A pharmacokinetic approach to model-guided design of infliximab schedules in ulcerative colitis patients
Aproximación al diseño de un programa de administración de infliximab a pacientes con colitis ulcerosa basado en un modelo famacocinético
Alejandro Pérez-Pitarch1,2, Rafael Ferriols-Lisart1,2, Manuel Alós-Almiñana1,2 and Miguel Mínguez-Pérez2,3
1 Pharmacy Department. Hospital Clínico Universitario. Valencia, Spain
2 Universidad de Valencia
3 Digestive Medicine Department. Hospital Clínico Universitario. Valencia, Spain
ABSTRACT
Background: Infliximab, an anti-tumour necrosis factor approved for treatment of Crohn's disease and ulcerative colitis, is administered at predefined interdose-intervals. On insufficient response or loss of response, treatment can be intensified. The lack or loss of response is likely related to complex pharmacokinetics of infliximab.
Aims: To explore optimal dosing strategies of infliximab in treatment-naïve patients with ulcerative colitis through predictive Monte Carlo simulations based on a validated population PK model.
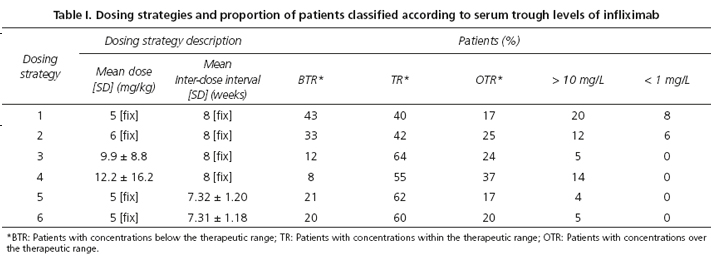
Methods: A population of 2,000 treatment-naïve patients was generated by Montecarlo simulation. Six dosing strategies for maintenance therapy were simulated on this population. Strategies 1 and 2 consisted on 5 mg/kg and 6 mg/kg doses, respectively, and 8 weeks inter-dose interval. Strategies 3 and 4 used Individualized doses, adjusted to albumin level, sex and body weight, and a fix inter-dose interval of 8 weeks to achieve a target trough concentration of 5 mg/L or 6 mg/L, respectively. Strategies 5 and 6 used a fix dose of 5 mg/kg and individualized inter-dose intervals, adjusted to the same covariates, to achieve a target concentration, of 5 mg/L or 6 mg/L, respectively.
Results: Strategies 2-6 reached trough levels statistically higher than strategy 1 (p < 0.05). Strategy 5 proved to be the best dosing strategy. It was associated with a higher proportion of responder patients than strategy 1 (62 % vs. 40 %) without reaching higher peak concentrations.
Conclusions: Optimization of maintenance treatment of colitis with infliximab by a pharmacokinetic approach could benefit infliximab-naive patients with ulcerative colitis.
Key words: Infliximab. Pharmacokinetics. Colitis. Ulcerative.
Introduction
Infliximab is an anti-tumour necrosis factor approved for treatment of Crohn's disease and ulcerative colitis. Treatment with infliximab is started by intravenous doses of 5 mg/kg at 0, 2, and 6 weeks (induction therapy) and continued every 8 weeks as maintenance therapy. Empiric data have shown that response to infliximab is highly heterogenous. Induction therapy fails, approximately, in 30-40 % of patients and increase up to 50 % over time (secondary failure) (1,2). On insufficient response or loss of response, treatment can be intensified by increasing the dose or by decreasing the dosing interval (3). The lack or loss of response is multifactorial, but is likely related to the molecular structure and complex pharmacokinetics of infliximab. Undetectable through serum concentrations of infliximab have been reported in the absence of antibodies against infliximab in 16-39 % of patients. These data indicate that other factors influence the pharmacokinetic profile of infliximab (1,4).
