My SciELO
Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Cited by Google
Cited by Google -
 Similars in
SciELO
Similars in
SciELO -
 Similars in Google
Similars in Google
Share
Ars Pharmaceutica (Internet)
On-line version ISSN 2340-9894
Ars Pharm vol.56 n.3 Granada Jul./Sep. 2015
https://dx.doi.org/10.4321/S2340-98942015000300006
ARTÍCULO DE REVISIÓN
Protein and Peptide in Drug Targeting and its Therapeutic Approach
Las proteínas y péptidos en la orientación de fármacos y su abordaje terapéutico
Raj K. Keservani1, Anil K. Sharma2 and Urmila Jarouliya3
1. School of Pharmaceutical Sciences, Rajiv Gandhi Proudyogiki Vishwavidyalaya, Bhopal, India-462036.
2. Department of Pharmaceutics, Delhi Institute of Pharmaceutical Sciences and Research, New Delhi, India-110017.
3. School of Studies in Biotechnology, Jiwaji University, Gwalior (M.P), 474011, India.
ABSTRACT
Aim: The main aim of this review article is to provide information like advantages of protein and peptides via different routes of drug administration, targeted to a particular site and its implication in drug delivery system.
Methods: To that aim, from the web sites of PubMed, HCAplus, Thomson, and Registry were used as the main sources to perform the search for the most significant research articles published on the subject. The information was then carefully analyzed, highlighting the most important results in the development of protein and peptide drug targeting as well as its therapeutic activity.
Results: In recent years many researchers use protein and peptide as a target site of drug by a different delivery system. Proteins and peptides are used as specific and effective therapeutic agents, due to instability and side effects their use is complicated. Protein kinases are important regulators of most, if not all, biological processes. Abnormal activity of proteins and peptides has been implicated in many human diseases, such as diabetes, cancer and neurodegenerative disorders.
Conclusions: It is concluded that the protein and peptide were used in drug targeting to specific site and also used in different diseased states like cancer, diabetes, immunomodulating, neurodegenerative effects and antimicrobial activity.
Key words: Protein, Peptide, Controlled release, Bioerodible polymer.
RESUMEN
Objetivo: El objetivo principal de este artículo de revisión es proporcionar información sobre las ventajas de las proteínas y péptidos a través de diferentes vías de administración de fármacos, dirigidos a un sitio en particular y su implicación en el sistema de administración de fármacos.
Métodos: Con ese objetivo, los sitios web de PubMed, HCAplus, Thomson, se utilizaron como las principales fuentes para realizar la búsqueda de los artículos de investigación más importantes publicados sobre el tema. La información fue luego cuidadosamente analizada, destacando los resultados más importantes en el desarrollo de proteína y péptido de direccionamiento de drogas, así como su actividad terapéutica.
Resultados: En los últimos años muchos investigadores utilizan las proteínas y los péptidos como un sitio diana de fármaco por un sistema de administración diferente. Las proteínas y los péptidos se utilizan como agentes terapéuticos específicos y eficaces, debido a la inestabilidad y los efectos secundarios de su uso es complicado. Las proteínas quinasas son reguladores importantes de la mayoría, si no todos, los procesos biológicos. La actividad anormal de proteínas y péptidos se ha implicado en muchas enfermedades humanas, tales como diabetes, cáncer y trastornos neurodegenerativos.
Conclusión: Finalmente concluyó que la proteína y el péptido se utilizaron en fármaco que se dirige al sitio específico y también se utiliza en diferentes estados de enfermedad como el cáncer, la diabetes, como sustancias inmunomoduladora, efectos neurodegenerativos y actividad antimicrobiana.
Palabras clave: proteína, péptido, liberación controlada, polímero bioerosionable.
Introduction
Protein and peptide enhance the successfully delivery of the drug to the desired site of action. In past twenty years many biopharmaceutical process development aspects have been studied by researchers. For the production of many pharmaceutical proteins that have been well characterized and overcome the problem associated with cell culture, purification, recovery and fermentation 1. Thus, the successful development of protein and peptide formulations is totally depending on the study of in vitro, in vivo drug characterization and its intended application1. The recently protein and peptide show great progress to understand the erosion mechanism of biodegradable polymer and the preparation of controlled release devices. The results provide useful information on the microstructure and chemical environment inside these polymers during erosion. Many protein and peptide cannot be administered orally because it degraded inside the gastrointestinal tract (GI tract) 2 due to their short half life in the body fluids.
These limitations forced the preparation of controlled release dosage form and enhance the protein and peptide stability and drug activity for long period of time after its application. Processing of this substance into dosage forms at all times not easily achieved. Many of them have limited chemical and physical stability. Common instabilities are irreversible aggregation (J) 3, oxidation 4 or conformational changes 5 all of which may affect activity. The controlled release devices for buccal, oral, nasal, rectal, parenteral, vaginal and transdermal routes have been prepared 6,7. In the last few decades number of bioactive protein and peptide have discovered due to the progress in the field of biology and biochemistry. Insulin is a classical example, which is mostly used in medical therapy. The rapid development of biotechnology and progress in peptide and protein chemistry allowed the mass production of many compounds and made their broad introduction into medical therapy possible 8.
In biological processes protein and peptides play important role such as regulator in most cases not all. Their abnormal activity has been implicated as causal factors in many human diseases, including cancer, diabetes and neurodegenerative disorders 9-11. Pharmaceutical carriers were used to increase the stability, improve their efficacy and decrease undesired side effects and to assist intracellular delivery of the drugs in most of the cases 8,12.
Structure of protein and peptides
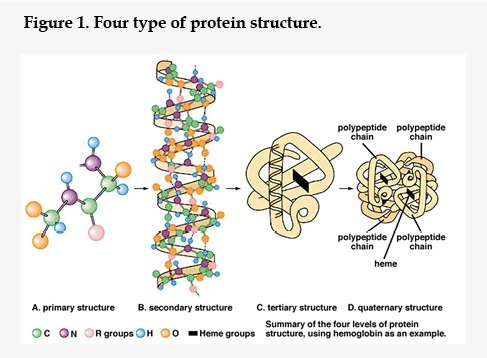
The Protein are complex of large molecules, consist of one or more amino acids in specific order. The protein and peptide acquire three dimensional conformations and protein structure is directly related to its function. It means that if the structure or shape of protein disrupted the function will also disrupted. There are four types of protein structures13 (Figure 1), namely, (a) primary structure (b) secondary structure (c) tertiary structure (d) quaternary structure.
