Mi SciELO
Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Accesos
Accesos
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en
SciELO
Similares en
SciELO -
 Similares en Google
Similares en Google
Compartir
Revista de Osteoporosis y Metabolismo Mineral
versión On-line ISSN 2173-2345versión impresa ISSN 1889-836X
Rev Osteoporos Metab Miner vol.14 no.2 Madrid abr./jun. 2022 Epub 12-Sep-2022
https://dx.doi.org/10.4321/s1889-836x2022000200005
ORIGINALS
Effect of treatment with denosumab for 24 months in individuals with recent spinal cord injury with osteoporosis
1Rheumatology Department. Germans Trias i Pujol University Hospital. Badalona (Spain)
2Guttmann Neuro-rehabilitation Hospital. Autonomous University of Barcelona. Badalona (Spain)
3Department of Public Health. University of Barcelona (Spain)
4Nuclear Medicine Department. Clinic Hospital of Barcelona (Spain)
5Clinic Hospital of Barcelona (Spain)
Introduction:
Osteoporosis development is a frequent complication associated with spinal cord injury (SCI), especially at the sublesional level. However, at present, data on its treatment are scarce.
Aim:
To analyze bone mineral density (BMD) and bone turnover markers (BTM) after 2-year treatment with denosumab in individuals with SCI-related osteoporosis.
Methods:
Prospective study including patients with recent SCI and related osteoporosis treated with denosumab for 24 months. In all patients, BTMs (bone ALP, sCTX and PINP), 25-OH-vitamin D levels and lumbar and femoral BMD were assessed at baseline and at 12 and 24 months.
Results:
13 patients (aged 39±15 years) with recent SCI (mean duration of 15 months) and osteoporosis treated with denosumab for 24 months were included. Patients showed a significant increase in BMD at lumbar spine and proximal femur after 12 months of treatment with denosumab, with a further increase in BMD at 24 months of follow-up, reaching an increase of 9.1% in lumbar spine, 4.4% in femoral neck and 5.3% total femur, respectively. BTM significantly decrease at 12 months and remained decreased at 24 months of follow-up. No skeletal fractures or treatment-related adverse events were observed during follow-up.
Conclusions:
Treatment with denosumab during 24 months increases lumbar and femoral BMD and decreases BTMs in patients with recent SCI. Denosumab may be a promising therapeutic option in SCI-related osteoporosis.
Key words denosumab; osteoporosis; spinal cord injury; bone mineral density; bone turnover markers
INTRODUCTION
After a spinal cord injury (SCI) there is a marked loss of bone mass and an increase in remodeling that leads to the development of osteoporosis and skeletal fractures, especially below the level of the injury1-3. Thus, more than 50% of patients with complete SCI develop densitometric osteoporosis one year after SCI1, which can reach 81% of patients after more than 5 years of SCI4. However, despite the high incidence of osteoporosis and fractures, the therapeutic approach to these patients is clearly deficient, since less than 10-20% of them receive anti-osteoporotic treatment2,5.
There are few studies that analyze the effect of antiosteoporotic treatment on osteoporosis associated with SCI. In this sense, treatment with oral or intravenous bisphosphonates, especially zoledronate, has been shown to reduce the loss of bone mineral density (BMD) in this process. However, in patients with recent SCI, in whom there is a rapid and marked loss of BMD associated with an increase in bone turnover, its efficacy is lower, especially at the infra-lesional level, in the lower limbs6-9, where most fragility fractures occur in these patients2. Along the same lines, teriparatide, a bone-forming treatment, has also not shown efficacy in preventing bone loss in this process10. All this indicates the need to improve the therapeutic approach in these patients, not only at the advanced stages of the disease, but also early after SCI, when the magnitude of bone loss is greater, thus preventing associated long-term complications.
Denosumab, a monoclonal antibody directed against RANK-ligand, is an essential mediator for osteoclast differentiation and survival, with a marked antiresorptive effect and demonstrated effectivity in the treatment of postmenopausal and male osteoporosis11. It offers a remarkably positive effect on cortical bone, such as the proximal femur or distal forearm11. Therefore, the use of denosumab could be especially indicated in treating patients with SCI and osteoporosis. In fact, an increased expression of RANKL was observed in an animal model of mice with SCI12, suggesting a potential therapeutic role for denosumab in this clinical situation. Similarly, in a recent exploratory study that included a limited number of patients with SCI, a preventive effect of denosumab on bone loss a few months after SCI was observed13. We previously reported a positive effect of this type of treatment in patients with SCI and osteoporosis during a 12-month follow-up period14.
