My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
The European Journal of Psychology Applied to Legal Context
On-line version ISSN 1989-4007Print version ISSN 1889-1861
The European Journal of Psychology Applied to Legal Context vol.8 n.2 Madrid Jul. 2016
https://dx.doi.org/10.1016/j.ejpal.2016.01.001
Testosterone and attention deficits as possible mechanisms underlying impaired emotion recognition in intimate partner violence perpetrators
La testosterona y los déficits de atención como posibles mecanismos que subyacen a las carencias en el reconocimiento emocional de los hombres que ejercen violencia contra la mujer en las relaciones de pareja
Ángel Romero-Martínez, Marisol Lila and Luis Moya-Albiol
University of Valencia, Spain
This work was supported by the Ministry of Economy and Competitiveness and the National Plan on Drugs of the Spanish Ministry of Health, Social Services, and Equality [PNSD/2012/001], by the Master in Neuro-criminology (ADEIT, Universitat de València) and the Science Policy Unit at the Department of Education of the Valencian regional government (PROMETEOII/2015/020).
ABSTRACT
Several studies have reported impairments in decoding emotional facial expressions in intimate partner violence (IPV) perpetrators. However, the mechanisms that underlie these impaired skills are not well known. Given this gap in the literature, we aimed to establish whether IPV perpetrators (n = 18) differ in their emotion decoding process, attentional skills, and testosterone (T), cortisol (C) levels and T/C ratio in comparison with controls (n = 20), and also to examine the moderating role of the group and hormonal parameters in the relationship between attention skills and the emotion decoding process. Our results demonstrated that IPV perpetrators showed poorer emotion recognition and higher attention switching costs than controls. Nonetheless, they did not differ in attention to detail and hormonal parameters. Finally, the slope predicting emotion recognition from deficits in attention switching became steeper as T levels increased, especially in IPV perpetrators, although the basal C and T/C ratios were unrelated to emotion recognition and attention deficits for both groups. These findings contribute to a better understanding of the mechanisms underlying emotion recognition deficits. These factors therefore constitute the target for future interventions.
Key words: Attention. Cortisol. Emotion recognition. Testosterone. Intimate partner violence.
RESUMEN
Diversos estudios hablan de carencias en la descodificación de expresiones emocionales faciales en aquellos sujetos que cometen violencia de pareja (VP). No obstante, no se conocen bien los mecanismos subyacentes al déficit de estas capacidades. A la vista de esta laguna en investigación, hemos intentado establecer si las personas que cometen violencia de pareja (n = 18) se diferencian en el proceso de descodificación emocional, capacidad de atención y niveles de testosterona (T), cortisol y cociente T/C, comparados con los controles (n = 20), así como analizar el papel moderador del grupo y de las variables hormonales en la relación entre capacidad de atención y proceso de descodificación emocional. Nuestros resultados han demostrado que quienes perpetran violencia de pareja muestran un peor reconocimiento emocional y mayor coste en el cambio de atención que los controles. No obstante, no había diferencias en la atención a los detalles y en las variables hormonales. Por último, la pendiente que predice el reconocimiento emocional a partir de los déficits en el cambio de atención era más pronunciada a medida que aumentaban los niveles de testosterona, sobre todo en los sujetos que perpetran violencia de pareja, aunque el cortisol basal y el cociente T/C no guardaban relación con el reconocimiento de emociones y déficit de atención en ambos grupos. Estos resultados contribuyen a explicar los mecanismos que subyacen a los déficits de reconocimiento de emociones. Estos factores son pues el objetivo de intervenciones futuras.
Palabras clave: Atención. Cortisol. Reconocimiento de emociones. Testosterona. Violencia de pareja.
The ability to recognize the affective content of external cues is crucial to interpersonal interactions and social competence as it allows individuals to accurately process potentially threatening stimuli, and to establish appropriate social interactions by enabling rapid decoding of the affective state of others from their facial expressions (Carton, Kessler, & Pape, 1999; Kinsbourne & Bemporad, 1984; Morrison & Bellack, 1981; Moya-Albiol, Herrero, & Bernal, 2010; Semrud-Clikeman, Guy, Griffin, & Hynd, 2000; Tannock, Martinussen, & Frijters, 2000). Hence, impairments in this ability can lead to interpersonal problems such as violence due to its importance to understanding social cues.
