 ]]>
]]>

Effects unrelated to anti-inflammation of lipid emulsions containing fish oil in parenteral nutrition for adult patients
Efectos no relacionados con la antiinflamación de las emulsiones lipídicas que contienen aceite de pescado en la nutrición parenteral para pacientes adultos
Javier Mateu-de-Antonio1 and Marta Florit-Sureda2
1Pharmacy Department. Hospital del Mar. Barcelona, Spain.
2Pharmacy Department. Corporació Sanitària Parc Taulí. Sabadell, Barcelona. Spain
ABSTRACT
Several reviews and meta-analyses on modulated inflammatory and immunologic responses after the administration of omega-3 polyunsaturated fatty acids (PUFAs) in different diseases and conditions have been published. However, omega-3 PUFAs exert several other actions which are not directly related to immunologic or inflammatory responses. The aim of this paper was to review the effects which are not directly related to immunologic and inflammatory responses of intravenous lipid emulsions (IVLEs) containing fish oil (FO) in parenteral nutrition (PN) for adult patients. IVLEs containing FO could have a role in the prevention of alterations in liver enzyme tests (LETs) or PN-associated liver disease (PNALD). Studies using FO doses of ≥ 0.150 mg/kg/day or IVLEs with high FO concentration reported more positive results than those with lower doses. Once PNALD was developed, the use of IVLEs exclusively composed of FO at doses of 0.25-1 g of FO/kg/day for several weeks could attenuate or even eradicate cholestasis and liver alteration. IVLEs containing FO seemed to have faster blood clearance, and this could be beneficial for some patients. Some studies also suggested a possible improvement of respiratory function by the administration of these IVLEs. In general, IVLEs containing FO were safe. Their use did not increase oxidative stress but, in contrast, increased plasma tocopherol content. They did not alter insulin sensitivity or glycemic control, and studies have found no relevant clinical effect on platelet aggregation or hemostasis. In conclusion, the use of IVLEs containing FO in PN may be beneficial with regard to older IVLEs, in addition to the modulation of systemic inflammation response.
Key words: Fish oils. Intravenous fat emulsions. Parenteral nutrition. Omega-3 fatty acids.
RESUMEN
Se han publicado varias revisiones y metaanálisis sobre la modulación de las respuestas inflamatorias e inmunológicas por la administración de ácidos grasos poliinsaturados (AGPI) omega-3. Sin embargo, los AGPI omega-3 ejercen otras acciones no directamente relacionadas con estas respuestas. El objetivo de este trabajo es revisar los efectos de las emulsiones lipídicas intravenosas (ELIV) que contienen aceite de pescado (AP) en la nutrición parenteral (NP) de pacientes adultos. Estas emulsiones pueden tener un papel importante en la prevención de las alteraciones del perfil hepático o de la enfermedad hepática asociada a la NP (EHANP) en comparación con las ELIV sin AP. Los estudios que usaron dosis ≥ 0,150 mg/kg/día presentaron resultados más positivos que aquellos con dosis menores. Una vez se ha presentado la EHANP, el uso de una ELIV compuesta exclusivamente de AP, a dosis de 0,25-1 g de AP/kg/día durante varias semanas, podría atenuar o incluso revertir la alteración hepática y la colestasis cuando la administración de la NP es crónica. Las ELIV con AP parecen tener un aclaramiento plasmático más rápido, lo que podría suponer una ventaja en ciertos pacientes. Algunos estudios también sugieren que estas emulsiones podrían mejorar la función respiratoria. En general, estas ELIV que contienen AP son muy seguras, no aumentan el estrés oxidativo e incluso incrementan el nivel de alfa-tocoferol plasmático. Asimismo, no alteran la sensibilidad a la insulina o el control glicémico y tampoco se ha demostrado que afecten clínicamente a la agregación plaquetaria o a la hemostasia. En conclusión, el uso de ELIV con AP en la NP podría suponer algunas ventajas frente a la ELIV sin AP, además de la modulación de la respuesta inflamatoria sistémica.
Palabras clave: Aceites de pescado. Emulsiones lipídicas intravenosas. Nutrición parenteral. Ácidos grasos omega-3.
Introduction
]]> In adult patients, the administration of omega-3 polyunsaturated fatty acids (PUFAs) from fish oil (FO) has been recommended to reduce inflammatory response. Several studies and reviews have been published on this topic in the last years. In a recent meta-analysis on patients with systemic inflammatory response syndrome, the addition of omega-3 PUFAs was associated with lower mortality and might have shortened the hospital length of stay (LOS). No differences between parenteral and enteral routes of administration regarding outcomes were found (1). In critically ill patients, the use of FO did not reduce mortality, but, on the contrary, it significantly diminished infectious complications and showed a tendency to reduce the days on mechanical ventilation and the LOS. Authors did not find differences in any endpoint amongst trials of parenteral and enteral nutritional strategies either (2). In pancreatitis, the use of omega-3 PUFAs has been shown to reduce mortality, infectious complications and LOS, especially when used parenterally (3).These effects have been related to the immunomodulatory and anti-inflammatory action of omega-3 PUFAs. Their use, especially by parenteral route, diminishes several inflammatory biomarkers in both acutely and chronically ill patients (4). They also reduce interleukin-6, increase albumin and, additionally, decrease the C-reactive protein to albumin ratio in colorectal cancer patients (5).
