Nutrient-mediated modulation of incretin gene expression; a systematic review
Modulación de la expresión de genes de incretinas mediada por nutrientes; revisión sistemática
R. Martínez-Rodríguez and A. Gil
Departament of Biochemistry and Molecular Biology II. Institute of Nutrition and Food Technology. Centre of Biomedical Research. University of Granada. Granada. Spain.
]]>
ABSTRACT
Incretins are a cluster of hormones which are secreted and released into the bloodstream after food intake by gut enteroendocrine cells, reaching to pancreas where produce a potentiating effect on insulin release. The aim of this study was to perform a systematic review of incretins gene expression mediated by nutrients using specific search equations in the PubMed database. The two most relevant incretins are GLP-1 and GIP, which come from proglucagon and proGIP precursor respectively. GLP-1 is mainly synthesized and released by ileum and colon L cells, in contrast to GIP which does it by K cells in duodenum and proximal jejunum. It has been shown that canonical Wnt signalling pathway is closely related to the production of these hormones, since transcription factor TCF7L2 affects proglucagon and proGIP gene expression in L and K enteroendocrine cells. On the other hand, it has been shown that the hexosamine biosynthetic pathway can produce N-linked glycosylation of -catenin, an essential component of canonical Wnt signalling. This process hinders β-catenin phosphorylation and, thereby prevents proteasome degradation. Increasing glucose concentration enhances the hexosamine pathway and thus β-catenin glycosylation. This causes a β-catenin cytoplasmic accumulation allowing entry into nucleus, where it exerts its action by binding to a clump of molecules and transcription factors, allowing to express the target genes, including the incretin hormones. There is also evidence that glucose, through the hexosamine pathway, can induces autocrine activation of Wnt signalling pathway by stimulating secretion of Wnt proteins.
Key words: Gene expression regulation. Wnt proteins. Glycosylation.
RESUMEN
Las incretinas son una serie de hormonas que tras una ingesta de alimentos son secretadas y liberadas al torrente sanguíneo por células enteroendocrinas del intestino, llegando al páncreas, donde producen un efecto potenciador en la liberación de insulina. El objetivo de este trabajo ha sido realizar una revisión sistemática de la modulación de la expresión génica de las incretinas mediada por nutrientes utilizando ecuaciones específicas de búsqueda en la base de datos PubMed. Las dos incretinas más relevantes son el péptido análogo al glucagón 1 (GLP-1) y el péptido insulinotrópico dependiente de glucosa (GIP), que provienen de los precursores proglucagón y proGIP, respectivamente. GLP-1 es mayoritariamente sintetizado y secretado por las células L del íleon y del colon, a diferencia de GIP que lo hace por las células K de duodeno y yeyuno proximal. Se ha demostrado que la ruta canónica de señalización Wnt está estrechamente relacionada con la producción de estas hormonas, ya que el factor de transcripción TCF7L2 influye en la expresión génica de proglucagón y proGIP en células enteroendocrinas L y K. Por otra parte, se ha demostrado que la ruta biosintética de las hexosaminas es capaz de glicosilar la β-catenina, componente fundamental de la señalización canónica Wnt, lo que interfiere en la fosforilación de esta proteína, impidiendo así su degradación en el proteasoma. El aumento de la concentración de glucosa incrementa la ruta de las hexosaminas y de esta manera la glicosilación de la β-catenina. Esto produce una acumulación de esta proteína en el citoplasma celular y permite su entrada al núcleo, donde ejerce su acción al unirse a una serie de moléculas y factores de transcripción, permitiendo de este modo que se expresen los genes diana, entre los que se encuentran los de las hormonas incretinas. También hay evidencias de que la glucosa, a través de la ruta de las hexosaminas, es capaz de inducir la activación autocrina de la ruta de señalización Wnt estimulando la secreción de proteínas Wnt.
Palabras clave: Incretinas. Regulación de la expresión génica. Proteínas Wnt. Glicosilación.
Abbreviations
APC: Adenomatous polyposis coli.
β-Trcp: β-Transducin repeat-containing protein β. ]]>
CBP: CREB-binding protein.
CK1: Casein kinase 1.
DPP IV: Dipeptidyl peptidase IV.
GFAT: Glutamine:fructose-6-phosphate amidotransferase.
GIP: Glucose-dependent insulinotropic peptide.
GLP-1: Glucagon-like peptide-1.
GPR: G protein-coupled receptor.
GSK3: Glycogen synthase kinase 3.
HAT: Histone-acetyltransferase.
HBP: Hexosamine biosynthetic pathway. ]]>
HMT: Histone-methyltransferase.
LEF: Lymphoid enhancer factor.
LRP: Low density lipoprotein receptor-related protein.
OGA: O-GlcNAc hydrolase.
OGT: O-GlcNAc transferase.
PC: Prohormone convertase.
