Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista de Osteoporosis y Metabolismo Mineral
versión On-line ISSN 2173-2345versión impresa ISSN 1889-836X
Rev Osteoporos Metab Miner vol.13 no.4 Madrid nov./dic. 2021 Epub 12-Dic-2022
https://dx.doi.org/10.4321/s1889-836x2021000400002
SPECIAL ARTICLE
Sequential treatment in osteoporosis. New trends
1Rheumatology Service. University Hospital Parc Taulí (UAB). Sabadell (Spain)
2Parc Taulí Research Institute (I3PT) (Spain)
3Obstetrics and Gynecology Service. Cruces University Hospital (Spain)
4University of the Basque Country. Baracaldo (Spain)
Treatment for osteoporosis is usually long term, so it is often necessary to implement a sequential treatment strategy. The clinician must know not only how to select the best available therapy in each clinical situation, but also how to discontinue or change treatment at a certain point in the evolution of the disease.
Here, we briefly review the mechanisms of action and the consequences of discontinuing each of the drugs for osteoporosis, as well as what happens with the different treatment sequences.
Discontinuation of denosumab has clearly negative consequences for the skeleton, and only bisphosphonates, due to its particular remaining antiresorptive effect, could be discontinued for a limited time (drug holidays).
Switching from antiresorptive to another antiresorptive with a different mechanism of action is an option that may be favorable in the management of some patients with osteoporosis. Switching from antiresorptive to anabolic may be associated with an initial bone mineral density loss that does not appear to have negative consequences on antifracture efficacy. Starting with anabolic (teriparatide or romosozumab) and subsequently switching to antiresorptive is the best treatment sequence, so it could be the preferred option in patients who present a very high risk of fracture.
Key words sequential therapy; osteoporosis; treatment; antiresorptive; bone formation
INTRODUCTION
Osteoporosis is a chronic systemic disease characterized by a deterioration in bone density and/or quality, which predisposes to a greater risk of fracture1.
All treatments for osteoporosis have shown greater or lesser efficacy in reducing the risk of fracture, especially in postmenopausal women2. This beneficial effect occurs from the modification of the bone remodeling process with the consequent increase in bone mineral density (BMD) and/or a modification of the bone's microarchitecture, although this requires a prolonged treatment over years in most cases.
Although treatment with antiresorptives, menopausal hormone therapy [THM], selective estrogen receptor modulators [SERM], bisphosphonates [BP] and denosumab [DMAB]) can be maintained for at least 5-10 years, the balance between risk and benefit should always be taken into account, since in prolonged treatments with the highest antiresorptive potency drugs (BP and DMAB) an increased risk of some very infrequent complications such as osteonecrosis of the maxilla or atypical femur fracture has been described3,4.
In the case of bone-forming drugs (teriparatide [TPTD] and abaloparatide [ABL]) or dual-effect drugs (romosozumab [ROMO]), the duration of treatment is limited to a shorter period of time. TPTD and ABL (the latter not authorized for marketing in Europe) are not recommended to be administered for more than 2 years5,6, and ROMO should not be administered beyond 12 months7.
In recent years, the term sequential treatment has been gaining importance in the management of patients with osteoporosis, that is, the sequential use of different treatments to achieve maximum efficacy with the least risk of complications8.
ADVANTAGES OF A SEQUENTIAL TREATMENT
In managing patients with osteoporosis, a sequential treatment strategy over the years is often required. After using a treatment for a certain period of time, it can be beneficial to discontinue or replace it with a different one, as a sequence.
There are several reasons that justify sequential therapy in patients with osteoporosis:
Osteoporosis is a chronic disease that requires long-term treatment, probably for many years.
Some treatments for osteoporosis have a maximum recommended duration (for example, 2 years for TPTD and ABL or 1 year for ROMO)
Some treatments are associated with some infrequent complications, especially if they are used for more than a certain time (for example, atypical femur fractures are more frequent in patients treated with BP for more than 5 years).
With some of the osteoporosis treatments there is a loss in BMD gain if a different one is not administered after its discontinuation.
In some cases, greater efficacy is achieved when a sequence of 2 or more drugs is used than if a single drug is used in the same period of time.
Patients tend to get tired and lose adherence to treatments, when they are administered very chronically for years, without changes in the administration guidelines9.
Sometimes there is an inadequate response to treatment, either due to a loss of BMD or the development of new fractures despite following the treatment correctly; or simply an insufficient response to reach a therapeutic objective ("treat-to-target" strategy).
In all these situations we should propose a change in the treatment for osteoporosis. Although in some cases a temporary interruption might be better, in most situations it would be more appropriate to substitute a different treatment.
It is thus very important that the clinician knows the advantages and disadvantages of the different sequences of treatments in the patient with osteoporosis.
MECHANISM OF ACTION OF DRUGS FOR OSTEOPOROSIS
For the proper management of the patient with osteoporosis, knowing how to select the best available therapy in each clinical situation is crucial, as well as discontinuing or changing when the risk-benefit balance recommends. It is therefore essential to be very familiar with the mechanism of action, effectiveness and safety of each of the drugs.
Menopausal hormone therapy (THM)
Treatment with estrogens, alone or in combination with progestogens (depending on whether the patient retains her uterus or not), acts through the activation of nuclear estrogen receptors (ERα and ERβ) distributed in different body tissues. ERα receptors predominate in bone, their stimulation on the one hand inhibits osteoclastogenesis from the inhibition of the ligand of the activating receptor for nuclear factor kappa B (RANKL) and the stimulation of osteoprotegerin (OPG)10, and, on the other hand, effects on bone formation have been described, through the stimulation of factors such as IGF-1 or the inhibition of sclerostin11. In bone remodeling, MHT has an antiresorptive effect, with the consequent increase in BMD and a decrease in the risk of vertebral, non-vertebral and hip fracture, especially when administered in the first 5-10 years after the menopause12.
However, given the much debated risk of breast cancer and cardiovascular complications in prolonged estrogen treatments only confirmed in treatments started beyond the age of 60, it is currently assumed that MHT is reserved for women with premature ovarian failure and for those under 60 years of age with overt climacteric symptoms, in which the benefits clearly outweigh the risks13.
Selective estrogen receptor modulators (SERMs)
SERMs are a group of drugs with agonist activity at bone estrogen receptors, inhibiting resorption, and antagonist at estrogen receptors in the breast and endometrium, which is why they can provide the beneficial effect of estrogens in patients with osteoporosis, minimizing the effects of adverse effects on other organs14. Raloxifene (RLX) and bazedoxifene are the SERMs indicated in women with postmenopausal osteoporosis, since they increase BMD and reduce the risk of vertebral fracture. The unproven efficacy in reducing the risk of non-vertebral or hip fracture and the increased risk of venous thromboembolism restrict its use to postmenopausal women under 70 years of age at risk of vertebral fracture and low risk of hip fracture and venous thrombosis15
Bisphosphonates (BP)
BPs are compounds derived from inorganic pyrophosphate with a high affinity for bone hydroxyapatite, which, when taken up by endocytosis by the osteoclast, inhibit farnesyl pyrophosphate synthetase and lead to cell apoptosis, with the consequent inhibitory effect on bone resorption16.
Alendronate (ALN) and risedronate (RIS), taken by mouth, and zoledronic acid (ZOL) by intravenous route, are the BP most widely used and recommended by clinical guidelines, given the demonstrated efficacy both in increasing BMD and in reducing the risk of vertebral, non-vertebral and hip fracture15,17. Ibandronate (IBN) is another oral BP, which, although it offers the advantage of its monthly administration, has only shown a reduction in the risk of vertebral fracture17.