Retrospective studies have shown a relationship between efficacy and serum trough level of infliximab (5). A growing body of evidence suggests that both short- and long-term treatment outcomes are improved by achieving and maintaining adequate serum drug concentrations, but optimal trough serum level of infliximab is not well defined. For patients with ulcerative colitis treated with infliximab, a detectable trough serum infliximab (≥ 1,4 mg/L) predicts clinical remission, endoscopic improvement and a lower risk for colectomy (4). Bortlik et al. observed that patients with Crohn's disease and infliximab levels above 3 mg/L at the beginning of maintenance phase had lower likelihood to lose clinical response compared to individuals with lower levels (6). In a post-hoc analysis of ACCENT I trial, a serum trough level of infliximab of 3.5 mg/L had the best sensitivity and specificity to predict the long-term benefit to infliximab (7). Nevertheless, high doses had been associated with an increased risk for infections and malignancies. Based on these considerations, a target range of infliximab trough concentrations of 3-7 mg/L have been suggested as safe and effective (6,8). Therapeutic drug monitoring of infliximab concentrations can be useful to identify those patients who will benefit from dose escalation and those who will be best served by switching to an alternative drug class due to the development of anti-drug antibodies.
Many studies have shown a high inter-individual variability in serum infliximab concentrations leading to varied exposure and efficacy. Factors such as presence of anti-infliximab antibodies, concomitant use of immunomodulating agents, disease activity or body mass have been suggested to be partially responsible for the variability of infliximab serum concentration (9). Pharmacokinetic differences between patients with Crohn's disease or ulcerative colitis may or may not be explained by the previously mentioned factors. Reinisch et al. suggested that there may be a more rapid consumption of the therapeutic antibody in ulcerative colitis because involvement of the intestine is continuous and inflammatory burden may be higher than in Crohn's disease (2).
Fasanmade et al. identified four covariates (body weight, albumin, sex and antibodies to infliximab) influencing the PK properties of infliximab in patients with ulcerative colitis in a validated population pharmacokinetic (PK) model (10). Body weight was the most significant covariate in volume of distribution of infliximab. Low albumin levels predict higher infliximab clearance. Sex was a potential covariate on both clearance and volume of distribution, and clearance was 47.1 % higher for patients positive to antibodies to infliximab compared with those were non-positive. In this context, administration of the same maintenance dose of infliximab to all patients, (the "one size fits all" rule), without regard to potential covariates that influence infliximab pharmacokinetics can provide highly variable serum concentrations.
The objective of the work presented in this paper was to improve our understanding of infliximab dosing and explore optimal maintenance dosing strategies through predictive Monte Carlo simulations based on a validated population PK model, which was constructed from phase III clinical trial data (10). Here we report the probability of maintaining steady state infliximab levels in the target range in naïve patients to infliximab with ulcerative colitis with several dosing regimens of infliximab.
Methods
The available literature was reviewed in order to select a robust pharmacokinetic model of infliximab in ulcerative colitis patients. The PK model proposed by Fasanmade et al. was the only model validated in patients with moderately to severely active ulcerative colitis (8). It was a two-compartment model with albumin, sex, antibody-to-infliximab positivity and body weight as key covariates influencing infliximab pharmacokinetics.
]]> A population of 2000 naïve patients was generated by Montecarlo simulation with demographic characteristics equivalent to those of the population in which the pharmacokinetic model was validated. Weight and albumin distributions were simulated with a log normal and normal distribution, respectively. Patients were naïve to infliximab treatment so it was assumed that no antibodies to infliximab had been developed at the start of the treatment. The pharmacokinetic model was implemented in Berkeley Madonna® software (University of California, Berkeley, USA) and concentration-time profiles were generated for the different dosing schemes.Six dosing strategies for maintenance therapy were simulated for naïve patients (patients with no previous infliximab administration). Induction phase was always equal (5 mg/kg at 0, 2 and 6 weeks). Strategy 1 and 2 were classified as fixed dosing strategies. They consisted on fixed doses (mg/kg) and fixed inter-dose intervals (τ) for all patients. All patients simulated with strategy 1 received a dose of 5 mg/kg of infliximab and 8 weeks inter-dose interval, according to EMA and FDA label schedule. Similarly, strategy 2 consisted on 6 mg/kg and 8 weeks inter-dose interval for all patients.
