The effects of Agaricus sylvaticus fungi dietary supplementation on the metabolism and blood pressure of patients with colorectal cancer during post surgical phase
Efectos de la suplementación dietética con hongos agaricus sylvaticus en el metabolismo y la presión arterial en pacientes con cáncer colorrectal en la fase postoperatoria
R. Costa Fortes1 and M.a R. Carvalho Garbi Novaes2
1Science and Education School Sena Aires. University of Brasilia - DF. Paulista University - DF. Brazil.
2School of Medicine. Institute of Health Science (ESCS/FEPECS/SESDF). University of Brasilia - DF. Brazil.
ABSTRACT
Introduction: Metabolic, biochemical and enzymatic alterations are common in patients with cancer. Medicinal fungi has been used as adjuvants in cancer therapy due to its immunomodulatory and nutritional effects.
Objective: The objective of this study was to evaluate the metabolic and blood pressure effects on patients with colorectal cancer after dietary supplementation with Agaricus sylvaticus.
Methods: The methodology used was a randomized, double-blind, placebo-controlled clinical trial conducted at the Base Hospital of the Federal District-Brazil. Samples of 56 patients with colorectal cancer, divided into two groups: Agaricus sylvaticus (30 mg/kg/day) and placebo. Three serum evaluations were conducted throughout the treatment: glucose, total cholesterol, triglycerides, uric acid, urea, creatinine, alkaline phosphatase; total, direct and indirect bilirubin; aspartate aminotransferase and alanine aminotransferase; immunoglobulins A (IgA), G (IgG) and M (IgM); total proteins and protein fractions; blood pressure levels were measured as well. The results were analyzed by Microsoft Excel 2003 and SPSS 14.0 programs, Student's t test, F statistical test, with p < 0.05.
Results: The Agaricus sylvaticus group presented a significant reduction of fasting plasma glucose (p = 0.02), total cholesterol (p = 0.01), creatinine (p = 0.05), aspartate amino-transferase (p = 0.05), alanine aminotransferase (p = 0.04), IgA (p = 0.0001), IgM (p = 0.02), systolic blood pressure (p = 0.0001) and diastolic blood pressure (p = 0.0001). These alterations were not observed in the placebo group.
Conclusions: Results suggest that dietary supplementation with Agaricus sylvaticus is capable of providing metabolic benefits to the biochemical, enzymatic and blood pressure parameters of patients with colorectal cancer in the postsurgical phase.
Key words: Agaricus sylvaticus fungi. Metabolic alterations. Blood pressure. Cancer.
RESUMEN
]]> Introducción: Las alteraciones en los parámetros bioquímicos enzimáticos son comunes en pacientes con cáncer. El hongo medicinales se ha utilizado como coadyuvante para el tratamiento contra el cáncer debido a sus sustancias imunomoduladoras.Palabras clave: Hongos Agaricus sylvaticus. Alteraciones metabólicas. Presión arterial. Cáncer.
Introduction
Colorectal cancer is one of the most common types of malignancies12 with around 600,000 cases diagnosed annually worldwide.3
The factors that contribute to the development of colorectal cancer have yet to be entirely clarified.1 Alterations in the natural history of the disease, changes in eating habits and advances in diagnosis and treatment have been proposed.3
]]> Metabolic abnormalities are common in patients with cancer, which culminates in alterations of the biochemical and enzymatic parameters with consequent increase in mortality.4-6The main goals for colorectal cancer patients with no chance of cure include: local control of disease, sphincter preservation; improved quality of life and prolonged survival.7
With the purpose of improving the quality of life of cancer patients, adjuvant therapeutic strategies along with traditional treatments, have been tested in recent years with the use of edible fungi, particularly Agaricaceae. 8-11
Scientific evidence have attested the presence of bioactive substances in Agaricaceae fungi with essential nutritional and metabolic properties including: glucans, proteoglucans, ergosterol, lectins and arginine.8,9,12,13
Agaricus sylvaticus fungi, belonging to the family Agaricaceae, are part of a Brazilian species used in clinical trials conducted in immunosuppressed patients and has attracted considerable attention due to the presence of pharmacologically active substances capable of acting as adjuvants in cancer therapy.5,14
The purpose of this study is to assess metabolic and blood pressure effects of a dietary supplementation with Agaricus sylvaticus in postsurgical patients with colorectal cancer, treated at a public hospital in the Federal District, Brazil.
Methods
Study design
The study consists of a randomized, double-blind placebo-controlled clinical trial, which was approved by the Ministry of Health Research Ethics Committee -Federal District-Brazil, under protocol 051/2004. Terms of free consent were obtained from patients, who voluntarily participated after acknowledging study procedures. Work was developed at the Ambulatory Care of Proctology in a Tertiary-Level Base Hospital of the Federal District in Brazil. The study was carried out from November 2004 to July 2006.
]]> The sampleThe sample consisted of patients with cancer of the large intestine, divided in two groups: placebo and sup plemented with Agaricus sylvaticus fungi, having complied with the following criteria for inclusion: patients aged twenty or older, with colorectal cancer in post-surgery phase, from three months to two years of surgical intervention. Were excluded: pregnant women, breast feeding infants, physically disabled patients, patients submitted to alternative therapy, patients with any other non transmissible chronic disease or undergoing metastasis processes.
Agaricus sylvaticus extract
The Agaricus sylvaticus was first described in Switzerland and has wide geographical distribution, occurring naturally in Brazil. Its identification was confirmed by fungus expert Dr. David N. Pegler of the Royal Botanic Gardens of London whose paper was provided by the Botany Institute of the State Environment Secretariat, Sao Paulo on 10/11/1995.