PPAR: Peroxisome proliferator activated receptor.
TCF: T-cell transcription factor.
WRE: Wnt-responsive element.
]]> Introduction
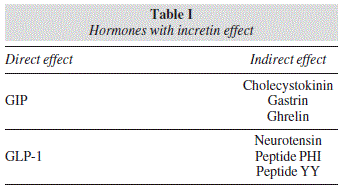
The term incretin refers to the effect of insulin released in response to food intake. Incretin hormones directly or indirectly increase this insulin secretion by β cells of pancreatic islets of Langerhans.1,2 Various hormones exert this effect in an indirect manner in organisms3-7 (table I), but only two of them act in a direct manner, making them the most relevant. These are glucose-dependent insulinotropic peptide (GIP), previously known as gastric inhibitory peptide, and glucagon-like peptide-1 (GLP-1).8 Besides enhancing pancreatic insulin release, these hormones play other roles in the organism. GLP-1 suppresses glucagon secretion, inhibits gastric emptying, and reduces appetite and food intake.9 It also appears to improve and increase the proliferation of pancreatic β cells, protect myocytes from ischemic damage, regulate glucose homeostasis, and exert an effect on immune function.10 For its part, GIP has been directly related to obesity, lipid metabolism,10,11 and bone remodeling, and it plays a regulatory role in the CNS in the proliferation of neural progenitor cells and behavior modification.10 A recent rat study added a possible relationship between GIP and cold acclimation.12
The incretin hormones GIP and GLP-1 belong to a superfamily of glucagon peptides and therefore have amino acid sequences in common. GIP is a peptide of 42 amino acids derived from its precursor, ProGIP, while GLP-1 includes peptides of 30 and 31 amino acids derived from its precursor, proglucagon. The "incretin effect" has been studied with glucose, observing a much greater insulin response after the oral versus intravenous intake of glucose, indicating that GIP and GLP-1 secretion takes place in the digestive tract13 and is mediated by two types of enteroendocrine cells of the gastrointestinal epithelium. Thus, K cells, mainly localized in the duodenum and proximal jejune, synthesize and release GIP, while L cells, largely found in the ileum and colon, synthesize and release GLP-1.14 A small amount of proximal small intestine cells are also known to express both GLP-1 and GIP.15 It is estimated that the indirect effects of incretins are responsible for up to 70% of the insulin secretion stimulated by glucose in healthy individuals.14 A change in blood glucose concentration derived from food intake amplifies the secretion by pancreatic β cells of GIP and GLP-1 into the blood stream, thereby increasing the release of insulin at the time when it is most needed.13 These effects on insulin release and glucose homeostasis improvement have raised expectations about the utilization of these hormones in the treatment of type two diabetes, and various therapies have been developed, mainly based on GLP-1.
The fact that these hormones are released when the food arrives in the digestive system and the capacity of some nutrients to increase or decrease this release16-18 suggest the existence of a system that regulates their production. However, there have been few studies on this issue, and the nutrient-mediated modulation of incretin gene expression remains hypothetical. The objective of this systematic review of the available literature was to search for an incretin gene expression regulatory mechanism that is influenced by a nutrient and therefore supports this hypothesis.
Method
The study was initiated with a search for literature reviews that would allow the issue to be approached from a wide perspective. We explored the PubMed database of the U.S. National Center for Biotechnology Information (NCBI). The first search equation used was "Gene Expression Profiling"[Mesh] OR "Gene Expression"[Mesh] OR "Gene Expression Regulation, Developmental"[Mesh] OR "Gene Expression Regulation"[Mesh]) AND "Incretins"[Mesh]. This initial search yielded nine articles, of which three were reviews. After reading the abstracts of each paper, one of them was selected for a full examination.13
]]> Once the bases of knowledge on incretins were established, a search was performed for articles related to our study hypothesis, using the equation (("Incretins/agonists"[Mesh] OR "Incretins/analogs and derivatives"[Mesh] OR "Incretins/biosynthesis"[Mesh] OR "Incretins/genetics"[Mesh] OR "Incretins/metabolism"[Mesh] OR "Incretins/physiology"[Mesh] OR "Incretins/secretion"[Mesh] OR "Incretins/therapeutic use"[Mesh] )) AND "Gene Expression Regulation"[Mesh]) NOT Review. This search yielded six reports, but only one provided useful data on the regulation of gene expression in incretin synthesis.34 We then used an alternative route (("Incretins/ biosynthesis" [Mesh] OR "Incretins/genetics"[Mesh] OR "Incretins/metabolism"[Mesh]) NOT ("review"[Publication Type] OR "review literature as topic"[MeSH Terms] OR "review"[All Fields]) AND ("2006/04/01"[PDat] : "2011/03/30"[PDat]) AND ("humans"[MeSH Terms] AND "2006/04/01"[PDat]: "2011/03/30" [PDat])), which generated 65 articles. Although some of these were not very relevant, their reading led to other papers of more interest. The search equation "Gene expression" AND "Incretins" AND "nutrient" also produced no conclusive result, and it was concluded that no authors had directly addressed the issue of interest in this study.Given the lack of studies on nutrient-mediated incretin gene regulation, a search was conducted on the synthesis of incretins, using (("Incretins/biosynthesis"[Mesh]) NOT Review). Various articles referred to Wnt signaling as a regulator of incretin synthesis, and the search was then focused on the relationship between nutrients and the Wnt signaling pathway, using "Wnt Proteins"[Mesh] AND "Nutrients". We found a comment in the Biochemical Journal on an original study highly relevant to our study hypothesis.39 The remaining studies consulted were drawn from reference lists in the selected articles and from different web pages.