This study reports our experience in patients with recent complete motor SCI treated with denosumab over 24 months.
PATIENTS AND METHODS
Study design and patient selection
This study is part of a prospective observational study with the main objective of analyzing the effect of recent SCI (<6 months) on bone mass loss and bone metabolism in these patients1. The patients were consecutively recruited at the Guttmann Neurohabilitation Institute, and subsequently referred to the Metabolic Bone Pathology Unit of the Rheumatology Service of the Hospital Clínic de Barcelona. Antiosteoporotic treatment was indicated in those patients who presented densitometric OP during follow-up. In patients with 25-OH-vitamin D deficiency ([25-OHD] <20 ng/ml), vitamin D supplements were indicated. The study was approved by the ethics committee of the Hospital Clínic de Barcelona and the Guttmann Neurorehabilitation Institute. All patients signed the informed consent prior to their inclusion.
In this study, we present data on the effect of anti-osteoporotic treatment with denosumab on BMD evolution of bone turnover markers (BTM) in individuals with SCI who developed osteoporosis during follow-up and completed 24 months of treatment with denosumab, with 13 patients included.
METHODS
All patients underwent a clinical and analytical assessment with BTM quantification and bone densitometry at baseline and at 12 and 24 months of follow-up.
Osteoporosis risk factors, body mass index (BMI), and injury characteristics were collected, including the level of SCI (tetraplegia/paraplegia), the presence of spasticity, and the severity of SCI according to the scale of AIS15 that classifies according to motor and sensory involvement in 5 categories: A: complete motor and sensory SCI; B: complete motor and partial sensory SCI; C and D: partial motor and sensory; E: no motor or sensory lesion. The incidence of skeletal fractures and potential adverse effects during follow-up were also collected.
Analytical determinations
Analytical determinations included: creatinine, calcium and phosphate by automated methods. The values of 25OHD (Liason DiaSorin) and the following BRMs were quantified: bone alkaline phosphatase (bone FA by IDS, Vitro), type I procollagen amino-terminal propeptide (PINP by Cobas e411, Roche) and type I collagen carboxyterminal telopeptide. I (CTX by Cobas e411 automated method, Roche).
Bone mineral density
Lumbar spine and proximal femur BMD (femoral neck and total femur) were quantified by dual X-ray absorptiometry (DXA; Lunar Prodigy, Radiation Corporation Madison, WI) at baseline, and at 12- and 24-month follow-up. The densitometric categories were defined according to WHO criteria (normal BMD, osteopenia and osteoporosis)16.
Statistic analysis
The results have been expressed as the mean ± standard deviation of the mean (SD). The differences between means of the continuous variables were analyzed using Student's t-test and the differences between proportions using the Chi-square. To compare paired variables (baseline and 12 months; 12 months and 24 months; baseline and 24 months) the Wilcoxon non-parametric test was used. To assess the association between analytical and densitometric variables, the Pearson correlation coefficient was used. The value p<0.05 was considered statistically significant.
RESULTS
The clinical characteristics of the individuals included in the study are shown in table 1.
Table 1. Clinical, analytical and densitometric characteristics of patients with SCI at baseline
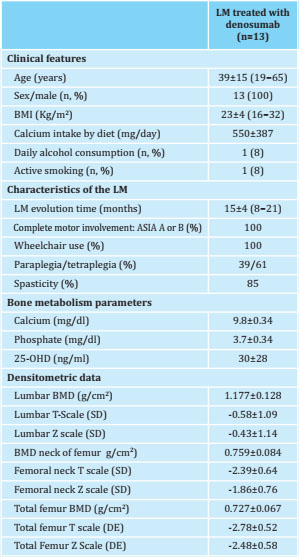
Results expressed as mean ± SD, n and %.LM: spinal cord injury; BMI: Body Mass Index; 25-OHD: 25-OH-vitamin D; SD: standard deviation.
In all, 13 men were included, with a mean age of 39±15 years at 15±4 months after having suffered SCI. All patients had severe SCI (ASIA A or B) and 61% had tetraplegia. Most of them had spastic-type SCI (85%) and all of them required a wheelchair to get around. The main cause of SCI was traffic accident (85%). One patient presented SCI attributed to precipitation and another due to a sports accident. All the patients included in the study had developed osteoporosis during the initial follow-up period (prior to starting anti-osteoporotic treatment with denosumab).