Some infrequent complications associated with prolonged treatment with BP have been described, such as osteonecrosis of the maxilla and atypical fracture of the femur, with incidences of 0.1-1/10,000 and 1-2/10,000 patient-years, respectively15.
Denosumab (DMAB)
DMAB is a completely human monoclonal antibody, administered subcutaneously, with a potent antiresorptive activity, through the blockade of RANKL18. DMAB has been shown to continuously increase BMD, for at least 10 years, and reduce the risk of vertebral, non-vertebral, and hip fractures19. DMAB has demonstrated superiority to BP in terms of increases in BMD, and its prolonged treatment has also been associated with osteonecrosis of the maxilla and atypical femur fracture, although also with a very low incidence20.
Teriparatide (TPTD) and abaloparatide (ABL)
TPTD and ABL are synthetic parathormone analogues (PTH 1-34 and PTHrP 1-34 respectively) with bone-forming activity and approved for treating osteoporosis with high risk of fracture (the EMA has only approved TPTD). Both drugs bind to the PTH receptor (although with affinity to different conformational states) and inhibit sclerostin, thereby stimulating osteoblastogenesis, and decreasing apoptosis of osteoblasts. Later, and to a lesser degree, they increase RANKL secretion and therefore bone resorption21
Both treatments produce an increase in bone mass and improve the microarchitecture of the skeleton, especially in the trabecular bone, increasing bone strength and reducing the risk of fracture.
Both drugs are indicated in the treatment of patients with osteoporosis at high risk of fracture and have shown superiority over BP22, but should not be administered beyond 2 years according to the technical data sheet.
Romosozumab (ROMO)
ROMO is a humanized antibody for subcutaneous administration with a dual effect on bone remodeling, as it inhibits sclerostin and secondarily RANKL, producing a rapid, but transitory, increase in bone formation (osteoformer) associated with a more sustained decrease over time of bone formation. resorption. As a consequence, with this treatment there is a marked increase in BMD (greater than with TPTD) and a decrease in the risk of fracture23,24.
Unlike what happens with TPTD or ABL, the increase in bone formation that occurs with ROMO is mainly due to a marked increase in bone shaping (bone formation after activation of the lining cells in quiescent areas, without a process of prior resorption).
The beneficial effects of ROMO are both on the trabecular bone and on the cortical bone.
However, in its development, doubts have appeared in relation to cardiovascular safety, which have not yet been resolved. In the ARCH study, comparing with ALN, a higher incidence of serious cardiovascular events was observed in women treated with ROMO25.
ROMO has been approved by the EMA, although as of the writing of this article it is not yet marketed in Spain.
According to the technical data sheet, it can only be administered for 12 months and is contraindicated in patients with a history of myocardial infarction or stroke7. In addition, the cardiovascular risk of patients should be assessed, based on risk factors, before and during its administration.
DISCONTINUING OSTEOPOROSIS TREATMENTS
THM discontinuation
Discontinuation of estrogen treatment involves increased remodeling in some patients, with a rapid decrease in BMD and progressive loss of anti-fracture efficacy, which can be prevented with the administration of BP26.
SERM treatment discontinuation
Discontinuation of treatment with X-ray is accompanied by a loss of BMD in both the lumbar spine and the femur, although less than that produced with estrogen interruption and proportional to the physiological loss that occurs with age27.
Discontinuation of BP treatment
BPs are the only drugs for the treatment of osteoporosis with a residual effect on the skeleton after its discontinuation, the duration of which will depend on the affinity for hydroxyapatite of each BP, ranging between 1-2 years for RIS and IBN and 2-3 years for ALN and ZOL28.
After discontinuation of treatment with BP, there is no immediate increase in remodeling, but rather this increase will appear progressively over time, which implies a stability or slight loss of BMD, while this residual effect lasts28,29.
This property of BP justifies the possibility of considering a temporary suspension of treatment or therapeutic vacations in patients with low risk of fracture28.
Discontinuation of DMAB treatment
DMAB has a reversible RANKL inhibition effect, so that discontinuation of treatment produces a rapid and marked increase in bone remodeling, with elevation of formation and resorption markers, as early as 9 months after the last dose, reached levels even higher than those prior to treatment, and which did not normalize until after 24 months30. This "rebound" effect on remodeling is accompanied by a rapid loss of BMD, which can reach the values prior to the start of treatment (figure 1)30, and, in some patients, an increased risk of vertebral fractures, particularly multiple ones31. The incidence of vertebral fractures in women with postmenopausal osteoporosis who discontinue DMAB has been estimated between 8.5-10.5% in the 12-18 months after discontinuation32,33, although some of these fractures could be due not only to the effect "Rebound", but rather the return to a high-risk situation due to previous vertebral fractures that patients had before starting treatment with DMAB.
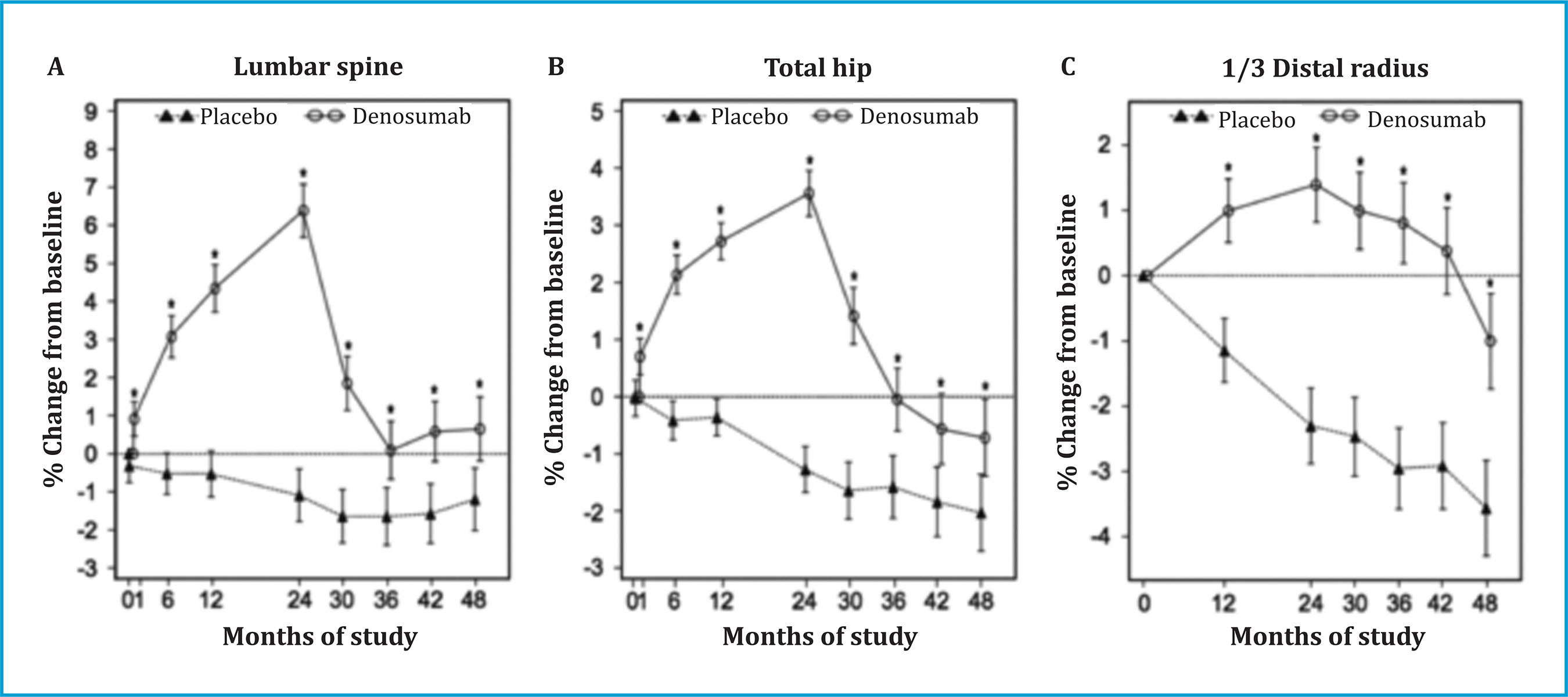
Figure 1. Changes in bone mineral density of the lumbar spine (A), total hip (B) and 1/3 radius (C) during the 24 months of treatment with denosumab and during the 24 months after discontinuation. (Modified from Bone HG, et al.) 30
A systematic review of the literature carried out by a European Calcified Tissue Society (ECTS) working group demonstrated that the risk factors for the presentation of multiple vertebral fractures in patients who discontinue DMAB are young age, having prevalent vertebral fractures, a duration of treatment greater than 2.5 years, a greater gain in hip BMD during treatment and a greater decrease in hip BMD after discontinuation34. Some case series indicate that previous treatment with BP could mitigate the rebound effect of remodeling (lower increase in markers) that occurs after discontinuation with DMAB, although it is not clear whether this attenuation prevents loss of BMD and fractures34.