Strategies 3 to 6 were classified as individualized schedules. Strategies 3 and 4 used individualized doses, adjusted to albumin level, sex and body weight, and a fix inter-dose interval of 8 weeks to achieve a target trough concentration of 5 mg/L or 6 mg/L, respectively. Strategies 5 and 6 used a fix dose of 5 mg/kg and individualized inter-dose intervals to achieve a target trough concentration, of 5 mg/L or 6 mg/L, respectively. Trough level was defined as serum concentration immediately before an infliximab infusion.
Serum trough and peak concentrations of infliximab were obtained from the simulated concentration-time profiles. Therapeutic range was defined as trough concentrations between 3-7 mg/L. Patients were classified according to trough levels as: a) Patients with concentrations within the therapeutic range (TR); b) patients with concentrations below the therapeutic range (BTR); and c) patients with concentrations over the therapeutic range (OTR). The main goal was to reach the highest proportion of TR with a low proportion of OTR. Additionally, peak levels were analysed to detect extreme values that could be associated to adverse events. Peak level was defined as serum concentration immediately after an infliximab infusion.
Strategy 1 (5 mg/kg/8 weeks) was included as a reference point since it is the schedule recommended by the EMA guidelines.
Statistical analysis was performed using IBM SPSS Statistics 19.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics were stated as mean and standard deviation (SD) values for continuous variables. Categorical variables were analysed using Cochran-Mantel-Haenszel χ2 test or the Fisher's exact test. Continuous variables were evaluated with analysis of variance (ANOVA). Mean and 95 % confidence interval (95 % CI) were computed for the simulations. Two sided p-values < 0.05 were considered statistically significant. Monte Carlo simulation was performed using the Berkeley Madonna® software.
Results
No statistical differences were found between the characteristics of the simulated patients and the characteristics of the population used in validation of the PK model (p > 0.05) (8). Albumin value was 4.11 (95 % CI: 2.9-5.4) g/dL, weight 81.7 kg (95 % CI: 36.4-163.7) and 60 % of the patients were males.
The outcomes and descriptions of the simulated strategies are shown in table I. More patients were classified as TR with the individualized strategies (strategies 3-6) than with strategy 1, and statistically significant (p < 0.05) differences were detected. No differences were found among individualized strategies, but strategy 4 was the individualized strategy that achieved the highest proportion of OTRpatients and patients with serum trough levels of infliximab higher than 10 mg/L.
]]>
Patients on strategy 3 received a mean dose of 9.9 ± 8.8 mg/kg/8 weeks and 12.2 ± 16,2 mg/kg/8 weeks with strategy 4. Patients in strategies 5 and 6 received a fix dose of 5 mg/kg but with a similar interval dose, approximately, 7.3 ± 1.2 weeks.
Table II shows mean serum trough and peak levels of infliximab for each strategy evaluated. All strategies reached trough levels statistically higher than strategy 1 (p < 0.05). Peak levels of strategies 5 and 6 were similar to strategy 1. Patients on strategies 3 and 4 reached higher peak concentrations than strategy 1. Strategy 5 was associated with a low variability in the estimation of serum infliximab levels (CV of 49 % and 22 % for trough and peak levels). Strategies 1 and 2 showed high variability in trough levels and strategies 3 and 4 showed high variability for peak levels. Serum level distributions of infliximab are graphed in figure 1.
]]> Strategy 5 seems to be the best strategy. It was associated with a better proportion of TR patients without reaching higher peak concentrations than with the strategy 1 (5 mg/kg/8 weeks).
Discussion
A critical step in dose selection is an understanding of the pharmacokinetic goals for treatment derived from PK modelling, to maximize the likelihood of a favourable clinical response as well as to minimize the probability of exposure related toxicities. Appropriate models can provide a useful starting point for predicting the exposure and the time course of a drug for different dosage regimens in a target population.