The Agaricus sylvaticus fungus, family Agaricaceae, popularly known as the "Sun Mushroom" or "Cogumelo do Sol", was acquired from a producer from the Tapirai region in the countryside of Sao Paulo State. The fungus extract was obtained by soaking dehydrated material in hot water for 30 minutes, liquefied, bolted and dried in a drying box. The chemical composition of the final solution was analyzed by the Japan Food Research Laboratories Center by HPLC method and results attested the presence of carbohydrates (18.51 g/100 g), lipids (0.04 g/100 g), ergosterol (624 mg/100 g), proteins (4.99 g/100 g), amino-acids (arginine-1.14%; lysine-1.23%; histi-dine-0.51%, phenylalanine-0.92%, tyrosine-0.67%, leucine-1.43%, methionine-0.32%, valine-1.03%, alanine-1,28%, glicine-0.94%, proline-0.95%, glutamic acid-3.93%, serine-0.96%, threonine-0.96%, aspartic acid-1.81%, tryphtophan-0.32%, cysteine-0.25%) and micronutrients in trace quantities.
The dried extract was compressed into tablets, following pharmacotechnical procedures and the dosage supplemented to the control group was the equivalent of 30 mg/kg/day, divided into two daily intakes (six tablets daily, three in the morning three in the afternoon, between meals), taking into consideration the average weight of the studied population during a six-month period. The placebo group received the same number of tablets with resembling ingredients and the same amount of calories (starch), but without Agaricus sylvaticus extract.
Clinical evolution
Patients were monitored for six months. During the first three months, consultations were scheduled every 15 days for clinical evaluation, and towards the final months, every 30 days.
At first, all patients remained on their usual diet but, throughout treatment, received general orientation on how to maintain a healthy diet. After six months of monitoring, all patients were prescribed a personal diet and were sent to other health professionals when necessary.
Three laboratory tests were performed for analysis of metabolic, biochemical and enzymatic profile of patients: one before the beginning of the supplementation, one after three months of treatment and one at the end of the treatment (after six months).
]]> The tests performed were glucose, total cholesterol (TC), triglycerides (TG), uric acid, urea, creatinine, alkaline phosphatase, total (TB), direct (DB) and indirect (IB) bilirubin, aspartate aminotransferase (AST) and alanine aminotransferase (ALT), immunoglobulins A (IgA), G (IgG) and M (IgM), total proteins and protein fractions.In addition to these tests, blood pressure levels were assessed at every consultation. However, for statistical purposes, the average of confronted results was weighed at three distinct stages: before beginning supplementation, after three and six months subsequently.
All patients were contacted by researchers weekly by telephone to clarify doubts, check adequate use of the mushroom and confirm appointments in view of guaranteeing major adhesion to treatment and continuity of the study.
Dropouts were deemed as: patients who only showed up for the first appointment; those who did not come to consultations during the six-month period; those who had less than three examinations. Patients who died before the end of the treatment were not included in the sample.
Laboratorial evaluation
The collection of blood was realized upon the criterion of 12h fasting patients. Collected material was placed in vacuum tubes for serum obtention, having followed protocols recommended by the Brazilian Society of Pathology for Collection of Venus Blood.15
Examinations were carried out at the Base Hospital Laboratory of Clinical Pathology of the Health Ministry-Federal District. Collected samples were centrifuged and analyzed in a 3000 TARGA device-Random Access Chemistry Analyzer, using Wiener kits while following laboratorial procedures.
Analyses of glucose, total cholesterol, triglycerides and uric acid were determined by the enzymatic colorimetric method. Urea, creatinine and alkaline phosphatase were analyzed by enzyme kinetic method together with total bilirubin and bilirubin fractions by DPD method. AST and ALT were analyzed by optimized UV method (IFCC). IgA, IgG and IgM were measured by immunoturbidimetry. Total protein and protein fractions by colorimetric biuret method and after determination of these fractions, the albumin/ globulin ratio (A/G) were calculated.
Test results were analyzed according to standardized reference values of the State Health Secretariat Laboratory-Federal District.
Blood pressure assessment
]]> To measure blood pressure, we used an aneroid sphygmomanometer and Tycos® stethoscope previously calibrated for adults. Measurements were constantly taken in the right arm with the patient seated. The systolic blood pressure (SBP) was recorded in the first Korotkoff phase (appearance of the noise), and diastolic blood pressure (DBP) was recorded in the fifth Korotkoff phase (disappearance of the noise). All patients were submitted to two or three measurements of blood pressure during consultations, whereby values obtained in the second blood pressure measurement were considered as the final analysis. The VI Joint National Committee-JNC16 criterion was used for defining arterial hypertension (mmHg). Thus, the individuals with SBP > 140 mmHg and/or DBP > 90 mmHg were considered hypertensive, and those with SBP < 140 mmHg and/or DBP < 90 mmHg normotensive.Statistical analysis
Values presented were compared and analyzed using the t-student and F statistical test, realized by the Microsoft, Excel version 2003 and SPSS (Statistical Package of the Social Sciences, SPSS Inc., Chicago, USA) for Windows version 14.0 programs with value of significance for p < 0.05.
In the placebo group, observations concluded that, 46.43% (n = 13) of patients were between the age of 45 and 60; 42.86% (n = 12) were between the age of 60 and 80 and only 10.71% (n = 3) were between 30 and 45 years old. There were no individuals under the age of 30 (table I). In the supplemented group, 46.43% (n = 13) of patients were between the age of 60 and 80; 35.71% (n = 10) were between 45 and 60 years old; 10.71% (n = 03) were under the age of 30 and only 7.14% (n = 2) were aged between 30 and 45 (table I).
Results
Characteristics of study population
A total of 92 patients with colorectal cancer who met the criteria for inclusion and exclusion agreed to participate in the study. Of these, 22 who were part of the pilot study were dismissed before the end of the study, and consequently, excluded from the final sample. Seventy patients started the study, and of these, four died and ten withdrew for various reasons. The final sample consisted of 56 patients (24 men and 32 women), stages I (n = 12), II (n = 16) and III (n = 28), separated into two groups: placebo (n = 28) and supplemented with Agaricus sylvaticus fungi (n = 28) (fig. 1).