However, omega-3 PUFAs from FO exert several other actions which are not directly related to immunologic and inflammatory responses. To our knowledge, these effects have not been reviewed in sum.
The aim of this paper was to review the effects which are not directly related to immunologic and inflammatory responses of intravenous lipid emulsions (IVLEs) containing FO as part of parenteral nutrition (PN) in adult patients. We divided these effects in potential benefits and possible concerns.
Potential benefits
PREVENTION OF ALTERATIONS IN LIVER ENZYME TESTS (LETS) OR PARENTERAL NUTRITION-ASSOCIATED LIVER DISEASE (PNALD)
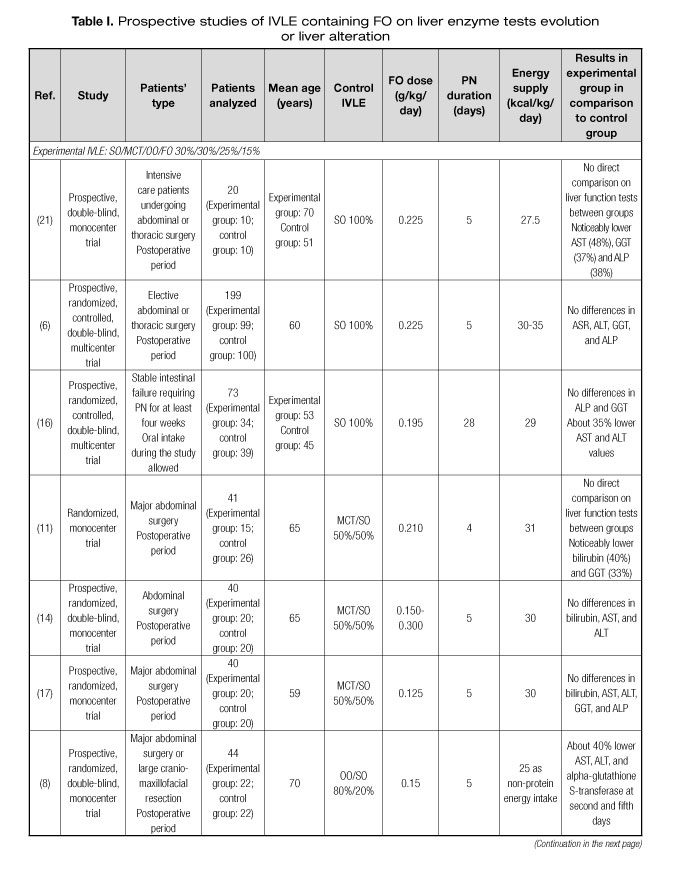
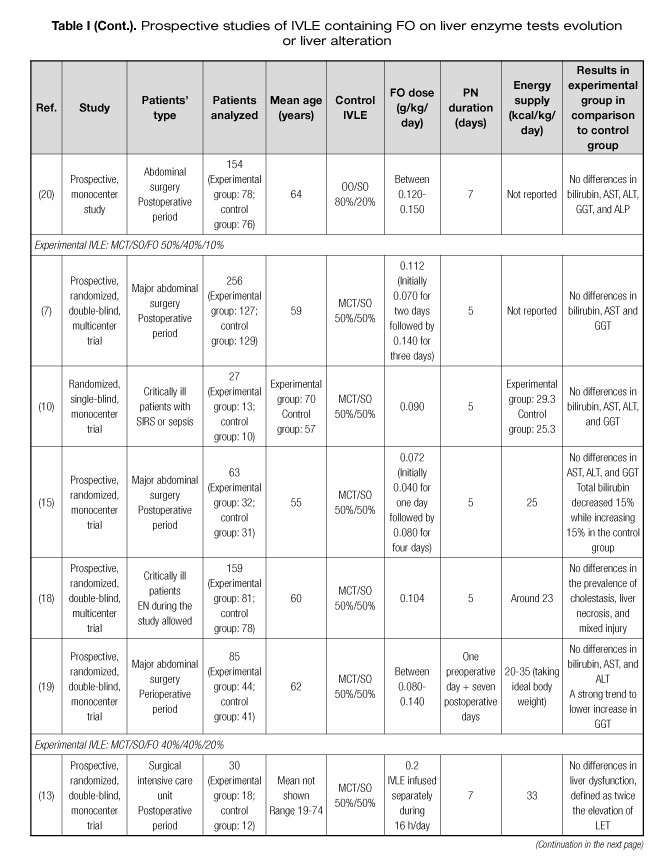
Several prospective studies have been published assessing the preventive effect of IVLEs containing FO on alterations in LETs or PNALD (6-21). Characteristics of these studies are shown in table I. Most of the studies were focused on middle-aged surgical or critically ill patients. Seven (44%) of the 16 studies found some positive results for LETs or liver ultrasound tests in patients receiving IVLEs containing FO (8,11,12,15,16,19,21). Studies using FO in doses of ≥ 0.150 mg/kg/day presented more positive results than those with lower doses. In five (63%) of the eight articles using doses ≥ 0.150 mg/kg/day some positive results in LETs were observed, whereas only two (25%) of the eight studies using lower doses reported some positive results. This hypothesis could be reinforced by two recent retrospective studies. In a large retrospective study including more than 1,500 patients receiving PN, a higher FO dose was associated with a greater decrease in gamma-glutamyl transferase (GGT) and alkaline phosphatase (ALP) in a multivariate adjusted model (22). Additionally, in a propensity score-matched study, a higher percentage of FO administered by PN was associated with lower LET elevation during PN (23).
