Toxicidad hepática recurrente secundaria a metilprednisolona intravenosa
Recurrent acute liver toxicity from intravenous methyprednisolone
M. Rivero Fernández, J. M. Riesco1, V. F. Moreira, A. Moreno1, A. López San Román, G. Arranz y L. Ruiz del Árbol
Servicios de Gastroenterología y 1Anatomía Patológica. Hospital Ramón y Cajal. Madrid
Dirección para correspondencia
]]>
RESUMEN
Las reacciones adversas hepáticas relacionadas con la administración de fármacos (hepatotoxicidad) son cuadros relativamente frecuentes que presentan una amplia variabilidad clínica e histológica. La identificación precoz de estos cuadros es fundamental en la práctica clínica debido a su potencial gravedad. En la mayoría de los casos la suspensión del fármaco desencadenante es suficiente para la resolución del cuadro clínico.
A pesar de que los esteroides son utilizados en una amplia variedad de situaciones clínicas, la notificación de cuadros de hepatotoxicidad secundaria a esteroides intravenosos es excepcional.
Presentamos el caso clínico de una mujer diagnosticada de esclerosis múltiple, que recibió metilprednisolona a altas dosis en forma de "pulsos" intravenosos como tratamiento de las reagudizaciones de su enfermedad y presentó 3 brotes recurrentes de hepatitis de predominio hepatocelular con un patrón clínico, analítico e histológico compatible con toxicidad hepática aguda secundaria a metilprednisolona intravenosa. En el tercer episodio se realizó una biopsia hepática que demostró un patrón de hepatitis aguda con necrosis líticas confluentes, histología no descrita previamente en pacientes tratados con esteroides intravenosos.
Palabras clave: Metilprednisolona. Hepatotoxicidad. Esclerosis múltiple.
ABSTRACT
Adverse drug reactions (hepatotoxicity) are a frequent cause of acute liver injury with a wide clinical and histological spectrum. An early recognition of drug-related liver disease has been considered essential in clinical practice due to potential risks. In most cases exposure discontinuation improves the clinical picture.
Steroids are used in a variety of clinical settings. However, intravenous steroids have rarely been associated with hepatotoxicity. We report the case of a middle-aged woman with multiple sclerosis who received a bolus of methylprednisolone on three occasions for the management of relapsing disease, with the development of repeated episodes of elevated liver enzymes after corticoid administration. In the third episode a liver biopsy was performed, which showed acute hepatitis with bridging necrosis; such histological picture has not been described before in patients treated with intravenous steroids.
Key words: Methylprednisolone. Hepatotoxicity. Multiple sclerosis.
Introducción
La hepatotoxicidad inducida por fármacos es una de las etiologías más comunes de hepatitis aguda en nuestro medio y representa la causa más frecuente de fallo hepático fulminante en EE. UU. (1). Sin embargo, las tasas reales de hepatitis secundaria a fármacos son desconocidas (2). La expresión clínica de estos cuadros es muy variable, oscilando entre alteraciones asintomáticas de las enzimas hepáticas hasta fallo hepático agudo fulminante y su diagnóstico precisa de un alto índice de sospecha y se sustenta en una correcta anamnesis y la exclusión de las causas más comunes potencialmente responsables (3,4). Las escalas o algoritmos diagnósticos son herramientas útiles para valorar objetivamente la relación de causalidad.
Los cuadros de hepatotoxicidad secundaria al uso de esteroides son poco frecuentes en la práctica clínica. Una de las formas de aplicación de esteroides a altas dosis es la pulsoterapia o "pulsos" intravenosos, alternativa terapéutica muy común en el tratamiento de las reagudizaciones graves de pacientes con enfermedades autoinmunes.
A nuestro juicio, el caso clínico expuesto representa el primer caso confirmado histológicamente de hepatotoxicidad recurrente en relación al uso de metilprednisolona administrada en "pulsos" intravenosos con patrón de hepatitis aguda con necrosis.