In the placebo group (n = 28), the average age was 59.14 ± 12.95 years. In relation to gender, 57.1% (n = 16) were female and 42.9% (n = 12) male. Patients supplemented with Agaricus sylvaticus (n = 28) were between 56.34 ± 15.53 years of age. In relation to gender, female represented 57.1% (n = 16) and male 42.9% (n = 12).
In the placebo group, observations concluded that, 46.43% (n = 13) of patients were between the age of 45 and 60; 42.86% (n = 12) were between the age of 60 and 80 and only 10.71% (n = 3) were between 30 and 45 years old. There were no individuals under the age of 30 (table I). In the supplemented group, 46.43% (n = 13) of patients were between the age of 60 and 80; 35.71% (n = 10) were between 45 and 60 years old; 10.71% (n = 03) were under the age of 30 and only 7.14% (n = 2) were aged between 30 and 45 (table I).
As for the stadium, 50.00% (n = 14) of patients in both groups belonged to stadium III. In stadium II, 35.71% (n = 10) of patients belonged to the placebo group and 21.43% (n = 6) belonged to the group supplemented with Agaricus sylvaticus. In stadium I, 14.29% (n = 4) of patients belonged to the placebo group and 28.57% (n = 8) to the supplemented group (table I).
]]> It was observed that 7.14% (n = 02) of patients were smokers. In the placebo group, 17.86% (n = 5) were ex-smokers and 75.00% (n = 21) non-smokers. In the supplemented group, 28.57% (n = 8) were ex-smokers and 64.29% (n = 18) had never smoked (table I).Regarding alcoholism, 17.86% (n = 5) of patients supplemented with Agaricus sylvaticus related alcoholic beverage consumption, while in the placebo group this number reached 10.71% (n = 3) (table I).
In both groups 7.14% (n = 2) of patients practiced some physical activity (table I). After six months of treatment, an increased number of patients were observed to have adhered to physical activity, 21.43% (n = 6) and 35.71% (n = 10) in the placebo and Agaricus sylvaticus groups, respectively.
Effects of Agaricus sylvaticus in glucose and lipid metabolism
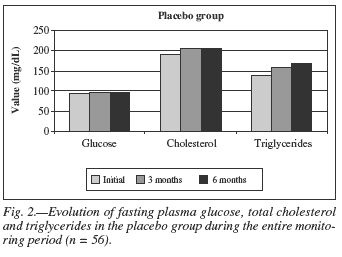
Initially, the placebo group had an average of fasting glucose concentration of 94.36 ± 15.34 mg/dL, and after three months a significant increase to 98.12 ± 15.54 mg/dL (p = 0.03) occurred, remaining at normal levels (70-110 mg/dL); during the sixth months of supplementation a significant increase was observed once again in plasma glucose concentration of 98.52 ± 9.03 mg/dL (p = 0.01), remaining within normal levels (fig. 2). Similarly, the group supplemented with Agaricus sylvaticus had an average glucose concentration of 95.92 ± 11.64 mg/dL in the beginning, which after three months was reduced to 94.88 ± 12.24 mg/dL, though statistically non-significant (p = 0.65). During sixth months of supplementation, a more significant reduction of fasting plasma glucose levels occurred; from 94.88 ± 12.24 mg/dL to 92.86 ± 6.82 mg/dL (p = 0.01) (fig. 3).
As for the analysis of total cholesterol (reference value = 140-200 mg/dL) the following results were observed: the placebo group had an initial total cholesterol of 192.71 ± 35.70 mg/dL, after three months it was observed an increase (from 192.71 ± 35.70 mg/dL to 204.68 ± 56.09 mg/dL, p = 0.08) and in the sixth months, there was again an increase (from 192.71 ± 35.70 mg/dL to 203.32 ± 48.78 mg/dL, p = 0.08), but these alterations were non-relevant statistically (figure 2). The Agaricus sylvaticus group had initially a total cholesterol of 207.36 ± 52.67 mg/dL, within three months 194.68 ± 44.66 mg/dL, and after six months 191.11 ± 39.72 mg/dL, with significant reduction of total cholesterol, with p = 0.05 and p = 0.01, respectively (fig. 3).
In the placebo group, it was observed a significant increase of serum TG (reference value = 65-150 mg/dL) within three months (from 137.93 ± 59.19 mg/dL to 157.54 ± 68.45 mg/dL, p = 0.01) and after six months (from 137.93 ± 59.19 mg/dL to 169.65 ± 89.13 mg/dL, p = 0.0001) (fig. 2). These alterations were not observed in the supplemented group that presented decreased TG within three months (from 181.64 ± 187.62 mg/dL to 174.79 ± 168.17 mg/dL, p = 0.33) and after six months (from 181.64 ± 187.62 mg/dL to 168.04 ± 146.91 mg/dL, p = 0.18) of treatment (fig. 3).
]]> Effects of Agaricus sylvaticus on biochemical and enzymatic parametersIn the placebo group, a significant increase was observed in serum urea levels within three months (p= 0.0001), and a non-significant increase (p = 0.30) within six months of monitoring. Opposite results were found in the Agaricus sylvaticus group throughout treatment. However, these changes are not statistically relevant (table II).
In relation to serum creatinine, a non-significant increase was observed in the placebo group after three and six months of monitoring, with p = 0.38 and p = 0.37, respectively. These results were not found in the group supplemented with Agaricus sylvaticus, which presented a significant reduction of serum creatinine levels within six months of supplementation, p = 0.05 (table II).
Upon analyzing serum uric acid, alkaline phos-phatase and total direct and indirect bilirubin levels, neither group showed statistically relevant alterations during the whole monitoring period (table II).