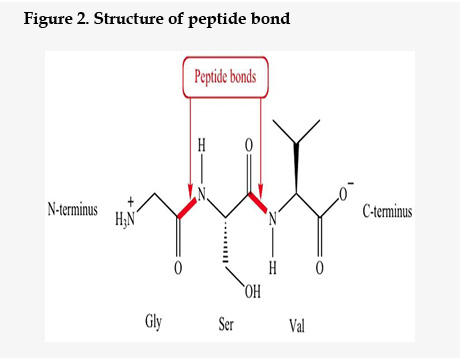
A target peptide (Table 1) is short peptide chain of amino acids (usually 3-70) that directs the transport of protein and peptide to specific region in the cell, such as including mitochondria, nucleus, endoplasmic reticulum (ER), apolpast, chloroplast, peroxisome and plasma membrane. Signal peptidase enzyme cleaved the target peptide from protein after those proteins were transported14. The amino acids joined together by amide linkage15 known as peptide bond (Figure 2).
Advantages of protein and peptides in different routes
Protein and peptides delivery of drugs provides several advantages when drug delivered by various routes, some of the mentioned below:
Buccal route
When the drug delivered via buccal routes it provides much less pain, irritation in case of long term of treatment. It can be easily attached and removed without any discomfort and easily accessible and acceptable by patients 16.
Nasal route
Nasal route is simple, convenient and provide rapid onset of drug action and also avoid the first pass metabolism 13,17.
Rectal route
A large dose of drug can be administered by rectal route, drugs can be target to lymphic system, it avoids presystemic or first pass metabolism and suitable for drugs that cause nausea/vomiting and irritation in GI tract on oral administration 16.
Pulmonary route
Pulmonary route of drug administration provides direct route to the circulation, safe route for drug entry even in patient with lung disease. Less dose required, less pain or discomfort and improve patient compliance 13,18,19.
Transdermal route
Transdermal route provides controlled and sustained release of drugs. Self administered and improve patient compliance due to its convenience and ease of use. Reduced side effects and avoid the hepatic first pass effect and gastrointestinal breakdown of drugs. Quick abrupt terminations of drug effect by easily remove the delivery system from the skin surface 20-23.
Peptide and protein drugs - brief overview and delivery problems
Many proteins and peptides acquire biological activity that makes them potent therapeutics. The enzymes represent an important and, probably, the best investigated group of protein drugs 24-26. The insulin peptide hormones used most likely than that of other hormone. To the treatment of pituitary and gastrointestinal (GI) tumors, somatostatin analogs peptides such as lanreotide, octreotide and vapreotide become available in the clinics 27. Antibodies against certain cancer-specific ligands can also be considered as protein anti cancer drugs 28,29. Still, the use of proteins and peptides as therapeutic agents is hampered by the whole set of their intrinsic properties associated with their nature as complex macromolecules, which are, as a rule, foreign to the recipient organism.
Stability of protein may vary at different condition of physiological pH and temperature, most of the peptide especially protein show low stability at normal physiological pH and temperature. Due to receptor interaction most of the protein and peptide and antibodies exert their action extracellularly. Many others, however, have their targets inside the cell. In the latter case, low permeability of cell membranes to macromolecules often represents an additional obstacle for the development of peptide-based and protein-based drug formulations 30.
Peptides as competitive inhibitors
The amino acid sequences responsible for the protein kinases specificity to substrate phosphorylation which leads to phosphorylation called as consensus sequences. The numbers and types of interactions with residues surrounding the phosphorylation site vary considerably among kinases, reflecting differences in sequence specificity. By virtue of that consensus sequence can serve as a template for a peptide that will acts as a competitive inhibitor 31,32.
Peptides competing with anchoring and targeting proteins
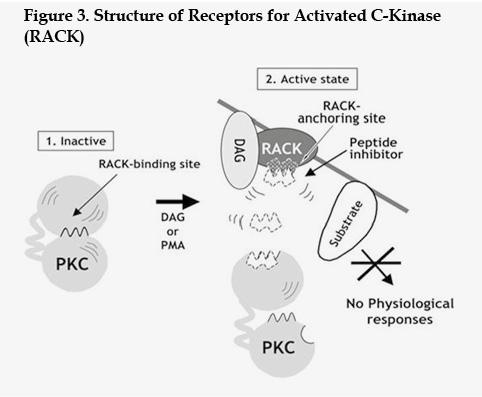
Modular binding partners target some Ser/Thr kinases to their specific substrates via changes in cellular localization. Binding of a peptide to this targeting domain may prevent the interaction of the kinase with its substrates. The Protein kinase C isoforms are known to translocate from cytosol to particulate fractions in response to various stimuli. A family of adaptor molecules, termed RACKs (Figure 3) (from Receptors for Activated C-Kinase), interact with the various Protein kinase C isozymes and facilitate their cellular translocation 33,34. The RACK binding domain is a short sequence located in the Protein kinase C's N terminal-sub domain and is different in each isoform. Thus, the translocation of Protein kinase C isoforms could be disrupted selectively using peptides derived from the specific RACK docking region 35,36. The use of such peptides has enabled analysis of the functions of individual Protein kinase C isozymes. For example, peptides derived from the C-2 region of PKC-β II prevented hormone-induced membrane translocation of PKC β II and inhibited insulin-induced Xenopusoocyte maturation 37.
Formulation development considerations
Novel delivery system includes the complete characterization of the drug properties and drug stability in different formulations, is the main key for development of any protein and peptide formulation. Typically, a formulation scientist will begin by considering the physicochemical properties of the protein such as the isoelectric point, molecular weight, glycosylation or other post-translational modification, and overall amino acid composition. These properties along with any known behavior of the drug in different solutions (e.g. different buffers, cofactors, etc.) as well as its in vivo behavior should guide the choice of formulation components for testing in the initial screen of candidate formulations. The potential candidate formulations are composed of U. S. Food and Drug Administration (FDA) approved buffer components, excipients, and any required cofactors (e.g. metal ions). Often, the first choice of candidate formulations is based upon the previous experience of the formulation scientist with other proteins or peptides and, in many cases; a simple phosphate buffered saline solution may be one of the initial candidates. The pH of the solution impacts major degradation pathway. Thus, the initial formulations also assess the pH dependence of the degradation reactions and the mechanism for degradation can often be determined from the pH dependence 38. The stability of each solution quickly analyzed by the formulation scientist. Rapid screening methods usually involve the use of accelerated stability at elevated temperatures (e.g. 40oC) 39-42.
Many proteins and peptides shows extensive degradation without affecting their safety and efficacy, such as recombinant human growth hormone (rhGH) is fully bioactive and nonimmunogenic but 70 % deamidated, this value of degradation not acceptable by regulatory agency standard for therapeutic protein 43.