Caso clínico
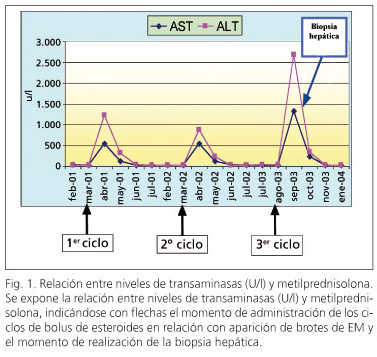
Mujer de 57 años diagnosticada a los 44 años de esclerosis múltiple (EM) que recibió 3 cursos de 6-metilprednisolona a dosis de 1.000 mg intravenosa (i.v.) administrada en forma de "pulsos" por exacerbación de su EM. La paciente previamente presentaba una analítica hepática rigurosamente normal, no reconocía ingesta habitual de alcohol y no tomaba fármacos potencialmente hepatotóxicos. A los 3 días de recibir el último "pulso" de 6-metilprednisolona i.v. presentó la siguiente alteración del perfil hepático en el análisis de rutina: GOT (AST): 543 U/l (N < 50), GPT (ALT): 1223 U/l (N < 40), BT: 1,2 mg/dl, GGT: 71 U/l (N < 50), FA: 113 U/l (N < 120), LDH: 423 U/l con posterior normalización de dichos valores 3 meses más tarde tras suspender la administración del fármaco. Doce meses después la paciente presentó un nuevo episodio de exacerbación de su EM por lo que recibió nuevamente un ciclo de 3 bolos de 6-metilprednisolona (1 g/día i.v. durante 3 días) con reaparición de la alteración del perfil hepático a los 5 días tras concluir la administración del tratamiento (GOT: 541 U/l, GPT: 883 U/l, GGT: 82 U/l, FA: 100 U/l) con posterior normalización analítica a los 4 meses. Diecisiete meses tras la segunda exposición, recibió nuevamente "bolos" de 3 días de 6-metilprednisolona i.v. a las mismas dosis, presentando un nuevo episodio de elevación asintomática de las enzimas hepáticas (GOT: 1328 U/l, GPT: 2685 U/l, GGT: 56 U/l, FA: 115 U/l) con posterior normalización de los parámetros bioquímicos al mes de suspender el tratamiento. El resumen de la evolución de los parámetros analíticos del paciente se presentan en la figura 1.
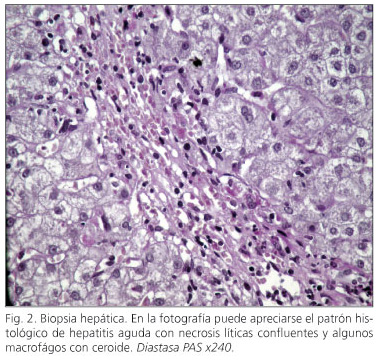
Durante la fase aguda de estos cuadros de alteración de la bioquímica hepática, se determinaron la serología del VHB (HBsAg, IgM HBcAc), VHC (PCR, anti-VHC, VHC-RNA) y VHA (anti-HA IgM e IgG) que fueron negativas, al igual que los anticuerpos antinucleares, antimitocondriales, antimúsculo liso, anti-LKM, antigliadina y antiendomisio. El cobre y la ceruloplasmina séricos, junto con la cupruria de 24 horas, la ferritina, la TSH y la α1-antitripsina estuvieron en rango normal. Las ecogra-fías abdominales en relación con los 3 episodios de alteración de la bioquímica hepática no mostraron hallazgos patológicos. Un mes después de la detección del último brote de hepatitis aguda se decidió la realización una de una biopsia hepática donde se identificaron lesiones histológicas características de hepatitis aguda con presencia de necrosis líticas e hiperplasia macrofágica con ceroide (Fig. 2).
]]>
La conjunción de datos clínicos, analíticos e histológicos, una vez excluidas otras causas de potencial alteración del perfil hepático sugirieron el diagnóstico de hepatitis aguda tóxica recurrente secundaria a metilprednisolona intravenosa.
Discusión
Las reacciones adversas hepáticas relacionadas con la administración de fármacos resultan de vital importancia en la práctica clínica habitual debido a su potencial gravedad. Representan la causa más frecuente de restricción de uso y retirada de medicamentos del mercado farmacéutico (5).
Un estudio realizado en Francia sugiere que la tasa de incidencia anual de daño hepático inducido por fármacos es de 13,9 por cada 100.000 habitantes (6).
La mayoría de los fármacos utilizados en la práctica clínica son potencialmente hepatotóxicos. La toxicidad hepática es habitualmente idiosincrásica, determinada por la susceptibilidad individual y no se relaciona con la dosis de fármaco (7).
Existen varios patrones de hepatotoxicidad: citotóxico (citolítico), colestásico, mixto y esteatósico (8) y se ha sugerido que las formas citolíticas evolucionan con mayor frecuencia a daño hepático fulminante (9). El diagnóstico precisa de un alto índice de sospecha y se sustenta en una correcta anamnesis y la exclusión de las causas más comunes potencialmente responsables. Entre los criterios favorables al diagnóstico de hepatotoxicidad se incluye la demostración de una rápida mejoría clínica y biológica tras la retirada del fármaco (dechallenge) y la reaparición de las alteraciones tras la re-exposición al agente causal (rechallenge). En la actualidad se utilizan la escala de Naranjo (10), la escala de CIOMS/RUCAM (11) y la escala diagnóstica clínica (CDS) (12) para valorar la relación de causalidad.