Several studies have reported impairments in decoding emotional facial expressions in intimate partner violence (IPV) perpetrators (Holtzworth-Munroe & Smutzler, 1996; Romero-Martínez, Lila, Catalá-Miñana, Williams, & Moya-Albiol, 2013; Romero-Martínez, Lila, Sariñana-González, González-Bono, & Moya-Albiol, 2013; Romero-Martínez & Moya-Albiol, 2013). In fact, one study reported that IPV perpetrators are less able to recognize standardized neutral and disgust facial expressions than non-violent men, although they did not differ in interpreting other facial expressions conveying, for example, sadness, anger, fear, happiness, and surprise (Babcock, Green, & Webb, 2008), suggesting that IPV perpetrators may present deficits in recognizing their wives’ or partners’ emotions, thus increasing the risk of violent reactions. In a similar line, another study demonstrated that IPV perpetrators with diminished sensitivity to their wives’ or partners’ expressions of fear presented a high risk of perpetrating IPV (Marshall & Holtzworth-Munroe, 2010). However, the mechanisms which underlie the impaired emotion recognition skills are not well known.
One possible mechanism underlying impaired emotion recognition could be attention deficits, as has been reported in several studies based on people with attention deficit/hyperactivity disorder (ADHD) (Fuermaier et al., 2014; Noordermeer et al., 2015; Williams et al., 2008). For some emotions, people with ADHD spend less time looking at relevant areas of faces and take longer to detect an emotion compared with people without ADHD (Serrano, Owens, & Hallowell, 2015). This could be partially explained by the fact that individuals with ADHD tend to show substantially larger switch costs and pay less attention to detail than those without ADHD (Cepeda, Cepeda, & Kramer, 2000). In social contexts, subjects must rapidly and accurately switch their attention focus from one side of the face to the other and pay attention to micro facial expressions. For this reason, attention deficits could affect the detection of subtle facial expressions.
ADHD in adults is one of the predictive and treatable risk factors for delinquency, including IPV (Buitelaar, Posthumus, & Buitelaar, 2015; Fang, Massetti, Ouyang, Grosse, & Mercy, 2010; Wymbs, Dawson, Suhr, Bunford, & Gidycz, 2015). Nevertheless, little is known about the role of attention deficits in the emotion decoding process of IPV perpetrators. Only one study has reported deficits in sustained and divided attention in IPV perpetrators (Cohen et al., 2003), but there is a gap in the literature analyzing other attention deficits such as switch costs and attention to detail in IPV perpetrators, and whether these attention deficits could explain their diminished emotion recognition skills.
In addition to attention biases as a possible explanation for emotion recognition deficits, hormonal parameters could modulate neural processes affecting facial emotion processing, which may interfere with social functioning (Little, 2013). Firstly, sex steroids such as testosterone (T) have been related to improvements in male detection of angry faces (Derntl et al., 2009; Stanton, Wirth, Waugh, & Schultheiss, 2009; van Honk & Schutter, 2007a; Wirth & Schultheiss, 2007), but those studies failed to find an association between T levels and faces with a neutral or positive value. However, T levels have been negatively related to the capacity to infer the thoughts and feelings of others (Ronay & Carney, 2013). Indeed, Ronay and Carney (2013) explain these associations as a mechanism to predispose and facilitate the adoption of violence in order to maintain their status. Finally, converging evidence suggests that T levels have a negative relationship with cortisol (C) levels (Terburg, Morgan, & van Honk, 2009). Hence, it seems logical that C levels will present an inverse relationship with emotion recognition. One study in fact demonstrated that people with high C levels oriented their attention away from masked angry faces (van Honk et al., 1998). Nevertheless, there is a gap in the analysis of how these hormones and the ratio between them affect emotion recognition and attention skills in IPV perpetrators.
Bearing all of the above in mind, the main aim of this paper is to confirm whether IPV perpetrators show deficits in emotion recognition and attention (high attention switch costs and less attention to detail) that differentiate them from the general population, and in addition, these variables should be positively related. These hypotheses are congruent with previous studies demonstrating that IPV perpetrators present poor emotion recognition skills (Gracia, Rodríguez, & Lila, 2015; Holtzworth-Munroe & Smutzler, 1996; Romero-Martínez, Lila, Catalá-Miñana et al., 2013; Romero-Martínez, Lila, Sariñana-González et al., 2013; Romero-Martínez & Moya-Albiol, 2013) and attention deficits in comparison with the non-violent population (Romero-Martínez & Moya-Albiol, 2013), and that the two variables are positively related (Fuermaier et al., 2014; Noordermeer et al., 2015; Williams et al., 2008), especially in IPV perpetrators. Additionally, this study investigated the potential moderating effect of group (IPV perpetrators vs. controls) and basal hormonal parameters (T, C and T/C ratio) on the potential relationship of attention deficits with emotion recognition skills. Previous studies have found that violent populations tend to present a bias toward negative faces and that T levels were positively and C negatively correlated with angry faces and unrelated to other kinds of emotional stimuli (Derntl et al., 2009; Stanton et al., 2009; van Honk & Schutter, 2007b; van Honk et al., 1998; Wirth & Schultheiss, 2007). Hence, we hypothesized that high basal T and low basal C levels improve general emotion recognition, and that these associations are significant only in IPV perpetrators.
