Manejo de la infección por el VHC en la enfermedad renal crónica
Management of HCV infection in chronic kidney disease
S. Aoufi Rabih1, R. García Agudo2
1Servicio de Aparato Digestivo. Complejo Hospitalario La Mancha-Centro. Alcázar de San Juan, Ciudad Real
2Servicio de Nefrología. Complejo Hospitalario La Mancha-Centro. Alcázar de San Juan, Ciudad Real
Dirección para correspondencia
]]>
RESUMEN
La prevalencia de la infección crónica por el virus de la hepatitis C (VHC) en pacientes con enfermedad renal crónica es mayor que en la población general. En hemodiálisis, se estima una prevalencia del 13%, con una amplia variabilidad geográfica y entre las unidades de un mismo país. La biopsia hepática es una herramienta útil para decidir el inicio de la terapia antiviral y excluir causas concomitantes de disfunción hepática, como la hepatopatía grasa no alcohólica, cuya incidencia está en auge, y la hemosiderosis, que pueden afectar a la progresión de la enfermedad y condicionar la respuesta al tratamiento antiviral; además, la vía transyugular se puede utilizar para medir el gradiente de presión venoso hepático y confirmar la existencia de hipertensión portal. La hepatitis crónica por el VHC ha demostrado reducir la supervivencia en hemodiálisis y en el trasplante renal, así como la supervivencia del injerto. Constituye la cuarta causa de mortalidad y la principal causa de disfunción hepática postrasplante renal. El VHC se comporta como un factor de riesgo independiente para la aparición de proteinuria, aumenta el riesgo de desarrollar diabetes, una glomerulonefritis de novo o una nefropatía crónica del injerto, de empeorar la enfermedad hepática y de provocar un mayor número de infecciones. También se ha descrito un incremento de la frecuencia de hepatitis colestásica fibrosante que, junto a la evolución acelerada a cirrosis, puede elevar significativamente la morbimortalidad y conllevar la necesidad de un trasplante hepático. Además, la inmunosupresión en el trasplante renal predispone a la reactivación del VHC. Sin embargo, como la farmacocinética del interferón y la ribavirina está alterada en la insuficiencia renal y su uso tiene efectos adversos sobre la función y la supervivencia del injerto, la terapia combinada se limita a los individuos no trasplantados con un filtrado glomerular estimado mayor de 50 ml/min y en diálisis suele emplearse el interferón en monoterapia. El hecho de que una cuarta parte de los pacientes VHC-positivos evaluados para trasplante renal tenga fibrosis en puente o cirrosis en la biopsia hepática puede renovar el planteamiento del tratamiento pretrasplante renal.
Palabras clave: Virus de la hepatitis C, Enfermedad renal crónica, Hemodiálisis, Biopsia hepática, Trasplante renal, Interferón, Ribavirina.
ABSTRACT
The prevalence of chronic infection with the hepatitis C virus (HCV) in patients with chronic kidney disease is higher than in the general population. The estimated prevalence is 13% in haemodialysis, with wide variations geographically and between units in the same country. A liver biopsy is a useful tool for deciding whether to start antiviral therapy and to exclude concomitant causes of liver dysfunction. Examples of this include nonalcoholic fatty liver disease, whose incidence is on the rise, and haemosiderosis, which may affect the progression of the disease and determine the response to antiviral therapy. In addition, the transjugular approach can be used to measure the hepatic venous pressure gradient and confirm the existence of portal hypertension. Chronic hepatitis due to HCV has been shown to reduce survival in haemodialysis, renal transplantation and graft survival. It is the fourth leading cause of death and the leading cause of post-renal transplantation liver dysfunction. HCV behaves as an independent risk factor for the occurrence of proteinuria; it increases the risk of developing diabetes mellitus, de novo glomerulonephritis and chronic allograft nephropathy; it leads to a deterioration in liver disease and causes a greater number of infections. An increased frequency of fibrosing cholestatic hepatitis has also been described which, together with the rapid evolution to cirrhosis, can significantly increase morbidity and mortality and lead to the need for liver transplantation. In addition, immunosuppression in renal transplantation predisposes a reactivation of HCV. However, as the pharmacokinetics of interferon and ribavirin is impaired in kidney failure and their use has adverse effects on function and graft survival, a combination therapy must be limited to non-transplanted individuals with an estimated glomerular filtration rate greater than 50ml/min, and with the interferon being used as monotherapy in dialysis. The fact that a quarter of HCV-positive patients evaluated for a renal transplant have bridging fibrosis or cirrhosis in the liver biopsy may renew renal pre-transplant treatment planning.
Key Words: Hepatitis C virus, Chronic kidney disease, Hemodialysis, Liver biopsy, Renal transplantation, Interferon, Ribavirin.
Introducción
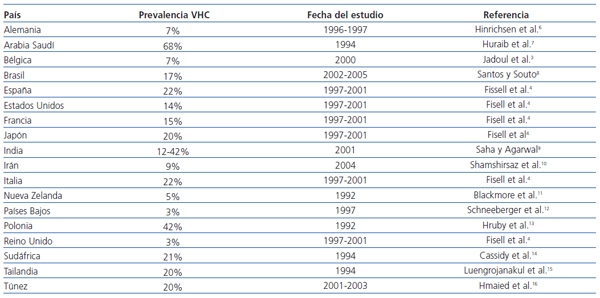
]]> La Organización Mundial de la Salud estima la prevalencia global de la infección crónica por el virus de la hepatitis C (VHC) en el 3%, con una amplia variabilidad geográfica: menor del 5% en la mayor parte de los países del norte de Europa, alrededor del 10% en el sur de Europa y Estados Unidos y del 10-50% y hasta el 70% en muchos países en vías de desarrollo, incluyendo algunas zonas de Asia, Latinoamérica y norte de África. Se ha logrado disminuir la incidencia de la infección por el VHC a menos del 1-2% en países desarrollados1-3.La infección por el VHC en pacientes con enfermedad renal crónica (ERC) estadio 5 es mayor que en la población general. En hemodiálisis, se observa una prevalencia del 13%, con una variabilidad del 1-70%4 (tabla 1). Asimismo, la prevalencia del VHC es altamente variable entre las unidades de hemodiálisis de un mismo país5. En España, la prevalencia de la infección por el VHC en hemodiálisis en 1997-2001 se estimó en el 22%4.