At 12 months from the start of treatment with denosumab, a significant increase in BMD was observed in all locations analyzed: lumbar spine (+7.47±3.67%, p=0.001) and femoral neck (+3.03±3.73, p=0.019) (table 2 and figure 1). Likewise, at 12 months, a significant decrease was observed in all the ROM: bone FA (-41±22%, p=0.003); PINP (-53±26%, p=0.001) and CTX (-59±29%, p=0.002) (figure 2).
Table 2. BMD development and bone remodeling markers at 12 and 24 months of treatment with denosumab
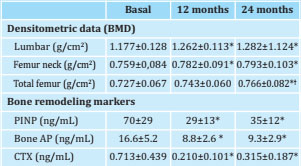
Results expressed as mean ± SD.* p<0.05 compared to baseline values.† p<0.05 compared to values at 12 months. BMD: bone mineral density; P1NP: type I procollagen amino-terminal propeptide; Bone AP: bone alkaline phosphatase; CTX: carboxy-terminal telopeptide of type I collagen.
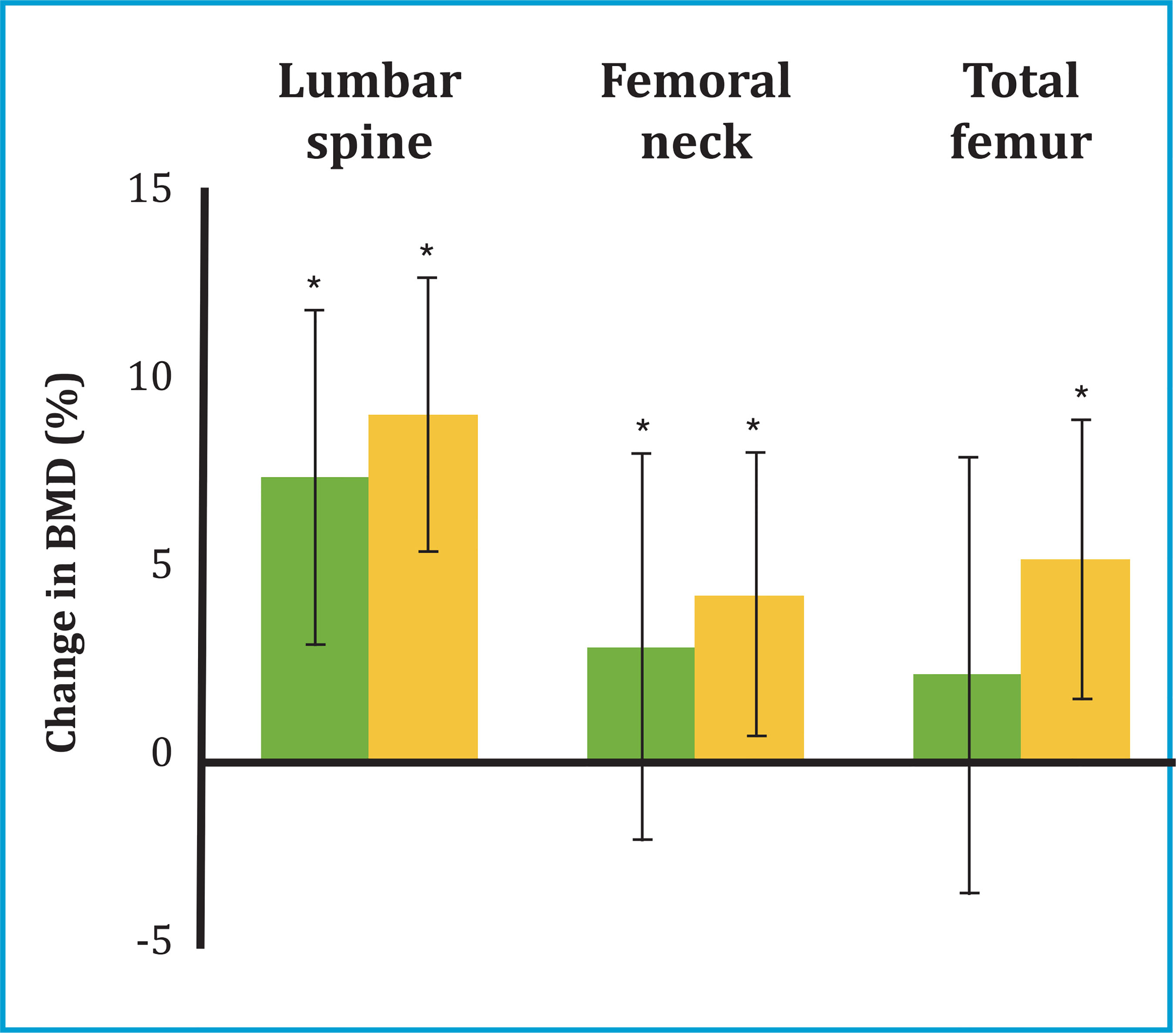
* p<0.05 compared to baseline values.