The administration of another potent antiresorptive treatment such as BP in patients who discontinue DMAB appears to have partial efficacy on the rebound effect that occurs. Experts recommend that patients who have received ABDM for less than 2.5 years and are at low risk of fracture can be treated with an oral BP for a minimum of 1-2 years (depending on bone markers and BMD). However, patients who have received DMAB for a longer time, those still at high risk of fracture, or those who cannot tolerate oral BP should receive a dose of ZOL 6 months after the last dose of DMAB, which could be repeated at 3-6 months, depending on the levels of bone markers34.
Experts conclude that the indication for DMAB should be carefully assessed, especially in young patients, and that in those who discontinue treatment, a BP should be administered 6 months after the last DMAB injection, while there are no more data from new clinical trials.
Based on the data from the systematic review by Tsourdi et al.34, we present a practical decision algorithm in patients receiving treatment with DMAB (figure 2).
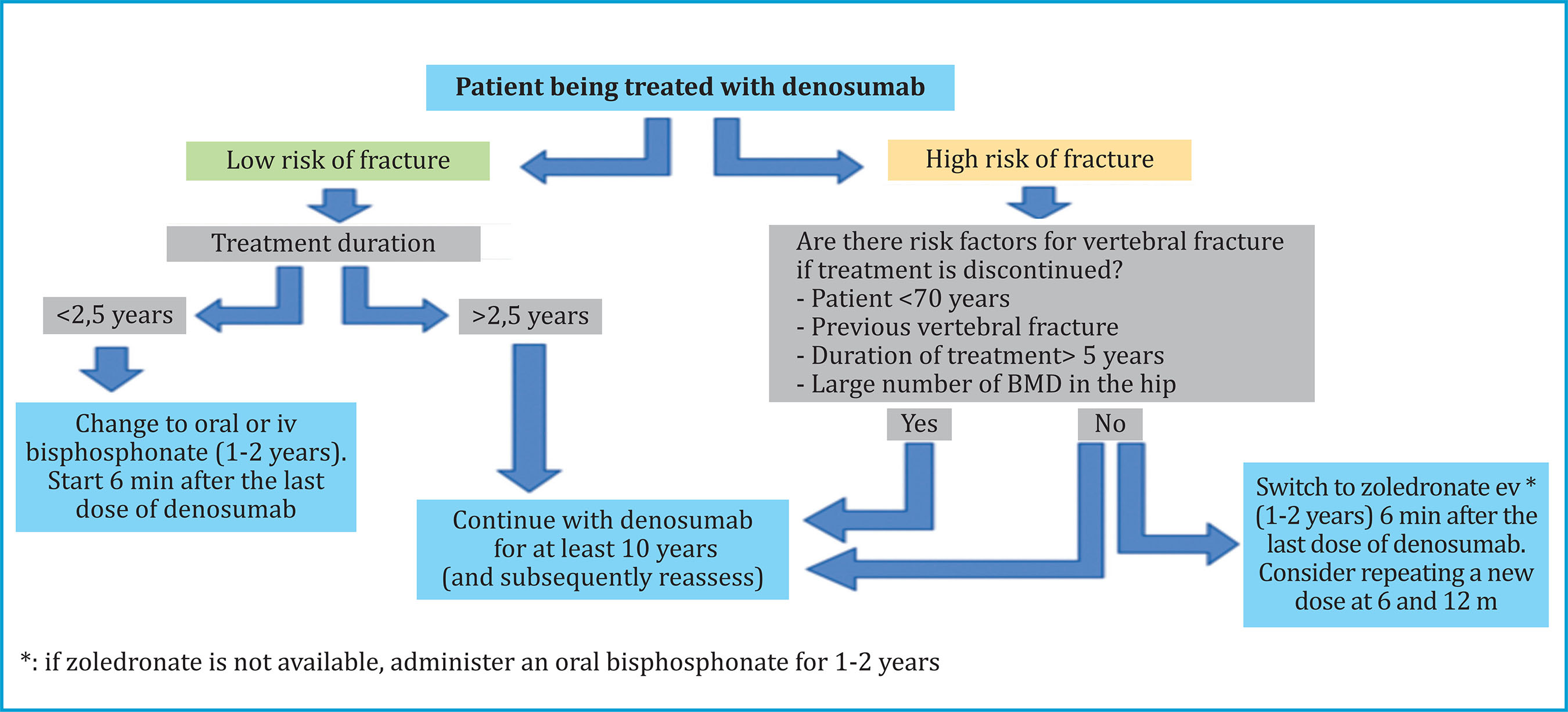
Figure 2. Decision algorithm in patients receiving denosumab treatment for osteoporosis. (Adapted from Tsourdi E, et al.) 34
In case an invasive dental procedure is necessary during treatment with DMAB, experts recommend that it be performed after the 5th month from the last dose, and the next dose is administered as soon as the surgical wound has healed34.
Discontinuation of treatment with TPTD and ABL
Discontinuation of TPTD (and probably ABL) is accompanied by a loss of BMD in the spine and femur in the subsequent 12 months, being more marked in postmenopausal women (7.1%) than in men (4.1%)35. The administration of an antiresorptive after discontinuation of TPTD maintains or even increases BMD36,37.
ROMO treatment discontinuation
The effect of ROMO on bone remodeling is reversible, and its discontinuation is accompanied by a normalization of training and a "rebound" effect of resorption, which translates into a rapid loss of BMD in both the lumbar spine and the hip. (figure 3)38. The previous administration of ALN seems to mitigate this "rebound" effect and attenuate the loss of BMD.