Method
Participants
The final sample was composed of 38 healthy male volunteers (18 IPV perpetrators and 20 controls). The IPV perpetrators were recruited from participants in the CONTEXTO psycho-educational and community-based treatment program (mandatory for male abusers) at the Department of Social Psychology of the University of Valencia (Spain). Having been sentenced to less than two years in prison and having no previous criminal record, participants were given a suspended sentence on the condition that they attended an intervention program (Catalá-Miñana, Lila, Conchell, Romero-Martínez, & Moya-Albiol, 2013; Lila, Gracia, & Murgui, 2013; Lila, Oliver, Catalá-Miñana, & Conchell, 2014; Lila, Oliver, Galiana, & Gracia, 2013). Participating IPV perpetrators were initially classified as type II according to Gottman, Jacobson, Rushe, and Shortt's (1995) classification because their criminal record indicated that the violence perpetrated against their partner was not premeditated, but was described as impulsive violence in reaction to marital or relationship conflicts.
We advertised in the University of Valencia for male volunteers for the control group, establishing contact by email and then screening applicants in interviews. The inclusion criteria for controls were: having no physical or mental problems, having similar anthropometrical and demographic characteristics to the IPV perpetrators, and not having perpetrated severe violence, defined as assaulting a partner or other individual outside the home or engaging in any severely violent act.
All participants were right handed, lived in Valencia (Spain), and gave written informed consent. The experiment was performed in accordance with the Helsinki Declaration and approved by the University of Valencia Ethics Committee.
Procedure
Each participant took part in three sessions in the psychobiology laboratories of the University of Valencia. In the first sessions, they were interviewed to exclude any individuals with organic diseases. In all cases, the second sessions were held between 4:00 and 7:00 p.m. After arriving at the laboratory, participants were taken to a room where they signed informed consent forms, their height and weight were measured and two saliva samples were collected to assess T and C levels. Finally, in the last sessions, participants completed the ‘reading the mind in the eyes’ test (henceforth, the eyes test) and the inattention subscales of the Autism Spectrum Quotient questionnaire (see below). For further details, see Romero-Martínez, Lila, Catalá-Miñana et al. (2013), Romero-Martínez, Lila, Sariñana-González et al. (2013).
Emotion Recognition
The revised version of the eyes test was administered. This task is considered to be an advanced theory of mind test that contains 36 black and white photographs of the eye region of faces of different male and female actors. Subjects were asked to attribute the mental state of the actors by choosing which of four words best described what the person in the photo was thinking or feeling. Scores are calculated as the total number of correct choices for all 36 photographs (Baron-Cohen, Wheelwright, Hill, Raste, & Plumb, 2001).
Inattention Measures
We used the attention switching and attention to detail subscales of the Spanish version of the adult Autism Spectrum Quotient (AQ) (Baron-Cohen, Wheelwright, Skinner, Martin, & Clubley, 2001). The attention switching subscale was used to measure difficulty in attention switching or deficient set shifting (Polderman et al., 2013); the attention to detail subscale was used as a high precision measurement to detect small variations in the perceived stimulus. The subscale consists of 10 items rated on a 4-point scale: definitely agree, slightly agree, slightly disagree, and definitely disagree. Ratings were recoded as 1 if the person reported inattentive symptoms and as 0 if they showed no symptoms. Scores could therefore range from 0 to 10, obtained by adding the frequency of inattentive symptoms, with a high score corresponding to more or severe symptoms.
Hormonal Parameters
Saliva was collected directly from the mouth using Salivette devices for C (Sarstedt, Rommelsdorf, Germany) and sterile glass tubes for T samples. In all cases, participants were informed that it was important to follow the instructions for saliva sampling in order to obtain meaningful data. All samples were collected in the same order, first those for assessing C, then those for T, and frozen at -20 °C until analysis. Salivary T levels were assessed by chemiluminescence immunoassays using testosterone saliva ELISA kits (Diagnostics Biochem Canada) and salivary C levels by radioimmunoassay using Coat-A-Count cortisol kits (DPC-Siemens Medical Solutions Diagnostics). As the T was expressed in pmol/L and the C in nmol/L, they were transformed in the same scale in order to obtain the ratio. For more details, see Romero-Martínez, Lila, Catalá-Miñana et al. (2013), Romero-Martínez, Lila, Sariñana-González et al. (2013).