The application of Monte Carlo simulations based on population pharmacokinetic concepts can be valuable to test treatment schedules. These simulations are useful to estimate serum concentrations of a given treatment and evaluate the suitability of different dosing regimens (9,10). However, clinical studies are needed to confirm the findings.
During the induction phase, patients have higher trough levels than in the maintenance phase. Patients receiving the same maintenance dosing have shown large interindividual variability in trough levels of infliximab. Concentrations of infliximab are influenced by several variables that can fluctuate over time and among individuals. However, after taking these factors into consideration infliximab concentration still vary considerably among patients receiving the same dose of infliximab. Serum albumin concentration is a predictive factor of infliximab pharmacokinetics and clinical response in patients with ulcerative colitis. Thus, patients with low albumin levels (< 3.5 g/dL) have been associated with higher infliximab clearance and lower infliximab concentrations. The effect on clinical response could be relevant (11).
The present study shows that an individualized maintenance therapy of infliximab could be a better strategy than fixed maintenance therapy to achieve potentially therapeutic concentrations. Only serum albumin concentration, weight and sex were used in the calculation of doses and inter-dose intervals. In this study, patients with serum trough levels between 3-7 mg/L achieved the target concentration, but treatment success does not only depend on this factor. Thus, Steenholdt et al. found patients with inflammatory bowel disease and infliximab trough levels above the established cut-off level that had lost clinical response (12). They hypothesized that loss of response was caused by a pharmacodynamic problem, for example, due to TNF-independent disease. On the other hand, a proportion of patients maintained long-term remission despite sub-therapeutic infliximab levels or lost clinical response even though they had high serum levels of drug (3,12,13).
It is not known what the optimal trough level for infliximab is. Bortlik et al. observed that patients with Crohn's disease and infliximab trough levels > 3 mg/L at the start of maintenance regimen (weeks 14-22) had about 66 % lower likelihood to lose their clinical response until the end of the follow-up (median of 25 months) compared to individuals with subtherapeutic levels (4). Cornillie et al. proposed a trough level of 3.5 mg/L as the cut-off value for trough levels associated with the best sensitivity and specificity to predict the long-term benefit to infliximab (5). After induction therapy with 5 mg/kg infliximab, 25 % and 33 % of patients sustained week 14 response to infliximab 5 or 10 mg/kg/8 weeks without dose escalation, respectively, through week 54. Patients with durable sustained response with infliximab 5 mg/kg maintenance treatment had higher post-induction trough levels (4 mg/L) than patients without durable sustained response. Serum infliximab trough levels ≥ 3.5 mg/L and ≥ 60 % C-reactive protein decrease were significantly associated with durable sustained response(5).
Some algorithms have been proposed to guide therapy in patients with inflammatory bowel disease receiving infliximab (14). These algorithms use as lower limit of trough level a value of 3 mg/L (6,7). However, some authors suggest lower values. Seow et al. found that a detectable trough serum infliximab (> 1.4 mg/L) predicts clinical remission, endoscopic improvement and a lower risk for colectomy in patients with ulcerative colitis (3). Steinholdt et al. proposed an optimal cut-off value of 0.8 mg/L for patients with ulcerative colitis but this value was observed in a small cohort of patients (n = 21) (12). The authors used a clinically validated fluid-phase RIA that is not influenced to the same extent by the potential artefacts encountered in solid-phase enzyme-linked immunosorbent assays (EIA). Commercial assays to measure infliximab are based in bridging-type EIAs. The cause of the variable findings regarding clinical relevance of infliximab levels may be related to the assay employed. False-positive and false-negative results can be caused by non-specific binding of antibodies, rheumatic factor or complement factor to infliximab or to the matrix effects (12,15).