Molecular mechanisms of incretin secretion and synthesis regulation
The enteroendocrine L cells that synthesize and release GLP-1 are found dispersed throughout the small and large intestine, with a higher density in the distal ileum and colon. In contrast, the anatomically distinct K cells that produce GIP are largely found in the duodenum. This may explain the different profiles of blood GLP-1 and GIP concentrations after food intake. Depending on the composition of the food, GLP-1 secretion can show a biphasic profile, with an early stage shortly after food intake that lasts for 15-30 min and a late phase that persists for 1-2 hours or longer. GIP secretion is produced after a similar brief delay and can remain elevated for various hours. In humans, glucose can be detected in the proximal duodenum within 5 minutes after the intake of an overload of liquid glucose, correlating with the time that elapses before the first rapid rise in blood GLP-1 and GIP levels.13 Hence, these cells possess nutrient-detection mechanisms that permit the prompt release and possibly the synthesis of the hormones.
Incretin secretion
GIP is released rapidly upon the arrival of nutrients to the duodenum because of the elevated local density of K cells. However, human studies on glucose infusion showed that GLP-1 is only stimulated when the rate of glucose entry into the duodenum exceeds the duodenal absorption capacity, i.e., when the nonadsorbed glucose filters to the jejune and can stimulate L cells in this region.19 Secretion is stimulated not only by carbohydrates but also by the other two macronutrients, lipids and proteins.20-23 Both GLP-1 and GIP are rapidly degraded after their secretion by the enzyme dipeptidyl peptidase IV (DPP-IV).24,25
Several highly ubiquitous transmembrane receptors with seven domains are widely activated by amino acids, carbohydrates, and free fatty acids and are expressed in various tissues, including the gastrointestinal tract. It has been hypothesized that these receptors may act as food intake modulation sensors, e.g., releasing incretin hormones into the intestine.26 GIP and GLP-1 receptors are G protein-coupled receptors (GPRs), which are integral membrane proteins with seven α helices,27,28 although enteroendocrine cells express various types of GPR coupled to Gs and Gq that induce incretin secretion. These receptors act by inducing signaling pathways, such as AMPc and protein kinese (PKC) in order to increase the calcium concentration via calcium channels, thereby producing the release of incretins.13
Incretin production
Hormone processing
]]> GLP-1 is cleaved after translational processing of the proglucagon gene product, which is expressed not only in L cells but also in pancreatic cells and a subset of brain-stem neurons. Alternative production of the prohormone is largely attributable to the differential expression of prohormone convertase (PC) enzymes. Pancreatic a cells express PC2 and generate glucagon, whereas L cells in the intestine express PC1/3 and produce the bioactive peptides GLP-1, GLP-2, and oxyntomodulin. The carboxyl-terminal fraction of proglucagon I is almost completely processed to GLP-1 and GLP-2 in L cells, while the N-terminal is released as active peptide oxyntomodulin and the rest as glycentin.14,29,30 Analysis of gene expression in L GLUTag cells, an experimental model, showed significantly increased mRNA levels of proglucagon and PC1/3 after incubation with glucose versus lactate. In contrast, the mRNA of PC2 was not regulated by glucose after 12 h of incubation, while both glucose and lactate tended to increase its expression after 24 h. These data show that glucose is not only a primary stimulator of GLP-1 release but also a positive transcriptional modulator of proglucagon and PC- 1/3 mRNA levels in L GLUTag cells.31Unlike proglucagon, the proGIP precursor in K cells is not believed to contain sequences of bioactive peptides besides GIP. The processing of proGIP in K cells, as in the case of proglucagon in L cells, is attributed to the action of PC1/3.30
Synthesis regulation
Various studies have reported that incretin gene expression is mainly regulated by the Wnt signaling pathway. One member of this pathway, transcription factor TCF7L2 or TCF-4 (T-cell transcription factor, not to be confused with transcription factor 4), was found to influence the gene expression of both proglucagon and proGIP in enteroendocrine cells in the gut.32-35 Furthermore, GLP-1 itself stimulates the Wnt pathway in an autocrine manner, and none of these effects have been described in pancreatic islet cells.14
WNT SIGNALING PATHWAY
Wnt signaling is an extremely complex process that has to be meticulously controlled in time and space. It is critically important for cell differentiation, cell-cell communication, and organ formation and growth. Wnt proteins are glycoprotein ligands secreted by cells. They contain a conserved domain of around 300 amino acids interrupted by a sequence of 21 to 23 cysteine residues that have a characteristic spatial pattern. They are poorly soluble in water due to a lipid chain (palmitoyl) bound to a conserved cysteine residue. Many cells, especially undifferentiated or inflammatory cells secrete Wnt proteins, and others also secrete them in response to stimuli. The effect of Wnt proteins depends on their concentration. They are transported over long distances and can act on cells distant from their production site. Wnt is activated by both paracrine and autocrine signaling.36 There are two types of Wnt signaling, but the canonic signaling pathway is more relevant to the present study.