Data Analysis
After confirming the normality of the data using the Kolmogorov–Smirnov test, t-tests with Levene's test for equality of variances were used to check for significant differences in age, BMI, AQ subscales, and hormonal parameters between groups (IPV and controls).
Correlation analyses were performed to analyze the direct association of emotion recognition with attention deficits and hormonal parameters. We then selected the related variables and performed a regression analysis. When an attention deficit was found to predict the emotion recognition, we assessed whether the hormonal parameters moderated this relationship. In order to assess the moderating role of the hormonal parameters (T, C, and T/C ratio), we followed the method of Aiken and West (1991), Dawson (2014), and Dawson and Richter (2006) to plot the interaction effects. In Step 1, we included the attention deficit (attention switch costs or low attention to detail), as the independent variable (IV 1). In Step 2, we added a hormonal parameter (IV 2) as predictor. Finally, in Step 3, we added the interaction term ‘attention deficit * hormonal parameter’ to investigate whether the relationship between attention deficit and emotion recognition was moderated by a specific hormonal parameter of the participants.
Data analyses were carried out using IBM SPSS Statistics for Windows (version 19.0). The threshold for statistical significance was set at p values ≤ .05. Average values are reported in the tables as mean ± SD.
Results
Descriptive characteristics of the IPV perpetrators are presented in Table 1. As can be seen, the only differences found were between groups in AQ switch attention (t37= 2.55, p = .015, d = 0.83) and eyes test (t37 = 1.82, p = .051, d = 0.60), with IPV perpetrators presenting greater difficulties to switch their attention and lower scores in eyes test than the controls. Nevertheless, they did not differ in AQ attention to detail (t37 = 1.11, p = .274, d = 0.35) and/or testosterone levels (t37 = 1.68, p = .102, d = 0.55).
Table 1. Mean and SD of Descriptive Characteristics, Psychological Questionnaires,
and Hormonal Parameters for IPV Perpetrators and Controls. 
* p < .05, ** p < .01.
Relationships between variables are summarized in Table 2.
Table 2. Pearson Correlations between AQ Attention Deficits, Testosterone,
and Eyes Test Performance for IPV Perpetrators and Controls. 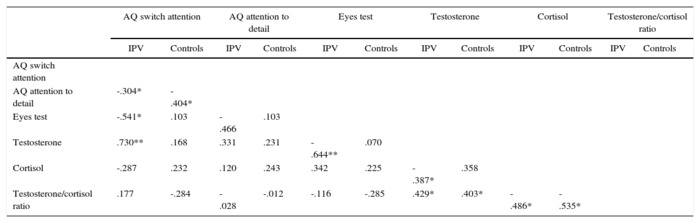
* p < .05, ** p < .01.
Moderation Models
Does ‘group’ play a moderating role in the relationship between switch attention deficits and emotion recognition? The introduction of ‘group’ together with ‘AQ switch attention’ in Step 2 significantly increased (p < .05) the amount of variance explained: adj R2 = .171, ΔR2 = .050, F(2, 38) = 4.91, p = .013. In Step 3, the inclusion of the interaction term "group * AQ switch attention" also significantly increased (p < .05) the amount of variance explained: adj R2 = .271, ΔR2 = .114, F(3, 38) = 5.71, p = .003. Hence, the slope predicting emotion recognition from deficits in attention switching became higher especially in IPV perpetrators (β = .891, p < .01) (Figure 1).

Figure 1. Relationship between AQ Switch Cost and Emotion Recognition
for IPV Perpetrators and Control Group.
Does ‘testosterone’ play a moderating role in the relationship between switch attention deficits and emotion recognition? The introduction of ‘testosterone’ together with ‘AQ switch attention’ in Step 2 significantly increased (p < .01) the amount of variance explained: adj R2 = .265, ΔR2 = .139, F(2, 38) = 7.86, p = .001. In Step 3, the inclusion of the interaction term ‘group * AQ switch attention’ also significantly increased (p < .05) the amount of variance explained: adj R2 = .319, ΔR2 = .069, F(3, 38) = 6.94, p = .001. Hence, the slope predicting emotion recognition from deficits in attention switching became higher as testosterone levels increased (β = -.107, p < .01).