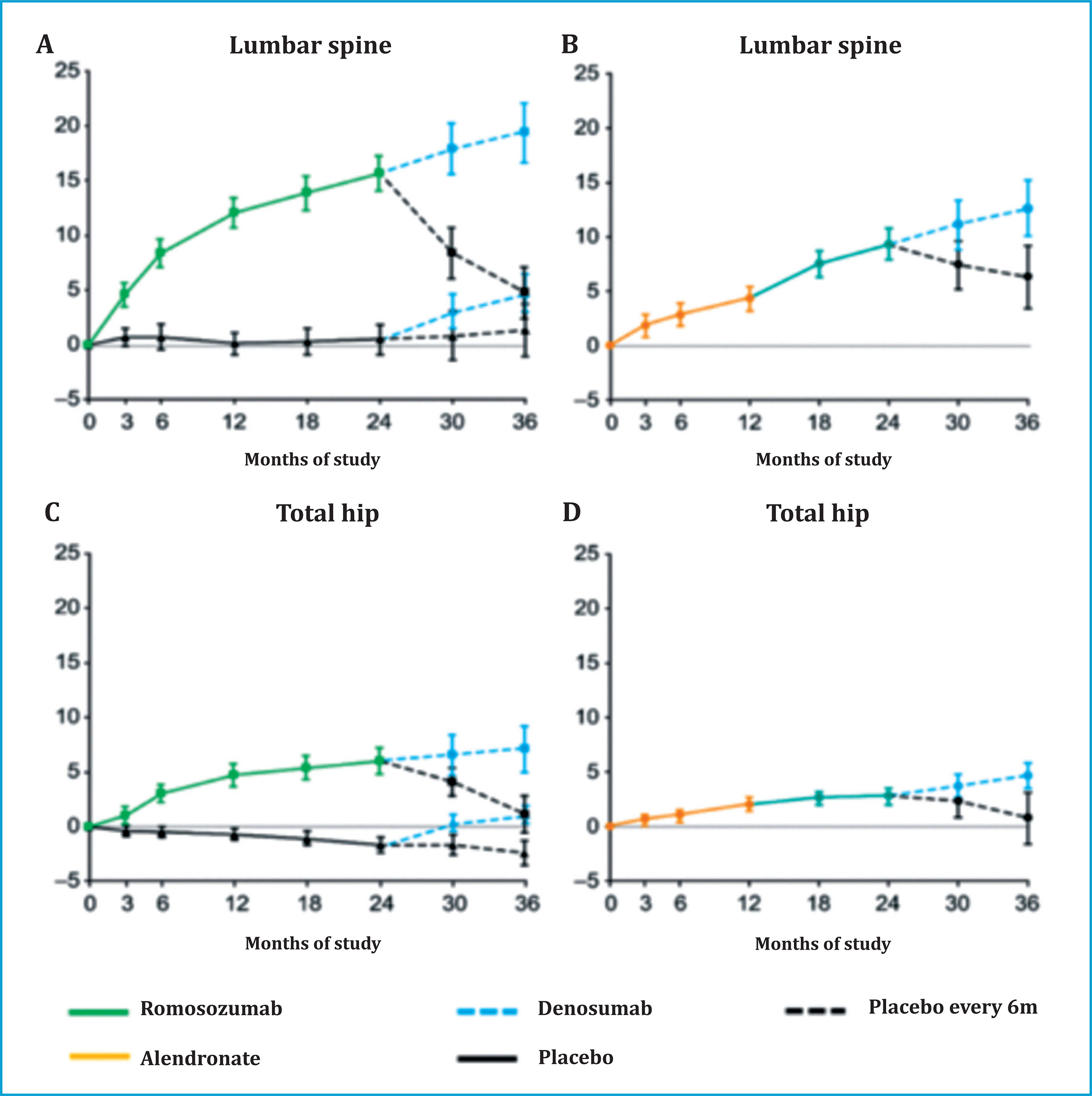
Figure 3. Changes in bone mineral density in the lumbar spine (A) and total hip (C) during treatment with romosozumab and after discontinuation. The loss of BMD is less in patients who had previously received alendronate (B and D). (Modified from McClung MR, et al.)38
ANTIRESORPTIVE FOLLOWED BY ANOTHER ANTIRESORPTIVE
The administration of DMAB after having received ALN achieves greater suppression of remodeling and greater gain in BMD than the same treatment time with ALN alone39. The ALN-DMAB sequence also appears more efficient than the ALN-ZOL sequence, at least in terms of DMO gain40.
Therefore, in patients treated with oral BP who present a therapeutic failure or do not reach the goal of treatment, a reasonable option could be the transition to DMAB or ZOL, as long as its indication is clear and the administration of bone former.
On the other hand, and as mentioned above, in order to minimize the rebound effect that occurs on bone remodeling after discontinuation of DMAB, it seems advisable to administer a BP (oral or intravenous) at 6 months of the last injection of DMAB (figure 2).
ANTIRESORPTIVE FOLLOWED BY OSTEOFORMER
Previous treatment with BP, especially with those with a higher affinity for hydroxyapatite (ALN and ZOL), seems to attenuate the BMD gain that occurs with TPTD41, observing a decrease in the first 6 months of treatment42, although anti-fracture efficacy seems to be maintained as demonstrated in a post-hoc analysis of the VERO study43.
However, the transition from RLX to TPTD does not seem to attenuate the osteoforming effect or the BMD gain of the latter42.
In the DATA-Switch study, the women who received DMAB for 2 years and switched to TPTD presented an increase in bone remodeling, with a marked decrease in BMD in the hip in the first year, but which recovered in the second year, returning to BMD values at the onset of TPTD (figure 4)44. Although there is no evidence whether this transient loss of BMD is accompanied by an increased risk of fracture, some experts suggest not discontinuing treatment with DMAB if TPTD (or ABL) is to be indicated, that is, combination therapy45.
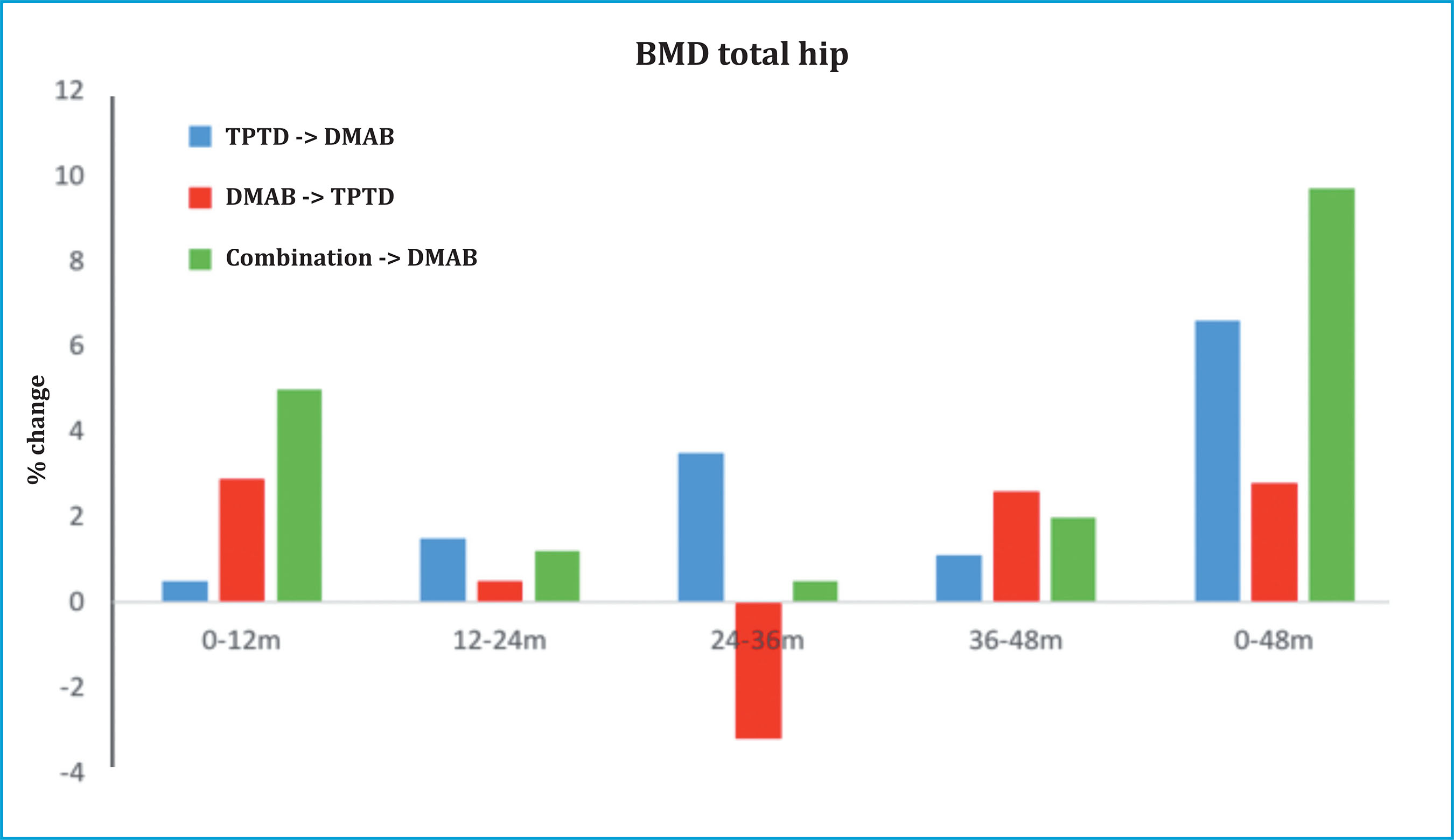
Figure 4. DATA-Switch study. The denosumab-teriparatide sequence produces a marked decrease in bone mineral density in the hip in the first year of transition, which recovers in the second year (red). The teriparatide-denosumab sequence achieves a marked gain in BMD in the hip (blue), similar to the combination therapy followed by denosumab (green). (Adapted from Leder BZ, et al.)44
In the STRUCTURE study, it was observed that, as occurs in the transition to TPTD, patients who received ALN and switched to ROMO had a lower increase in BMD than patients who received ROMO without prior antiresorptive treatment46.