Arias et al. evaluated the importance of trough levels and antibodies on the long-term efficacy of infliximab therapy in ulcerative colitis (16). Low trough levels of infliximab after induction phase were associated with higher risk for loss of response and discontinuation (p = 0.01). A cut-off value of 7.19 mg/L for trough level at week 14 was identified for a sustained response.
]]> The published studies evaluate the importance of serum trough levels but there is little information about the clinical relevance of peak levels (17). Optimal use of a drug implies the correct dosage with the lowest trough level to exert the full clinical effect of the drug and peak levels that not exceed concentrations associated with toxicity (2). In our study, peak concentrations of strategies 5 and 6 were similar to the reference strategy.There are several limitations to our study. First, the results were obtained from a simulated population and clinical studies are needed to confirm the findings. Secondly, simulated patients were naïve to infliximab administration and the influence of the development of anti-infliximab antibodies on serum trough levels of infliximab was not considered. Loss of clinical response has been correlated with the appearance of antibodies to infliximab. Most of the patients who develop anti-infliximab antibodies do so within the first year of treatment (18). Some authors have observed a need for infliximab dose intensification in patients with inflammatory bowel disease (19,20). Earlier and a higher rate of dose intensification were required for patients with ulcerative colitis in another study (20). Also, it was assumed that this trough levels are necessary for all patients with ulcerative colitis, with or without immunodulators associated and independent of their clinical evolution.
Implementation of individualised dosing schedules in clinical practice has technical limitations. Application of pharmacokinetic principles for population model-based individualised dosing requires the intervention of a specialised pharmacokinetics unit. The development of dosing algorithms based on factors affecting pharmacokinetics of drugs is another possibility. Even if personalised regimens were employed, therapeutic drug monitoring is still recommended because population pharmacokinetic models explain only part of the pharmacokinetic variability. Nevertheless, if personalised regimens were employed, the expected proportion of naïve patients with potentially therapeutic concentrations is higher than the one achieved with standard maintenance dosing schedules. Individualized strategies, such as 5 and 6, would achieve better trough serum concentrations of infliximab. In these patients, primary treatment failures would probably not be due to subtherapeutic drug levels, but due to other reasons.
In conclusion, optimising maintenance therapy of colitis with infliximab by a pharmacokinetic approach could be of benefit in naïve patients with ulcerative colitis. Further clinical studies are needed in order to confirm if an individualized maintenance therapy, adjusted to a target trough level, is more useful from a clinical point of view.
References
1. Agency EM. Remicade: EPAR - Product Information. In; 2009. [ Links ]
2. Rutgeerts P, Vermeire S, Van Assche G. Predicting the response to infliximab from trough serum levels. Gut 2010;59:7-8. [ Links ]
]]>3. Seow CH, Newman A, Irwin SP, Steinhart AH, Silverberg MS, Greenberg GR. Trough serum infliximab: A predictive factor of clinical outcome for infliximab treatment in acute ulcerative colitis. Gut 2010;59:49-54. [ Links ]
4. Bortlik M, Duricova D, Malickova K, Machkova N, Bouzkova E, Hrdlicka L, et al. Infliximab trough levels may predict sustained response to infliximab in patients with Crohn's disease. J Crohns Colitis 2013;7:736-43. [ Links ]