Scientists must consider potential conditions and all the major degradation route of optimization to fulfill the regulatory requirements for a stable formulation. Surfactant is added in case of aggregation or sugar can prevent denaturation process, which leads to irreversible aggregation. The use of amine buffers (like Tris, ammonium, or imidazole) may slow the deamidation rate when degradation rate dominant 44. In another degradation pathway, for example, by adding surfactants or other polymers to prevent aggregation, the residual peroxide in the surfactant may cause a more rapid oxidation 45.
To decrease the deamidation rate pH of the formulation must be reduced in some cases. Reduction of pH may also alter the protein solubility, since many proteins have isoelectric points at or near the optimal pH (pH 5-6) for minimizing the deamidation rate. For each protein formulation, all the degradation pathways must be evaluated and often a balance must be achieved between the different degradation pathways. The researchers have options to develop solid formulation like lyophilized powder. The removal of excess water from the formulations decreases rate of the degradation in process of deamidation and hydrolysis. The residual moisture in a solid protein or peptide formulation can contribute to the physical stability of the protein by preventing its denaturation and subsequent aggregation upon reconstitution. Recent studies on lyophilization of proteins without excipients required some residual water for stability purpose 46. Different routes of protein and peptide delivery and polylactide applications presented in Table 2 and Table 3 respectively.
The basic concept for drug delivery from bioerodible polymers
Polymer matrix was represented by two dimensional computational grids, and each of grid point represents either an amorphous or crystalline part of the polymer matrix. The erosion of polymer mainly depends on two features (a) contact with the degradation medium (b) crystalline or amorphous nature of matrix. Polymer that's not contact with water will not erode. Pixels on the surface of the polymer matrix or next to an eroded neighbor have contact to water. The amorphous parts of polymer erode faster than the crystalline matrix 62.
Controlled release devices for the delivery of proteins and peptides from polymers
Biodegradable and non degradable polymers have been used in controlled release delivery of proteins and peptides. Direct diffusion through a non degradable polymer matrix is not possible due to the high molecular weight of proteins 63. By the introduction of a network of pores in manufacturing, however, the release of such large compounds does occur 64. Another possibility is the creation of hydrophilic pathways using swellable polymers, or embedding the compounds into gels. In the case of biodegradable polymers, pores are created upon erosion of the polymer matrix enabling the release of proteins from the dosage form. All of these options have certain advantages and disadvantages. Embedding suspended compounds into a non-degradable matrix prevents some of the protein from being released 65, or might cause some instability due to the intense contact with organic solvents 66. Their disadvantage with respect to parenteral application, however, is the need for removing such systems after therapy. By using gels as a carrier, the protein may be released very quickly if not combined with some other sort of material67,68. Degradable polymers change their properties substantially during erosion, a characteristic which may or may not be beneficial for the stability of proteins and peptides. In general, decisions about the suitability of a release device for specific proteins or peptides and appropriate manufacturing technology must be made on a case by case basis. In the following section, they provide a number of examples from the progress they made in the delivery of proteins from polymers.
Biodegradable polymers for immunization
In seventies, the idea of polymers were using as antigens releasing carriers for the simulation of response 69. Due to their progress and ease of application, potential efficient carriers (microspheres) emerged to enhance immune response 64. They have so far been used for a number of vaccines 70-72. These microspheres were made from polymers based on poly-lactic acid (PLA) and its copolymers with glycolic acid (PLGA). For these applications, loading a polymer with a high molecular weight protein requires adjusting the desired release rate and preventing the protein's loss of activity. In an attempt to develop a controlled release system for vaccination against tetanus, Sluzky et al. investigated the design of microspheres with a desired release rate and methods to maintain the imunogenicity of the processed tetanus toxoid 73.
Application of protein and peptides in diseases
Protein and peptides widely used in the management and treatment of various diseases such as cancer managements74, cancer therapy75, diabetes therapy76, Antimicrobial activity77,78, Peptides and peptidomimetics can function as immunomodulating agents by blocking the immune response79,80, neurodegenerative diseases 81 etc.
Cancer therapy and managements
Gene et al., describes a number of peptide based therapies we have developed to target theses pathways, and which are currently being tested in preclinical models. Therapeutics are based on a synthetic polymeric carrier elastin-like polypeptide (ELP), which can be synthesized in various sequences and sizes to stabilize the therapeutic peptide and avoid crossing the placental interface. This prevents fetal exposure and potential developmental effects 82.
Targeted delivery by cell-targeting peptides (CTPs) with the ability to recognize cancer cells is particularly attractive for cancer therapy; the use of these peptides has increased the specificity and efficacy of drug delivery while reducing side effects in a model 83. PEGA, a homing peptide used in the diagnosis of breast cancer, this peptide conjugated to the cell-penetrating peptide pVEC was taken up by different breast cancer cells 84. Peptide D2A21 as therapeutic for several types of cancer and has been developing this peptide gel formulation as a wound healing product to treat infected burns and wounds. A TAT peptide derived from the N-terminus of p53 has been used in the application of several tumor suppressor and apoptotic genes, TAT was fused to a peptide derived from the VHL tumor suppressor gene that inhibits insulin-like growth factor-I receptor (IGF-IR) signaling in renal cell carcinomas 85. Another study done by Su et al., observed that naturally existing anti-cancer bioactive peptide obtained from liver (ACBP-L) alone or in combination with the low dose Cisplatin improves quality of life in the xenograft tumor model having human gastric cancer 86. The bioactive peptide RRM-MV obtained by the resonant recognition model and Myxoma virus (RRM-MV), mimicks the bioactivity of specific MV proteins and evaluated the suitability of RRM-MV for human skin cancer therapy 87.
Diabetes therapy
In the study by Kaspar et al. (2013), recent successes in the development of peptides as therapeutics, most notably glucagon-like peptide 1 receptor (GLP-1R) agonists such as Lixisenatide (Lyxumia1), a GLP-1R agonist, was approved as a treatment for type 2 diabetes88. It competes with two other GLP-1R agonist peptides, exena-tide and liraglutide, for type 2 diabetes, they are administered subcutaneously. The GLP-1R agonist administered s.c. weekly is being compared to other diabetes drugs (i.e. exenatide, insulin glargine, metformin, sitagliptin, liraglutide) or placebo in type 2 diabetes patients. It acts (e.g. cell-targeting peptides, cell-penetrating peptides) by linking the peptides to various molecular formats (e.g. peptides linked to small molecules, lipids, carbohydrates, biopolymers, polyethylene glycol or proteins) and GLP-1R is able to normalize the levels of blood glucose in Type 2 diabetes through several mechanisms: the release of insulin is stimulated and the release of glucagon is suppressed89. In the another study by Wahren et al., C-peptide activates insulin receptor tyrosine kinase phosphorylation, and glycogen synthase kinase 3 phosphorylation , with downstream effects leading to GLUT mobilization, promotion of amino acid uptake, and glycogen synthesis, suggesting that C-peptide signaling may cross-talk with the insulin pathway at the level of the insulin receptor 90. Several clinical studies, with C-peptide replacement in patients with type 1 diabetes, show beneficial effects on somatic and autonomic Diabetic peripheral neuropathy (DPN), also the C-peptide mediates reduction of glomerular hyperfiltration and decrease of urinary albumin excretion in the diabetic state 91. Another protein peptide in diabetic study is G-protein-coupled receptors (peptide-binding GPCRs) that play an important role in the pathophysiology of vascular dysfunction during diabetes92.