Although anabolic treatment with TPTD or ROMO seems more effective in patients not previously treated with antiresorptive drugs, patients who, despite receiving antiresorptive treatment, present a high risk of fracture, probably obtain a greater benefit with the change to bone-forming treatment, except in the case of DMAB, which seems better to maintain a combination therapy with TPTD46.
The DMAB-ROMO sequence could be more favorable, since it has been reported that a second 12-month cycle of ROMO in patients who had received 12 months of DMAB after a first 2-year cycle of ROMO does increase spinal BMD lumbar (2.3%) and maintain hip BMD (figure 5)47. However, it must be taken into account that these results have been extracted from a cohort of only 14 patients, and in whom DMAB had only been administered for one year.
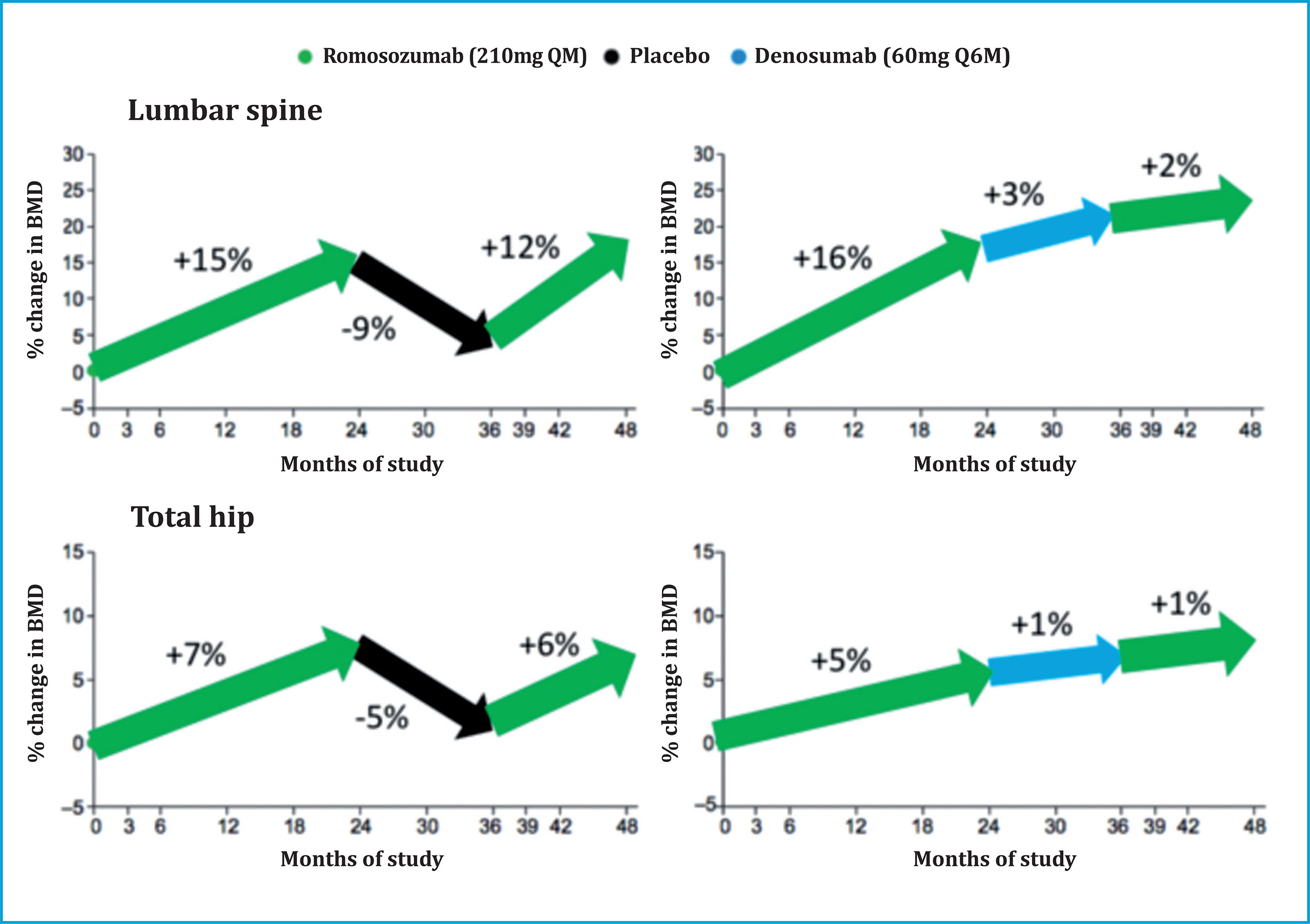
Figure 5. Sequential denosumab-romosozumab treatment after a 2-year cycle of romosozumab succeeds in increasing bone mineral density in the lumbar spine and hip. (Adapted from Kendler DL, et al.)47
OSTEOFORMER FOLLOWED BY ANTIRESORPTIVE
Sequential TPTD-RLX treatment has been shown to be beneficial. RLX manages to maintain or even increase the BMD gain achieved with previous treatment with TPTD48.
Equally beneficial is the TPTD-BP sequence. The BMD gain achieved with TPTD can be increased if, after its discontinuation, a BP is administered, also maintaining the anti-fracture efficacy49.
But the TPTD-DMAB sequence is probably the one that provides the highest BMD gain (18% in the lumbar spine and 8% in the femoral neck after 2 years of treatment with TPTD followed by another 2 years of treatment with DMAB)44.
Depending on the clinical characteristics of the patient and the risk of fracture, we could select one antiresorptive or another after completing the two years of treatment with TPTD (figure 6).