In the Agaricus sylvaticus group, there was a significant reduction of AST and ALT after six months of treatment, with p = 0.05 and p = 0.04, respectively. These results were not found in the placebo group (table II).
A significant reduction was detected in IgA (p = 0.0001) and IgM (p = 0.02) and non-significant reduction in IgG (p = 0.20) in the Agaricus sylvaticus group after six months of treatment, facts not observed in the placebo group (table II).
In relation to total proteins and protein fractions, a significant reduction of total protein was observed in the placebo group within six months (p = 0.01) while there was non-significant reduction (p > 0.05) of albumin and globulin within three and six months of monitoring. In the group supplemented with Agaricus sylvaticus, a significant increase of total protein was observed after three and six months (p = 0.0001), of globulin (p = 0.02) within three months, and an insignificant increase of other parameters during the treatment. Analyzing the A/G ratio in both groups, no statistically relevant alteration was observed (table II).
Effects of Agaricus sylvaticus on blood pressure
Initially the placebo group had an average SBP of 127.31 ± 18.01 mm/Hg, after three months an in significant increase to 128.65 ± 17.86 mm/Hg, (p = 0.33) occurred, and in six months a significant increase to 134.23 ± 20.82 mm/Hg (p = 0.01) was observed. The supplemented group initially had an average SBP of 127.88 ± 15.37 mm/Hg, after three and six months a significant reduction to 119.23 ± 13.83 mm/Hg (p = 0.0001) and 120.42 ± 12.77 mm/Hg (p = 0.0001), respectively was observed.
The average DBP in the placebo group before starting treatment was 77.12 ± 10.41 mm/Hg with a significant increase to 88.08 ± 11.23 mm/Hg (p = 0.0001) after three months and 80.38 ± 10.38 mm/Hg (p = 0.02) after six months of monitoring. The Agaricus sylvaticus group initially had an average DBP of 82.50 ± 8.63 mm/Hg with significant reduction within three (75.38 ± 9.89 mm/Hg, p = 0.0001) and six months (76.35 ± 8.19 mm/Hg, p = 0.0001) of supplementation.
]]>Discussion
The highest prevalence of patients in the present study belongs to the female gender, which corroborates with the estimates of the National Cancer Institute that there is a predominance of colorectal cancer among women when compared to men.17
The average age was 56 and 59 years old in the Agaricus sylvaticus and placebo groups respectively. Such results are confirmed by literature, which indicates the highest incidence of colorectal cancer among patients over 50 years of age.1
Fifty percent of patients in the placebo and Agaricus sylvaticus groups belonged to stadium III, by TNM (primary tumor, regional lymph nodes and metastasis at a distance) and Duke's classification systems of malignant tumors of the International Union Against Cancer (UICC).18,19 Similar results were found in other studies, showing a delay in diagnosing the majority of patients, thus significantly jeopardizing the prognostic.20
Information on both groups verified the presence of smokers and consumers of alcoholic beverage even in the presence of the disease. Scientific evidences have shown that exposure to smoking may increase the risk of colorectal cancer development as well as high mortality from this disease. Similarly, studies indicate that excessive consumption of alcoholic beverages is a risk factor for adenomas and colorectal cancer.1
In this study, only a small percentage of patients belonging to the placebo and Agaricus sylvaticus groups stated to have practiced some physical activity before starting treatment. After six months, observation showed a growing interest in physical activity by both groups; however, the supplemented group showed a higher number of patients. Studies have demonstrated that physical activity reduces the risk of colorectal cancer whose main mechanism seems to be the improvement of insulin resistance1. Other probable mechanism related to this effect includes: reduction of intestinal transit time, reduction of contact of carcinogens with intestinal cells, improvement on prostaglandin levels, modification of the biliary acid metabolism and a boosted immune system.21
Alterations in carbohydrate, protein, and lipid metabolism have been reported in patients with cancer5,12 such as: glucose intolerance, decreased insulin secretion, insulin resistance, increase in synthesis and turnover of glucose, elevated Cori cycle activity, increased protein turnover, increase in hepatic protein synthesis, elevated muscle catabolism, depletion of lipid deposits, increased lipolysis, increase of glycerol and fatty acids turnover, reduced lipogenesis and hyperlipidemia.12 Nevertheless, it has been hypothesized that the tumor itself is responsible for these metabolic abnormalities and for alterations in nutrient intake with significant repercussion on the nutritional status of oncological patients.5
Mechanisms related to the process of oncogenesis involve the secretion of cytokines, regulatory and counterregulatory hormones, such as tumor necrosis factor-alpha (TNF-α), interleukin 1 (IL-1), interleukin 2 (IL-2), interleukin 6 (IL-6), interferon-gamma (ITF-y), glucagon, cortisol, catecholamines, growth hormone, serotonin, leptin, lipid-mobilizing factor (LMF), and proteolysis-inducing factor (PIF).12
Scientific evidence have demonstrated that bioactive compounds present in Agaricaceae fungi exert antitumor, anticarcinogenic, antimutagenic, immunomodula-tory,9,22 hypolipidemic, hypocholesterolemic,14 hypoglycemic,5 hypotensive23 activities, and others that act in bioregulation and preservation of body homeostasis.9
]]> Glucans, proteoglucans, lectins, ergosterol, triter-penes, arginine, among other active substances, are commonly present in medicinal fungi.5,11,24 The mechanisms of action of these substances are not entirely elucidated in the literature, but evidences suggest that these bioactive compounds may modulate the process of carcinogenesis as it initiates, modulate promotion and progression stages thus bringing additional benefits to patients with various types of cancer.11In the present study, a significant reduction of fasting glycemia was observed in the Agaricus sylvaticus group after six months of supplementation. Opposite results were found in the placebo group, where a significant increase in glycemic levels was attested. A study to determine whether Agaricus blazei Murrill improves insulin resistance in type 2 diabetic patients treated with metformin and glicazide showed significant reduction of insulin resistance index and elevated adiponectin concentrations in the supplemented group compared to the placebo group25. Experimental studies carried out with rats with type 1 diabetes have demonstrated that the administration of medicinal fungi has important hypoglycemic action through secretion and/or action of insulin.26
The main substance responsible for the metabolic attributes of Agaricaceae fungi is the β-glucan,9 a soluble alimentary fibers capable of bringing down peaks of insulin and postprandial glucose and its respective curves, promoting beneficial effects towards glucose tolerance.27
Hypotheses suggest that the intrinsic mechanisms of these fibers retards gastric emptying, reduces absorption of carbohydrates, and stimulates the secretion of gastrointestinal inhibiting peptide, cholecystokinin and enteric glucagon hormones which together with parassympatic stimulation promote retardation of gastric emptying, increasing intestinal motility and the release of insulin by the pancreas.28