In relation to the IVLE used, the most studied one is a multicomponent oil emulsion containing soybean oil (SO) 30%, middle-chain triglycerides (MCT) 30%, olive oil (OO) 25%, and FO 15%. Four (50%) of the eight studies using this IVLE resulted in positive outcomes (8,11,16,21). The second most studied IVLE is an emulsion containing SO 40%, MCT 50%, and FO 10%. Two (40%) of the five studies with this IVLE rendered some positive results (15,19). Only one study tested an IVLE containing exclusively FO 100% administered in high doses, which lead to improved liver ultrasound test and better lactate dehydrogenase (LDH) in septic patients (12). Two additional studies tested a combination of an IVLE exclusively composed of FO 100% and an already marketed IVLE without FO. No differences in liver parameters were found (9,13).
These studies have several limitations regarding clinical practice. The first limitation is the length of the study period. PN lasted from four to eight days in most trials. This lapse could be shorter than the usual PN course in clinical settings. Only one study had a four-week duration, resulting in an improved LET using a multicomponent oil emulsion containing FO 15% in doses of > 0.150 g/kg/day (16). Another important limitation is the age of the patients under assessment, who were younger than many patients requiring PN nowadays, usually older than 70.
In summary, IVLEs with FO in doses of FO > 0.150 g/kg/day or with high proportion of FO could reduce the incidence of alterations in LETs or PNALD.
TREATMENT OF PNALD
Treatment with IVLEs containing FO has not been thoroughly studied in adults after the development of PNALD. The first case was described by Jurewitsch et al. (24). A 75-year-old female required PN after a massive intestinal resection that led to short bowel syndrome. She developed hepatomegaly, altered LET, and an abnormal liver biopsy after ten weeks of PN containing an IVLE of SO 100%. After replacing the initial IVLE by an IVLE of FO 100%, in doses of 0.25 g FO/kg/day, liver span decreased, LET dropped, and liver biopsy normalized in approximately 20 weeks. To our knowledge, two additional cases have been described so far in patients requiring chronic PN (25,26). They developed severe liver alterations that could not be reversed except by replacing the initial IVLE (SO 100% and OO/SO 80%/20%, respectively) with an IVLE of FO 100%. The doses of FO used were about 1g/kg/day.
Three studies focused on patients presenting LET alteration and PNALD. The first one is a retrospective study in 54 patients receiving long term PN that assessed the effect of adding taurine to PN on LET (27).
Authors concluded that a synergistic effect could exist between taurine and a multicomponent IVLE of SO/MCT/OO/FO 30%/30%/25%/15% by reducing an altered LET. However, this effect was not accurately analyzed.
]]> The second was an open-label study on 15 adults who developed cholestasis while receiving PN with IVLE of SO 100% (28). This treatment was partially replaced by an IVLE of FO 100% at doses of 0.15-0.2 g of FO/kg/day for at least one month. In this period, bilirubin and ALT dropped and liver biopsies showed a marked decrease in the degree of cholestasis and inflammation.The last study focused on the effect of different IVLEs in ten adults with PNALD secondary to long-term PN. Additionally, an in vitro study assessed the effect of these IVLEs on human hepatocytes (29). The use of an IVLE of FO 100% at 1 g/kg/day for four months restored LET, fibrotic and inflammatory markers to normal values. This FO 100% emulsion showed not only a potent in vitro anti-inflammatory effect but also a possible direct antifibrotic effect in the liver.
As a concern, it has been hypothesized that the administration of IVLE with FO 100% as an exclusive source of fat does not provide the required essential fatty acid amount when used for long periods (29). However, it has not been possible to prove this point clinically (25).
The substitution of the initial IVLE by one exclusively composed by FO or with high amounts of it and administered at doses of 0.25-1 g of FO/kg/day during several weeks could attenuate or even revert LET alterations or PNALD in patients under chronic PN.
PLASMA LIPID CLEARANCE
Prospective studies collecting data on plasma clearance of IVLEs containing FO are presented in table II (6-8,12-14,16,17,19-21,30-34).
Three studies on healthy subjects assessed lipid oxidation or lipid plasma clearance specifically (31-33). These studies resulted in a 15-25% faster clearance or 25-45% shorter half-life of the experimental IVLE containing FO, although emulsions under assessment had different composition and FO content.