The canonic Wnt signaling pathway (fig. 1) was well defined by McDonald, Tamai, and He.37 It functions by regulating the β-catenin transcriptional coactivator. In the absence of Wnt, plasma β-catenin is constantly degraded by action of the Axin complex, which is composed of the tumor suppressor protein adenomatous polyposis coli (APC), casein kinase 1 (CK1), and glycogen synthase kinase 3 (GSK3). CK1 and GSK3 sequentially phosphorylate the amino-terminal region of β-catenin, producing its recognition by β-Trcp, a subunit of ubiquitin E3 with ligase activity, and its consequent ubiquitination and degradation in the proteasome. This continuous elimination of β-catenin prevents its entry into the nucleus, and the target genes of the Wnt pathway are therefore repressed by the DNAbound T-cell factor/lymphoid enhancer factor (TCF/ LEF) family. The Wnt/β-catenin signaling pathway is activated when a ligand binds to the Frizzled (Fz) transmembrane receptor and its co-receptor, low density lipoprotein receptor-related protein 6 or 5 (LRP6/5). Formation of the Wnt-Fz-LRP6/5 complex along with recruitment of the Disheveled (Dvl) scaffolding protein results in LRP phosphorylation and activation and anchorage of the Axin complex to the receptors. This event permits inhibition of Axin complex-mediated β-catenin phosphorylation and thereby achieves the stabilization of β-catenin, which accumulates in the cytoplasm and can travel to the nucleus to form complexes with TCF/LEF and activate the target gene expression of Wnt,37 leading to the synthesis of GLP-1 and GIP in enteroendocrine L and K cells, respectively.
]]> In 2008, Anagnostou and Shepherd published a study in which they identified an important link between glucose detection and the Wnt/β-catenin signaling pathway,38 indicating that this pathway responds to physiological concentrations of nutrients. One of the first results obtained suggested that glucose regulates β-catenin levels. They used macrophage cell lines (J774.2 and RAW264.7), because these had previously shown a response to glucose. Anagnostou and Shepherd also observed that the β-catenin in these cells represents a cytosolic and nuclear pool of β-catenin that is involved in the transduction of Wnt signals and TCF dependent transcriptional activity. They maintained these cell lines during a period of glucose starvation and found that the restoration of glucose levels produced a rapid and dose-dependent increase in β-catenin in both cell lines. Changes in β-catenin were observed between 5 mM and 20 mM of glucose, indicating that this effect can be exerted within the physiological range of glucose concentrations.38
HEXOSAMINE PATHWAY
Anagnostou and Shepherd reported that the hexosamine pathway is involved in the regulation of β-catenin levels. Their first evidence was that the above-mentioned effects were blocked by drugs that inhibit glutamine: fructose- 6-phosphate amidotransferase (GFAT). GFAT is known to require glutamine as amino group donor, and the effect of glucose disappeared when glutamine was not present. Further proof of the influence of the hexosamine pathway was their finding that the effects of glucose could be replicated by administering low levels of glucosamine (GlcN), which can directly enter the hexosamine pathway downstream from GFAT.38
The hexosamine biosynthetic pathway (HBP) results in the production of UDP-N-acetyl glucosamine (UDPGlcNAc) and other hexosamine nucleotides. UDP-Glc- NAc is the main product and sole donor for O-binding of a single molecule of N-acetyl glucosamine (O-GlcNAc) to many cytoplasmic and nuclear proteins. Upon entry into the cell, the glucose is rapidly phosphorylated to glucose-6-phosphate, which can be oxidized via glycolysis, converted to pentose phosphates, or stored as glycogen. In glycolysis, G6P is isomerized to fructose- 6-phosphate (F6P) before activation of the pathway. Approximately 2.5% of the F6P, and consequently the glucose, is diverted to HBP. As noted above, GFAT catalyzes the formation of glucosamine-6-phosphate, with glutamine as amine donor and F6P as acceptor substrate in the first and rate-limiting step of the pathway. Subsequently, addition of an acetyl group produces N-acetylglucosamine- 6-phosphate, which is rapidly modified to UDP-GlcNAc. The utilization of glucosamine and acetyl in the first two steps potentially links the metabolism of amino acids and fatty acids to HBP.39