Immunomodulating agents
The immunomodulatory action of biopeptides is related to the stimulation of proliferation of human lymphocytes and macrophages phagocytic activity 93. Lactoferrin (milk protein peptide) and its derivatives affect the production of cytokines involved in immune reactions of the organism94. LF-derived peptides have clinical applications due to their chemopreventive and immunomodulatory properties 95. α-lactalbumin (whey protein fraction of bovine milk) has immunomodulatory properties, whereas products of its degradation by trypsin and chemotrypsin (f1 - 5, f17 - 31-SS-f109 -114 and f61 - 68-SS-f75 - 80) or pepsin exhibit both immnunomodulatory and antimicrobial properties against bacteria, viruses and fungi 96. Acidic peptides derived from β-Lactoglobulin under the action of peptidases of Lactobacillus paracasei decreased the stimulation of lymphocytes and regulated production of IL-10, IFN-γ and IL-4 this indicates eliminating allergic reaction in cow milk 97. Casein fraction and its derived peptide (isracidin) of milk total protein, is a rich source of bioactive peptides that stimulate and aid the immune system 98. In the study by Rodrigo and Albani, Peptides derived from the E. coli heat shock protein (hsp) dnaJ are antigenic in human autoimmune disorder rheumatic arthritis 99. T cell recognition of these peptides is associated with TH-I type and pro-inflammatory responses, including production of TNF-α and IFN-g in the pathogenesis of autoimmune inflammation. Another studied peptide LL-37 is a human antimicrobial peptide derived from cathelicidin has anti-endotoxic activity, induces chemokine production, promotes IL-1b secretion, but inhibits inflammatory responses to certain TLR ligands 100,101. Neutrophils LL-37 and defensins are produced by neutrophils, stored within neutrophil granules and play an important microbicidal role in phagolysosomes.
Neurodegenerative diseases
There are currently no effective drugs for the treatment of neurodegenerative diseases; potential therapeutic targets for symptomatic treatments of neurodegenerative diseases may include neuroprotective factors, neurotrophins and neuroprotective peptides 102,103. Peptides like NAP (NAPVSIPQ) derived from activity of neuroprotective protein(NAP) and ADNF-9 peptide derived from activity-dependent neurotrophic factor (ADNF) in the treatment of Alzheimer's disease (AD) and Amyotrophic lateral sclerosis (ALS) disease. ADNF-9 has been found to protect against Aβ, apolipoprotein E deficiencies, and oxidative stress, as well as increasing synapse formation. Colivelin another protein peptide also has potential neuroprotecrive effect in neurodegenerative diseases such as AD 104. NAP mimics the neuroprotective activity of ADNP in its ability to cross then blood-brain barrier, interact with tubulin, enhance assembly of microtubules, and promote neuronal outgrowth in AD and ALS disease 104. In the another discovery of poly Q binding protein (QBP1) peptide, these QBP1 specifically binds to the expanded polyQ stretch and inhibits its misfolding and aggregation, resulting in suppression of neurodegeneration in cell culture and animal models of the polyQ diseases (Huntington's disease, spinocerebellar ataxia different types, dentatorubral pallidoluysian atrophy, and spinobulbar muscular atrophy (SBMA)] 106.
Antimicrobial activity
Antimicrobial peptides (AMPs) are widely distributed in nature, are found in species ranging from bacteria and insects to mammals 107. Most of these peptides are synthesized as a prepropeptide consisting of an N-terminal signal sequence, a pro segment and a C-terminal cationic peptide that demonstrates antimicrobial activity after it is cleaved from the rest of the protein 108. Compared to conventional anti-infective agents, some AMP may kill bacteria but also simultaneously neutralize released pathogenic factors, like lipopolysaccharide (LPS) or lipoprotein (LP). AMPs have anti-Gram-positive and -negative effects 109 as well as antiviral and anti-yeast effects. Peptides have shown potential and desirable therapeutic properties like antimicrobial, antiviral, anticancer and contraceptive activities. Members of the major groups of antimicrobial peptides have been classified mainly on the basis of their biochemical (net charge) and/or structural features (linear/circular/amino acid composition).
Cationic peptides
This is the largest group and the first to be reported, being widely distributed in animals and plants. On the basis of their structural features, cationic peptides can be divided as well into three different classes: (1) linear peptides forming-helical structures (such as Cecropins) ; (2) cysteine-rich open ended peptides containing single or several disulfide bridges (such as Defensins); and (3) molecules rich in specific amino acids such as proline, glycine, histidine and tryptophan (such as amino-acid enriched class) 110. Some other cationic peptides include, Thionins (plant peptides) 111 and histone derived compounds, beta-hairpin, cathelicidins and thrombocidins.
Anionic peptides
This is a smaller group of antimicrobial peptides which includes; Neuropeptide derived molecules,Oxygen-binding proteins, Aromatic dipeptides and Aspartic-acid-rich molecules. Some other AMPs Bacterial ribosomes synthesize antimicrobial peptides which are generally called as bacteriocins (in bacteria), cecropins, Drosomycin and metchnikowin (in insect). These peptides however, have certain properties in common. They all have an affinity for membrane lipids and their specificity for microbial membranes in many cases has been shown to be related to the positive charge on the peptide favouring interaction with the exposed anionic lipids of microorganisms. The peptides may form pores in the membrane allowing for leakage of ions and other materials from the cell. The activity of the peptide is explained by mechanisms like carpet, barrel stave, toroidal along with these mechanisms, it shows an intracellular killing activity which affects the nucleic acid of the microorganism 112.