Do ‘group’ and ‘testosterone’ play a moderating role in the relationship between switch attention deficits and emotion recognition? The introduction of these variables (group and testosterone levels) together with ‘AQ switch attention’ in Step 2 significantly increased (p < .05) the amount of variance explained: adj R2 = .291, ΔR2 = .183, F(3, 38) = 6.21, p < .002. In Step 3, the inclusion of the interaction term ‘group*testosterone * AQ switch attention’ also significantly increased (p < .05) the amount of variance explained: adj R2 = .361, ΔR2 = .081, F(4, 38) = 6.36, p < .001. Hence, the slope predicting emotion recognition from deficits in attention switching became higher as testosterone levels increased, especially in IPV perpetrators (β = .527, p < .05).
Discussion
Compared with controls, IPV perpetrators showed poor emotion recognition and high attention switch costs. There were, however, no differences between groups in basal T and C levels, T/C ratio and/or attention to detail scores. On the other hand, we obtained that the slope predicting emotion recognition from deficits in attention switching became higher as T levels increased, especially in IPV perpetrators. Nevertheless, the basal C and T/C ratio were unrelated to emotion recognition and attention deficits for both groups.
Our findings suggest that emotion recognition impairments appear to be a specific deficit in IPV perpetrators, especially in type II or reactive IPV perpetrators. This is consistent with previous research in this field (Babcock et al., 2008; Holtzworth-Munroe & Smutzler, 1996; Marshall & Holtzworth-Munroe, 2010; Romero-Martínez, Lila, Catalá-Miñana et al., 2013; Romero-Martínez, Lila, Sariñana-González et al., 2013; Romero-Martínez & Moya-Albiol, 2013). We also found that those emotional decoding impairments could be partially explained by self-reported impairments in attentional set shifting or switch costs. In fact, IPV perpetrators reported larger switch costs than controls, although they did not differ in attention to detail. In line with these results, previous studies have demonstrated that violent offenders show significant deficits in attentional set shifting or larger attention switch costs in comparison with non-violent population (Bergvall, Wessely, Forsman, & Hansen, 2001). In fact, this attention deficit might explain not only the impaired emotion decoding process but also the executive domain deficits defined as a worse performance in tests like the Wisconsin Card Sorting Test, which has been previously reported in IPV perpetrators (Gracia et al., 2015; Romero-Martínez, Lila, Catalá-Miñana et al., 2013; Romero-Martínez, Lila, Sariñana-González et al., 2013; Romero-Martínez & Moya-Albiol, 2013). Nonetheless, it is important to note that in our research we analyzed attention deficits with self-reports, although these subscales have been employed as successful inattention measures in previous studies in several populations (Polderman et al., 2013; Rusconi, Ferri, Viding, & Mitchener-Nissen, 2015; Sizoo et al., 2009). For this reason future studies should consider, in addition to self-reports, more implicit measures as well as neuropsychological tests in order to reinforce the value of the measurements.
Regarding the hormonal parameters, our research also demonstrated that only T played an important role in the impaired emotion decoding process observed in IPV perpetrators. In fact, high T levels were associated with worse emotion recognition, especially in IPV perpetrators. According to previous studies, high T levels are associated with high attention and detection of angry faces (Montoya, Terburg, Bo, & van Honk, 2012). Our results did not support this hypothesis, however, which could be explained by several reasons. Firstly, previous studies manipulated T levels by exogenous administration, whereas in our study we used basal endogenous hormonal levels without external manipulation. Secondly, previous studies were more interested in determining participant arousal and/or activation to several images than in their ability to discriminate those images; furthermore, their results are based on non-violent populations. Regarding C levels, only one previous study has reported an inverse relationship between angry face detection and basal C levels (van Honk et al., 1998). Nevertheless, our results failed to find a significant relationship between C and emotion recognition, and we did not obtain a significant relationship between the T/C ratio and the emotion decoding process or the attention deficits. As in the case of T, these differences could be explained by methodological variations between studies in this field, such as different sample sizes, cognitive tests and/or hormonal measurements. Moreover, a recent study with IPV perpetrators analyzed the relationship between basal T and C levels and the T/C ratio with personality traits and proneness to anger expression, obtaining a differential pattern of relationships between variables (Romero-Martínez, Lila, & Moya-Albiol, 2015). Hence, our results reinforce the need to study these hormonal parameters together rather than analyze in isolation their relationships with cognitive and psychological variables (Sollberger & Ehlert, 2015), in order to offer a deeper understanding of the underlying mechanism that could explain proneness to violence.