The supplemented group with Agaricus sylvaticus showed significant reduction of TC levels and non-significant reduction of TG levels throughout the treatment period, facts not observed in the placebo group. Research has shown that several medicinal fungi constitute a natural diet essential for the control of dyslipidemia. The potential lipid-lowering effect of polysac-charides extracted from Pholiota nameko fungi, including the ability to reduce the serum concentration of TC, TG, low-density lipoprotein (LDL) and phospholipids levels, as well as capacity to increase high-density lipoprotein (HDL), was established in hyper-lipidemic experimental models.29 Other studies conducted with laboratory animals have also shown that certain species of fungi are capable of significantly reducing the very low-density lipoprotein (VLDL), atherogenic index and activity of 3-hydroxy-3-methylglutaryl-coenzyme A reductase enzyme.30,31 An experimental study has found abnormal levels of triglycerides in animals with solid Walker 256 tumor, whereas the group treated with Agaricus sylvaticus showed significant reduction when compared to the animals treated with placebo, indicating beneficial effects of a therapy with this mushroom5. Percario et al32 verified the preventive effects of atherosclerosis in rabbits after supplementation with Agaricus sylvaticus and demonstrated potent antioxidant action of this fungus as it prevented the development of atheromatous plaques in the supplemented group.
β-glucans are capable of lowering serum cholesterol and other seric lipids,30 although the active mechanisms of these substances have not been totally detailed in literature.12 It has been widely recognized that this property is multifactorial, involving biliary acids and cholesterol increase in fecal excretion,30 alterations in intestinal absorption and secretion of pancreatic and/or intestinal hormones, alterations in lipoprotein and bile acids metabolism, increase in viscosity of small intestine with reduced absorption and/or re-absorption of biliary acids and cholesterol and/or triglyceride, and production of short-chain fatty acids, particularly propionate, a fermentable metabolite of soluble fibers.33 Triterpenes also contribute to explain the hypolipidemic effects, since it acts as inhibitor of enzymes such as cholesterol synthase and angiotensin-converting enzyme.34
Abnormal levels of urea and creatinine have been widely studied in oncological patients.5 These patients often display skeletal muscle catabolism, in consequence do not preserve the skeletal muscle protein and do not release large amounts of nitrogen in the form of urea. The gluconeogenesis, which converts amino acids into glucose, is linked to the rate at which ammonium ions in the liver are converted into either urea or glutamine.35 The catabolism of body proteins and oxidation of released amino acids in this process generate ammonia, which in turn, is predominantly incorporated into urea by the liver. This catabolism increases with tumor progression, which culminates in increased production of urea. During periods of severe metabolic stress, protein catabolism and urea production may increase significantly.5,35 Therefore, daily intake of protein is essential for the preservation of muscle mass and body nitrogen reserves, and the measurement of creatinine levels has been shown to be a useful marker of protein to evaluate nutritional status.5
In the present study, a significant increase was observed in urea levels in the placebo group and a significant reduction of creatinine in the supplemented group, implying that the nutrients present in Agaricus sylvaticus, particularly proteins and amino acids may reduce the catabolic state common in oncological patients. In an experimental study, rats inoculated with Walker 256 tumor presented higher levels of urea compared to the control animals, but the animals that received Agaricus sylvaticus solution presented a significant reduction in blood urea levels compared to those treated with placebo. Similarly, alterations were observed in creatinine levels in animals with Walker 256 tumor compared to control animals.5 Fungi from the Agaricaceae family are rich in arginine, a nutrient with important immunomodulatory property and essential to the preservation of muscle mass and nitrogen reserves5, since it is able to enhance the immune system through growth hormone release.36
Significant alterations in serum uric acid, alkaline phosphatase, total and fraction bilirubin levels, have not been found in either group during the entire monitoring period. However, studies show that certain medicinal fungi are capable of significantly reducing alkaline phosphatase, total bilirubin and hepatic collagen content, consisting of a promising antifibrotic agent.37
The activity of AST and ALT in the liver, decreases in cachexic cancer patients, indicating a reduction in alanine and aspartic acid through degrading processes. In repletion, with a normal protein diet, the hepatic levels of such enzymes are restored to normal. Therefore, in cancer patients these hepatic enzymes increase during protein depletion and decrease on repletion, indicating that cancer is more efficient in competing with the liver by intake of amino acids as an energetic source and nitrogenated.35
]]> In the present study it was observed a significant reduction of AST and ALT, in the supplemented group with Agaricus sylvaticus, whereas in the placebo group these alterations were not found. Wang et al.38 demonstrated that Ganoderma lucidum polysaccharides may significantly improve the serum hepatic enzyme levels of ALT and AST in rats with immune hepatic injury induced by bacillus Calmette-Guerin (BCG). Hi et al.,14 in order to assess the effect of concentrated extract of Agaricus sylvaticus in rats inoculated with Pristane, showed that the group of animals inoculated with this drug and supplemented with the fungus, presented little neoplastic alterations compared to the control group. Albumin, ALT and AST levels were within reference values, suggesting a protective effect of Agaricus sylvaticus, without producing toxic effect in studies with animals. Gonzaga et al.22 in an experiment with rats transplanted with sarcoma 180, did not observe renal (urea) nor hepatic (ALT and AST) alterations in rats treated with Agaricus blazei Murrill only or in combination with 5-FU (fluorouracil). The authors, however, observed significant antitumoral effect by inhibition of tumor growth promoted by the Agaricus blazei Murrill, reinforcing the potential anticarcinogenic effect of these medicinal fungi. Hsu et al.39 observed a significant reduction in hepatic enzyme levels, with consequent normalization in patients with chronic hepatitis B and with abnormal liver function after receiving Agaricus blazei Murrill extracts. These studies have proved the hepatoprotective effects attributed to medicinal fungi whose probable mechanism of action may be the inhibition of hepatic oxidative stress due to its antioxidant action and ability to fight against free radi-cals.40In the present study the supplemented group with Agaricus sylvaticus showed a significant reduction of IgA and IgM and, in the placebo group no significant alterations were found. However, opposite results were found in other studies conducted in oncological patients undergoing chemotherapy and supplemented with other medicinal fungi.41,42 In vivo studies have revealed that extracts of specific fungi posses non-significant immunological effect on individuals with normal parameters; however, they are able to restore the depressed immunological responsiveness caused by tumors, reaching normal levels.16 These factors may partially explain the results found in immunoglobulin levels of patients supplemented with Agaricus sylvaticus in this study.