5. Cornillie F, Hanauer SB, Diamond RH, Wang J, Tang KL, Xu Z, et al. Postinduction serum infliximab trough level and decrease of C-reactive protein level are associated with durable sustained response to infliximab: A retrospective analysis of the ACCENT I trial. Gut 2014;63:1721-7. [ Links ]
6. Zandvliet ML, van Bezooijen JS, Bos MA, Prens EP, van Doorn M, Bijen I, et al. Monitoring antigen-specific biologics: current knowledge and future prospects. Ther Drug Monit 2013;35:588-94. [ Links ]
7. Yarur AJ, Abreu MT, Deshpande AR, Kerman DH, Sussman DA. Therapeutic drug monitoring in patients with inflammatory bowel disease. World J Gastroenterol 2014;20:3475-84. [ Links ]
]]>8. Fasanmade AA, Adedokun OJ, Ford J, Hernandez D, Johanns J, Hu C, et al. Population pharmacokinetic analysis of infliximab in patients with ulcerative colitis. Eur J Clin Pharmacol 2009;65:1211-28. [ Links ]
9. Mould DR, Upton RN. Basic concepts in population modeling, simulation, and model-based drug development. CPT Pharmacometrics Syst Pharmacol 2012;1:e6. [ Links ]
10. Samtani MN, Flamm R, Kaniga K, Nandy P. Pharmacokinetic-pharmacodynamic-model-guided doripenem dosing in critically ill patients. Antimicrob Agents Chemother 2010;54:2360-4. [ Links ]
11. Fasanmade AA, Adedokun OJ, Olson A, Strauss R, Davis HM. Serum albumin concentration: A predictive factor of infliximab pharmacokinetics and clinical response in patients with ulcerative colitis. Int J Clin Pharmacol Ther 2010;48:297-308. [ Links ]
12. Steenholdt C, Bendtzen K, Brynskov J, Thomsen OO, Ainsworth MA. Cut-off levels and diagnostic accuracy of infliximab trough levels and anti-infliximab antibodies in Crohn's disease. Scand J Gastroenterol 2011;46:310-8. [ Links ]
]]>13. Maser EA, Villela R, Silverberg MS, Greenberg GR. Association of trough serum infliximab to clinical outcome after scheduled maintenance treatment for Crohn's disease. Clin Gastroenterol Hepatol 2006;4:1248-54. [ Links ]
14. Khanna R, Sattin BD, Afif W, Benchimol EI, Bernard EJ, Bitton A, et al. Review article: A clinician's guide for therapeutic drug monitoring of infliximab in inflammatory bowel disease. Aliment Pharmacol Ther 2013;38:447-59. [ Links ]
15. Steenholdt C, Ainsworth MA, Tovey M, Klausen TW, Thomsen OO, Brynskov J, et al. Comparison of techniques for monitoring infliximab and antibodies against infliximab in Crohn's disease. Ther Drug Monit 2013;35:530-8. [ Links ]
16. Arias M.T VCN, Drobne D, Ferrante M, Cleynen i, Ballet V, Rutgeerts P, et al. Importance of trough levels and antibodies on the long-term efficacy of infliximab therapy in ulcerative colitis. In: European Crohn's and Colitis Organisation - ECCO. Barcelona; 2012. [ Links ]
17. Yamada A, Sono K, Hosoe N, Takada N, Suzuki Y. Monitoring functional serum antitumor necrosis factor antibody level in Crohn's disease patients who maintained and those who lost response to anti-TNF. Inflamm Bowel Dis 2010;16:1898-904. [ Links ]
]]>18. Ungar B, Chowers Y, Yavzori M, Picard O, Fudim E, Har-Noy O, et al. The temporal evolution of antidrug antibodies in patients with inflammatory bowel disease treated with infliximab. Gut 2014;63:1258-64. [ Links ]
19. Magro F, Rodrigues-Pinto E, Lopes S, Macedo G. Earlier need of infliximab intensification in ulcerative colitis than in Crohn's disease. J Crohns Colitis 2014;8:1331-2. [ Links ]
20. Casteele N. Vande CG, Ballet V, Van Assche G, Gils A, Vermeire S, Rutgeerts P. Individualised infliximab treatment using therapeutic drug monitoring: A prospective controlled Trough level Adapted infliXImab Treatment (TAXIT) trial. In: European Crohn's and Colitis Organisation - ECCO. Barcelona; 2012. [ Links ]
![]() Correspondence:
Correspondence:
Alejandro Pérez-Pitarch ]]>
Pharmacy Department
Hospital Clínico Universitario
Avda. Blasco Ibañez, 17
46010 Valencia, Spain
e-mail: alejandroperezpitarch@gmail.com
Received: 24-10-2014
Accepted: 23-12-2014