La mayoría de los cuadros mejoran suprimiendo el fármaco desencadenante.
Las alteraciones de la función hepática tras el uso de esteroides pueden responder a varios mecanismos: reactivación de infecciones hepáticas virales (13), esteatosis/esteatohepatitis o hepatitis inmunoalérgica (en relación al excipiente de las preparaciones parenterales) (14). Además, en los pacientes con esclerosis múltiple, como en el caso de nuestra paciente, debe considerarse el diagnóstico de hepatitis autoinmune cuando existe alteración de los parámetros de la bioquímica hepática (15). Iancu y cols. sugirieron que la hepatomegalia tras el uso de esteroides era el resultado de la acumulación reversible de glucógeno y no tanto de la existencia de daño o disfunción hepática (16).
]]> Revisando la literatura hemos encontrado únicamente 5 casos publicados de hepatotoxidad inducida por esteroides intravenosos. Dos casos de hepatitis fulminante asociada a prednisolona, ambos con histología de esteatohepatitis no alcohólica, a diferencia de nuestro caso (17,18).El tercero de los casos, el de un varón de 76 años que sufrió la aparición de un fallo hepático fulminante no dispone de confirmación histológica (19).
Sólo hemos encontrado dos casos publicados de episodios recurrentes de elevación de enzimas hepáticas en relación a la administración de pulsos de esteroides; el primero de ellos se trata de una mujer de 46 años diagnosticada de esclerosis múltiple, que presentó 3 episodios de elevación asintomática de las enzimas hepáticas tras recibir pulsos de metilprednisolona aunque durante ninguno de estos episodios se realizó biopsia hepática (20). El segundo de ellos, el de una mujer de 48 años con EM que presentó alteración de las enzimas a las 6 semanas de recibir 3 ciclos de metilprednisolona, presentó cambios histológicos sugerentes de hepatitis inmunoalérgica (infiltrado mixto del parénquima hepático por linfocitos, eosinófilos y células plasmáticas) (21).
En los casos citados en los que se dispuso de biopsia hepática, en ninguno se observó un patrón histológico como el presentado en nuestro caso.
En nuestro caso, la relación temporal entre la administración del fármaco y la aparición de alteraciones en la analítica hepática (de forma repetida), con posterior normalización tras la retirada del esteroide (dechallenge y rechallenge), junto con la exclusión de otros factores potencialmente hepatotóxicos y los hallazgos histológicos de la biopsia hepática, apoyan el diagnóstico de hepatitis aguda tóxica por esteroides. Al aplicar los algoritmos diagnósticos de causalidad obtuvimos una puntuación de 9 puntos en la escala de Naranjo (relación "definitiva") y una puntuación de 12 en las escala de la CIOMS (relación "altamente probable"). Creemos además que nuestra comunicación, a diferencia del resto de casos publicados previamente, es relevante ya que, además de tratarse de episodios recurrentes, representa el primer caso histológicamente confirmado de hepatitis aguda con necrosis asociada al uso de metilprednisolona en forma de "pulsos" intravenosos. Este hecho adquiere mayor interés si se tiene en cuenta que este patrón histológico identifica aquellas formas que tienden a evolucionar clínicamente de una forma más desfavorable (22).
Llama la atención que los casos de toxicidad hepática secundaria a esteroides intravenosos son poco numerosos, aunque potencialmente muy graves y en ocasiones recurrentes, por lo que es fundamental reconocer el cuadro clínico-histológico, suspender el tratamiento precozmente y tener este hecho muy presente en posteriores administraciones del fármaco por la posibilidad de desarrollo de fallo hepático asociado al patrón de necrosis confluente (23). Además, creemos que este caso ilustra la posibilidad de que un mismo fármaco produzca un espectro histológico lesivo diferente según las características del paciente.
