;11:E486-92. [ Links ]
14. Handley TP, McCaul JA, Ogden GR. Dyskeratosis congenita. Oral Oncol 2006;42:331-6. [ Links ]
15. Tomita K, Ishii M. A case of Cole- Engman syndrome: Effect of etretinate on the oral leukoplakia. Acta Dermatol Kyoto 1992;87:567. [ Links ]
![]() Correspondence:
Correspondence:
Dr. Ajit Auluck
Department of Oral Medicine and Radiology
Manipal College of Dental Sciences
Light House Hill Road
Happankata
Mangalore 575001
Karnataka, India
E-mail: drajitauluck@gmail.com
Received: 25-10-2006
Accepted: 23-02-2007