Numerous studies focus on multipotential activity of lactoferricin (Lfcin) that is a product of hydrolytic degradation of lactoferrin (LF). Lactoferrin itself exhibits a strong bactericidal activity through its ability to bind iron. This peptide shows a considerably higher antimicrobial activity than the native protein 113. Lactoferrampin (Lfampin) is another peptide derived from lactoferrin. It has a wide spectrum of antifungal and antibacterial properties. The peptide exerted antifungal (against Candida) activity higher than LF and was also active against Bacillus subtilis, Escherichia coli and Pseudomonas aeruginosa 114. Lactoferrin and its derivatives show the antibacterial activity in vitro against various pathogens, e.g. Clostridium perfringens, Candida albicans, Haemophilus influenzae, Helicobacter pylori, Pseudomonas aeruginosa, Staphylococcus aureus, Streptoccccus mutans, Vibrio cholerae as well as antiviral activity against hepatitis C and B virus; HIV-1; poliovirus; rotavirus; and herpes simplex virus 115. Hydrolysis of αs2-casein (by chymosin acting) results in releasing casocidin, this peptide shows antibacterial properties. It was also reported that antibacterial peptides obtained from αs2-casein (i.e. f183-207 and f164-179) inhibit growth of both Gram-positive and Gram-negative bacteria. In the study by Rivas et al., resulted that Penetratin, the peptide derived from the drosophila transcription factor antennapedia and the HIV1-Tat derived nonapeptide, belong to the arginine-class of peptides. These peptides exhibit antimicrobial or antifungal properties 116.
Conclusions
Several variables play important role in the design, development and delivery methods of protein and peptides. To make successful protein and peptide drugs, the relationship between the routes of administration, pharmacokinetics, toxicity and clinical indication should be carefully balanced. Many novel systems like liposome and polymer offers smart alternative to solid and liquid dosage form. New delivery often requires invasive method but noninvasive delivery routes such as transdermal or pulmonary delivery may give up capable results for protein and peptides in future. The delivery of DNA and RNA in gene therapy and antisense therapy respectively is addressed by formulations scientist. A new type of system such as biodegradable nanosphere is available in near future because presently they are the focus for research by scientists. Finally we concluded that the protein and peptide have versatile applications in the field of drug delivery.
Fundings
No funding agency for the research work.
Competing interest
Author declares no conflict of interest.
Acknowledgements
Authors are thankful to the Mr. Adi Dev Bharti, for valuable suggestion.
References
1. Cleland J, et al. In Formulation and Delivery of Proteins and Peptides; ACS Symposium Series; Amer. Chem. Societ. Washington, DC. 1994; 1: 1-2. [ Links ]
2. Luft FC, Lang GR, Aronoff H, Ruskoaho M, Toth M, Ganten D, Sterzel RB, Unger T. Atriopeptin III kinetics and pharmacodynamics in norma and anephric rats. J. Pharmacol. Exp. Ther. 1986; 236:416-428. [ Links ]
3. Zhang F, Liu CL, Hu BR. Irreversible aggregation of protein synthesis machinery after focal brain ischemia. J. Neurochem. 2006; 98(1):102-112. [ Links ]
4. Witkop B. Nonenzymatic Methods For The Preferential and Selective Cleavage and Modification Of Proteins. Adv. Protein. Chem. 1961;16:221-321. [ Links ]
5. Marcritchie F. Adv. Proteins at interfaces. Protein. Chem. 1978;32:283-326. [ Links ]
6. Zhou XH, Li WP. Peptide and protein drugs. I Therapeutic applications, absorption and parenteral administration. Int. J. Pharm. 1991a; 75(2-3):97-115. [ Links ]
7. Zhou XH, Li WP. Peptide and protein drugs. I Therapeutic applications, absorption and parenteral administration. Int. J. Pharm. 1991a; 75: 117-130. [ Links ]
8. Sadee W. Protein drugs: A revolution in therapy. Pharm. Res. 1986; 3(1): 3-6. [ Links ]
9. Blume JP, Hunter T. Oncogenic kinase signalling. Nature. 2001; 411(6835):355-365. [ Links ]
10. Cohen P. Protein kinases-the major drug targets of the twenty-firstcentury. Nat. Rev. Drug. Discov. 2002; 1(4):309-15. [ Links ]
11. Saltiel AR, Pessin JE. Insulin signaling pathways in time andspace. Trends Cell. Biol. 2002; 12(2): 65-71. [ Links ]
12. Torchilin VP. Recent advances with liposomes as pharmaceuticalcarriers. Nat. Rev. Drug. Discov. 2005; 4:145-160. [ Links ]
13. Vyas SP, Khar KR. Targeted and controlled drug delivery, Novel carrier system, CBS publishers and distributors, New Delhi. 2002; 505-537. [ Links ]
14. Rapoport T. Protein translocation across the eukaryotic endoplasmic reticulum and bacterial plasma membranes. Nature. 2007; 450 (7170):663-669. [ Links ]
15. Martin RB. Free energies and equilibria of peptide bond hydrolysis and formation. Biopolymers. 1998; 45:351-353. [ Links ]
16. Chein YW. Novel drug delivery systems, second edition, 1992; 50:637-679. [ Links ]
17. Chien YW, Chang SF. Intranasal drug delivery for systemic medication. Crit. Rev. Ther. Drug. Carrier. Syst. 1987; 4:67-194. [ Links ]
18. Banga AK, Chein YW. Systemic delivery of therapeutic peptides and proteins. Int. J. Pharm. 1988; 48:15-50. [ Links ]
19. Wieriks J. Resorption of alpha amylase upon buccal application. Arch. Int. Pharmacodyn. Ther. 1964; 151:126-135. [ Links ]
20. Tregear RT. The permeability of skin to albumin, dextrans and Polyvinylpyrrolidone. J. Invest. Dermatol. 1996; 46:24-27. [ Links ]
21. Menasche. Pharmacological studies on elastin peptides (kappa-elastin). Blood clearance, percutaneous penetration and tissue distribution. Pathol. Biol. 1981; 29:548-554. [ Links ]