Tabla 1. Prevalencia de la infección por el VHC en hemodiálisis 
En los trasplantados renales, la prevalencia de la infección por el VHC varía entre el 7 y el 40%, también con una amplia variabilidad geográfica y demográfica4,17-19.
Hasta un 55-85% de los infectados por el VHC evoluciona hacia la cronicidad20-23 y, de éstos, el 5-25% desarrolla cirrosis a los 25-30 años20,24. Los sujetos con cirrosis tienen un riesgo mayor de presentar un hepatocarcinoma que la población no cirrótica. En España, el VHC es actualmente el principal factor de riesgo asociado con la aparición de hepatocarcinoma25, aunque el riesgo varía según el grado de afectación hepática: es menor del 1% anual en pacientes con hepatitis crónica sin fibrosis significativa y se incrementa del 3 al 7% anual en la cirrosis26. Una vez que se presenta la cirrosis hepática, el riesgo de que el hepatocarcinoma se desarrolle continúa a pesar de obtener una respuesta viral sostenida al tratamiento27. Se han identificado varios factores de progresión a cirrosis: edad avanzada, obesidad, inmunosupresión, consumo de alcohol mayor de 50 g al día28-31, y se ha descrito una evolución más rápida a cirrosis en los trasplantados renales32-34.
Histología hepática: papel de la biopsia
]]> La biopsia hepática tiene un valor sustancial en la evaluación de la gravedad de la enfermedad hepática en la infección crónica por el VHC, en relación con el grado de fibrosis y la actividad necroinflamatoria, así como para excluir otras causas concomitantes de disfunción hepática, como la hepatopatía grasa no alcohólica, cuya incidencia está en auge, y la hemosiderosis; estas entidades pueden afectar a la progresión de la enfermedad y condicionar la respuesta al tratamiento35-38. Las Guías KDIGO (Kidney Disease Improving Global Outcomes)39 recomiendan la biopsia hepática en el estudio de hepatopatía de los pacientes candidatos a trasplante renal; la Guía de la AASLD (American Association for the Study of Liver Diseases) la limita a los pacientes VHC-positivos con genotipos 1 y 4, pero la considera innecesaria en los genotipos 2 y 340, dado que más del 80% de los pacientes (con función renal normal) consiguen una respuesta viral sostenida.El índice METAVIR evalúa la actividad necroinflamatoria (grado) y la fibrosis (estadio). Consiste en un sistema de codificación de dos letras y dos números: A =actividad histológica (A0 = ausencia de actividad, A1 = actividad leve, A2 = actividad moderada, A3 = actividad intensa) y F = fibrosis (F0 = ausencia de fibrosis, F1 = fibrosis portal sin tabiques, F2 = fibrosis portal con pocos tabiques, F3 = numerosos tabiques sin fibrosis, F4 = fibrosis)41. Se requiere una biopsia hepática de alta calidad, de al menos 2 cm de longitud y con más de 5 espacios porta, para calcular un adecuado índice METAVIR42,43.
Según las Guías KDIGO, los pacientes en lista de espera de trasplante renal que no responden o rechazan el tratamiento antiviral deben someterse a una biopsia hepática cada tres-cinco años según el índice METAVIR que presenten al inicio (cada tres años para un índice METAVIR de tres y cada cinco años para un índice METAVIR de 1-2)39. No hay evidencia que apoye esta recomendación, aunque se ha demostrado que la enfermedad hepática progresa en los pacientes en diálisis17,34. Los marcadores de daño hepático (por ejemplo, la GPT) no reflejan exactamente la gravedad histológica de la hepatopatía en la población con ERC y hasta un 25% de los pacientes con infección por el VHC evaluados para trasplante renal tiene fibrosis en puente o cirrosis en la biopsia hepática (METAVIR > 3)44-49. No hay estudios definitivos que hayan analizado si el estadio histológico de la biopsia pretrasplante predice la enfermedad hepática postrasplante y sus resultados; sin embargo, la presencia de cirrosis en la biopsia hepática pretrasplante se ha asociado con un 26% de supervivencia a los 10 años50. Varios estudios han demostrado que el 19-64% de los trasplantados renales con infección por el VHC tiene enfermedad hepática postrasplante, en comparación con sólo el 1-30% de los pacientes no infectados18,50-56. La mayoría de los estudios son retrospectivos y tratan de pacientes sin biopsia hepática pretrasplante, lo que podría resultar en una infraestimación de enfermedad hepática avanzada, dado el aumento en el índice de hepatopatía descompensada. Los estudios sin biopsia hepática pretrasplante pero con biopsia hepática secuencial en el postrasplante han demostrado que la histología hepática puede progresar en un 20% de los pacientes57,58. Dado que existe un 6-8% de riesgo de mortalidad anual en los pacientes en lista de espera de trasplante renal59, parece razonable establecer una monitorización de su enfermedad hepática para constatar que siguen siendo candidatos a trasplante renal en las mismas condiciones. El daño hepático antes del trasplante renal es un factor predictor independiente de una pobre supervivencia a largo plazo50.
La coagulopatía secundaria a la disfunción hepatocelular y la trombocitopenia por hipertensión portal e hiperesplenismo conllevan un mayor riesgo de sangrado60. En pacientes con ERC, en presencia de ascitis y debido al riesgo añadido de hemorragia incrementado por la disfunción plaquetaria asociada a la uremia, la anticoagulación en hemodiálisis y el frecuente tratamiento antiagregante, se recomienda la biopsia hepática por vía transyugular o transfemoral, que además puede aportar información diagnóstica adicional, como el gradiente de presión venoso hepático, para confirmar la existencia de hipertensión portal39,40.
Se ha constatado un empleo aumentado de desmopresina (DDAVP, 0,3 μg/kg) inmediatamente antes de la biopsia hepática en pacientes con ERC, aunque no se ha determinado el nivel de creatinina sérica o el filtrado glomerular a partir del cual se debe indicar su uso61.