The reversible modification of the proteins by the addition and removal of GlcNAc (analogous to the addition and removal of phosphate) also shows a complex interaction with O-phosphate modification. The target sites of N-acetyl-O-glycosylation are the phosphorylation sites themselves or are very close to them. The GlcNAc cycle is completed thanks to two enzymes. One of these is O-GlcNAc transferase (OGT), which catalyzes the O-binding of GlcNAc to serine and threonine residues of the target protein. The other is O-GlcNAc hydrolase (OGA), which eliminates the hexosamine fraction.39
Because of the wide functionality of the target proteins, the hexosamine pathway is also known as the hexosamine signaling pathway. The final product, UDP-GlcNAc, is a potent inhibitor of GFAT and modulates the affinity of OGT for specific substrates.39
Anagnostou and Shepherd38 utilized the N-glycosylation inhibitors tunicamycin and 2-deoxy-D-glucose (2DOG) and demonstrated that this glycosylation is responsible for the effect of glucose on β-catenin, but the mechanism underlying this effect has not been elucidated. It was initially attributed to an increase in catenin synthesis, but this was ruled out by the finding that β-catenin mRNA levels were not affected by glucose or glucosamine or by the absence of glutamine; therefore, another explanation must be sought.39
RELATIONSHIP BETWEENWNT AND HEXOSAMINES
Phosphorylation of β-catenin by GSK3 is an essential step in its signaling for degradation in the proteasome, as commented above. It was first thought that low GSK3 activity levels may explain the increase in β-catenin, but this has been ruled out. However, the phosphorylated/total β-catenin ratio was found to decrease with glucose or glucosamine treatments, indicating a reduced GSK3-mediated phosphorylation and a consequent stabilization of β-catenin.38 One explanation, based on the results of the latter study and the physiolo gy of the hexosamine pathway, is depicted in figure 2. The effect of glucose on Wnt signaling is that N-acetyl-O-glycosylation of β-catenin hinders GSK3- mediated O-phosphorylation due to a simple structural cause, and this leads to a conformational change that allows it to evade the destruction complex and subsequent degradation in the proteasome, thereby increasing the concentration of β-catenin in the cytoplasm.
]]>
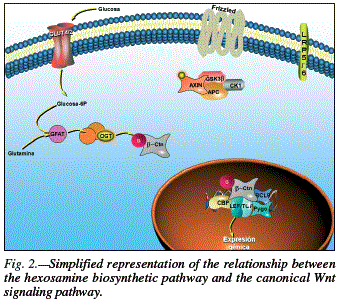
Besides its effects on β-catenin, glucose also induces autocrine activation of the Wnt canonical signaling pathway. Glucose may stimulate the secretion of Wnt proteins, given that the secretion of some Wnts is known to require N-glycosylation, and autocrine regulation of Wnt signaling has been observed. Another hypothesis is that glucose may regulate the N-glycosylation of Fz proteins or their co-receptors (LRP5/6). This may alter Wn signaling, because some receptors are functionally regulated by N-glycosylation. Thus, there is evidence that LRP6 is N-glycosylated, and this affects its localization and signaling capacity. In order to test whether, with either hypothesis, extracellular Wnt signaling was required for the effect of glucose on β-catenin, Anagnostou and Shepherd first utilized Dkk1, which blocks the interaction between Wnt and the Frizzed-LPR complex, and then utilized sFRP2, which also blocks Wnt signaling. The effect of glucose on β-catenin was not produced in either case; hence, the Wnt signaling pathway may be involved in this effect. The researchers next used a medium containing Wnt3a and tunicamycin (which blocks LRP6 phosphorylation) and found a glucose-induced increase in β- catenin due to an autocrine activation of the Wnt signaling system involving the hexosamine pathway and protein N-glycosylation.38
Gene expression regulation
The TCF/LEF family of DNA-bound transcription factors is the main companion of β-catenin in gene regulation. TCF represses the gene expression, interacting with the repressor Groucho (known as TLE1 in humans), which promotes histone deacetylation and chromatin compaction. The stabilization of β-catenin induced by Wnt and its accumulation in the nucleus leads TCF to form a complex with β-catenin, which displaces Groucho and binds to other gene coactivators. TCF proteins are high-mobility group (HMG) DNA-binding transcription factors that not only bind to a DNA consensus sequence designated the Wntresponsive element (WRE), CCTTTGWW (with W representing both T and A) but also produce a DNA flexion that can change the local chromatin structure. TCF7L2 acts as both repressor and activator.37