22. Brunette BR, Marreco D. Comparison between the iontophoretic and passive transport of thyrotropin releasing hormone across excised nude mouse skin. J Pharm. Sci. 1986; 75:738-743. [ Links ]
23. Siddiqui O, Sun Y, Liu JC, Chein YW. Facilitated transdermal transport of insulin. J Pharm. Sci. 1987; 76:341-345. [ Links ]
24. Wolf M, Ransberger K. Enzyme-therapy. Vantage Press. 1972. [ Links ]
25. Holcenberg JS, Roberts J. (ed.) Enzymes as Drugs, Wiley. 1981. [ Links ]
26. Torchilin VP. (ed.) Immobilized Enzymes in Medicine, Springer-Verlag. 1991. [ Links ]
27. Froidevaux S, Eberle A N. Somatostatin analogs andradiopeptides in cancer therapy. Biopolym. 2002; 66:161-183. [ Links ]
28. Baselga J, Albanell J. Mechanism of action of anti-HER2monoclonal antibodies. Ann. Oncol. 2001; 12 (1):S35-S41. [ Links ]
29. Marshall H. Anti-CD20 antibody therapy is highly effective in thetreatment of follicular lymphoma.Trends Immun. 2001; 22 183-184. [ Links ]
30. Varga CM. Receptor-mediated targeting of gene deliveryvectors:insights from molecular mechanisms for improved vehicle design. Biotechnol. Bioeng. 2000; 70:593-605. [ Links ]
31. Hanks SK, Hunter T. The eukaryotic proteinkinase superfamily:kinase (catalytic) domain structure and classification. Faseb J. 1995; 9(8):576-96. [ Links ]
32. Pinna LA, Ruzzene, M. How do protein kinases recognize their substrates? Biochim. Biophys. Acta. 1996; 1314(3):191-225. [ Links ]
33. Ron D, Mochly-Rosen D. An autoregulatory region in proteinkinase C:the pseudo anchoring site.Proc. Natl. Acad. Sci. USA1995; 92(2):492-496. [ Links ]
34. Schechtman D, Mochly RD. Adaptor proteins in proteinkinase C-mediated signal transduction. Oncogene. 2001; 20(44):6339-6347. [ Links ]
35. Niv MY, Rubin H, Cohen J, Tsirulnikov L, Licht T, Peretzman-Shemer A. Sequence-based design of kinase inhibitors applicablefor therapeutics and target identification. J Biol Chem. 2004; 279(2):1242-55. [ Links ]
36. Mack E, Ziv E, Reuveni H, Kalman R, Niv MY, Jorns A. Prevention of insulin resistance and beta-cell loss by abrogatingPKCepsilon-induced serine phosphorylation of muscle IRS-1 in Psammomysobesus. Diabetes Metabol. Res. Rev. 2008; 24(7):577-584. [ Links ]
37. Ron D, Luo J, Mochly-Rosen D. C2 region-derived peptides inhibit translocation and function of beta protein kinase C in vivo. J. Biol. Chem. 1995; 270(41), 24180-24187. [ Links ]
38. Loudon GM. Mechanistic interpretation of pH-rate profiles. J. Chem Ed. 1991; 68:973-984. [ Links ]
39. Yoshioka S, Aso Y, Izutsu KI, Terao T. Application of accelerated testing to shelf-life prediction of commercial protein preparations. J. Pharm. Sci. 1994; 83 (3):454-456. [ Links ]
40. Cleland JL, Powell MF, Shire SJ. The development of stable protein formulations:a close look at protein aggregation, deamidation, and oxidation. Crit. Rev. Therap. Drug. Carr. Syst. 1993; 10:307-377. [ Links ]
41. Pearlman R, Nguyen T. Pharmaceutics of protein drugs. J. Pharm. Pharmacol. Sppl. 1992; 44 (1):178-185. [ Links ]
42. Gu KM, Erdo EΑ, Chiang H, Calderwood T, Tsai K, Visor GC, Duffy J, Hsu W, Foster LC. Stability of interleukin 1 beta (IL-1 beta) in aqueous solution:analytical methods, kinetics, products, and solution formulation implications. Pharm. Res. 1991; 8:485-490. [ Links ]
43. Skottner Α, Forsman Α, Skoog B, Kostyo JL, Cameron CM, Adamfio NΑ, Thorngren KG, Hagerman M. Biological characterization of charge isomers of human growth hormone. Acta. Endocrinol. 1988; 118:14-19. [ Links ]
44. Oliyai C, Schoneich C, Wilson GS, Borchardt RT. In:Topics in Pharmaceutical Sciences, Crommelin DJΑ, Miha KK, (Ed.) Med. Pharm. Scientific Publishers, Stuttgart. 1992; 23-46. [ Links ]
45. Hora MS, Rana RK, Wilcox CL, Katre NV, Hirtzer P, Wolfe SN., Thomson JW. Development of a lyophilized formulation of interlukin-2.Dev. Biol. Stand. 1991; 74:295-307. [ Links ]
46. Wearley LL. Recent progress in protein and peptide delivery by noninvasive routes. Crit. Rev. Therap Drug Carrier Syst. 1991; 8:331-394. [ Links ]
47. Oliyai R, Stella VJ. Prodrugs of peptides and protein for improved formulation and delivery. Annu. Rev. Pharmcol. Toxicol. 1993; 32:521-544. [ Links ]
48. Miyamoto S, Takaoka K, Okada T, Yoshikawa H, Hashimoto J, Suzuki S, Ono K. Polylactide acid -polyethylene glycol block copolymer:a new biodegradable synthetic carrier for bone morphogenetic protein. Clin. Orthopaedics Related Res. 1993; 294:333-343. [ Links ]
49. Gombotz WR, Pankey SC, Bouchard LS, Ranchalis J, Puolakkainen P. Controlled release of TGF-beta (1) from a biodegradable matrix for bone regeneration. J. Biomater. Sci. Polymer 1993; 5:49-63. [ Links ]
50. Heya, T, Okada H, Ogawa Y, Toguchi H. Factors influencing the profiles of TRH release from copoly (d,l-lactic/glycolic acid) microspheres. Int. J. Pharm. 1991; 72:199-205. [ Links ]
51. Mariette Β, Coudane J, Vert M, Gautier JC, Moneton P. Release of the GRF29NH2 analog of human GRF44NH2 from a PLA/GA matrix. J Contrl. Rel. 1993; 237-246. [ Links ]
52. Bodmer D, Kissel T, Traechslin E. Factors influencing the release of peptides and proteins from biodegradable parenteral depot systems. J. Contrl. Rel. 1992; 21:129-138. [ Links ]
53. Yamakawa I, Tsushima Y, Machida R, Watanabe S. Preparation of neurotensin analogue-containing poly(dl-lactic acid) microspheres formed by oil in water solvent evaporation. J. Pharm Sci. 1992; 81:899-903. [ Links ]
54. Sanchez Α, Vila-Jato JL, Alonso MJ. Development of biodegradable microspheres and nanospheres for the controlled release of cyclosporine A. Int. J. Pharm. 1993; 99(2-3):263-273. [ Links ]
55. Reid RH, Boedeker EC, McQueen CE, Davis D, Tseng LY, Kodak J, Sau K. Preclinical evaluation of microencapsulated CFA/II oral vaccine against enterotoxi- genic E. coli. Vaccine 1993; 11(2):159-167. [ Links ]