No se conoce en la actualidad la utilidad de los marcadores no invasivos (Índice de Forns, APRI, FIB-4) en el estudio del daño hepático en los pacientes con ERC e infección por el VHC62. Se habían creado expectativas referentes a la elastografía transitoria (FibroScan), que no ha logrado reemplazar a la biopsia: no ha sido aprobada por la FDA (Food and Drug Administration), su índice de error es mayor en pacientes obesos y puede estar sobrestimada en la hepatitis aguda, que se acompaña de gran actividad necroinflamatoria y baja o nula fibrosis63,64.
Tratamiento
El tratamiento de elección en la hepatitis crónica por el VHC es el interferón convencional o pegilado, en monoterapia o combinado con ribavirina. La indicación de tratamiento antiviral debe hacerse de forma individualizada, según la gravedad de la enfermedad hepática, la posibilidad de efectos adversos graves, la variabilidad en la respuesta al tratamiento, la presencia de comorbilidad (sobre todo insuficiencia renal) y la decisión del paciente40. Los individuos con ERC tienen niveles de transaminasas menores o incluso normales45,65-67 con respecto a la población sin ERC. Clásicamente se ha considerado que los sujetos con genotipo 1 y transaminasas persistentemente normales tenían una fibrosis hepática mínima y por ello no eran subsidiarios de tratamiento; hoy día se ha demostrado que hasta un 25% de estos pacientes tieneuna fibrosis significativa y su respuesta al tratamiento es similar a la de los pacientes con transaminasas elevadas68-74. Los pacientes con manifestaciones extrahepáticas han de ser tratados, independientemente de la gravedad de su enfermedad hepática75.
En los individuos con una función renal normal, la terapia antiviral tiene R como objetivo erradicar la infección por el VHC para mejorar la histología hepática, que a largo plazo resulta en una menor tasa de morbilidad y una mayor supervivencia. En los pacientes con ERC, el tratamiento del VHC es aún más relevante, pues la hepatitis crónica ha demostrado reducir la supervivencia en hemodiálisis y en trasplantados renales, así como la supervivencia del injerto renal18,50-56,76-78, en comparación con los pacientes no infectados, lo que se debe en parte a la progresión de la enfermedad hepática, la evolución acelerada a cirrosis y/o la aparición de carcinoma hepatocelular18,32-34,50,56,79. La infección por el VHC es la principal causa de disfunción hepática postrasplante renal y la cuarta causa de mortalidad en esta población33. El VHC se comporta como un factor de riesgo independiente para la aparición de proteinuria53,80, aumenta el riesgo de desarrollar una diabetes postrasplante81-83, una glomerulonefritis de novo84-87 o una nefropatía crónica del injerto, de empeorar la enfermedad hepática y de provocar un mayor número de infecciones39. Además, la inmunosupresión en el trasplante renal predispone a la reactivación del VHC. En particular, los esteroides se han asociado con un incremento de 10 a 100 veces de la carga viral88, por lo que deberían evitarse o minimizarse en los pacientes VHC-positivos89. También se ha descrito un aumento de la frecuencia de hepatitis colestásica fibrosante que, junto a la evolución acelerada a cirrosis, puede elevar significativamente la morbimortalidad y conllevar la necesidad de un trasplante hepático90,91.
]]> Sin embargo, la terapia antiviral en la ERC sigue siendo controvertida. No existen estudios comparativos sobre los que basar la decisión de un adecuado tratamiento antiviral. La mayoría de los estudios realizados en hemodiálisis han investigado el uso de interferón alfa convencional (3 MUI x tres veces/semana) o interferón alfa pegilado (α2a, 135 μg/semana, o α2b, 50 μg/semana o 0,5-1 μg/kg/semana) en monoterapia y arrojan resultados variables, en general una respuesta viral sostenida reducida (19-75%) y una intolerancia farmacológica considerable (el 30-50% de los pacientes en diálisis interrumpe la terapia)92-95. La AGA (American Gastroenterological Association) y la AASLD recomiendan el uso de dosis reducidas de interferón alfa pegilado en monoterapia, considerando la asociación de ribavirina como una contraindicación en los sujetos con un filtrado glomerular estimado menor de de 50 ml/min40,96,97-104. Hay escasa experiencia con el tratamiento combinado con ribavirina (200 mg x tres veces/semanao 200 mg/24 h) en diálisis, pero se han sugerido mejores resultados, aunque los estudios realizados son series de casos, escasas y con un número muy limitado de pacientes105-110. En un reciente metanálisis de los ensayos clínicos existentes sobre la terapia combinada en diálisis, alrededor de la mitad de los pacientes obtenían una respuesta viral sostenida111. El riesgo de anemia severa por hemólisis secundaria dificulta su uso; algunos investigadores la han empleado basándose en los niveles séricos del fármaco y han obtenido resultados dispares106-110,112, aunque los datos disponibles son alentadores y su uso puede estar indicado en centros en los que el tratamiento del paciente lo lleven a cabo entre hepatólogos y nefrólogos. El tratamiento antiviral en trasplantados renales es excepcional salvo en casos de alternativas terapéuticas limitadas o hepatitis colestásica severa90,91. El inconveniente principal del tratamiento antiviral antes del trasplante es el retraso de la inclusión del paciente en lista de espera, sin que además pueda asegurarse la erradicación del VHC por los bajos índices de respuesta.Por tanto, todos los pacientes en hemodiálisis con ARN del VHC detectable y un índice METAVIR F0-F2 deberían considerarse candidatos para el tratamiento con interferón alfa. Los pacientes con fibrosis en puente cirrosis compensada deberían recibir también terapia antiviral y convertirse en candidatos a trasplante renal si consiguen una respuesta viral sostenida; los pacientes con cirrosis descompensada deberían ser valorados para trasplante combinado de riñón e hígado40.