Thirteen clinical studies assessed clearance of IVLE with FO in hospitalized patients. Six of them (46%) reported higher clearance in the FO group as well (8,12,13,17,19,21).
Regarding the rest of clinical studies, six (46%) resulted in similar clearance to control emulsions (6,14,16,20,30,34), and only in one (8%) of them (7) triglyceridemia increased in the experimental group.
The most studied control IVLE was composed by MCT/SO 50%/50%. Seven (88%) of the eight studies which used this control resulted in faster lipid clearance of the IVLE containing FO. The remaining eight studies used control emulsions of SO 100% or OO/SO 80%/20%, three (38%) of them resulting in faster clearance for the IVLE containing FO.
]]> A multicomponent IVLE containing SO 30%, MCT 30%, OO 25% and FO 15% has been recently used as a substitutive emulsion in patients who presented moderate hypertriglyceridemia (250-400 mg/dL) during PN containing an IVLE of OO/SO 80%/20%. This substitution resulted in a reduction in triglyceridemia of about 70 mg/dL (about 25%) and allowed maintaining the caloric intake, thus improving nutrition parameters without affecting the hepatic profile (35).Taking all studies together, five (38%) out of 13 clinical studies and the three experimental studies resulted in faster clearance or shorter half-life in the IVLEs containing FO. The inclusion of FO in IVLEs could be beneficial to some patients as it improves lipid clearance.
RESPIRATORY GAS INTERCHANGE
Few trials have investigated the effect of IVLEs with FO in gas exchange and the oxygenation index (ratio PaO2/FiO2). In two studies respiratory function was improved, and a tendency to a shorter LOS during five days of PN in the group receiving an IVLE of MCT/SO/FO 50%/40%/10% in comparison to those being administered an IVLE of SO 100% (10,36) was observed. Doses of FO were 0.09 to 0.2 g/kg/day. Two more studies analyzed respiratory parameters in cardiac surgery patients. In one of them, 40 patients subject to elective coronary artery bypass grafting were randomized to receive a preoperative IVLE of FO 100% at doses of 10 g (about 0.11 g/kg) or saline. The results of this study showed that the extraction of oxygen and the uptake of lactate were markedly increased in the FO pretreated patients compared to the control group (37). In the second study, 23 patients were randomized to receive FO 100% 0.2 g/kg or saline administered two times on the evening before cardiopulmonary bypass and immediately before surgery. The FO group presented shorter ventilation time but no significant impact on the oxygenation index was observed (38).
On the other hand, no differences in relation to gas exchange parameters were found in a study on 16 patients with acute respiratory distress syndrome who were randomized in two different groups receiving IVLE containing either SO 100% or MCT/SO/FO 50%/40%/10%, administered at a dose of 0.12 g/kg/hour during 12 hours. The lack of beneficial results was attributed to the short duration of the treatment (39).
The mechanism by which FO could improve respiratory function was not clear (10).
OTHER EFFECTS
In a randomized controlled clinical trial, patients who had undergone orthotopic liver transplantation and received PN with an IVLE of SO/MCT/FO 40%/40%20%, at doses of FO 0.2 g/kg/day for seven days, presented better alanine aminotransferase (ALAT) profile and prognostic nutritional index, more reduced hepatic cell injury and higher prealbumin than patients receiving a similar PN without FO. In addition, the FO group had a decreased incidence of infectious complications and their post-transplant hospital stay was shorter (40). It has been hypothesized that IVLEs containing high amounts of FO are of benefit to patients who have undergone kidney transplantation and may require PN after severe complications (41).
In a single-arm phase II trial on patients with advanced pancreatic cancer, an IVLE containing FO (MCT/SO/FO 50%/40%/10%) was used to augment chemotherapy activity and improve quality of life. It was administered weekly as a standard intravenous infusion of 500 mL (FO 10 g) after the antitumor agent gemcitabine 1,000 mg/m2 for three weeks, followed by a rest week. The study showed improved activity of the antitumor agent and improvement of the quality of life (42).
]]> Possible concerns
OXIDATIVE STRESS
FO contains a high percentage of long chain omega-3 PUFAs, such as eicosapentaenoic acid and docosahexaenoic acid, susceptible to oxidation that could increase oxidative stress. To counteract peroxidation reactions, IVLEs containing FO are enriched with higher doses of alpha-tocopherol, a lipophilic antioxidant (43).
Several studies have demonstrated that IVLEs partly or totally composed of FO increase plasma levels of alpha-tocopherol in comparison to IVLEs with SO 100% due to their high content of this vitamin (7,16,21,44,45). Doses used in these studies ranged from 0.7 to 2 g of fat/kg/day. Comparisons with other IVLEs without FO have not been made.
In addition, some studies assessed the effect of IVFEs with FO on oxygen radical production, peroxidation capacity or antioxidant capacity. No differences have been detected in oxidative status using IVLEs with FO in comparison to control IVLEs, mostly SO 100% (17,44-46). However, these studies used different techniques and oxidation markers, and had small samples and short time lipid administration. Thus, the use of IVLEs with high PUFA content did not seem to increase oxidative stress.