Figure 1. Percentage change in BMD (± standard deviation) in the lumbar spine, total femur, and femoral neck at 12 (green bars) and 24 (orange bars) months after starting treatment with denosumab
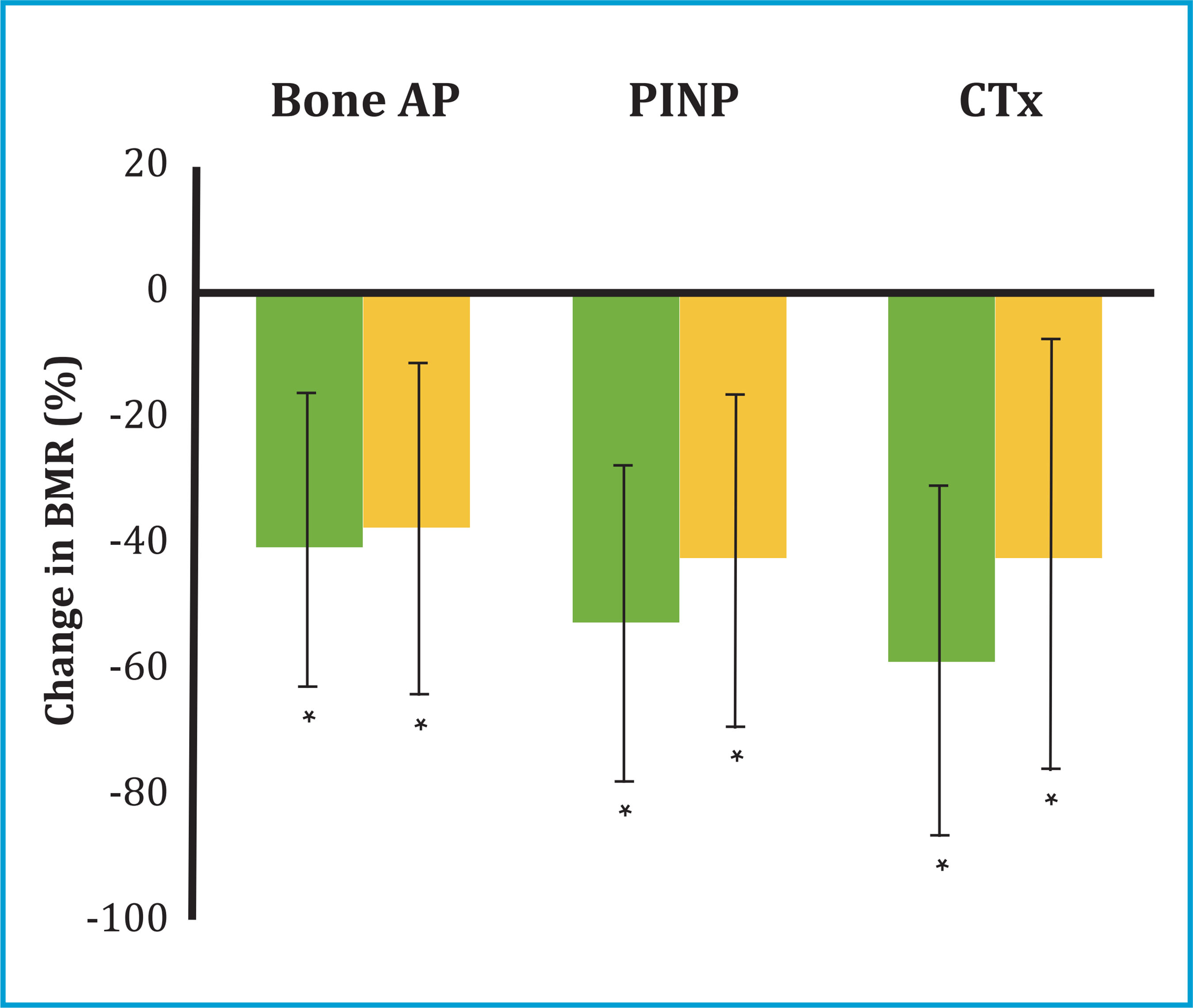
* p<0.05 compared to baseline values.