A la luz del impacto que la infección crónica por el VHC tiene en el trasplante renal, se recomienda que los pacientes con ERC sean tratados antes de ser sometidos a trasplante113,114. Sin embargo, a pesar de la evidencia que existe sobre los beneficios del tratamiento antiviral en los pacientes con hepatitis crónica por VHC y ERC antes del trasplante renal, sólo algunos protocolos de trasplante renal recomiendan el tratamiento frente al VHC y no suele estar catalogado como un criterio previo al trasplante115-117. De hecho, la evaluación pretrasplante renal de los pacientes VHC-positivos en tratamiento sustitutivo renal es la que pone de manifiesto que, además de no realizarse el tratamiento frente al VHC antes del trasplante, el seguimiento hepatológico de estos pacientes en diálisis puede ser en muchos casos casi inexistente. Es posible que esto se deba a que la complejidad que trae consigo el tratamiento de ERC haga que el nefrólogo asuma toda la patología del paciente en hemodiálisis.
Son necesarios más estudios para evaluar la situación clínica y el seguimiento de la hepatitis por VHC de los pacientes en hemodiálisis, con el fin de detectar mejoras e implicar tanto a nefrólogos como a hepatólogos en su manejo.
Referencias Bibliográficas
1. Finelli L, Miller JT, Tokars JI, Alter MJ, Arduino MJ. National surveillance of dialysisassociated diseases in the United States, 2002. Semin Dial 2005;18(1):52-61. [ Links ]
2. Espinosa M, Martín-Malo A, Ojeda R, Santamara R, Soriano S, Aguera M, et al. Marked reduction in the prevalence of hepatitis C virus infection in hemodialysis patients: causes and consequences. Am J Kidney Dis 2004;43(4):685-9. [ Links ]
]]>3. Jadoul M, Poignet JL, Geddes C, Locatelli F, Medin C, Krajewska M, et al. The changing epidemiology of hepatitis C virus (HCV) infection in haemodialysis: European multicentre study. Nephrol Dial Transplant 2004;19(4):904-9. [ Links ]
4. Fissell RB, Bragg-Gresaham JL, Woods JD, Jadoul M, Gillespie B, Hedderwick SA, et al. Patterns of hepatitis C prevalence and seroconversion in hemodialysis units from three continents: the DOPPS. Kidney Int 2004;65(6):2335-42. [ Links ]
5. Sivapalasingam S, Malak SF, Sullivan JF, Lorch J, Sepkowitz KA. High prevalence of hepatitis C infection among patients receiving hemodialysis at an urban dialysis center. Infect Control Hosp Epidemiol 2002;23(6):319-24. [ Links ]
6. Hinrichsen H, Leimenstoll G, Stegen G, Schrader H, Fölsch UR, Schmidt WE; PHV Study Group. Prevalence and risk factors of hepatitis C virus infection in haemodialysis patients: a multicentre study in 2796 patients. Gut 2002;51(3):429-33. [ Links ]
7. Huraib S, Al-Rashed R, Aldrees A, Aljefry M, Arif M, Al-Faleh FA. High prevalence of and risk factors for hepatitis C in haemodialysis patients in Saudi Arabia: a need for new dialysis strategies. Nephrol Dial Transplant 1995;10(4):470-4. [ Links ]
]]>8. Santos MA, Souto FJ. Infection by the hepatitis C virus in chronic renal failure patients undergoing hemodialysis in Mato Grosso state, central Brazil: a cohort study. BMC Public Health 2007;7:32. [ Links ]
9. Saha D, Agarwal SK. Hepatitis and HIV infection during haemodialysis. J Indian Med Assoc 2001;99(4):194-9. [ Links ]
10. Shamshirsaz AA, Kamgar M, Bekheirnia MR, Ayazi F, Hashemi SR, Bouzari N, et al. The role of hemodialysis machines dedication in reducing Hepatitis C transmission in the dialysis setting in Iran: a multicenter prospective interventional study. BMC Nephrol 2004;5(1):13. [ Links ]
11. Blackmore TK, Stace NH, Maddocks P, Hatfield P. Prevalence of antibodies to hepatitis C virus in patients receiving renal replacement therapy, and in the staff caring for them. Aust N Z J Med 1992;22(4):353-7. [ Links ]
12. Schneeberger PM, Keur I, Van Loon AM, Mortier D, De Coul KO, Van Haperen AV, et al. The prevalence and incidence of hepatitis C virus infections among dialysis patients in the Netherlands: a nationwide prospective study. J Infect Dis 2000;182(5):1291-9. [ Links ]
]]>13. Hruby Z, Sliwinski J, Molin I, Zalewska M, Knysz B, Czyz W, et al. High prevalence of antibodies to hepatitis C virus in three haemodialysis centres in south-western Poland. Nephrol Dial Transplant 1993;8(8):740-3. [ Links ]
14. Cassidy MJ, Jankelson D, Becker M, Dunne T, Walzl G, Moosa MR. The prevalence of antibodies to hepatitis C virus at two haemodialysis units in South Africa. S Afr Med J 1995;85(10):996-8. [ Links ]
15. Luengrojanakul P, Vareesangthip K, Chainuvati T, Murata K, Tsuda F, Tokita H, et al. Hepatitis C virus infection in patients with chronic liver disease or chronic renal failure and blood donors in Thailand. J Med Virol 1994;44(3):287-92. [ Links ]
16. Hmaied F, Ben Mamou M, Saune-Sandres K, Rostaing L, Slim A, Arrouji Z, et al. Hepatitis C virus infection among dialysis patients in Tunisia: incidence and molecular evidence for nosocomial transmission. J Med Virol 2006;78(2):185-91. [ Links ]
17. Pereira BJ, Natov SN, Bouthot BA, Murthy BV, Ruthazer R, Schmid CH, et al. Effects of hepatitis C infection and renal transplantation on survival in end-stage renal disease. The New England Organ Bank Hepatitis C Study Group. Kidney Int 1998;53(5):1374-81. [ Links ]