INSULIN SENSITIVITY AND GLUCOSE METABOLISM
Elevated plasma free fatty acid levels are known to contribute to peripheral insulin resistance by impairing insulin-receptor signaling, which leads to decreased cellular glucose uptake and serum hyperglycemia, and can also reduce insulin secretion in subjects with impaired glucose tolerance (47). So far, three studies have examined glycemic control as a primary objective when administering IVLEs with FO. In the first study, 24 patients were randomized to receive an IVLE containing either FO 100% or SO 100%, in doses of 0.25 g of fat/kg/day, as part of PN for four days. No differences were detected in glucose metabolism in patients receiving FO (30). In the second study, 44 patients were randomized to receive PN containing an IVLE with SO 100% or an IVLE with SO/FO 80%/20% at doses of 1 g fat/kg/day for five days. Blood glucose levels normalized to the insulin doses applied and did not differ significantly between both groups. However, blood glucose levels fell significantly over time in the SO group, whereas they remained high in the SO/FO group (34). In the third study, 11 subjects with type 2 diabetes mellitus received infusions of SO 100% or SO/FO 89%/11% randomly by four-hour isoglycemic hyperinsulinemic clamps. Both IVLEs similarly decreased insulin-mediated glucose utilization. The IVLE with FO increased plasma free fatty acids. However, no difference regarding glucose utilization, insulin secretion and total energy production was observed between groups (46).
Two additional studies had glycemic control as a secondary outcome. In a randomized trial with 44 patients comparing a multicomponent emulsion of SO/MCT/OO/FO 30%/30%/25%/15% (FO dose 0.15 g/kg/day) versus OO/SO 80%/20% for five days, glucose levels and insulin dosage did not differ significantly between groups (8). Another study assessed 23 patients who were randomized to receive FO 100% 0.2 g/kg or saline administered twice on the evening before cardiopulmonary bypass and immediately before surgery. The average glycemia during the first 24 hours after surgery was significantly lower in the FO group. However, insulin requirements and endogenous glucose production were similar in both groups in the first 24 hours (38).
In a retrospective study, the substitution of an IVLE of SO/OO 80%/20% by a multicomponent IVLE containing SO/MCT/OO/FO 30%/30%/25%/15% in patients presenting moderate hypertriglyceridemia during PN resulted in a slight increase of insulin requirements, but it was attributed to the previous hypertriglyceridemia (35).
IVLEs with FO seemed not to alter significantly insulin sensitivity or glycemic control in clinical practice.
]]> HEMOSTASISOmega-3 PUFAs contained in FO have an antithrombotic effect mainly mediated by inhibiting cyclooxygenase, which transforms arachidonic acid into thromboxane A2, a platelet activator, and reducing its formation, thus leading to decreased platelet aggregation. Early studies raised concern about the safety of high doses of these lipids due to an increased risk of bleeding (48) that could not be proved later. However, this concern still persists nowadays.
Several studies assessed hemostasis parameters in patients receiving IVLEs with FO. In an early study mainly focused in hemostasis, 44 patients were randomly administered IVLEs with SO 100% or SO/FO 80%/20% in a dose of 1 g of fat/kg/day (FO 0.2 g/kg/day) for five days. However, no differences were found between groups in relation to thromboplastin time, activated partial thromboplastin time, fibrinogen, antithrombin III, factors VIIa and XIIa, and platelet function (49). In a more recent study already mentioned above, 40 patients undergoing coronary artery bypass graft surgery were randomized to receive either FO IVLE as single doses of FO 10 g (about 0.11 g/kg) 100% or saline solution before surgery and again four hours before starting extracorporeal circulation. The results showed no differences in international normalized ratio (INR), activated partial thromboplastin time, bleeding volume, blood transfusion or fresh frozen plasma requirements, platelet count in postoperative period, and heparin or protamine use. Only in one of four tests platelets activity was statistically lower in the FO group, but with no negative effect (50).
Three additional studies evaluated certain hemostasis parameters, but lacking a detailed analysis. They found no differences in INR (16), partial thrombin time (10), and prothrombin time (7). Two of them showed no differences in platelet counts (10,16), but a moderate difference regarding platelet count recovery after surgery was found in the FO group in the earliest study (7). FO doses ranged from 0.09 to 0.195 g/kg/day.
In general, studies have found no relevant clinical effect on platelet aggregation or hemostasis when administering IVLE with FO.
Conclusions
The use of IVLEs containing FO in PN may have some advantages with respect to earlier IVLEs, not only in terms of modulation of systemic inflammation but in relation to further benefits. There are some evidences that IVLEs with FO may reduce the incidence of LET alterations during PN, eradicate PNALD, increase plasma lipid clearance, and improve respiratory gas exchange. Their use is safe, and possible concerns as alterations in oxidative stress, insulin sensitivity or hemostasis have not been confirmed so far.