A large number of β-catenin-associated coactivators have been identified. This multiprotein complex includes BCL9 and Pygopus (Pygo), a mediator (to initiate the transcription), p300/CBP and histone-acetyltransferases (HATs) TRRAP/TIP60, histone-methyltransferases (HMTs) MLL1/2, the SWI/SNF family of ATPases for chromatin remodeling, and the PAF1 complex for elongation of histone transcription and modification. Structurally, β-catenin comprises three domains: the amino-terminal, central, and carboxylterminal domains. The central domain is formed by 12 armadillo-type repeats. Although the central repeats of β-catenin are associated with TCF and the amino-terminal repeat binds to BCL9, the majority of coactivator complexes interact with the carboxyl-terminal portion of β-catenin, creating interaction between β-catenin, transcriptional apparatus, and chromatin.37 The sequence from armadillo repeat 10 to the carboxyl- terminal end (COOH) of β-catenin interacts with and binds to the CPB (CREB binding protein) domain. CBP acts as coactivator of β-catenin and they activate transcription together.40 In fact, the binding of TCF/β-catenin to WREs leads to a CBP-dependent histone acetylation and a significant genomic distance (30 kb), which suggests that the local recruitment of TCF/β-catenin results in a generalized chromatin modification.37
A recent study also related the peroxisome proliferator activated receptor β/δ (PPARβ/δ) to the Wnt pathway, 31 in which PPARβ/δ was observed to transcriptionally regulate proglucagon expression in enteroendocrine L cells through stimulation of the β-catenin/TCF-4 pathway. The authors identified a role for PPARβ/δ as positive regulator of GLP-1 signaling through increases in the gene expression of proglucagon and the GLP-1 receptor in GLP-1-producing enteroendocrine L cells and pancreas, respectively [31].
However, the key question is whether the glucoseinduced increase in β-catenin has functional consequences for gene expression. This was addressed by Anagnostou and Shepherd38 in experiments that showed increased β-catenin in the nucleus of the cells under study. They also demonstrated that glucose increases the transcriptional activity of the β-catenin/ TCF system, given that a series of β-catenin target genes were expressed after its addition. These results clearly demonstrate that glucose, through the hexosamine pathway and N-glycosylation, increases levels of functional β- catenin, which accumulates in the nucleus and binds to members of the TCF/LEF family to activate transcription of the target genes,38 including GLP-1 and GIP genes in enteroendocrine L and K cells.
These results can also be obtained in vivo, and a rapid 2-to 3-fold increase in β-catenin levels has been reported in liver, muscle, and adipose tissue after refeeding food-deprived rats and in the liver of insulinopenic and hyperglycemic rats with streptozotocin- induced diabetes.38 This indicates that changes in β-catenin take place in tissues involved in the regulation of glucose metabolism under relevant physiological conditions, and that these effects are also attributable to changes in glucose in vivo, permitting the framing of hypotheses on the presence of this regulation in incretin hormone-producing enteroendocrine L and K cells.
]]> Conclusion
The Wnt signaling pathway is responsible for regulation the gene expression of different proteins involved in innumerable metabolic processes. Synthesis of proglucagon and consequently of GLP-1 and GIP is known to be controlled by the TCF/LEF repressor system, on which β-catenin acts, in enteroendocrine K and L cells. Evidence that glucose can exert direct effects on the Wnt/β-catenin signaling pathway establishes a link between nutrients and signaling networks, which are capable of coordinating and regulating different metabolic responses. In this way, understanding of the process can improve our knowledge of the metabolism of insulin and glucose homeostasis. Furthermore, other nutrients are likely to be involved in modulating the gene expression of incretin hormones, and they warrant specific study.
Perspectives
The aim of this study was to establish the bases for a possible research line to test hypotheses on enteroendocrine K and L cells in the human gut and the production of GIP and GLP-1 incretin hormones. These observations, alongside other available evidence, indicate that Wnt/β-catenin signaling is of therapeutic interest, especially in relation to obesity, diabetes, and other associated metabolic diseases. In fact, diabetic drug treatments using these hormones are now available. A greater understanding of incretin production processes and their modulation by nutrients and other dietary components can help to improve treatments or identify alternative options.