]]>18. Hanafusa T, Ichikawa Y, Kishikawa H, Kyo M, Fukunishi T, Kokado Y, et al. Retrospective study on the impact of hepatitis C virus infection on kidney transplant patients over 20 years. Transplantation 1998;66(4):471-6. [ Links ]
19. Vosnides GG. Hepatitis C in renal transplantation. Kidney Int 1997;52(3):843-61. [ Links ]
20. Liang TJ, Rehermann B, Seeff LB, Hoofnagle JH. Pathogenesis, natural history, treatment and prevention of hepatitis C. Ann Intern Med 2000;132(4):296-305. [ Links ]
21.Thomas DL, Seeff LB. Natural history of hepatitis C. Clin Liver Dis 2005;9(3):383-98. [ Links ]
22. Strader DB, Seeff LB. The natural history of chronic hepatitis C infection. Eur J Gastroenterol Hepatol 1996;8(4):324-8. [ Links ]
]]>23. Seeff LB, Hoofnagle JH. National Institutes of Health Consensus Development Conference: management of hepatitis C: 2002. Hepatology 2002;36(5 Suppl 1):S1-2. [ Links ]
24. Seeff LB. Natural history of chronic hepatitis C. Hepatology 2002;36(5 Suppl 1):S35-S46. [ Links ]
25. Forner A, Ayuso C, Real MI, Sastre J, Robles R, Sangro B, et al. Diagnóstico y tratamiento del carcinoma hepatocelular. Med Clin (Barc) 2009;132(7):272-87. [ Links ]
26. Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: Incidente and risk factors. Gastroenterology 2004;127(5 Suppl 1):S35-50. [ Links ]
27. Bruno S, Stroffolini T, Colombo M, Bollani S, Benvegnu L, Mazzella G, et al. Sustained virological response to interferon-alpha is associated with improved outcome in HCV-related cirrhosis: A retrospective study. Hepatology 2007;45(3):579-87. [ Links ]
]]>28. Benhamou Y, Bochet M, Di Martino V, Charlotte F, Azria F, Coutellier A, et al. Liver fibrosis progression in human immunodeficiency virus and hepatitis C virus coinfected patients. The Multivirc Group. Hepatology 1999;30(4):1054-8. [ Links ]
29. Poynard T, Bedossa P, Opolon P. Natural history of liver fibrosis progression in patients with chronic hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC groups. Lancet 1997;349(9055):825-32. [ Links ]
30. Harris DR, Gonin R, Alter HJ, Wright EC, Buskell ZJ, Hollinger FB, et al. The relationship of acute transfusion-associated hepatitis to the development of cirrhosis in the presence of alcohol abuse. Ann Intern Med 2001;134(2):120-4. [ Links ]
31. Powell EE, Jonsson JR, Clouston AD. Steatosis: co-factor in other liver diseases. Hepatology 2005;42(1):5-13. [ Links ]
32. Marcelli D, Stannard D, Conte F, Held PJ, Locatelli F, Port FK. ESRD patient mortality with adjustment for comorbid conditions in Lombardy (Italy) versus the United States. Kidney Int 1996;50(3):1013-8. [ Links ]
]]>33. Maisonneuve P, Agodoa L, Gellert R, Stewart JH, Buccianti G, Lowenfels AB, et al. Cancer in patients on dialysis for end-stage renal disease: an international collaborative study. Lancet 1999;354(9173):93-9. [ Links ]
34. Nakayama E, Akiba T, Marumo F, Sato C. Prognosis of anti-hepatitis C virus antibody-positive patients on regular hemodialysis therapy. J Am Soc Nephrol 2000;11(10):1896-902. [ Links ]
35. Kleiner DE. The liver biopsy in chronic hepatitis C: a view from the other side of the microscope. Semin Liver Dis 2005;25(1):52-64. [ Links ]
36. Rubbia-Brandt L, Fabris P, Paganin S, Leandro G, Male PJ, Giostra E, et al. Steatosis affects chronic hepatitis C progression in a genotype specific way. Gut 2004;53(3):406-12. [ Links ]
37. Poynard T, Ratziu V, McHutchison J, Manns M, Goodman Z, Zeuzem S, et al. Effect of treatment with peginterferon or interferon alfa-2b and ribavirin on steatosis in patients infected with hepatitis C. Hepatology 2003;38(1):75-85. [ Links ]
]]>38. Olynyk JK, Reddy KR, Di Bisceglie AM, Jeffers LJ, Parker TI, Radick JL, et al. Hepatic iron concentration as a predictor of response to interferon alfa therapy in chronic hepatitis C. Gastroenterology 1995;108(4):1104-9. [ Links ]
39. Kidney Disease Improving Global Outcomes. Clinical practice guidelines for the prevention, diagnosis, evaluation, and treatment of hepatitis C in chronic kidney disease. Kidney Int 2008;73(Suppl 109):S53-S68. [ Links ]
40. Ghany MG, Strader DB, Thomas DL, Seeff LB; American Association for the Study of Liver Diseases. Diagnosis, management, and treatment of hepatitis C: an update. Hepatology 2009;49(4):1335-74. [ Links ]
41. Knodell RG, Ishak KG, Black WC, et al. Formulation and application of a numeral scoring system for assessing histological activity in asymptomatic chronic active hepatitis. Hepatology 1981;1(5):431-5. [ Links ]
42. Bravo AA, Sheth SG, Chopra S. Liver biopsy. N Engl J Med 2001;344(7):495-500. [ Links ]
]]>43. Regev A, Berho M, Jeffers LJ, Milikowski C, Molina EG, Pyrsopoulos NT, et al. Sampling error and intraobserver variation in liver biopsy in patients with chronic HCV infection. Am J Gastroenterol 2002;97(10):2614-8. [ Links ]
44. Martin P, Carter D, Fabrizi F, Dixit V, Conrad AJ, Artinian L, et al. Histopathological features of hepatitis C in renal transplant candidates. Transplantation 2000;69(7):1479-84. [ Links ]
45. Caramelo C, Ortiz A, Aguilera B, Porres JC, Navas S, Marriott E, et al. Liver disease patterns in hemodialysis patients with antibodies to hepatitis C virus. Am J Kidney Dis 1993;22(6):822-8. [ Links ]