Bibliografía
1. Ostapowicz G, Fontana RJ, Schidiot FV, et al. Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med 2002; 137: 947-54. [ Links ]
2. Lucena, MI, Andrade RJ. Increasing the detection of hepatotoxic response in clinical practice: a quality commitment. Rev Esp Enferm Dig 2005; 97: 145-53. [ Links ]
3. Andrade RJ, Robles M, Fernández-Castañer A, López-Ortega S, López-Vega M, Lucena MI. Assessment of drug-induced hepatotoxicity in clinical practice: A challenge for gastroenterologists. World J Gastroenterol 2007; 13: 329-40. [ Links ]
4. Andrade RJ, López-Ortega S. Hepatitis tóxicas. Rev Esp Enferm Dig 2006; 9: 701. [ Links ]
5. Bakke OM, Manocchia M, De Abajo F, Kaitin KI, Lasagna L. Drug safety discontinuations in the United Kingdom, the United States, and Spain from 1974 through 1993: a regulatory perspective. Clin Pharmacol Ther 1995; 58: 108-17. [ Links ]
6. Sgro C, Clinard F, Ouazir K, Chanay H, Allard C, Guilleminet C, et al. Incidence of drug-induced hepatic injuries: a French population-based study. Hepatology 2002; 36: 451-5. [ Links ]
7. Willis C, Maddrey MD. Drug-induced hepatotoxicity. J Clin Gastroenterol 2005; 39: 83-9. [ Links ]
8. Bissell DM, Gores GJ, Laskin DL, Hoofnagle JH. Drug-induced liver injury: mechanisms and test systems. Hepatology 2001; 33: 1009-13. [ Links ]
9. Andrade RJ, Lucena MI, Fernández MC, Pelaez G, Pachkoria K, García-Ruiz E, et al. Drug-induced liver injury: an analysis of 461 incidences submitted to the Spanish Registry over a 10-year period. Gastroenterology 2005; 129: 512-21. [ Links ]
10. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther 1981; 30: 239-45. [ Links ]
11. Danan G, Bénichou C. Causality assessment of adverse drug reactions to drugs I. A novel method based on the conclusions of international consensus meetings: application to drug-induced liver injuries. J Clin Epidemiol 1993; 46: 1323-30. [ Links ]
12. María V, Victorino R. Development and validation of a clinical scale for the diagnosis of drug-induced hepatitis. Hepatology 1997; 26: 664-9. [ Links ]
13. Shiota G, Harada K, Oyama K, Udgawa A, Nomi T, Tanaka K, et al. Severe exacerbation of hepatitis alter short-term corticosteroid therapy in a patient with "latent" chronic hepatitis B. Liver 2000; 20: 415-20. [ Links ]
14. Gerolami R, Mambrini P, Barthet M, Jean-Pastor MJ, Salducci J, Grimaud JC. Acute hepatitis caused by Solupred in a patient with Crohn disease. Gastroenterol Clin Biol 1997; 21: 236-7. [ Links ]
15. Nuñez O, De Andrés C, Álvarez E, García-Monzón C, Clemente G. Hepatitis autoinmune en pacientes con diagnóstico de esclerosis múltiple. Gastroenterol Hepatol 2004; 27: 521-4. [ Links ]
16. Iancu TC, Shiloh H, Dembo L. Hepatomegaly following short-term high-dose steroid therapy. J Pediatr Gastroenterol Nutr 1986; 5: 41-6. [ Links ]
17. Nanki T, Koike R, Miyasaka N. Subacute severe steatohepatitis during prednisolone therapy for systemic lupus erythematosis. Am J Gastroenterol 1999; 94: 3379. [ Links ]
18. Dourakis SP, Sevastionos VA, Kaliopi P. Acute severe steatohepatitis related to prednisolonone therapy. Am J Gastroenterol 2002; 97: 1074-5. [ Links ]
19. Weissel M, Hauff W. Fatal liver failure after high-dose glucocorticoid pulse therapy in a patient with severe thyroid eye disease. Thyroid 2000; 10: 521. [ Links ]
20. Hofstee H, Nannayakkara P, Stehouwer C. Acute hepatitis related to prednisolone. Eur J Int Med 2005; 16: 209-10. [ Links ]
21. Das D, Graham I, Rose J. Recurrent acute hepatitis in patient receiving pulsed methylprednisolone for multiple esclerosis. Indian J Gastroenterol 2006; 25: 314-6. [ Links ]
22. Fernández-Castañer A, García-Cortés M, Lucena MI, Borraz Y, Peláez G, Costa J, et al. Análisis de las causas, características y consecuencias de la reexposición al fármaco o compuesto responsable de un episodio de hepatotoxicidad. Rev Esp Enferm Dig 2008; 100: 278-84. [ Links ]
23. Andrade RJ, Robles M, Ortiz N, Lucena MI. Reexposición en hepatotoxicidad: ¿prueba de concepto o accidente terapéutico? Rev Esp Enferm Dig 2008; 100: 255-8. [ Links ]
![]() Dirección para correspondencia:
Dirección para correspondencia:
Miguel Rivero Fernández.
Servicio de Gastroenterología.
Hospital Ramón y Cajal.
Ctra. de Colmenar, km 9,100.
28034 Madrid.
e-mail: rivernautaes@yahoo.es