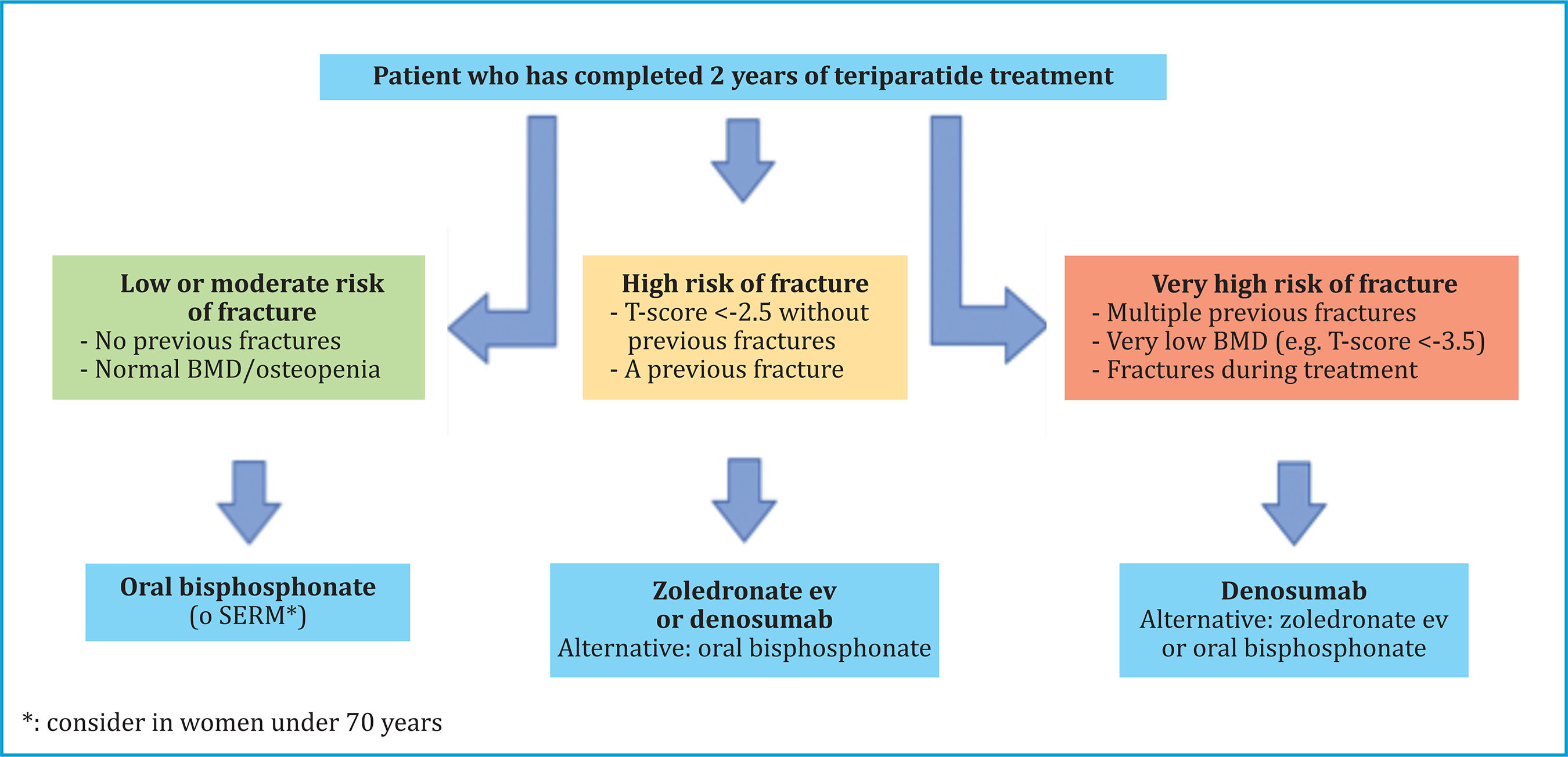
Figure 6. Decision algorithm in patients who have completed two years of treatment with teriparatide for osteoporosis
A study with 68 postmenopausal women has recently been published comparing the efficacy of 3 repeated cycles of 6 months of TPTD followed by 6 months of DMAB (total 36 months), versus a standard treatment of 18 months of TPTD followed by 18 months of DMAB. After 3 years of treatment, the patients who received standard sequential therapy achieved greater gains in BMD in the lumbar spine (16% vs 12%; p=0.04), with no changes in hip and radius BMD. Cyclical therapy achieved better results in hip and radius BMD at 18 months, so according to the authors, repeated cyclical treatment of TPTD and DMAB could be potentially useful in patients at imminent risk of fracture, especially in patients at higher risk of non-vertebral fracture50.
The ABL-BP sequence is also effective. In the extension of the ACTIVE study, the administration of ALN after ABL was shown to increase the BMD gain achieved with ABL and to maintain anti-fracture efficacy51.
There are also data on the ROMO-antiresorptive sequence. In the ARCH study, it was observed how the BMD gain achieved with ROMO was maintained after switching to ALN. Regarding anti-fracture efficacy, the reduction in the risk of vertebral fracture was maintained and the reduction in the risk of non-vertebral fracture was increased52.
In the FRAME study, the sequence 1 year of ROMO followed by 1 year of DMAB was equally beneficial in terms of gain in BMD and reduction of the risk of vertebral fracture. Although a lower incidence of clinical fractures and non-vertebral fractures was also observed with this sequential treatment than with a single year of ABMD, it must be said that the differences were not significant24.
THERAPEUTIC HOLIDAYS
In order to minimize the risk of long-term complications or improve patient adherence to treatment, a temporary interruption of antiresorptive treatment may be considered in the management of osteoporosis, but only with BP, both oral and intravenous, given its remaining effect on the skeleton29,52,53.
The working group on the management of patients with osteoporosis on prolonged treatment with BP of the American Society for Research in Bone Metabolism (ASBMR) recommends considering therapeutic holidays after at least 5 years of treatment with oral BP or 3 years of ZOL, but only in patients under 70 years of age, without fractures before or during treatment and who do not have a hip BMD in the osteoporosis range or present risk factors for fracture or a high risk according to FRAX28.
The duration of the therapeutic holiday will depend on the BP used, being greater in the case of BP with greater affinity for hydroxyapatite such as ALN (2 years) or ZOL (3 years), and lower (1-2 years) in the case of BP with lower skeletal affinity such as RIS or IBN28.
It should be remembered that a temporary interruption cannot be considered with any other antiresorptive, and that doing so with DMAB could expose the patient to a situation of high risk of fracture, especially vertebral, especially among patients with a previous vertebral fracture, as already mentioned. previously. It is precisely in these cases that one might ask the reasons for a suspension or interruption of treatment.
COMBINATION THERAPY
The combination of two antiresorptives has not been shown to be more effective than treatment with a single antiresorptive54.
Nor does the combination of osteoformer with BP provide greater efficacy. The combined treatment of TPTD and ALN appears to be even less effective than TPTD alone55. In any case, it could be somewhat more beneficial to add TPTD to treatment already started with BP or to add BP to treatment already started with TPTD56, although more studies are needed to confirm this assumption.
Combined TPTD and ZOL therapy has been shown to achieve a faster increase in BMD in both the lumbar spine and the hip, although with no greater gain in BMD at 12 months than TPTD in monotherapy57.
The most promising combination therapy is the simultaneous administration of TPTD and DMAB. In the DATA-Switch study, it was observed that the patients treated with the combination had greater increases in BMD in the spine and hip than the patients treated with each drug alone (figure 4)44.
According to the latest osteoporosis recommendations of the Spanish Society of Rheumatology, this combination could be justified in very selected cases of severe osteoporosis12.
CONCLUSIONS
Osteoporosis is a chronic disease that usually requires treatment for many years.
For optimal management of the disease, the clinician must know very well the mechanism of action, efficacy and safety of each of the drugs, as well as the differences that exist according to the order of administration of the same.
With the exception of BPs, which have a residual anti-fracture effect, discontinuation of treatment for osteoporosis is not favorable for the bone, being especially negative in the case of DMAB.
For this reason, therapeutic holidays can only be considered in patients treated with BP with a low risk of fracture.
The change from an antiresorptive to another antiresorptive with a different mechanism of action is an option that may be favorable in the management of some patients with osteoporosis.