References
]]>1. Wan X, Gao X, Bi J, Tian F, Wang X. Use of n-3 PUFAs can decrease the mortality in patients with systemic inflammatory response syndrome: A systematic review and meta-analysis. Lipids Health Dis 2015;14:23. [ Links ]
2. Manzanares W, Langlois PL, Dhaliwal R, Lemieux M, Heyland DK. Intravenous fish oil lipid emulsions in critically ill patients: An updated systematic review and meta-analysis. Crit Care 2015;19:167. [ Links ]
3. Lei QC, Wang XY, Xia XF, Zheng HZ, Bi JC, Tian F, et al. The role of omega-3 fatty acids in acute pancreatitis: A meta-analysis of randomized controlled trials. Nutrients 2015;7(4):2261-73. [ Links ]
4. Rangel-Huerta OD, Aguilera CM, Mesa MD, Gil A. Omega-3 long-chain polyunsaturated fatty acids supplementation on inflammatory biomakers: A systematic review of randomised clinical trials. Br J Nutr 2012;107(Suppl 2):S159-70. [ Links ]
5. Mocellin MC, Camargo CQ, Nunes EA, Fiates GM, Trindade EB. A systematic review and meta-analysis of the n-3 polyunsaturated fatty acids effects on inflammatory markers in colorectal cancer. Clin Nutr 2016;35(2):359-69. [ Links ]
]]>6. Mertes N, Grimm H, Furst P, Stehle P. Safety and efficacy of a new parenteral lipid emulsion (SMOFlipid) in surgical patients: A randomized, double-blind, multicenter study. Ann Nutr Metab 2006;50(3):253-9. [ Links ]
7. Wichmann MW, Thul P, Czarnetzki HD, Morlion BJ, Kemen M, Jauch KW. Evaluation of clinical safety and beneficial effects of a fish oil containing lipid emulsion (Lipoplus, MLF541): Data from a prospective, randomized, multicenter trial. Crit Care Med 2007;35(3):700-6. [ Links ]
8. Piper SN, Schade I, Beschmann RB, Maleck WH, Boldt J, Rohm KD. Hepatocellular integrity after parenteral nutrition: Comparison of a fish-oil-containing lipid emulsion with an olive-soybean oil-based lipid emulsion. Eur J Anaesthesiol 2009;26(12):1076-82. [ Links ]
9. Badía-Tahull MB, Llop-Talaverón JM, Leiva-Badosa E, Biondo S, Farran-Teixidó L, Ramón-Torrell JM, et al. A randomised study on the clinical progress of high-risk elective major gastrointestinal surgery patients treated with olive oil-based parenteral nutrition with or without a fish oil supplement. Br J Nutr 2010;104(5):737-41. [ Links ]
10. Barbosa VM, Miles EA, Calhau C, Lafuente E, Calder PC. Effects of a fish oil containing lipid emulsion on plasma phospholipid fatty acids, inflammatory markers, and clinical outcomes in septic patients: A randomized, controlled clinical trial. Crit Care 2010;14(1):R5. [ Links ]
]]>11. Hallay J, Olah AV, Fulesdi B, Kocsor M, Vegh T, Kovacs G, et al. Hepatobiliary response in postoperative lipid therapy in gastrointestinal surgery. Hepatogastroenterology 2010;57(102-3):1069-73. [ Links ]
12. Sungurtekin H, Degirmenci S, Sungurtekin U, Oguz BE, Sabir N, Kaptanoglu B. Comparison of the effects of different intravenous fat emulsions in patients with systemic inflammatory response syndrome and sepsis. Nutr Clin Pract 2011;26(6):665-71. [ Links ]
13. Han YY, Lai SL, Ko WJ, Chou CH, Lai HS. Effects of fish oil on inflammatory modulation in surgical intensive care unit patients. Nutr Clin Pract 2012;27(1):91-8. [ Links ]
14. Ma CJ, Sun LC, Chen FM, Lu CY, Shih YL, Tsai HL, et al. A double-blind randomized study comparing the efficacy and safety of a composite vs a conventional intravenous fat emulsion in postsurgical gastrointestinal tumor patients. Nutr Clin Pract 2012;27(3):410-5. [ Links ]
15. Wang J, Yu JC, Kang WM, Ma ZQ. Superiority of a fish oil-enriched emulsion to medium-chain triacylglycerols/long-chain triacylglycerols in gastrointestinal surgery patients: A randomized clinical trial. Nutrition 2012;28(6):623-9. [ Links ]
]]>16. Klek S, Chambrier C, Singer P, Rubin M, Bowling T, Staun M, et al. Four-week parenteral nutrition using a third generation lipid emulsion (SMOFlipid): A double-blind, randomised, multicentre study in adults. Clin Nutr 2013;32(2):224-31. [ Links ]
17. Wu MH, Wang MY, Yang CY, Kuo ML, Lin MT. Randomized clinical trial of new intravenous lipid (SMOFlipid 20%) versus medium-chain triglycerides/long-chain triglycerides in adult patients undergoing gastrointestinal surgery. J Parenter Enteral Nutr 2014;38(7):800-8. [ Links ]