In the supplemented group, there was a significant increase in total protein levels during the treatment, while opposite results were found in the placebo group. The A/G ratio did not change significantly in both groups. However, as we verified a significant reduction of total protein in the placebo group, it was found that this had occurred at the expense of a reduction in albumin with consequent increase in globulin, events not observed in the supplemented group. A study in patients with acute non-lymphocytic leukemia treated with chemotherapy and supplemented with Agaricus blazei Murrill showed significant improvement in serum albumin levels and A/G ratio.41 Other studies in patients with gastrointestinal cancer showed no changes in total protein and protein fractions, and A/G ratio in the supplemented group with Agaricus blazei Murrill, whereas the placebo group presented insignificant reduction of total protein and albumin, insignificant increase of globulin, and significant reduction of A/G ratio, suggesting that controlled immunity inhibits tumoral growth.42
It's important to point out that the referred dosages of ALT and AST in the Agaricus sylvaticus group of the present study, were compared to normal dosages of albumin since it may reflect an increase in the capacity of hepatic synthesis, indicating a possible improvement in this group as a result of supplementation with these medicinal fungi.
Scientific evidence shows that β-glucans act on the immune system through stimulation of natural killer cells, T lymphocytes, B lymphocytes and complementary cells, which lead to an increase in the number and function of macrophages and monocytes, with subsequent proliferation and/or production of antibodies and various cytokines such as IL-2, IL-6, TNF-α and y-ITF, preventing regeneration and metastasis of cancer.8,9
Both SBP and DBP were considerably reduced in the supplemented group during treatment, whereas in the placebo group it showed an important increase in blood pressures. A study in tracheostomized experimental models showed substantial reduction in mean arterial pressure as well as in heart rate after supplementation with Agaricus blazei Murrill.23 Another study showed that administration of Tricholoma mongolicum in rats, promoted a reduction in mean arterial pressure of 95.3 ± 7.4 mmHg.44 Investigators hypothesized that the probable mechanism of action refers to vasorelaxation via production of adenosine A2 receptors and/or production of nitric oxide.23,44 The gamma-aminobutyric acid (GABA) can be found in some species of medicinal fungi, which may also explain the reduction in mean arterial pressure by direct action on blood vessels or ganglionary blockage, with subsequent release of transmitters in sympathetic nerve terminals, or the somatory of these two effects. It is worth mentioning that certain fungi are rich in potassium and calcium, and since calcium may activate potassium channels in vascular smooth muscle, causing hyperpolarization and relaxation, it partly explains the reduction in blood pressure.23 Arginine also plays an important role in regulating cardiovascular function, as it is a precursor of nitric oxide, a potent neurotransmitter that has a vital role in the dilatation and constriction of small cerebral blood vessels.36
Despite the fact that the mechanisms of the bioactive compounds present in Agaricaceae fungi have not beentotally clarified in literature, the nutrients present in Agaricus sylvaticus may contribute to reducing the catabolic state in oncological patients and assist in the reduction of metabolic abnormalities, and biochemical, enzyme and blood pressure parameters in these patients.
Conclusion
The results suggest that dietary supplementation with Agaricus sylvaticus fungi is capable of exerting metabolic benefits in addition to the benefits in the biochemical, enzymatic and blood pressure parameters of patients with colorectal cancer in the postsurgical phase. Nevertheless, given the scarcity of studies in literature, further randomized clinical trials such as this, are still necessary to determine dosage and other clinical conditions for the adjuvant use of Agaricus sylvaticus to be beneficial.