56. Almeidia AJ, Alpar HO, Williamson D, Brown MRW. Poly(lactic acid) microspheres as immunological adjuvants for orally delivered cholera toxin B subunit. 643rd Meeting of the Biochemical Society (Warwick, UK). Biochem. Soc.Trans. 1992; 20:316S. [ Links ]
57. Singh M, Sing, O, Singh Α, Talwar GP. Immunogenicity studies on diphtheria toxoid loaded biodegradable microspheres. Int. J. Pharm. 1992; 85:R5-R8. [ Links ]
58. Jeffery H, Davis SS, O'Hagen DT. The preparation and characterization of poly (lactide-co-glycolide) microparticle. II. The entrapment of a model protein using a (water-in-oil)-in-water emulsion solvent evaporation technique. Pharm. Res. 1993; 10:362-368. [ Links ]
59. Alonso MJ, Cohen S, Park TG, Gupta RK, Siber GR, Langer R. Determinants of release rate of tetanus vaccine from polyester microspheres. Pharm. Res. 1993; 10 (7):945-953. [ Links ]
60. Stoeckemann K, Sandow J. J. Cancer Res. Clin. Oncology 1993; 119:457-462. [ Links ]
61. Cohen S, Yoshioka T, Lucarelli M, Hwang LH, Lange R. Controlled Delivery Systems for Proteins Based on Poly(Lactic/Glycolic Acid) Microspheres Pharm. Res. 1991; 8(6):713-720. [ Links ]
62. Gopferich A, Langer R. The influence of microstructure and monomer properties on the erosion mechanism of a class of polyanhydrides. J. Polymer Sci. 1993; 31:2445-2458. [ Links ]
63. Langer R, Folkman J. Polymer for the sustained release of proteins and other macromolecules. Nature, 1976; 263 (5580):797-800. [ Links ]
64. Siegel R, Kost J, Langer R. Mechanistic studies of macromolecular drug release from macroporous polymers. I. Experiments and preliminary theory concerning completeness of drug release J. Contr. Rel., 1989; 8(3):223-236. [ Links ]
65. Arakawa T, Kita Y, Carpenter F. Protein-Solvent Interactions in Pharmaceutical Formulations. Pharm. Res. 1991; 8:285-291. [ Links ]
66. Edelman ER, Mathiowit E, Langer R, Klagsbrun M. Controlled and modulated release of basic fibroblast growth factor. Biomaterials. 1991; 7(12):619-626. [ Links ]
67. Andrianov AK, Cohen S, Visscher KB, Payne LG, Allcock HR, Langer R. Controlled release using ionotropic polyphosphazene hydrogels J. Contr. Rel. 1993; 27 (1):69-73. [ Links ]
68. Kohn J, Niemi SM, Albert EC, Murphy JC, Langer R, Fox J. Single step immunization using a controlled release, biodegradable polymer with sustained adjuvant activity. J. Immunol. Method. 1986; 95:31-38. [ Links ]
69. Preis I, Langer R. A single-step immunization by sustained antigen release. J. Immunol. Method. 1979; 28:193-197. [ Links ]
70. O'Hagan DT, Rahman D, McGee JP, Jeffery H, Davies MC, Williams P, Davis SS, Challacombe S. Biodegradable microparticles as controlled release antigen delivery systems. Immunology. J Immunology. 1991; 73:239-242. [ Links ]
71. Singh M, Singh A, Talwar GP. Controlled Delivery of Diphtheria Toxoid Using Biodegradable Poly(D,L-Lactide) Microcapsules. Pharm. Res. 1991; 8:958-961. [ Links ]
72. Eldridge JH, Staas JK, Meulbroek JA, Tice TR, Gille RM. Biodegradable and biocompatible poly(DL-lactide-co-glycolide) microspheres as an adjuvant for staphylococcal enterotoxin B toxoid which enhances the level of toxin-neutralizing antibodies. Infect. Immun. 1991; 59:2978-2986. [ Links ]
73. Sluzky V, Klibano AM, Langer R. Mechanism of insulin aggregation and stabilization in agitated aqueous solutions. Biotech. Bioeng. 1992; 40:895-903. [ Links ]
74. Chen HY, Mollstedt O, Tsai MH, Kreider RB. Potential clinical applications of multi-functional milk proteins and peptides in cancer management. Curr Med Chem. 2014; 21(21):2424-2437. [ Links ]
75. Wangler C, Buchmann I, Eisenhut M, Haberkorn U, Mier W. Radiolabeled peptides and proteins in cancer therapy. Protein Pept Lett. 2007; 14(3):273-239. [ Links ]
76. Rekha MR, Sharma Chandra P. Oral delivery of therapeutic protein/peptide for diabetes - Future perspectives. Intl. J Pharm. 2013; 440 (1):48-62. [ Links ]
77. Wang G. Human antimicrobial peptides and proteins. Pharmaceuticals. 2014; 7(5):545-594. [ Links ]
78. Bartlomiej D, Marta D. New milk protein-derived peptides with potential antimicrobial activity:An approach based on bioinformatic studies. Int. J. Mol. Sci. 2014; 15:14531-14545. [ Links ]
79. Gokhale AS, Satyanarayanajois S. Peptides and peptidomimetics as immunomodulators. Immunotherapy. 2014; 6 (6):755-774. [ Links ]
80. Sarah CM, Olga MP, Hancock REW. Host defense peptides:front-line immunomodulators. Trends in Immunology. 2014; 35 (9):443-450. [ Links ]
81. Kishore U. Neurodegerative disease (ed.) In:Role of protein aggregation in neurodegenerative diseases. Tutar Y, Ozgur A, Tutar L. Chap. 2013; 3 (2013):55-76. [ Links ]
82. Gene L, Bidwell III, George EM. Maternally sequestered therapeutic polypeptides - a new approach for the management of preeclampsia. Frontiers in Pharmacology. 2014; 5(201):1-9. [ Links ]
83. Zhang XX, Eden HS, Chen X. Peptides in cancer nanomedicine: Drug carriers, targeting ligands and protease substrates. J. Control. Release. 2012; 159:2-13. [ Links ]
84. Regberg J, Srimanee A, Langel U. Applications of Cell-Penetrating Peptides for Tumor Targeting and Future Cancer Therapies. Pharmaceuticals. 2012; 5, 991-1007. [ Links ]
85. Harada, H, Kizaka-Kondoh S, Hiraoka M. Antitumor protein therapy; application of the protein transduction domain to the development of a protein drug for cancer treatment. Breast Cancer 2006; 13:16-26. [ Links ]
86. Su X, Dong C, Zhang J, Su L, Wang X, et al. Combination therapy of anti-cancer bioactive peptide with Cisplatin decreases chemotherapy dosing and toxicity to improve the quality of life in xenograft nude mice bearing human gastric cancer. Cell & Bioscience 2014; 4:7, 1-13. [ Links ]
87. Almansour NM, Pirogova E, Coloe PJ, et al. A bioactive peptide analogue for myxoma virus protein with a targeted cytotoxicity for human skin cancer in vitro. Journal of Biomedical Science 2012; 19 (65):1-13. [ Links ]