18. Grau-Carmona T, Bonet-Saris A, García-de-Lorenzo A, Sánchez-Álvarez C, Rodríguez-Pozo A, Acosta-Escribano J, et al. Influence of n-3 polyunsaturated fatty acids enriched lipid emulsions on nosocomial infections and clinical outcomes in critically ill patients: ICU lipids study. Crit Care Med 2015;43(1):31-9. [ Links ]
19. Ma CJ, Wu JM, Tsai HL, Huang CW, Lu CY, Sun LC, et al. Prospective double-blind randomized study on the efficacy and safety of an n-3 fatty acid enriched intravenous fat emulsion in postsurgical gastric and colorectal cancer patients. Nutr J 2015;14:9. [ Links ]
20. Martínez-Lozano F, Palacio P, Serrano JM, Caballero C, Gómez MJ, Sánchez C. Comparative study on the efficacy and safety of parenteral nutrition formulated with two different fat emulsions: SMOFlipid® compared with Clinoleic®. Preliminary study. Nutr Hosp 2016;33(3):515-21. [ Links ]
]]>21. Antebi H, Mansoor O, Ferrier C, Tetegan M, Morvan C, Rangaraj J, et al. Liver function and plasma antioxidant status in intensive care unit patients requiring total parenteral nutrition: Comparison of 2 fat emulsions. J Parenter Enteral Nutr 2004;28(3):142-8. [ Links ]
22. Badía-Tahull MB, Llop-Talaverón J, Leiva-Badosa E. Impact of intravenous lipid emulsions on liver function tests: Contribution of parenteral fish oil. Nutrition 2015;31(9):1109-16. [ Links ]
23. Llop-Talaverón JM, Badía-Tahull MB, Leiva-Badosa E, Ramón-Torrel JM. Parenteral fish oil and liver function tests in hospitalized adult patients receiving parenteral nutrition: A propensity score-matched analysis. Clin Nutr 2016;5. [ Links ]
24. Jurewitsch B, Gardiner G, Naccarato M, Jeejeebhoy KN. Omega-3-enriched lipid emulsion for liver salvage in parenteral nutrition-induced cholestasis in the adult patient. J Parenter Enteral Nutr 2011;35(3):386-90. [ Links ]
25. Burns DL, Gill BM. Reversal of parenteral nutrition-associated liver disease with a fish oil-based lipid emulsion (Omegaven) in an adult dependent on home parenteral nutrition. J Parenter Enteral Nutr 2013;37(2):274-80. [ Links ]
]]>26. Venecourt-Jackson E, Hill SJ, Walmsley RS. Successful treatment of parenteral nutrition-associated liver disease in an adult by use of a fish oil-based lipid source. Nutrition 2013;29(1):356-8. [ Links ]
27. González-Contreras J, Villalobos Gámez JL, Gómez-Sánchez AI, García-Almeida JM, Enguix Armanda A, Rius Díaz F, et al. Cholestasis induced by total parenteral nutrition: Effects of the addition of taurine (Tauramin®) on hepatic function parameters; possible synergistic action of structured lipids (SMOFlipid®). Nutr Hosp 2012;27(6):1900-7. [ Links ]
28. Xu Z, Li Y, Wang J, Wu B, Li J. Effect of omega-3 polyunsaturated fatty acids to reverse biopsy-proven parenteral nutrition-associated liver disease in adults. Clin Nutr 2012;31(2):217-23. [ Links ]
29. Pastor-Clerigues A, Marti-Bonmati E, Milara J, Almudever P, Cortijo J. Anti-inflammatory and anti-fibrotic profile of fish oil emulsions used in parenteral nutrition-associated liver disease. PLoS One 2014;9(12):e115404. [ Links ]
30. Tappy L, Berger MM, Schwarz JM, Schneiter P, Kim S, Revelly JP, et al. Metabolic effects of parenteral nutrition enriched with n-3 polyunsaturated fatty acids in critically ill patients. Clin Nutr 2006;25(4):588-95. [ Links ]
]]>31. Simoens CM, Deckelbaum RJ, Massaut JJ, Carpentier YA. Inclusion of 10% fish oil in mixed medium-chain triacylglycerol-long-chain triacylglycerol emulsions increases plasma triacylglycerol clearance and induces rapid eicosapentaenoic acid (20:5n-3) incorporation into blood cell phospholipids. Am J Clin Nutr 2008;88(2):282-8. [ Links ]
32. Carpentier YA, Hacquebard M, Portois L, Dupont IE, Deckelbaum RJ, Malaisse WJ. Rapid cellular enrichment of eicosapentaenoate after a single intravenous injection of a novel medium-chain triacylglycerol:fish-oil emulsion in humans. Am J Clin Nutr 2010;91(4):875-82. [ Links ]
33. Schlotzer E, Kanning U. Elimination and tolerance of a new parenteral lipid emulsion (SMOF): A double-blind cross-over study in healthy male volunteers. Ann Nutr Metab 2004;48(4):263-8. [ Links ]
34. Wendel M, Rössel T, Bergmann S, Otto S, Ragaller M, Zimmermann T, et al. Impact of total parenteral nutrition including omega-3 fatty acids on the regulation of plasma lipoproteins and glycemic control after major abdominal surgery. e-SPEN 2007;2(5):e103-e10. [ Links ]