Figure 2. Percentage change in BMR (± standard deviation) at 12 (green bars) and 24 months (orange bars) after starting denosumab treatment
At 24 months of treatment, an additional increase in BMD was observed in all locations. Thus, the patients achieved a total increase in BMD at 24 months of +9.1±4.4% in the lumbar spine (p=0.002); +4.4±5.1% in the neck of the femur (p=0.033) and +5.3±5.7% in the total femur (p=0.011) (table 2 and figure 1). The BTMs persisted decreased at 24 months with an overall decrease in bone FA (-38±27%, p=0.003); PINP (-43±27%, p=0.001) and CTX (-42±35%, p=0.005) (table 2 and figure 2).
BMD evolution was not related to changes in BTM or 25-OHD values. No patient presented skeletal fragility fractures during follow-up or adverse effects associated with treatment.
DISCUSSION
The results of this study confirm the efficacy of denosumab in the treatment of osteoporosis associated with recent onset SCI, not only in the prevention of bone loss but also in the sustained increase in BMD after 24 months of treatment. Thus, treatment with denosumab for 24 months was associated with a progressive and significant increase in bone mass in all skeletal locations, both in the lumbar spine and at the sub-lesional level, in the proximal femur, and with a sustained decrease in BTM during the 24-month treatment period.
The results presented indicate that treatment with denosumab in patients with osteoporosis associated with SCI not only prevents the loss of bone mass, but even partially reverses this loss, reporting an increase of +7.47% in the lumbar spine and +3% in femoral neck at 12 months of treatment. In addition, and as expected, the patients achieved a greater increase in bone mass after the second year of treatment with denosumab, up to +9% in the lumbar spine and +5% in the proximal femur. While untreated patients, according to the literature, present sustained BMD losses between 2% and 21%1,17-20, depending on the location evaluated (spine and/or proximal or distal femur), and the time of evolution of SCI, observing the greatest losses of BMD during the first 1-2 years after its establishment19-20.
Although this is an observational study that includes a small number of patients, it is important to point out that denosumab produced an increase in BMD, not only in the lumbar spine, but also at the sub-lesional level, in the neck of the femur and in the total femur, and that was 9.1% in the lumbar spine, 4.4% in the neck of the femur and 5.3% in the total femur after two years of treatment. To date, this is the only anti-osteoporotic treatment that has been associated with a BMD increase in patients with OP associated with a recent SCI. Our patients had complete motor SCI with a mean onset time of 15 months, which usually coincides with the period of greatest bone loss3,19-20, in which, with the exception of denosumab13. There does not seem to be an effective anti-osteoporotic treatment, particularly to prevent infralesional loss in lower limbs. In this sense, treatment with antiresorptive drugs, such as oral and/or intravenous bisphosphonates (including alendronate, pamidronate or zoledronate), or bone formers, such as teriparatide, have only been shown to attenuate the loss of bone mass in the lower limbs after a recent SCI6-10,21-22. However, in patients with long-standing SCI, in whom the magnitude of bone loss and bone turnover has decreased, bisphosphonates seem to have a preventive effect6,7. Although, the need to carry out new studies that include a greater number of patients with longer follow-up time to evaluate the treatment of these patients has been indicated23.
Likewise, treatment with denosumab was associated with a decrease in the values of all the BTMs analyzed in this study (bone FA, PINP and CTX), with a decrease of the order of ~40% after 24 months of treatment, a finding that we have observed in patients with SCI with a similar evolution time who did not undergo this treatment, in whom an increase in BTM persists24. This decrease was similar in magnitude to that reported in the treatment of postmenopausal osteoporosis and in men with this type of therapy11,25.
On the other hand, although this is an observational study that includes a small number of patients, no side effects related to denosumab treatment or the development of new skeletal fractures were observed during the 24-month follow-up.