]]>References
1. Fortes RC, Recova VL, Melo AL, Novaes MRCG. Hábitos dietéticos de pacientes com cáncer colorretal em fase pós-operatória. Rev Bras Cancerol 2007; 53 (3): 277-89. [ Links ]
2. Martellucci J, Civitelli S, Dhamo A, Tanzini G. Familial colorectal cancer: a concept revisited. Colorectal Dis 2009; 11 (2): 133-7. [ Links ]
3. Mehrkhani F, Nasiri S, Donboli H, Meysamie A, Hedayat A. Prognostic factors in survival of colorectal cancer patients after surgery. Colorectal Dis 2009; 11 (2): 157-61. [ Links ]
4. Araújo FF, Silva CC, Fortes RC. Terapia nutricional enteral em pacientes oncológicos: uma revisáo da literatura. Com Ciencias Saúde 2008; 19 (1): 61-70. [ Links ]
]]>5. Taveira VC, Reis MA, Silva MF, Novaes MRCG. Hematological and metabolic effects of dietary supplementation with Agaricus sylvaticus fungi on rats bearing solid Walker 256 tumor. Exp Biol Med 2008; 233 (11): 1341-47. [ Links ]
6. Toscano BAF, Coelho MS, Abreu HB, Logrado MHG, Fortes RC. Cáncer: implica tes nutricionais. Com Ciencias Saúde 2008; 19 (2): 171-180. [ Links ]
7. Santos Jr JCM. Cáncer ano-reto-cólico: aspectos atuais III -cáncer de reto - terapéutica neoadjuvante. Rev bras Coloproct 2008; 28 (1): 108-18. [ Links ]
8. Novaes MRCG, Fortes RC. Efeitos antitumorais de cogumelos comestíveis da familia Agaricaceae. Rev Nutr Bras 2005; 4 (4): 207-17. [ Links ]
9. Fortes RC, Taveira VC, Novaes MRCG. The immunomodulator role of -D-glucans as co-adjuvant for cancer therapy. Rev Bras Nutr Clin 2006; 21 (2): 163-8. [ Links ]
]]>10. Sullivan R, Smith JE, Rowan NJ. Medicinal mushrooms and cancer therapy. Spring 2006; 49 (2): 159-70. [ Links ]
11. Fortes RC, Recova VL, Melo AL, Novaes MRCG. Qualidade de vida de pacientes com cáncer colorretal em uso de suplementagáo dietética com fungos Agaricus sylvaticus após seis meses de segmento: ensaio clínico aleatorizado e placebo-controlado. Rev bras Coloproct 2007; 27 (2): 130-38. [ Links ]
12. Fortes RC, Novaes MRCG. Efeitos da suplementagáo dietética com cogumelos Agaricaceae e outros fungos medicinais na terapia contra o cáncer. Rev Bras Cancerol 2006; 52 (4): 363-71. [ Links ]
13. Hetland G, Johnson E, Lyberg T, Bernardshaws S, Tryggestad AMA, Grinde EtB. Effects of the medicinal mushroom Agaricus blazei Murril on immunity, infection and cancer. Scandinavian Journal of Immunology 2008; 68: 363-70. [ Links ]
14. Hi EMB, Azevedo MRA, Bach EE, Ogata TRP. Efeito protetor do Agaricus sylvaticus em fígado de ratos do tipo wistar inoculado com pristane. Saúde Coletiva 2008; 05 (21): 76-79. [ Links ]
]]>15. Recomendacòes da Sociedade Brasileira de Patologia Clinica/ Medicina Laboratorial para Coleta de Sangue Venoso. 1a ediçao. Sao Paulo. 2005. pp. 01-76. [ Links ]
16. The Sixth Report of the Joint National Committee on prevention, detection, and treatment of high blood pressure. Arch Intern Med 1997; 157: 2413-46. [ Links ]
17. Brasil. Ministério da Saúde. Secretaria de Atencao à Saúde. Coordenacao de Prevençao e Vigilancia. Instituto Nacional de Cáncer. Estimativa 2010: Incidencia de cáncer no Brasil. Rio de Janeiro: 17. 98 p.: il. [ Links ]
18. Diniz BSO, Lacerda-Filho A. Prevencao secundária do cáncer colorretal em individuos assintomáticos de baixo risco. Rev Med Minas Gerais 2004; 14 (1): 46-52. [ Links ]
19. Brasil. Ministério da Saúde. Secretaria de Atençao à Saúde. Instituto Nacional de Cáncer. TNM: classificaçao de tumores malignos. 6. ed. Rio de Janeiro: 19. p. 254. [ Links ]
]]>20. Saad-Hossne R, Prado RG, Bakonyi Neto A, Lopes PS, Nascimento SM, Santos CRV et al. Estudo retrospectivo de pacientes portadores de cáncer colorretal atendidos na Faculdade de Medicina de Botucatu no periodo de 2000-2003. Rev bras Coloproct 2005; 25 (1): 31-37. [ Links ]
21. Brasil. Ministério da Saúde. Secretaria de Assistencia à Saúde. Instituto Nacional de Cáncer. Falando sobre cáncer de intestino/ Instituto Nacional de Cáncer. Sociedade Brasileira de Coloproctologia. Colégio Brasileiro de Cirurgioes. Associacao Brasileira de Colite Ulcerativa e Doenca de Crohn. Colégio Brasileiro de Cirurgia Digestiva. Sociedade Brasileira de Endoscopia Digestiva. Sociedade Brasileira de Cancerologia. Sociedade Brasileira de Oncologia Clinica. Rio de Janeiro: 21. 36 p.: il. [ Links ]
22. Gonzaga MLC, Bezerra DP, Alves APNN, Alencar NMN, Mesquita RO, Lima MW et al. In vivo growth-inhibition of sarcoma 180 by an α-(1-4)-glucan-β-(1-6)-glucan-protein complex polysaccharide obtained from Agaricus blazei Murril. J Nat Med 2009; 63: 32-40. [ Links ]