Lastly, the main limitation of our study is the small sample size; for this reason, the findings should be considered preliminary. Additionally, we only used a single hormonal measurement with low reliability. Future studies should therefore assess several more reliable hormonal measurements and explore these patterns in larger samples. Nevertheless, our data is novel as no studies have previously analyzed the underlying mechanisms that could explain the impairments in the emotion decoding process, a relevant cognitive process for behavior and/or emotional regulation. It would also be useful to analyze whether or not type I IPV perpetrators present a cognitive profile similar to type II perpetrators.
In conclusion, this study shows that attention switch costs and T could be indirect modulators of aggressive behavior through their effects on the processing of emotional information. Our novel data could contribute to a deeper understanding of the factors involved in IPV perpetration and the risk of becoming violent, such as cognitive deficits and hormonal parameters. Finally, greater knowledge of the relationships between these variables could be useful in developing psychotherapeutic intervention programs for IPV perpetrators.
Conflict of Interest
The authors of this article declare no conflict of interest.
References
1. Aiken L.S., West S.G. Multiple regression: Testing and interpreting interactions. Newbury Park, CA: Sage, 1991. [ Links ]
2. Babcock J.C., Green C.E., Webb S.A. Decoding deficits of different types of batterers during presentation of facial affect slides. Journal of Family Violence. 2008;23:295-302. [ Links ]
3. Baron-Cohen S., Wheelwright S., Hill J., Raste Y., Plumb I. The "reading the mind in the eyes" Test revised version: A study with normal adults, and adults with Asperger syndrome or high-functioning autism. Journal of Child Psychology and Psychiatry. 2001;42:241-251. [ Links ]
4. Baron-Cohen S., Wheelwright S., Skinner R., Martin J., Clubley E. The autism-spectrum quotient (AQ): evidence from Asperger syndrome/high-functioning autism, males and females, scientists and mathematicians. Journal of Autism and Developmental Disorders. 2001;31:5-17. [ Links ]
5. Bergvall A.H., Wessely H., Forsman A., Hansen S. A deficit in attentional set-shifting of violent offenders. Psychological Medicine. 2001;31:1095-1105. [ Links ]
6. Buitelaar N.J., Posthumus J.A., Buitelaar J.K. ADHD in Childhood and/or Adulthood as a Risk Factor for Domestic Violence or Intimate Partner Violence: A Systematic Review. Journal of Attention Disorders. (first published on May 20, 2015). pii: 1087054715587099. [ Links ]
7. Carton J.S., Kessler E.A., Pape C.L. Nonverbal decoding skills and relationship well-being in adults. Journal of Nonverbal Behavior. 1999;23:91-100. [ Links ]
8. Catalá-Miñana A., Lila M., Conchell R., Romero-Martínez A., Moya-Albiol L. ¿Se benefician de los programas de intervención que no tratan específicamente el consumo de alcohol los maltratadores con problemas de consumo abusivo?. Psychosocial Intervention. 2013;22:135-143. [ Links ]
9. Cepeda N.J., Cepeda M.L., Kramer A.F. Task switching and attention deficit hyperactivity disorder. Journal of Abnormal Child Psychology. 2000;28:213-226. [ Links ]
10. Cohen R.A., Brumm V., Zawacki T.M., Paul R., Sweet L., Rosenbaum A. Impulsivity and verbal deficits associated with domestic violence. Journal of the International Neuropsychological Society. 2003;9:760-770. [ Links ]
11. Dawson J.F. Moderation in management research: What, why, when, and how. Journal of Business and Psychology. 2014;29:1-19. [ Links ]
12. Dawson J.F., Richter A.W. Probing three-way interactions in moderated multiple regression: Development and application of a slope difference test. Journal of Applied Psychology. 2006;91:917-926. [ Links ]
13. Derntl B., Windischberger C., Robinson S., Kryspin-Exner I., Gur R.C., Moser E., Habel U. Amygdala activity to fear and anger in healthy young males is associated with testosterone. Psychoneuroendocrinology. 2009;34:687-693. [ Links ]
14. Fang X., Massetti G.M., Ouyang L., Grosse S.D., Mercy J.A. Attention-deficit/hyperactivity disorder, conduct disorder, and young adult intimate partner violence. Archives of General Psychiatry. 2010;67:1179-1186. [ Links ]
15. Fuermaier A.B., Tucha L., Koerts J., Aschenbrenner S., Weisbrod M., Lange K.W., Tucha O. Cognitive complaints of adults with attention deficit hyperactivity disorder. Archives of Clinical Neuropsychology. 2014;28:1104-1122. [ Links ]
16. Gottman J.M., Jacobson N.S., Rushe R.H., Shortt J.W. The relationship between heart rate reactivity, emotionally aggressive behavior, and general violence in batterers. Journal of Family Psychology. 1995;9:227-248. [ Links ]
17. Gracia E., Rodriguez C.M., Lila M. Preliminary evaluation of an analog procedure to assess acceptability of intimate partner violence against women: the Partner Violence Acceptability Movie Task. Frontiers in psychology. 2015; [ Links ]6(1567).