There are few studies that assess the effect of anti-osteoporotic treatment in the medium term in individuals with SCI. Most studies published to date include only 624 months of treatment with teriparatide, bisphosphonates, or denosumab6-10,13,14,18,21,22.
This is the first observational study that assesses the effect of anti-resorptive treatment with denosumab for 2 years in individuals with OP associated with SCI.
Although the small number of patients and the absence of a control group constitute limitations of the study, it is important to highlight that it is a homogeneous cohort of patients, which includes men with recently established complete traumatic SCI with six-monthly follow-up, and in the that all patients experienced a marked and rapid loss of infralesional BMD after SCI1.
Therefore, despite the characteristics of the study, we consider that these results provide useful information in the management of osteoporosis associated with this entity.
In conclusion, patients with OP associated with recent onset SCI treated with denosumab for 24 months show a significant increase in BMD at the lumbar and femoral levels. Therefore, denosumab could be a promising therapeutic option in this clinical situation. Studies that include a larger number of patients and with a longer follow-up time are needed to analyze the long-term effect of this treatment on this condition.
Bibliografía
1 Gifre L, Vidal J, Carrasco J, Filella X, Ruiz-Gaspà S, Muxi A, Portell E, Monegal A, Guañabens N, Peris P. Effect of recent spinal cord injury on Wnt signaling antagonists (sclerostin and Dkk-1) and their relationship with bone loss. A 12-month prospective study. J Bone Miner Res. 2015; 30:1014-1021 [ Links ]
2 Gifre L, Vidal J, Carrasco J, Portell E, Puig J, Monegal A, Guañabens N, Peris P. Incidence of skeletal fractures after traumatic spinal cord injury: a 10-year follow-up study. Clin Rehabil. 2014;28: 361-369. [ Links ]
3 Frey-Rindova P, de Bruin ED, Stüssi E, Dambacher MA, Dietz V. Bone mineral density in upper and lower extremi- ties during 12 months after spinal cord injury measured by peripheral quantitative computed tomography. Spinal Cord. 2000;38:26-32. [ Links ]
4 Shojaei H, Soroush MR, Modirian E. Spinal cord injury-induced osteoporosis in veterans. J Spinal Disord Tech. 2006;19:114-117. [ Links ]
5 Weaver FM, Le B, Ray C, Miskevics S, Gonzalez B, Carbone LD. Predicting osteoporosis medication receipt in Veterans with a spinal cord injury: A retrospective cohort study. J Spinal Cord Med. 2019;42:760-767. [ Links ]
6 Bryson JE, Gourlay ML. Bisphosphonate use in acute and chronic spinal cord injury: a systematic review. J Spinal Cord Med. 2009;32:215-225. [ Links ]
7 Chang KV, Hung CY, Chen WS, Lai MS, Chien KL, Han DS. Effectiveness of bisphosphonate analogues and functional electrical stimulation on attenuating post-injury osteoporosis in spinal cord injury patients- a systematic review and meta-analysis. PLoS One 2013;8:e81124. [ Links ]
8 Bauman WA, Cirnigliaro CM, La Fountaine MF, , Martinez L, Kirshblum SC, Spungen AM. Zoledronic acid administration failed to prevent bone loss at the knee in persons with acute spinal cord injury: an observational cohort study. J Bone Miner Metab. 2015;33 (4):410-421. [ Links ]
9 Edwards WB, Haider IT, Simonian N, Barroso J, Schnitzer TJ. Durability and delayed treatment effects of zoledronic acid on bone loss after spinal cord injury: a randomized, controlled trial. J Bone Miner Res. 2021 Jul 19. doi: 10.1002/jbmr.4416. PMID: 34278611. [ Links ]
10 Gordon KE, Wald MJ, Schnitzer TJ. Effect of parathyroid hormone combined with gait training on bone density and bone architecture in people with chronic spinal cord injury. PM R 2013; 5:663-671. [ Links ]