46. Cotler SJ, Diaz G, Gundlapalli S, Jakate S, Chawla A, Mital D, et al. Characteristics of hepatitis C in renal transplant candidates. J Clin Gastroenterol 2002;35(2):191-5. [ Links ]
47. Glicklich D, Thung SN, Kapoian T, Tellis V, Reinus JF. Comparison of clinical features and liver histology in hepatitis C-positive dialysis patients and renal transplant recipients. Am J Gastroenterol 1999;94(1):159-63. [ Links ]
]]>48. Pol S, Romeo R, Zins B, Driss F, Lebkiri B, Carnot F, et al. Hepatitis C virus RNA in anti-HCV positive hemodialyzed patients: significance and therapeutic implications. Kidney Int 1993;44(5):1097-100. [ Links ]
49. Sterling RK, Sanyal AJ, Luketic VA, Stravitz RT, King AL, Post AB, et al. Chronic hepatitis C infection in patients with end stage renal disease: characterization of liver histology and viral load in patients awaiting renal transplantation. Am J Gastroenterol 1999;94(12):3576-82. [ Links ]
50. Mathurin P, Mouquet C, Poynard T, Sylla C, Benalia H, Fretz C, et al. Impact of hepatitis B and C virus on kidney transplantation outcome. Hepatology 1999;29(1):257-63. [ Links ]
51. Breitenfeldt MK, Rasenack J, Berthold H, Olschewski M, Schroff J, Strey C, et al. Impact of hepatitis B and C on graft loss and mortality of patients after kidney transplantation. Clin Transplant 2002;16(2):130-6. [ Links ]
52. Bruchfeld A, Wilczek H, Elinder CG. Hepatitis C infection, time in renal replacement therapy, and outcome after kidney transplantation. Transplantation 2004;78(5):745-50. [ Links ]
]]>53. Morales JM, Domínguez-Gil B, Sanz-Guajardo D, Fernández J, Escuin F. The influence of hepatitis B and hepatitis C virus infection in the recipient on late renal allograft failure. Nephrol Dial Transplant 2004;19(Suppl 3):iii72-6. [ Links ]
54. Gentil MA, Rocha JL, Rodríguez-Algarra G, Pereira P, López R, Bernal G, et al. Impaired kidney transplant survival in patients with antibodies to hepatitis C virus. Nephrol Dial Transplant 1999;14(10):2455-60. [ Links ]
55. Legendre C, Garrigue V, Le Bihan C, Mamzer-Bruneel MF, Chaix ML, Landais P, et al. Harmful long-term impact of hepatitis C virus infection in kidney transplant recipients. Transplantation 1998;65(5):667-70. [ Links ]
56. Murthy BV, Muerhoff AS, Desai SM, Yamaguchi J, Mushahwar IK, Schmid CH, et al. Impact of pretransplantation GB virus C infection on the outcome of renal transplantation. J Am Soc Nephrol 1997;8(7):1164-73. [ Links ]
57. Kamar N, Rostaing L, Selves J, Sandres-Saune K, Alric L, Durand D. Natural history of hepatitis C virus-related liver fibrosis after renal transplantation. Am J Transplant 2005;5(7):1704-12. [ Links ]
]]>58. Zylberberg H, Nalpas B, Carnot F, Skhiri H, Fontaine H, Legendre C, et al. Severe evolution of chronic hepatitis C in renal transplantation: a case control study. Nephrol Dial Transplant 2002;17(1):129-33. [ Links ]
59. U.S. Renal Data System, USRDS 2002 Annual Data Report: Atlas of End-Stage Renal Disease in the United States, in, Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2002. [ Links ]
60. Caldwell SH, Hoffman M, Lisman T, Macik BG, Northup PG, Reddy KR, et al. Coagulation disorders and hemostasis in liver disease: pathophysiology and critical assesment of current management. Hepatology 2006;44(4):1039-46. [ Links ]
61. DiMichele DM, Hathaway WE. Use of DDAVP in inherited and acquired platelet dysfunction. Am J Hematol 1990;33(1):39-45. [ Links ]
62. Campbell MS, Reddy KR. Review article: the evolving role of liver biopsy. Aliment Pharmacol Ther 2004;20(3):249-59. [ Links ]
]]>63. Arena U, Vizzutti F, Corti G, Ambu S, Stasi C, Bresci S, et al. Acute viral hepatitis increases liver stiffness values measured by transient elastography. Hepatology 2008;47(2):380-4. [ Links ]
64. Sagir A, Erhardt A, Schmitt M, Häussinger D. Transient elastography is unreliable for detection of cirrhosis in patients with acute liver damage. Hepatology 2008;47(2):592-5. [ Links ]
65. Espinosa M, Martín-Malo A, Álvarez de Lara MA, Soriano S, Aljama P. High ALT levels predict viremia in anti-HCV-positive HD patients if a modified normal range of ALT is applied. Clin Nephrol 2000;54(2):151-6. [ Links ]
66. Guh JY, Lai YH, Yang CY, Chen SC, Chuang WL, Hsu TC, et al. Impact of decreased serum transaminase levels on the evaluation of viral hepatitis in hemodialysis patients. Nephron 1995;69(4):459-65. [ Links ]
67. Fabrizi F, Lunghi G, Finazzi S, Colucci P, Pagano A, Ponticelli C, et al. Decreased serum aminotransferase activity in patients with chronic renal failure: impact on the detection of viral hepatitis. Am J Kidney Dis 2001;38(5):1009-15. [ Links ]
]]>68. Martinot-Peignoux M, Boyer N, Cazals-Hatem D, Pham BN, Gervais A, Le Breton V, et al. Prospective study on anti-hepatitis C virus-positive patients with persistently normal serum alanine transaminase with or without detectable serum hepatitis C virus RNA. Hepatology 2001;34(5):1000-5. [ Links ]
69. Shiffman ML, Diago M, Tran A, Pockros P, Reindollar R, Prati D, et al. Chronic hepatitis C in patients with persistently normal alanine transaminase levels. Clin Gastroenterol Hepatol 2006;4(5):645-52. [ Links ]