23. Singi G, Damasceno DD, Andréa EDD, Alexandre GMB, Singi MB, Alves LC et al. Efeitos agudos da aplicaçao endovenosa do cogumelo-do-sol (Agaricus blazei Murril) sobre a pressao arterial média e a freqüencia cardiaca de ratos anestesiados. Rev Bras Farmacogn 2006; 16 (4): 480-84. [ Links ]
24. Novaes MRCG, Novaes LCG, Melo AL, Recova VL. Efeitos do cogumelo Agaricus sylvaticus no sistema hematopoético de ratos com tumor ascitico de Walker 256. Rev Bras Cancerol 2008; 54 (2): 147-52. [ Links ]
]]>25. Hsu C-H, Liao Y-L, Lin S-C, Hwang K-C, Chou P. The mushroom Agaricus blazei Murril in combination with metformin and glicazide improves insulin resistance in type 2 diabetes: a randomized, Double-blinded, and placebo-controlled clinical trial. J Altern Complement Med 2007; 13 (1): 97-102. [ Links ]
26. Hwang H-J, Kim S-W, Lim J-M, Joo J-H, Kim H-O, Kim H-M, et al. Hypoglicemic effect of crude exopolysaccharides produced by a medicinal mushroom Phellinus baumii in streptozotocin-induced diabetic rats. Life Sciences 2005; 76: 3069-80. [ Links ]
27. Behall AM, Schofield DJ, Hallfrisch JG, Liljeberg-Elmstahl HGM. Consumption of both resistant starch and -glucan improves postprandial plasma glucose and insulin in women. Diabetes Care 2006; 29 (5): 976-81. [ Links ]
28. Catalani LA, Kang EMS, Dias MCG, Maculevicius J. Fibras alimentares. Rev Bras Nutr Clin 2003; 18 (4): 178-82. [ Links ]
29. Li H, Zhang M, Ma G. Hypolipidemic effect of the polysaccharide from Pholiota nameko. Nutrition 2009; 1-7. [ Links ]
]]>30. Cheung PCK. Plasma and hepatic cholesterol levels and fecal neutral sterol excretion are altered in hamsters fed straw mushroom diets. J Nutr 1998; 128 (9): 1512-16. [ Links ]
31. Yang BK, Park JB, Song CH. Hypolipidemic effect of an exo-biopolymer produced from a submerged mycelial culture of Hericium erinaceus. Biosci Biotechnol Biochem 2003; 67 (6): 1292-98. [ Links ]
32. Percario S, Odorizzi VF, Souza DR, Pinhel MA, Gennari JL, Gennari MS et al. Edible mushroom Agaricus sylvaticus can prevent the onset of atheroma plaques in hypercholesterolemia rabbits. Cell Mol Biol 2008; 17 (54): 1055-61. [ Links ]
33. Fukushima M, Ohashi T, Fujiwara Y, Sonoyama K, Nakano M. Cholesterol-lowering effects of maitake (Grifola frondosa) fiber, shiitake (Lentinus edodes) fiber, and enokitake (Flammulina velutipes) fiber in rats. Exp Biol Med 2001; 226 (8): 758-65. [ Links ]
34. Taveira VC, Novaes MRCG. Consumo de cogumelos na nutriçao humana: uma revisao da literatura. Com Ciencias Saude 2007; 18 (4): 315-22. [ Links ]
]]>35. Lai H-S, Lee J-C, Lee P-H, Wang S-T, Chen W-J. Plasma free amino acid profile in cancer patients. Seminars in Cancer Biology 2005; 15 (4): 267-76. [ Links ]
36. Novaes MRCG, Novaes LCG, Taveira VC. Pharmacological effects of Agaricales fungi: a review of evidence. Rev Cien Med Campinas 2007; 16 (2): 87-95. [ Links ]
37. Park EJ, Ko G, Kim J, Sohn DH. Antifibrotic effects of a polysaccharide extracted from Ganoderma lucidum, glycyrrhizin, and pentoxifylline in rats with cirrhosis induced by biliary obstruction. Biol Pharm Bull 1997; 20 (4): 417-20. [ Links ]
38. Wang X, Zhao X, Li D, Lou Y-Q, Lin Z-B, Zhang G-L. Effects of Ganoderma lucidum polysaccharide on CYP2E1, CYP1A2 and CYP3A activities in BCG-immune hepatic injury in rats. Biol Pharm Bull 2007; 30 (9): 1702-06. [ Links ]
39. Hsu C-H, Hwang K-C, Chiang Y-H, Chou P. The mushroom Agaricus blazei Murril extract normalizes liver function in patients with chronic hepatitis B. J Altern Complement Med 2008; 14 (3): 299-301. [ Links ]
]]>40. Shi Y, Sun J, He H, Guo H, Zhang S. Hepatoprotective effects of Ganoderma lucidum peptides against D-galactosamine-induced liver injury in mice. Journal of Ethnopharmacology 2008; 117: 415-19. [ Links ]
41. Hui TX, Guo LZ, Jing W, Ito H, Shimura K, Zhi WJ. Clinical observation on treatment of acute non lymphocytic leukemia with Agaricus blazei Murril. Nutr Notebook 1988; 886. [ Links ]
42. Jing W, Min MX, Zheng CR, Zhi WJ, Ito W, Shimura K. Observation on treatment effect of Agaricus blazei Murril against alimentary tract tumor. Nutr Notebook 1988; 866-82. [ Links ]
43. Chu KKW, Ho SSS, Chow AHL. Coriolus versicolor: a medicinal mushroom with promising immunotherapeutic values. J Clin Pharmacol 2002; 42: 976-84. [ Links ]
44. Wang HX, Ooi VE, Ng TB, Chiu KW, Chang ST. Hypotensive and vasorelaxing activies of a lectin from the edible mushroom Tricholomamongolicum. Pharmacol Toxicol 1996; 79 (6): 318-23. [ Links ]
]]>
![]() Correspondence:
Correspondence:
Renata Costa Fortes.
Science and Education School Sena Aires.
University of Brasilia.
QI 14. CJ J. CS 26. Guará 1/DF.
CEP: 71.015-100 Brazil.
E-mail: renatacfortes@yahoo.com.br
Recibido: 21-VII-2010. ]]> Aceptado: 13-X-2010.
]]>