11 McClung MR, Lewiecki EM, Cohen SB, Bolognese MA, Woodson GC, Moffett AH, Peacock M, Miller PD, Lederman SN, Chesnut CH, Lain D, Kivitz AJ, Holloway DL, Zhang C, Peterson MC, Bekker PJ; AMG 162 Bone Loss Study Group. Denosumab in postmenopausal women with low bone mineral density. N Engl J Med. 2006;354:821-831. [ Links ]
12 Jiang SD, Jiang LS, Dai LY. Effects of spinal cord injury on osteoblastogenesis, osteoclastogenesis and gene expression profiling in osteoblasts in young rats. Osteoporos Int. 2007;18:339-349. [ Links ]
13 Cirnigliaro CM, La Fountaine MF, Parrott JS, Kirshblum SC, McKenna C, Sauer SJ, Shapses SA, Hao L, McClure IA, Hobson JC, Spungen AM, Bauman WA. Administration of Denosumab Preserves Bone Mineral Density at the Knee in Persons With Subacute Spinal Cord Injury: Findings From a Randomized Clinical Trial. JBMR Plus. 2020; 4(8):e10375. [ Links ]
14 Gifre L, Vidal J, Carrasco JL, Muxi A, Portell E, Monegal A, Guañabens N, Peris P. Denosumab increases sublesional bone mass in osteoporotic individuals with recent spinal cord injury. Osteoporos Int. 2016;27:405-410. [ Links ]
15 Waring WP 3rd, Biering-Sorensen F, Burns S, Donovan W, Graves D, Jha A, Jones L, Kirshblum S, Marino R, Mulcahey MJ, Reeves R, Scelza WM, Schmidt-Read M, Stein A. _2009 review and revisions of the international standards for the neurological classification of spinal cord injury. J Spinal Cord Med. 2010;33:346-352. [ Links ]
16 Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser. 1994;843:1-129. [ Links ]
17 Garland DE, Adkins RH, Stewart CA. Five-year longitudinal bone evaluations in individuals with chronic complete spinal cord injury. J Spinal Cord Med. 2008;31:543-550. [ Links ]
18 Gilchrist NL, Frampton CM, Acland RH, Nicholls MG, March RL, Maguire P, Heard A, Reilly P, Marshall K. Alendronate prevents bone loss in patients with acute spinal cord injury: a randomized, double-blind, placebo-controlled study. J Clin Endocrinol Metab. 2007;92:1385-1390. [ Links ]
19 Giangregorio L, McCartney N. Bone loss and muscle atrophy in spinal cord injury: epidemiology, fracture prediction, and rehabilitation strategies. J Spinal Cord Med. 2006;29:489-500. [ Links ]
20 Jiang SD, Dai LY, Jiang LS. Osteoporosis after spinal cord injury. Osteoporos Int. 2006;17:180-192 [ Links ]
21 Goenka S, Sethi S, Pandey N, Joshi M, Jindal R. Effect of early treatment with zoledronic acid on prevention of bone loss in patients with acute spinal cord injury: a randomized controlled trial. Spinal Cord. 2018;56:1207-1211 [ Links ]
22 Wu Y, Wang F, Zhang Z. The efficacy and safety of bisphosphonate analogs for treatment of osteoporosis after spinal cord injury: a systematic review and meta-analysis of randomized con- trolled trials. Osteoporos Int. 2021 Jan 2. doi: 10.1007/s00198-020-05807-0. Epub ahead of print. PMID: 33386876. [ Links ]
23 Soleyman-Jahi S, Yousefian A, Maheronnaghsh R, Shokraneh F, Zadegan SA, Soltani A, Hosseini SM, Vaccaro AR, Rahimi-Movaghar V. Evidence-based prevention and treatment of osteoporosis after spinal cord injury: a systematic review. Eur Spine J. 2018;27: 1798-1814. [ Links ]
24 L Gifre, J Vidal, JL Carrasco, A Muxi, E Portell, A Monegal, N Guañabens, P Peris. Denosumab Increases Sublesional Bone Mass In Osteoporotic Patients With Recent Spinal Cord Injury. ASBMR, Seattle (WA), USA, 2015. J Bone Miner Res. 2015;30 (suppl 1): MO0363. [ Links ]
25 Orwoll E, Teglbjærg CS, Langdahl BL, Chapurlat R, Czerwinski E, Kendler DL, Reginster JY, Kivitz A, Lewiecki EM, Miller PD, Bolognese MA, McClung MR, Bone HG, Ljunggren Ö, Abrahamsen B, Gruntmanis U, Yang YC, Wagman RB, Siddhanti S, Grauer A, Hall JW, Boonen S. A randomized, placebo-controlled study of the effects of denosumab for the treatment of men with low bone mineral density. J Clin Endocrinol Metab. 2012;97:3161-3169. [ Links ]
Received: March 14, 2022; Accepted: May 11, 2022











 texto en
texto en 