70. Boccato S, Pistis R, Noventa F, Guido M, Benvegnù L, Alberti A. Fibrosis progression in initially mild chronic hepatitis C. J Viral Hepat 2006;13(5):297-302. [ Links ]
71. Persico M, Persico E, Suozzo R, Conte S, De Seta M, Coppola L, et al. Natural history of hepatitis C virus carriers with persistently normal aminotransferase levels. Gastroenterology 2000;118(4):760-4. [ Links ]
72. Yu ML, Dai CY, Lee LP, Hou NJ, Hsieh MY, Huang JF, et al. A 24-week course of high-dose interferon-alpha plus ribavirin for Taiwanese chronic hepatitis C patients with persistently normal or near-normal alanine aminotransferase levels. Liver Int 2006;26(10):1187-95. [ Links ]
]]>73. Jacobson IM, Ahmed F, Russo MW, Lebovics E, Dieterich DT, Esposito SP, et al. Interferon alfa-2b [correction of alpha-2b] and ribavirin for patients with chronic hepatitis C and normal ALT. Am J Gastroenterol 2004;99(9):1700-5. [ Links ]
74. Zeuzem S, Diago M, Gane E, Reddy KR, Pockros P, Prati D, et al. Peginterferon alfa-2a (40 kilodaltons) and ribavirin in patients with chronic hepatitis C and normal aminotransferase levels. Gastroenterology 2004;127(6):1724-32. [ Links ]
75. Yee HS, Curie SL, Darling JM, Wright TL. Management and treatment of hepatitis C viral infection: recommendations from the Department of Veterans Affairs Hepatitis C Resource Center Program and the National Hepatitis C Program Office. Am J Gastroenterol 2006;101(10):2360-78. [ Links ]
76. Lee WC, Shu KH, Cheng CH, Wu MJ, Chen CH, Lian JC. Long-term impact of hepatitis B, C virus infection on renal transplantation. Am J Nephrol 2001;21 (4):300-6. [ Links ]
77. Aroldi A, Lampertico P, Montagnino G, Passerini P, Villa M, Campise MR, et al. Natural history of hepatitis B and C in renal allograft recipients. Transplantation 2005;79(9):1132-6. [ Links ]
]]>78. Morales JM, Marcén R, Andrés A, Domínguez-Gil B, Campistol JM, Gallego R, et al. Renal transplantation in patients with hepatitis C virus antibody. A long national experience. NDT Plus 2010;3(Suppl 2):ii41-ii46. [ Links ]
79. Gheith OA, Saad MA, Hassan AA, A-Eldeeb S, Agroudy AE, Esaza H, et al. Hepatic dysfunction in kidney transplant recipients: prevalence and impact on graft and patient survival. Clin Exp Nephrol 2007;11(4):309-15. [ Links ]
80. Hestin D, Guillermin F, Castin N, Le Faou A, Champigneulles J, Kessler M. Pretransplant hepatitis C virus infection: a predictor of proteinuria after renal transplantation. Transplantation 1998;65(5):741-4. [ Links ]
81. Bloom RD, Rao V, Weng F, Grossman RA, Cohen D, Mange KC. Association of hepatitis C with posttransplant diabetes in renal transplant patients on tacrolimus. J Am Soc Nephrol 2002;13(5):1374-80. [ Links ]
82. Kamar N, Mariat C, Delahousse M, Dantal J, Al Najjar A, Cassuto E, et al. Diabetes mellitus after kidney transplantation: a French multicentre observational study. Nephrol Dial Transplant 2007;22(7):1986-93. [ Links ]
]]>83. Fabrizi F, Martin P, Dixit V, Bunnapradist S, Kanwal F, Dulai G. Post-transplant diabetes mellitus and HCV seropositive status after renal transplantation: meta-analysis of clinical studies. Am J Transplant 2005;5(10):2433-40. [ Links ]
84. Roth D, Cirocco R, Zucker K, Ruiz P, Viciana A, Burke G, et al. De novo membranoproliferative glomerulonephritis in hepatitis C virus-infected renal allograft recipients. Transplantation 1995;59(12):1676-82. [ Links ]
85. Floege J. Recurrent glomerulonephritis following renal transplantation: an update. Nephrol Dial Transplant 2003;18(7):1260-5. [ Links ]
86. Choy BY, Chan TM, Lai KN. Recurrent glomerulonephritis after kidney transplantation. Am J Transplant 2006;6(11):2535-42. [ Links ]
87. Kamar N, Izopet J, Alric L, Guilbeaud-Frugier C, Rostaing L. Hepatitis C virus-related kidney disease: an overview. Clin Nephrol 2008;69(3):149-60. [ Links ]
]]>88. Humar A, Crotteau S, Cruessner A, Kandaswamy R, Gruessner R, Payne W, et al. Steroid minimization in liver transplant recipients: impact on hepatitis C recurrente and post-transplant diabetes. Clin Transplant 2007;21(4):526-31. [ Links ]
89. Pascual J, Crespo M, Mateos ML, Marcén R, Orofino L, Burgos FJ, et al. Reduced severity of acute rejection in hepatitis C virus positive renal allograft recipients: are milder immunosuppressive regimens advisable? Transplant Proc 1998;30(4):1329-30. [ Links ]
90. Kamar N, Ribes D, Izopet J, Rostaing L. Treatment of hepatitis C virus infection (HCV) after renal transplantation: implications for HCV-positive dialysis patients awaiting a kidney transplant. Transplantation 2006;82(7):853-6. [ Links ]
91. Toth CM, Pascual M, Chung RT, Graeme-Cook F, Dienstag JL, Bhan AK, et al. Hepatitis C virus-associated fibrosing cholestatic hepatitis after renal transplantation: response to interferon-alpha therapy. Transplantation 1998;66(9):1254-8. [ Links ]
92. Covic A, Maftei I-D, Mardare NGI, Ionita-Radu F, Totolici C, Tuta L, et al. Analysis of safety and efficacy of pegylated-interferon alpha-2a in hepatitis C virus positive hemodialysis patients: results from a large, multicenter audit. J Nephrol 2006;19(6):794-801. [ Links ]
]]>93. Tan SS, Abu Hassan MR, Abdullah A, Ooi BP, Korompis T, Merican MI. Safety and efficacy of an escalating dose regimen of pegylated interferon alpha-2b in the treatment of haemodialysis patients with chronic hepatitis C. J Viral Hepat 2010;17(6):410-8. [ Links ]