18. Holtzworth-Munroe A., Smutzler N. Comparing the emotional reactions and behavioral intentions of violent and nonviolent husbands to aggressive, distressed, and other wife behaviors. Violence and Victims. 1996;11:319-339. [ Links ]
19. Kinsbourne M., Bemporad B. Lateralization of emotions: A model and the evidence. In N. Fox, & R. J. Davidson (Eds.), The Psychobiology of Affective Development., Hillsdale, NJ: Erlbaum, [ Links ] 1984.
20. Lila M., Gracia E., Murgui S. Psychological adjustment and victim-blaming among intimate partner violence offenders: The role of social support and stressful life events. The European Journal of Psychology Applied to Legal Context. 2013;5:147-153. [ Links ]
21. Lila M., Oliver A., Catalá-Miñana A., Conchell R. Recidivism risk reduction assessment in batterer intervention programs: A key indicator for program efficacy evaluation. Psychosocial Intervention. 2014;23:217-223. [ Links ]
22. Lila M., Oliver A., Galiana L., Gracia E. Predicting success indicators of an intervention programme for convicted intimate partner violence offenders: the Contexto Programme. European Journal of Psychology Applied to Legal Context. 2013;5:73-95. [ Links ]
23. Little A.C. The influence of steroid sex hormones on the cognitive and emotional processing of visual stimuli in humans. Frontiers in Neuroendocrinology. 2013;34:315-328. [ Links ]
24. Marshall A.D., Holtzworth-Munroe A. Recognition of wives´ emotional expressions: a mechanism in the relationship between psychopathology and intimate partner violence perpetration. Journal of Family Psychology. 2010;24:21-30. [ Links ]
25. Montoya E.R., Terburg D., Bos P.A., van J. Testosterone, cortisol, and serotonin as key regulators of social aggression: A review and theoretical perspective. Motivation and Emotion. 2012;36:65-73. [ Links ]
26. Morrison R.L., Bellack A.S. The role of social perception in social skill. Behavior Therapy. 1981;12:69-79. [ Links ]
27. Moya-Albiol L., Herrero N., Bernal M.C. The neural bases of empathy. Revista de Neurología. 2010;50:89-100. [ Links ]
28. Noordermeer, S. D., Luman, M., Buitelaar, J. K., Hartman, C. A., Hoekstra, P. J., Franke, B., ...Oosterlaan, J. (2015). Neurocognitive Deficits in Attention-Deficit/Hyperactivity Disorder With and Without Comorbid Oppositional Defiant Disorder. Journal of Attention Disorders (first published on October 20, 2015). pii: 1087054715606216. [ Links ]
29. Polderman T.J., Hoekstra R.A., Vinkhuyzen A.A., Sullivan P.F., van S., Posthuma D. Attentional switching forms a genetic link between attention problems and autistic traits in adults. Psychological Medicine. 2013;43:1985-1996. [ Links ]
30. Romero-Martínez Á., Lila M., Catalá-Miñana A., Williams R.K., Moya-Albiol L. The contribution of childhood parental rejection and early androgen exposure to impairments in socio-cognitive skills in intimate partner violence perpetrators with high alcohol consumption. International Journal of Environmental Research and Public Health. 2013;10:3753-3770. [ Links ]
31. Romero-Martínez A., Lila M., Moya-Albiol L. The testosterone/cortisol ratio moderates the proneness to anger expression in antisocial and borderline intimate partner violence perpetrators. The Journal of Forensic Psychiatry & Psychology. 2015. http://dx.doi.org/10.1080/14789949.2015.1096415. [ Links ]
32. Romero-Martínez A., Lila M., Sariñana-González P., González-Bono E., Moya-Albiol L. High testosterone levels and sensitivity to acute stress in perpetrators of domestic violence with low cognitive flexibility and impairments in their emotional decoding process: a preliminary study. Aggressive Behavior. 2013;39:355-369. [ Links ]
33. Romero-Martínez A., Moya-Albiol L. Neuropsychology of perpetrators of domestic violence: the role of traumatic brain injury and alcohol abuse and/or dependence. Revista Neurología. 2013;57:515-522. [ Links ]