References
1. McIntyre N, Holdsworth CD, Turner DS. New interpretation of oral glucose tolerance. Lancet 1964; 2 (7349): 20-1. [ Links ]
2. McIntyre N, Holdsworth CD, Turner DS. Intestinal factors in the control of insulin secretion. The Journal of Clinical Endocrinology and Metabolism 1965; 25 (10): 1317-24. [ Links ]
3. Van der Burg MP, Guicherit OR, Frölich M, Gooszen HG. Insulinotropic effects of cholecystokinin, gastric inhibitory polypeptide and glucagon-like peptide-1 during perifusion of short-term cultured canine isolated islets. Regulatory Peptides 1995; 60 (1): 61-7. [ Links ]
4. Kelly KR, Brooks LM, Solomon TP, Kashyap SR, O'Leary VB, Kirwan JP. The glucose-dependent insulinotropic polypeptide and glucose-stimulated insulin response to exercise training and diet in obesity. American Journal of Physiology - Endocrinology and Metabolism 2009; 296 (6): E1269-74. [ Links ]
5. Rehfeld JF. Incretin physiology beyond glucagon-like peptide 1 and glucose-dependent insulinotropic polypeptide: cholecystokinin and gastrin peptides. Acta Physiologica (Oxford, England) 2011; 201 (4): 405-11. [ Links ]
6. Cui C, Ohnuma H, Daimon M, Susa S, Yamaguchi H, Kameda W, Jimbu Y, Oizumi T, Kato T. Ghrelin infused into the portal vein inhibits glucose-stimulated insulin secretion in Wistar rats. Peptides 2008; 29 (7): 1241-6. [ Links ]
7. Shuster LT, Go VL, Rizza RA, O´Brien PC, Service FJ. Potential incretins. Mayo Clinic Proceedings 1988; 63 (8): 794-800. [ Links ]
8. Arechavaleta R. El efecto fisiológico de las incretinas. Advanced Studies in Medicine 2006; 6 (7A): S581-S585. [ Links ]
9. Holst JJ, VilsbØll T, Deacon CF. The incretin system and its role in type 2 diabetes mellitus. Molecular and Cellular Endocrinology2009; 297 (1-2): 127-36. [ Links ]
10. Asmar M, Holst JJ. Glucagon-like peptide 1 and glucosedependentinsulinotropic polypeptide: new advances. Current Opinion in Endocrinology, Diabetes & Obesity 2010; 17 (1): 57-62. [ Links ]
11. Szalowska E, Meijer K, Kloosterhuis N, Razaee F, Priebe M, Vonk RJ. Sub-chronic administration of stable GIP analog in mice decreases serum LPL activity and body weight. Peptides 2011; 32 (5): 938-45. [ Links ]
12. Irwin N, Francis JM, Flatt PR. Alterations of glucose-dependent insulinotropic polypeptide (GIP) during cold acclimation. Regulatory Peptides 2011; 167 (1): 91-6. [ Links ]
13. Parker HE, Reimann F y Gribble FM. Molecular mechanisms underlying nutrient-stimulated incretin secretion. Expert Reviews in Molecular Medicine 2010; 12: e1. [ Links ]
14. Holst JJ. The Physiology of Glucagon-like Peptide 1. Physiological Reviews 2007; 87: 1409-1439. [ Links ]
15. Mortensen K, Christensen LL, Holst JJ, Orskov C. GLP-1 and GIP are colocalized in a subset of endocrine cells in the small intestine. Regulatory Peptides 2003; 114 (2-3): 189-96. [ Links ]
16. Fujita Y, Wideman RD, Speck M, Asadi A, King DS, Webber TD, Haneda M y Kieffer TJ. Incretin release from gut is acutely enhanced by sugar but not by sweeteners in vivo. American Journal of Physiology-Endocrinology and Metabolism 2009; 296: E473-E479. [ Links ]
17. Reimer RA. Meat hydrolysate and essential amino acidinduced glucagon-like peptide-1 secretion, in the human NCIH716 enteroendocrine cell line, is regulated by extracellular signal-regulated kinase1/2 and p38 mitogen-activated protein kinases. Journal of Endocrinology 2006; 191: 159-170. [ Links ]
18. Shimotoyodome A, Fukuoka D, Suzuki J, Fujii Y, Mizuno T, Meguro S, Tokimitsu I y Hase T. Coingestion of Acylglycerols Differentially Affects Glucose-Induced Insulin Secretion via Glucose-Dependent Insulinotropic Polypeptide in C57BL/6J Mice. Endocrinology 2009; 150 (5): 2118-26. [ Links ]
19. Schirra J, Katschinski M, Weidmann C, Schäfer T, Wank V, Arnold R y Göke B. Gastric emptying and release of incretin hormones after glucose ingestion in humans. Journal of Clinical Investigation 1996; 97 (1): 92-103. [ Links ]
20. Yoder SM, Yang Q, Kindel TL, Tso P. Stimulation of incretin secretion by dietary lipid: is it dose dependent? American Journal of Physiology-Gastrointestinal and Liver Physiology 2009; 297 (2): G299-305. [ Links ]