35. Mateu-de Antonio J, Florit-Sureda M. New strategy to reduce hypertriglyceridemia during parenteral nutrition while maintaining energy intake. J Parenter Enteral Nutr 2016;40(5):705-12. [ Links ]
]]>36. Wang X, Li W, Li N, Li J. Omega-3 fatty acids-supplemented parenteral nutrition decreases hyperinflammatory response and attenuates systemic disease sequelae in severe acute pancreatitis: A randomized and controlled study. J Parenter Enteral Nutr 2008;32(3):236-41. [ Links ]
37. Veljovic M, Popadic A, Vukic Z, Ilic R, Trifunovic Z, Antunovic M, et al. Myocardial protection during elective coronary artery bypasses grafting by pretreatment with omega-3 polyunsaturated fatty acids. Vojnosanit Pregl 2013;70(5):484-92. [ Links ]
38. Berger MM, Delodder F, Liaudet L, Tozzi P, Schlaepfer J, Chiolero RL, et al. Three short perioperative infusions of n-3 PUFAs reduce systemic inflammation induced by cardiopulmonary bypass surgery: A randomized controlled trial. Am J Clin Nutr 2013;97(2):246-54. [ Links ]
39. Sabater J, Masclans JR, Sacanell J, Chacon P, Sabin P, Planas M. Effects on hemodynamics and gas exchange of omega-3 fatty acid-enriched lipid emulsion in acute respiratory distress syndrome (ARDS): A prospective, randomized, double-blind, parallel group study. Lipids Health Dis 2008;7:39. [ Links ]
40. Zhu X, Wu Y, Qiu Y, Jiang C, Ding Y. Effects of omega-3 fish oil lipid emulsion combined with parenteral nutrition on patients undergoing liver transplantation. J Parenter Enteral Nutr 2013;37(1):68-74. [ Links ]
]]>41. Mateu-de Antonio J, Echeverria-Esnal D. Intravenous lipid emulsions in kidney transplant patients requiring parenteral nutrition. Nutrition 2016;32(3):397-8. [ Links ]
42. Arshad A, Isherwood J, Mann C, Cooke J, Pollard C, Runau F, et al. Intravenous omega-3 fatty acids plus gemcitabine: Potential to improve response and quality of life in advanced pancreatic cancer. J Parenter Enteral Nutr 2015. [ Links ]
43. Xu Z, Harvey KA, Pavlina TM, Zaloga GP, Siddiqui RA. Tocopherol and tocotrienol homologs in parenteral lipid emulsions. Eur J Lipid Sci Technol 2015;117(1):15-22. [ Links ]
44. Linseisen J, Hoffmann J, Lienhard S, Jauch KW, Wolfram G. Antioxidant status of surgical patients receiving TPN with an omega-3-fatty acid-containing lipid emulsion supplemented with alpha-tocopherol. Clin Nutr 2000;19(3):177-84. [ Links ]
45. Versleijen MW, Roelofs HM, Rombouts C, Hermans PW, Noakes PS, Calder PC, et al. Short-term infusion of a fish oil-based lipid emulsion modulates fatty acid status, but not immune function or (anti)oxidant balance: A randomized cross-over study. Eur J Clin Invest 2012;42(3):290-302. [ Links ]
]]>46. Mostad IL, Bjerve KS, Basu S, Sutton P, Frayn KN, Grill V. Addition of n-3 fatty acids to a 4-hour lipid infusion does not affect insulin sensitivity, insulin secretion, or markers of oxidative stress in subjects with type 2 diabetes mellitus. Metabolism 2009;58(12):1753-61. [ Links ]
47. Dresner A, Laurent D, Marcucci M, Griffin ME, Dufour S, Cline GW, et al. Effects of free fatty acids on glucose transport and IRS-1-associated phosphatidylinositol 3-kinase activity. J Clin Invest 1999;103(2):253-9. [ Links ]
48. Lorenz R, Spengler U, Fischer S, Duhm J, Weber PC. Platelet function, thromboxane formation and blood pressure control during supplementation of the Western diet with cod liver oil. Circulation 1983;67(3):504-11. [ Links ]
49. Heller AR, Fischer S, Rossel T, Geiger S, Siegert G, Ragaller M, et al. Impact of n-3 fatty acid supplemented parenteral nutrition on haemostasis patterns after major abdominal surgery. Br J Nutr 2002;87(Suppl 1):S95-101. [ Links ]
50. Veljovic M, Mihajlovic I, Subota V, Antunovic M, Jevdjic J, Udovicic I, et al. Effect of pretreatment with omega-3 polyunsaturated fatty acids (PUfAs) on hematological parameters and platelets aggregation in patients during elective coronary artery bypass grafting. Vojnosanit Pregl 2013;70(4):396-402. [ Links ]
]]>
![]() Correspondence:
Correspondence:
Javier Mateu-de-Antonio.
Pharmacy Department.
Hospital del Mar.
Passeig Marítim, 25-29.
08003 Barcelona, Spain
e-mail: 92976@hospitaldelmar.net
Received: 14/12/2016 ]]> Accepted: 23/12/2016
]]>