34. Ronay R., Carney D.R. Testosterone's Negative Relationship With Empathic Accuracy and Perceived Leadership Ability. Social Psychological and Personality Science. 2013;4:92-99. [ Links ]
35. Rusconi E., Ferri F., Viding E., Mitchener-Nissen T. XRIndex: a brief screening tool for individual differences in security threat detection in x-ray images. Frontiers in Human Neuroscience. 2015;9(439). [ Links ]
36. Semrud-Clikeman M., Guy K., Griffin J.D., Hynd G.W. Rapid naming deficits in children and adolescents with Reading Disabilities and Attention Deficit Hyperactivity Disorder. Brain and Language. 2000;74:70-83. [ Links ]
37. Serrano V.J., Owens J.S., Hallowell B. Where Children With ADHD Direct Visual Attention During Emotion Knowledge Tasks: Relationships to Accuracy, Response Time, and ADHD Symptoms. Journal of Attention Disorders.(first published online on July 3, 2015). pii: 1087054715593632. [ Links ]
38. Sizoo B.B., van den Brink W., Gorissen-van Eenige M., Koeter M.W., van Wijngaarden P.J., van der Gaag R.J. Using the Autism-spectrum quotient to discriminate Autism Spectrum Disorder from ADHD in adult patients with and without comorbid Substance Use Disorder. Journal of Autism and Developmental Disorders. 2009;39:1291-1297. [ Links ]
39. Sollberger S., Ehlert U. How to use and interpret hormone ratios. Psychoneuroendocrinology. 2015;63:385-397. [ Links ]
40. Stanton S.J., Wirth M.M., Waugh C.E., Schultheiss O.C. Endogenous testosterone levels are associated with amygdala and ventromedial prefrontal cortex responses to anger faces in men but not women. Biological Psychology. 2009;81:118-122. [ Links ]
41. Tannock R., Martinussen R., Frijters J. Naming speed performance and stimulant effects indicate effortful, semantic processing deficits in attention-deficit/hyperactivity disorder. Journal of Abnormal Child Psychology. 2000;28:237-252. [ Links ]
42. Terburg D., Morgan B., van J. The testosterone-cortisol ratio: A hormonal marker for proneness to social aggression. International Journal of Law and Psychiatry. 2009;32:216-223. [ Links ]
43. van Honk J., Schutter D.J. Testosterone reduces conscious detection of signals serving social correction: implications for antisocial behavior. Psychological Science. 2007;18:663-667. [ Links ]
44. van Honk J., Schutter D.J. Vigilant and avoidant responses to angry facial expressions. In E. Harmon-Jones & P. Winkielman (Eds.), Social Neuroscience, Integrating Biological and Psychological Explanations of Social Behavior, New York, NY: The Guilford Press, 2007. pp. 197-223. [ Links ]
45. van Honk J., Tuiten A., van den Hout M., Koppeschaar H., Thijssen J., de Haan E., Verbaten R. Baseline salivary cortisol levels and preconscious selective attention for threat. A pilot study. Psychoneuroendocrinology. 1998;23:741-747. [ Links ]
46. Williams, L. M., Hermens, D. F., Palmer, D., Kohn, M., Clarke, S., Keage, H., ...Gordon, E. (2008). Misinterpreting emotional expressions in attention-deficit/hyperactivity disorder: evidence for a neural marker and stimulant effects. Biological Psychiatry, 63, 917-26. [ Links ]
47. Wirth M.M., Schultheiss O.C. Basal testosterone moderates responses to anger faces in humans. Physiology & Behavior. 2007;90:496-505. [ Links ]
48. Wymbs B.T., Dawson A.E., Suhr J.A., Bunford N., Gidycz C.A. ADHD Symptoms as Risk Factors for Intimate Partner Violence Perpetration and Victimization. Journal of Interpersonal Violence. 2015. (Published first on line on May 28, 2015). pii: 0886260515586371. [ Links ].
![]() Correspondence:
Correspondence:
Ángel Romero-Martínez
Department of Psychobiology.
University of Valencia.
Avenida Blasco Ibañez, 21.
46010 Valencia, Spain.
Angel.Romero@uv.es
Received 14 November 2015
Accepted 14 January 2016