Although exchanging an antiresorptive for an osteoformer may be associated with a lower initial BMD gain (or even loss in the case of ABMD), this does not appear to have negative consequences on antifracture efficacy.
Starting with bone-forming treatment (TPTD or ROMO) and then continuing with an antiresorptive is the best treatment sequence, so it could be the preferred option in patients with a very high risk of fracture.
Bibliografía
1 NIH Consensus development panel on osteoporosis prevention, diagnosis, and therapy. JAMA. 2001; 285(6):785-95. [ Links ]
2 Yang L, Kang N, Yang JC, Su QJ, Liu YZ, Guan L, et al. Drug efficacies on bone mineral density and fracture rate for the treatment of postmenopausal osteoporosis: a network meta-analysis. Eur Rev Med Pharmacol Sci. 2019; 23(6): 2640-68. [ Links ]
3 Lu L, Lu L, Zhang J, Li J. Potential risks of rare serious adverse effects related to long-term use of bisphosphonates: An overview of systematic reviews. J Clin Pharm Ther. 2020; 45(1):45-51. [ Links ]
4 Pittman K, Antill YC, Goldrick A, Goh J, de Boer RH. Denosumab: Prevention and management of hypocalcemia, osteonecrosis of the jaw and atypical fractures. Asia Pac J Clin Oncol. 2017; 13(4): 266-76. [ Links ]
5 Ficha técnica de Forsteo. https://www.ema.europa.eu/en/documents/product-information/forsteo-epar-product-information_es.pdf. [ Links ]
6 Ficha técnica de Tymlos. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/208743lbl.pdf. [ Links ]
7 Ficha técnica de Evenity. https://www.ema.europa.eu/en/documents/product-information/evenity-eparproduct-information_es.pdf. [ Links ]
8 Guañabens N, Moro-Álvarez MJ, Casado E, Blanch-Rubió J, Gómez-Alonso C, Díaz-Guerra GM, et al. The next step after anti-osteoporotic drug discontinuation: an up-to-date review of sequential treatment. Endocrine. 2019; 64(3): 441-55. [ Links ]
9 Cadarette SM, Burden AM. Measuring and improving adherence to osteoporosis pharmacotherapy. Curr Opin Rheumatol. 2010; 22(4): 397-403. [ Links ]
10 Hofbauer LC, Schoppet M. Clinical implications of the osteoprotegerin/ RANKL/RANK system for bone and vascular diseases. JAMA. 2004; 292(4): 490-5. [ Links ]
11 Mirza FS, Padhi ID, Raisz LG, Lorenzo JA. Serum sclerostin levels negatively correlate with parathyroid hormone levels and free estrogen index in postmenopausal women. J Clin Endocrinol Metab. 2010; 95(4): 1991-7. [ Links ]
12 Anderson GL, Limacher M, Assaf AR, Bassford T, Beresford SA, Black H, et al. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women's Health Initiative randomized controlled trial. JAMA. 2004; 291(14): 1701-12. [ Links ]
13 de Villiers TJ, Pines A, Panay N, Gambacciani M, Archer DF, Baber RJ, et al. Updated 2013 International Menopause Society recommendations on menopausal hormone therapy and preventive strategies for midlife health. Climacteric. 2013; 16(3): 316-37. [ Links ]
14 Nelson ER, Wardell SE, McDonnell DP. The molecular mechanisms underlying the pharmacological actions of estrogens, SERMs and oxysterols: implications for the treatment and prevention of osteoporosis. Bone. 2013; 53(1): 42-50. [ Links ]
15 Naranjo Hernández A, Díaz Del Campo Fontecha P, Aguado Acín MP, Arboleya Rodríguez L, Casado Burgos E, Castañeda S, et al. Recommendations by the Spanish Society of Rheumatology on osteoporosis. Reumatol Clin. 2019; 15(4): 188-210. [ Links ]
16 Russell RG. Bisphosphonates: the first 40 years. Bone. 2011 ; 49(1): 2-19. [ Links ]
17 González-Macías J, Del Pino-Montes J, Olmos JM, Nogués X; en nombre de la comisión de redacción de las guías de osteoporosis de la SEIOMM. Clinical practice guidelines for posmenopausal, glucocorticoid-induced and male osteoporosis. Spanish Society for Research on Bone and Mineral Metabolism (3rd updated version 2014). Rev Clin Esp. 2015; 215(9): 515-26. [ Links ]
18 Dempster DW, Lambing CL, Kostenuik PJ, Grauer A. Role of RANK ligand and denosumab, a targeted RANK ligand inhibitor, in bone health and osteoporosis: a review of preclinical and clinical data. Clin Ther. 2012; 34(3): 521-36. [ Links ]
19 Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009; 361(8): 756-65. [ Links ]
20 Ismail A, Hassan BA, Ibrahim AA, Mirbaha S, Baratloo A. Denosumab and atypical femoral fractures: A scoping literatura review. Trauma Mon. 2018; 23(1): e42869. [ Links ]
21 Silva BC, Costa AG, Cusano NE, Kousteni S, Bilezikian JP. Catabolic and anabolic actions of parathyroid hormone on the skeleton. J Endocrinol Invest. 2011; 34(10): 801-10. [ Links ]
22 Kendler DL, Marin F, Zerbini CAF, Russo LA, Greenspan SL, Zikan V, et al. Effects of teriparatide and risedronate on new fractures in post-menopausal women with severe osteoporosis (VERO): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet. 2018; 391(10117): 230-40. [ Links ]
23 McClung MR, Grauer A, Boonen S, Bolognese MA, Brown JP, Diez-Perez A, et al. Romosozumab in postmenopausal women with low bone mineral density. N Engl J Med. 2014; 370(5): 412-20. [ Links ]
24 Cosman F, Crittenden DB, Adachi JD, Binkley N, Czerwinski E, Ferrari S, et al. Romosozumab treatment in postmenopausal women with osteoporosis. N Engl J Med. 2016; 375(16): 1532-43. [ Links ]
25 Saag KG, Petersen J, Brandi ML, Karaplis AC, Lorentzon M, Thomas T, et al. Romosozumab or alendronate for fracture prevention in women with osteoporosis. N Engl J Med. 2017; 377(15): 1417-27. [ Links ]
26 Ascott-Evans BH, Guanabens N, Kivinen S, Stuckey BG, Magaril CH, Vandormael K, et al. Alendronate prevents loss of bone density associated with discontinuation of hormone replacement therapy: a randomized controlled trial. Arch Intern Med. 2003; 14; 163(7): 789-94. [ Links ]
27 Neele SJ, Evertz R, De Valk-De Roo G, Roos JC, Netelenbos JC. Effect of 1 year of discontinuation of raloxifene or estrogen therapy on bone mineral density after 5 years of treatment in healthy postmenopausal women. Bone. 2002; 30(4): 599-603. [ Links ]
28 Adler RA, El-Hajj Fuleihan G, Bauer DC, Camacho PM, Clarke BL, Clines GA, et al. Managing 0steoporosis in patients on long-term bisphosphonate treatment: Report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2016; 31(1): 16-35. [ Links ]
29 Black DM, Schwartz AV, Ensrud KE, Cauley JA, Levis S, Quandt SA, et al. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA. 2006; 296(24): 2927-38. [ Links ]
30 Bone HG, Bolognese MA, Yuen CK, Kendler DL, Miller PD, Yang YC, et al. Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab. 2011; 96(4): 972-80. [ Links ]