21. Karamanlis A, Chaikomin R, Doran S, Bellon M, Bartholomeusz FD, Wishart JM, Jones KL, Horowitz M, Rayner CK. Effects of protein on glycemic and incretin responses and gastric emptying after oral glucose in healthy subjects. The American Journal of Clinical Nutrition 2007; 86 (5): 1364-8. [ Links ]
22. Hirasawa A, Tsumaya K, Awaji T, Katsuma S, Adachi T, Yamada M, Sugimoto Y, Miyazaki S, Tsujimoto G. Free fatty acids regulate gut incretin glucagon-like peptide-1 secretion through GPR120. Nature Medicine 2005; 11 (1): 90-4. [ Links ]
23. Carr RD, Larsen MO, Winzell MS, Jelic K, Lindgren O, Deacon CF, Ahrén B. Incretin and islet hormonal responses to fat and protein ingestion in healthy men. American Journal of Physiology-Endocrinology and Metabolism 2008; 295 (4): E779-84. [ Links ]
24. Deacon CF, Nauck MA, Meier J, Hücking K, Holst JJ. Degradation of endogenous and exogenous gastric inhibitory polypeptide (GIP) in healthy and in type 2 diabetic subjects as revealed using a new assay for the intact peptide. The Journal of Clinical Endocrinology and Metabolism 2000; 85: 3575-3581. [ Links ]
25. Vilsboll T, Agerso H, Krarup T, Holst JJ. Similar elimination rates of glucagon-like peptide-1 in obese type 2 diabetic patients and healthy subjects. The Journal of Clinical Endocrinology and Metabolism 2003; 88: 220-224. [ Links ]
26. Wellendorph P, Johansen LD, Bräuner-Osborne H. The emerging role of promiscuous 7TM receptors as chemosensors for food intake. Vitamins and Hormones 2010; 84: 151-84. [ Links ]
27. Harada N. Structure and function of incretin receptor. Nippon Rinsho 2001; 69 (5): 813-20. [ Links ]
28. Yip RG, Wolfe MM. GIP biology and fat metabolism. Life Sciences 2000; 66 (2): 91-103. [ Links ]
29. Dhanvantari S, Izzo A, Jansen E, Brubaker PL. Coregulation of glucagon-like peptide-1 synthesis with proglucagon and prohormone convertase 1 gene expression in enteroendocrine GLUTag cells. Endocrinology 2001; 142 (1): 37-42. [ Links ]
30. Meier JJ y Nauck MA. Glucose-dependent insulinotropic polypeptide/gastric inhibitory polypeptide. Best Practice & Research Clinical Endocrinology & Metabolism 2004; 18 (4): 587-606. [ Links ]
31. Daoudi M, Hennuyer N, Borland MG, Touche V, Duhem C, Gross B, Caiazzo R, Kerr-Conte J, Pattou F, Peters JM, Staels B, Lestavel S. PPAR beta /d Activation Induces Enteroendocrine L Cell GLP-1 Production. Gastroenterology 2011. [Epub ahead of print] [ Links ]
32. Jin T y Liu L. The Wnt Signaling Pathway Effector TCF7L2 and Type 2 Diabetes Mellitus. Molecular Endocrinology 2008; 22 (11): 2383-92. [ Links ]
33. Jin T. The WNT signalling pathway and diabetes mellitus. Diabetologia2008; 51 (10): 1771-80. [ Links ]
34. García-Martínez JM, Chocarro-Calvo A, Moya CM, García-Jiménez C. WNT/beta-catenin increases the production of incretins by entero-endocrine cells. Diabetologia 2009; 52 (9):1913-24. [ Links ]
35. Yi F, Brubaker PL, Jin T. TCF-4 mediates cell type-specific regulation of proglucagon gene expression by beta-catenin and glycogen synthase kinase-3beta. The Journal of Biological Chemistry 2005; 280 (2): 1457-64. [ Links ]
36. Gustafson B y Smith U. WNT signalling is both an inducer and effector of glucagon-like peptide-1. Diabetologia 2008; 51:768-1770. [ Links ]
37. MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Developmental Cell 2009; 17 (1): 9-26. [ Links ]
38. Anagnostou SH, Shepherd PR. Glucose induces an autocrine activation of the Wnt/beta-catenin pathway in macrophage cell lines. Biochemical Journal 2008; 416 (2): 211-8. [ Links ]
39. www.rgd.mcw.edu/wg/pathway/hexosamine_biosynthetic_pathway [ Links ]
40. Takemaru KI, Moon RT. The transcriptional coactivator CBP interacts with beta-catenin to activate gene expression. The Journal of Cell Biology 2000; 149 (2): 249-54. [ Links ]
![]() Correspondence:
Correspondence:
Ángel Gil. ]]>
Departamento de Bioquímica y Biología Molecular II.
Instituto de Nutrición y Tecnología de los Alimentos.
Centro de Investigación Biomédica.
Universidad de Granada. Campus de la Salud.
Avda. del Conocimiento, s/n.
18100 Armilla. Granada. España.
E-mail: agil@ugr.es
Recibido: 3-VIII-2011.
Aceptado: 8-VIII-2011.