94. Kokoglu OF, Ucmak H, Hosoglu S, Cetinkaya A, Kantarceken B, Buyukbese MA, et al. Efficacy and tolerability of pegylated-interferon alpha-2a in hemodialysis patients with chronic hepatitis C. J Gastroenterol Hepatol 2006;21(3):575-80. [ Links ]
95. Russo MW, Ghalib R, Sigal J, Joshi V. Randomized trial of pegylated interferon alpha-2b monotherapy in haemodialysis patients with chronic hepatitis C. Nephrol Dial Transplant 2006;21(2):437-43. [ Links ]
96. Dienstag JL, McHutchinson JG. American Gastroenterological Association technical review on the management of hepatitis C. Gastroenterology 2006;130(1):231-64. [ Links ]
97. Bocci V, Pacini A, Muscettola M, Paulesu L, Pessina GP, Santiano M et al. Renal filtration, absorption and catabolism of human alpha interferon. J Interferon Res 1981;1(3):347-52. [ Links ]
]]>98. Bino T, Madar Z, Gertler A, Rosenberg H. The kidney is the main site of interferon degradation. J Interferon Res 1982;2(2):301-8. [ Links ]
99. Kramer TH, Gaar GG, Ray CG, Minnich L, Copeland JG, Connor JD. Hemodialysis clearance of intravenously administered ribavirin. Antimicrob Agents Chemother 1990;34(3):489-90. [ Links ]
100. Glue P. The clinical pharmacology of ribavirin. Semin Liver Dis 1999;19(Suppl 1):17-24. [ Links ]
101. Uchihara M, Izumi N, Sakai Y, Yauchi T, Miyake S, Sakai T, et al. Interferon therapy for chronic hepatitis C in hemodialysis patients: increased serum levels of interferon. Nephron 1998;80(1):51-6. [ Links ]
102. Rostaing L, Chatelut E, Payen JL, Izopet J, Thalamas C, Ton-That H, et al. Pharmacokinetics of alpha IFN-2b in chronic hepatitis C virus patients undergoing chronic hemodialysis or with normal renal function: clinical implications. J Am Soc Nephrol 1998;9(12):2344-8. [ Links ]
]]>103. Glue P, Fang JW, Rouzier-Panis R, Raffanel C, Sabo R, Gupta SK, et al. Pegylated interferon-alpha 2b: pharmacokinetics, pharmacodynamics, safety, and preliminary efficacy data. Hepatitis C Intervention Therapy Group. Clin Pharmacol Ther 2000;68(5):556-67. [ Links ]
104. Gupta SK, Pittenger AL, Swan SK, Marbury TC, Tobillo E, Batra V, et al. Single-dose pharmacokinetics and safety of pegylated interferon alpha 2b in patients with chronic renal dysfunction. J Clin Pharmacol 2002;42(10):1109-15. [ Links ]
105. Luzon BA, Muir AJ, Heneghan MA. Safety and tolerability of pegylated interferon with or without low dose ribavirin for treatment of hepatitis C in hemodialysis. Hepatology 2005;42(Suppl):703A-704A. [ Links ]
106. Hakim W, Sheik S, Inayat I, Bia M, Caldwell C, Jain D, et al. Inicial HCV response in patients with end stage renal disease treated with combination pegylated interferon alfa-2a and ribavirin. Presented at: Digestive Disease Week; May 20-25, 2006; Los Angeles, CA. [ Links ]
107. Bruchfeld A, Lindahl K, Reichard O, Carlsson T, Schvarcz R. Pegylated interferon and ribavirin treatment for hepatitis C in haemodialysis patients. J Viral Herat 2006;13(5):316-21. [ Links ]
]]>108. Mousa DH, Abadía AH, Al-Shoail G, Al-Sulaiman MH, Al-Hawas FA, Al-Khader AA. Alpha-interferon with ribavirin in haemodialyzed patients with chronic hepatitis C: a prospective study. Presented at: 57th Annual Meeting of the American Association for the Study of Liver Diseases; October 27-31, 2006; Boston, MA. [ Links ]
109. Van Leusen R, Adang RP, De Vries RA, Cnossen TT, Konings CJ, Schalm SW, et al. Pegylated interferon alfa-2a (40 kD) and ribavirin in haemodialysis patients with chronic hepatitis C. Nephrol Dial Transplant 2008;23(2):721-5. [ Links ]
110. Carriero D, Fabrizi F, Uriel AJ, Park J, Martin P, Dieterich DT. Treatment of dialysis patients with chronic hepatitis C using pegylated interferon and low-dose ribavirin. Int J Artif Organs 2008;31(4):295-302. [ Links ]
111. Fabrizi F, Dixit V, Martin P, Messa P. Combined antiviral therapy of hepatitis C virus in dialysis patients: meta-analysis of clinical trials. J Viral Hepat 2010. doi: 10.1111/j.1365-2893.2010.01405.x. [Epub ahead of print] [ Links ]
112. Tan AC, Brouwer JT, Glue P, Van Leusen R, Kauffmann RH, Schalm SW, et al. Safety of interferon and ribavirin therapy in haemodialysis patients with chronic hepatitis C: results of a pilot study. Nephrol Dial Transplant 2001;16(1):193-5. [ Links ]
]]>113. Levey AS, Coresh J, Balk E, Kausz AT, Levin A, Steffes MW, et al. National Kidney Foundation practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Ann Intern Med 2003;139(2):137-47. [ Links ]
114. Barril G, González Parra E, Alcázar R, Arenas D, Campistol JM, Caramelo C, et al. Guía sobre enfermedades víricas en hemodiálisis. Nefrologia 2004;24(Suppl 2):43-66. [ Links ]
115. García García M, Oppenheimer F, Valencia J. Valoración y seguimiento de inclusión en lista de espera para trasplante renal. Nefrologia 2006;26(Suppl 8):60-9. [ Links ]
![]() Dirección para correspondencia:
Dirección para correspondencia:
Rebeca García Agudo, ]]>
Servicio de Nefrología,
Complejo Hospitalario La Mancha-Centro,
Avda. de la Constitución s/n,
13600, Alcázar de San Juan, Ciudad Real
E-mail: rgarciaagudo@hotmail.com
E-mail: rganefrologia@hotmail.com
Enviado a Revisar: 7 Dic. 2010
Aceptado el: 17 Ene. 2011