88. Kaspar Allan A. and Reichert, JM. Future directions for peptide therapeutics development. Drug Discovery Today, 2013; 1-11. [ Links ]
89. Andrea El-Ouaghlidi, and Michael A Nauck, Glucagon-like peptide 1:new. therapies for Type 2 diabetes. Diabetes voice, 2004; 49:2, 24-26. [ Links ]
90. Wahren J, Kallas A, et al. The clinical potential of C-peptide replacement in type-1 diabetes. 2012; 61:1-12. [ Links ]
91. Luppi P, Cifarelli V, Wahren J. C-peptide and long-term complications of diabetes. Pediatr Diabetes 2011; 12:276-292. [ Links ]
92. Carrillo-Sepulveda, MA, Matsumoto T, Nunes KP, and Webb RC. Therapeutic implications of peptide interactions with G-protein-coupled receptors in diabetic vasculopathy. Acta Physiol 2014; 211:20-35. [ Links ]
93. Clare DA, Catignani GL, Swaisgood HE. Biodefense properties of milk:the role of antimicrobial proteins and peptides. Current Pharmaceutical Design 2003; 29:1239-1255. [ Links ]
94. Moller NP, Scholz-Ahrens KE, Roos N, Schrezenmeir J. Bioactive peptides and proteins from foods:indication for health effects. European J. Nutr. 2008; 47, 171-182. [ Links ]
95. Krol J, Brodziak A, Litwińczuk Z, Szwajkowska M. Wykorzystanie białek serwatkowych w promocji zdrowia (Whey protein utilization in health promotion). In Polish, summary in English. Żywienie człowieka i metabolizm XXXVIII 2011; (1):36-45. [ Links ]
96. Kamau SM, Cheison SC, Chen W, Liu XM, Lu RR. Alpha-Lactalbumin:its production technologies and bioactive peptides. Comprehensive Reviews in Food Science and Food Safety 2010; 9:197-212. [ Links ]
97. Prioult G, Pecquet S, Fliss I. Stimulation of interleukin-10 production by acidic beta-lactoglobulin-derived peptides hydrolyzed with Lactobacillus paracasei NCC2461 peptidases. Clinical and Diagnostic Laboratory Immunology 2004; 11:266-271. [ Links ]
98. Szwajkowska M, Wolanciuk A, Barłowska J, Król J, Zygmunt Litwińczuk. Bovine milk proteins as the source of bioactive peptides influencing the consumers' immune system - a review. Animal Science Papers and Reports 2011; 29 (4):269-280. [ Links ]
99. Rodrigo TS, Salvatore A. Immunomodulatory Effects by a Heat Shock Protein dnaJ Derived Peptide in Rheumatoid Arthritis. Specific Immunotherapy of Chronic Autoimmune Diseases. 1999; 63-71. [ Links ]
100. Buchau AS. Schauber J, Hultsch T, Stuetz A, Gallo RL. Pimecrolimus enhances TLR2/6- induced expression of antimicrobial peptides in keratinocytes. J. Invest. Dermatol. 2008; 128:2646-2654. [ Links ]
101. Rosenberger CM, Gallo RL, Finlay BB. Interplay between antibacterial effectors:a macrophage antimicrobial peptide impairs intracellular Salmonella replication. Proc. Natl. Acad. Sci. USA 2004; 101:2422-2427. [ Links ]
102. Lu B, Pang PT, Woo NH. The yin and yang of neurotrophin action. Nat Rev Neurosci. 2005; 6:603-14. [ Links ]
103. Chao MV, Rajagopal R, Lee FS. Neurotrophin signalling in health and disease. Clin Sci (Lond). 2006; 110:167-73. [ Links ]
104. Chiba T, Yamada M, Sasabe J, Terashita K, Aiso S, et al. Colivelin prolongs survival of an ALS model mouse. Biochem Biophys Res Commun. 2006; 343:793-798. [ Links ]
105. Vulih-Shultzman I, Pinhasov A, Mandel S, et al. Activity dependent neuroprotective protein snippet NAP reduces tau hyperphosphorylation and enhances learning in a novel transgenic mouse model. J Pharmacol Exp Ther. 2007; 323:438-49. [ Links ]
106. Popiel HA, Burke J, Warren RJ, et al. The Aggregation Inhibitor Peptide QBP1 as a Therapeutic Molecule for the Polyglutamine Neurodegenerative Diseases. Journal of Amino Acids, 2011. [ Links ]
107. Velden WJ, Van Iersel, TM, Blijlevens NM, Donnelly JP. Safety and tolerability of the antimicrobial peptide human lactoferrin 1-11 (hLF1-11).In BMC Med. 2009; 7:44. [ Links ]
108. Bals R. Epithelial antimicrobial peptides in host defense against infection. Respirat. Research. 2000; 1 (3):141-150. [ Links ]
109. Schuerholz T, Brandenburg K, Marx G. The anti-inflammatory effect of the synthetic antimicrobial peptide 19-2.5 in a murine sepsis model:a prospective randomized study. Critical Care 2012; 16:207. [ Links ]
110. Bulet P, Hetru C, Dimarcq J. Hoffmann D. Antimicrobial peptides in insects; structure and function. Devel. Comparat. Immunol. 1999; 23(4-5):329-344. [ Links ]
111. Li SS, Gullbo J, Lindholm P, Larsson R, et al. Ligatoxin B, a new cytotoxic protein with a novel helix-turn-helix DNA-binding domain from the mistletoe Phoradendron liga. Biochem. J. 2002; 366 (2) :405-413. [ Links ]
112. Peravali JB, Kotra SR, Sobha K, et al. Antimicrobial peptides:an effective alternative for antibiotic therapy. Mintage J. Pharm. Med. Sci. 2013; 2 (2):1-7. [ Links ]
113. Oo TZ, Cole N, Garthwaite L, Mark D, Willcox P, Zhu H. Evaluation of synergistic activity of bovine lactoferricin with antibiotics in corneal infection. J. Antimicrob. Chemother. 2010; 65:1243-1251. [ Links ]
114. van der Kraan MIA, Nazmi K, et al. Lactoferrampin, an antimicrobial peptide of bovine lactoferrin, exerts its candidacidal activity by a cluster of positively charged residues at the C-terminus in combination with a helix-facilitating N-terminal partThe J. Biol. Chem. 2005; 386:137-142. [ Links ]
115. Pan Y, Rowney M, Guo P, Hobman P. Biological properties of lactoferrin:an overview. Austr. J. Dairy Techn. 2007; 62:31-42. [ Links ]
116. Rivas L, Luque-Ortega JR, Fernandez-Reyes M, Andreu D. Membrane-active peptides as anti-infectious agents. J. Appl. Biomed. 2010; 8:159-167. [ Links ]
![]() Correspondence:
Correspondence:
Raj K. Keservani
School of Pharmaceutical Sciences
Rajiv Gandhi Proudyogiki Vishwavidyalaya
Bhopal, India-462036
Mobile:+917897803904
rajksops@gmail.com
Received:19.04.2015
Accepted:26.06.2015