31 Cummings SR, Ferrari S, Eastell R, Gilchrist N, Jensen JB, McClung M, et al. Vertebral fractures after discontinuation of denosumab: A post hoc analysis of the randomized placebocontrolled FREEDOM trial and its extension. J Bone Miner Res. 2018; 33(2): 190-8. [ Links ]
32 McClung MR, Wagman RB, Miller PD, Wang A, Lewiecki EM. Observations following discontinuation of longterm denosumab therapy. Osteoporos Int. 2017; 28(5): 1723-32. [ Links ]
33 Zanchetta MB, Boailchuk J, Massari F, Silveira F, Bogado C, Zanchetta JR. Significant bone loss after stopping longterm denosumab treatment: a post FREEDOM study. Osteoporos Int. 2018; 29(1): 41-7. [ Links ]
34 Tsourdi E, Zillikens MC, Meier C, Body JJ, Gonzalez Rodriguez E, Anastasilakis et al. Fracture risk and management of discontinuation of denosumab therapy: a systematic review and position statement by ECTS. J Clin Endocrinol Metab. 2020; 26: dgaa756. [ Links ]
35 Leder BZ, Neer RM, Wyland JJ, Lee HW, Burnett-Bowie SM, Finkelstein JS. Effects of teriparatide treatment and discontinuation in postmenopausal women and eugonadal men with osteoporosis. J Clin Endocrinol Metab. 2009; 94(8): 2915-21. [ Links ]
36 Adami S, San Martin J, Muñoz-Torres M, Econs MJ, Xie L, Dalsky GP, et al. Effect of raloxifene after recombinant teriparatide [hPTH(1-34)] treatment in postmenopausal women with osteoporosis. Osteoporos Int. 2008; 19(1): 87-94. [ Links ]
37 Niimi R, Kono T, Nishihara A, Hasegawa M, Kono T, Sudo A. Efficacy of switsching from teriparatide to bisphosphonate or denosumab: A prospective, randomized, open-label trial. JBMR Plus. 2018; 2(5): 289-94. [ Links ]
38 McClung MR, Brown JP, Diez-Perez A, Resch H, Caminis J, Meisner P, et al. Effects of 24 months of treatment with romosozumab followed by 12 months of denosumab or placebo in postmenopausal women with low bone mineral density: A randomized, doubleblind, phase 2, parallel group study. J Bone Miner Res. 2018; 33(8): 1397-1406. [ Links ]
39 Kendler DL, Roux C, Benhamou CL, Brown JP, Lillestol M, Siddhanti S, et al. Effects of denosumab on bone mineral density and bone turnover in postmenopausal women transitioning from alendronate therapy. J Bone Miner Res. 2010; 25(1): 72-81. [ Links ]
40 Miller PD, Pannacciulli N, Brown JP, Czerwinski E, Nedergaard BS, Bolognese MA, et al. Denosumab or zoledronic acid in postmenopausal women with osteoporosis previously treated with oral bisphosphonates. J Clin Endocrinol Metab. 2016; 101(8): 3163-70. [ Links ]
41 Boonen S, Marin F, Obermayer-Pietsch B, Simões ME, Barker C, Glass EV, et al. Effects of previous antiresorptive therapy on the bone mineral density response to two years of teriparatide treatment in postmenopausal women with osteoporosis. J Clin Endocrinol Metab. 2008; 93(3): 852-60. [ Links ]
42 Ettinger B, San Martin J, Crans G, Pavo I. Differential effects of teriparatide on BMD after treatment with raloxifene or alendronate. J Bone Miner Res. 2004; 19(5): 745-51. [ Links ]
43 Geusens P, Marin F, Kendler DL, Russo LA, Zerbini CA, Minisola S, et al. Effects of teriparatide compared with risedronate on the risk of fractures in sub-groups of postmenopausal women with severe osteoporosis: The VERO trial. J Bone Miner Res. 2018; 33(5): 783-94. [ Links ]
44 Leder BZ, Tsai JN, Uihlein AV, Wallace PM, Lee H, Neer RM, et al. Denosumab and teriparatide transitions in postmenopausal osteoporosis (the DATA-Switch study): extension of a randomised controlled trial. Lancet. 2015; 386(9999): 1147-55. [ Links ]
45 Langdahl B. Treatment of postmenopausal osteoporosis with bone-forming and antiresorptive treatments: Combined and sequential approaches. Bone. 2020; 139: 115516. [ Links ]
46 Langdahl BL, Libanati C, Crittenden DB, Bolognese MA, Brown JP, Daizadeh NS, et al. Romosozumab (sclerostin monoclonal antibody) versus teriparatide in postmenopausal women with osteoporosis transitioning from oral bisphosphonate therapy: a randomised, open-label, phase 3 trial. Lancet. 2017; 390(10102): 1585-94. [ Links ]
47 Kendler DL, Bone HG, Massari F, Gielen E, Palacios S, Maddox J, et al. Bone mineral density gains with a second 12-month course of romosozumab therapy following placebo or denosumab. Osteoporos Int. 2019; 30(12): 2437-48. [ Links ]
48 Eastell R, Nickelsen T, Marin F, Barker C, Hadji P, Farrerons P, et al. Sequential treatment of severe postmenopausal osteoporosis after teriparatide: final results of the randomized, controlled European Study of Forsteo (EURO-FORS). J Bone Min Res. 2009; 24(4): 726-36 [ Links ]
49 Lindsay R, Scheele WH, Neer R, Pohl G, Adami S, Mautalen C, et al. Sustained vertebral fracture risk reduction after withdrawal of teriparatide in postmenopausal women with osteoporosis. Arch Intern Med. 2004; 164(18): 2024-30. [ Links ]
50 Cosman F, McMahon D, Dempster D, Nieves JW. Standard versus cyclic teriparatide and denosumab treatment for osteoporosis: A randomized trial. J Bone Miner Res. 2020; 35(2): 219-25. [ Links ]
51 Bone HG, Cosman F, Miller PD, Williams GC, Hattersley G, Hu MY, et al. ACTIVExtend: 24 months of alendronate after 18 months of abaloparatide or placebo for postmenopausal osteoporosis. J Clin Endocrinol Metab. 2018; 103(8): 2949-57. [ Links ]
52 Casado Burgos E. Nuevos datos sobre el tratamiento con bisfosfonatos: ¿son aconsejables unas vacaciones terapéuticas? [New data on biphosphonate therapy: are therapeutic holidays advisable?]. Reumatol Clin. 2011; 7 Suppl 2: S28-33. [ Links ]
53 Moro Álvarez MJ, Neyro JL, Castañeda S. Vacaciones terapéuticas en osteoporosis: estrategia en el tratamiento a largo plazo con bifosfonatos [Therapeutic holidays in osteoporosis: Longterm strategy of treatment with bisphosphonates]. Med Clin (Barc). 2016; 146(1): 24-9. [ Links ]
54 Wimalawansa SJ. Prevention and treatment of osteoporosis: efficacy of combination of hormone replacement therapy with other antiresorptive agents. J Clin Densitom. 2000; 3(2): 187-201. [ Links ]
55 Finkelstein JS, Wyland JJ, Lee H, Neer RM. Effects of teriparatide, alendronate, or both in women with postmenopausal osteoporosis. J Clin Endocrinol Metab. 2010; 95(4): 1838-45. [ Links ]
56 Leder BZ. Optimizing sequential and combined anabolic and antiresorptive osteoporosis therapy. JBMR Plus. 2018; 2(2): 62-8. [ Links ]
57 Cosman F, Eriksen EF, Recknor C, Miller PD, Guañabens N, Kasperk C, et al. Effects of intravenous zoledronic acid plus subcutaneous teriparatide [rhPTH(1-34)] in postmenopausal osteoporosis. J Bone Miner Res. 2011; 26(3): 503-11. [ Links ]
Received: January 19, 2021; Accepted: September 21, 2021











 texto en
texto en 


