ORIGINALES
Interdisciplinary recommendations document to improve adherence in patients with chronic inflammatory diseases: Adhing recommendations
Documento interdisciplinar de recomendaciones para mejorar la adherencia en pacientes con enfermedades inflamatorias crónicas: recomendaciones Adhing
Fernando Bermejo-San José1, Mireia Barceló-Bru2, Miquel Ribera-Pibernat3, José Luís Poveda-Andrés4 and Luciano Sanromán-Álvarez5 en representación del Grupo de Trabajo Recomendaciones Adhing
1Servicio de Digestivo. Hospital Universitario de Fuenlabrada, Fuenlabrada. Madrid.
2Unidad de Reumatología. Hospital Vall d'Hebron. Barcelona. ]]>
3Servicio de Dermatología. Hospital Universitari de Sabadell - Corporació Parc Taulí, Sabadell.
4Servicio Farmacia. Hospital Universitario y Politécnico la Fe. Valencia.
5Servicio de Ap. Dixestivo.Complexo Hospitalario Universitario de Vigo, Vigo. Spain.
Miquel Ribera hereby declares that he has received grants and payments for research, advisory activities and training from the following companies: Abbvie, Almirall, Janssen, Leo Pharma, MSD, Novartis and Pfizer.
ABSTRACT
Objective: Patient compliance in chronic inflammatory diseases is essential to achieve treatment effectiveness. The study objective is to develop a recommendations document for healthcare professionals to encourage treatment compliance.
Methods: A sequential methodology was followed: 1. Systematic review of EBM Reviews, Embase and MedLine without time restriction of interventions on compliance in chronic inflammatory diseases. Studies were selected according to previously defined criteria (confirmed diagnosis of chronic inflammatory disease of patients included, assessment of adherence interventions and experimental study design). 2. Three expert workshops (hospital pharmacy, nursing and medical specialties) to elaborate recommendations. 3. Expert online voting about the degree of agreement with recommendations. 4. Final face to face consensus workshop. ]]>
Results: A total of 1 115 papers were identified, 84 were fully reviewed and 30 were selected according to criteria. A preliminary list of 8 recommendations based on evidence was developed and discussed in the expert workshops. As a result, a new version of 14 recommendations was created. Later, online voting showed a high degree of agreement among experts: 7 out of 14 recommendations obtained unanimous approval; in the other 7, 1 to 3 experts were partially in disagreement. Recommendations without unanimous agreement were further reviewed and modified in the face to face meeting and final recommendations were approved unanimously.
Conclusions: This consensus statement gathers all relevant aspects to be interdisciplinary considered to detect, monitor and assess medication adherence, involving the patient in the process. Further studies are needed to assess the impact of interventions to improve compliance in patients with chronic inflammatory diseases both in terms of adherence and in health outcomes.
Key words: Patient compliance; Chronic disease; Inflammation; Recommendations.
RESUMEN
Objetivo: La adherencia terapéutica en los pacientes con enfermedades inflamatorias crónicas es fundamental para alcanzar la efectividad del tratamiento. El objetivo de este trabajo es crear un documento de recomendaciones para los profesionales de la salud que promueva la adherencia al tratamiento.
Métodos: Se siguió una metodología secuencial: 1. Revisión sistemática en Evidence Based Medicine Reviews (EBMR), Embase y MedLine de intervenciones en adherencia en enfermedades inflamatorias crónicas sin restriciones temporales. Los estudios se seleccionaron según criterios previamente definidos (pacientes diagnosticados de enfermedad inflamatoria crónica, evaluación de intervenciones para la mejora de la adherencia y diseño experimental de estudio). 2. Reunión con tres grupos de expertos (farmacia hospitalaria, enfermería y especialidades médicas) para generar recomendaciones. 3. Votación electrónica de los expertos sobre el grado de acuerdo con las recomendaciones. 4. Reunión presencial final de las recomendaciones.
Resultados: Se identificaron 1.115 citas, se revisaron 84 y se seleccionaron 30 de acuerdo a los criterios de elegibilidad. Se redactó un listado inicial de 8 recomendaciones basadas en la evidencia, que se discutió en las reuniones de expertos y dio lugar a una nueva versión de 14 recomendaciones. La posterior votación electrónica de esta versión evidenció un elevado grado de acuerdo entre los expertos: en 7 recomendaciones, acuerdo unánime; en las 7 restantes, entre 1 y 3 expertos parcialmente en desacuerdo. En la reunión presencial se volvieron a revisar y modificar las recomendaciones, sin acuerdo unánime, que finalmente se aprobaron por unanimidad.
Conclusiones: Estas recomendaciones recogen los aspectos relevantes para detectar, monitorizar y evaluar la adherencia terapéutica de forma multidisciplinar, implicando de forma activa al paciente. Son necesarios nuevos estudios para evaluar el impacto de las intervenciones para mejorar la adherencia en los pacientes con enfermedades inflamatorias crónicas y su influencia tanto en los aspectos relativos al cumplimiento como en los resultados en salud.
Palabras clave: Adherencia terapéutica; Enfermedad crónica; Inflamación; Recomendaciones.
]]> Contribution to Scientific Literature
In a social context of polymedicated patients, with an ageing population and a tendency to chronic conditions, the concepts of self-care, treatment adherence, and health outcomes have become particularly relevant. It is necessary to prepare recommendations targeted to and designed by healthcare professionals, in order to improve treatment adherence in their patients, and therefore improve disease control and their overall health status.
The innovative contribution of our study is its approach, which allows us to cover a need unmet so far: developing recommendation guidelines in order to increase patient adherence to treatments for inflammatory conditions.
This study was conducted sequentially, through systematic literature review of interventions on treatment adherence for chronic inflammatory conditions, without any time limitations. The result of said review was the selection of different studies according to previously defined criteria. Meetings were set up with three groups of experts from different settings (hospital pharmacy, nursing, and different medical specialties), to provide the recommendations with a multidisciplinary and plural perspective. Finally, a series of structured recommendations were established, highlighting the need for actions targeted to identifying the causes for lack of patient adherence to treatment, and to define informative and educational actions, as well as reminders (emails, apps, etc.). Another suggestion from the conclusions was to set up new lines of research, with studies assessing the implementation of guidelines for treatment adherence and their monitoring, in order to determine the health outcomes derived of said implementation, also assessing the opinion of patient associations.
Introduction
In recent years, the treatment armamentarium for chronic inflammatory conditions has been increased with the launch of new biologic drugs, which have improved the progression of said diseases, with a positive impact on patient satisfaction with treatments and on their quality of life. However, for any drug to reach its optimal levels of efficacy, it is essential to create awareness in patients about the importance of treatment adherence. In the case of biologic drugs, the treatment adherence rate for inflammatory diseases would be between 22 and 99%1,2, depending on the drug and the method for assessing adherence; though generally it is estimated at around 70%.
The attitude of patients regarding adherence will be mainly determined by their knowledge about their disease and its treatment; on the other hand, sociodemographical aspects are not so important. These aspects can also explain why a patient can be compliant and have good adherence to treatments for one disease, while not being compliant with another treatment (for another concomitant disease, or the same one)3. Therefore, interventions targeted to improving adherence must be, as far as possible, individualized for each patient; and to this aim, it is necessary to try and learn the causes for the lack of adherence in a tailored way, in order to select the adequate strategies for each patient4,5.
The objective of this study has been the preparation of recommendations, supported by the main healthcare professionals (hospital pharmacy, nursing, and medical specialties) involved in managing patients with chronic inflammatory diseases, who encourage treatment adherence from their clinical practice.
]]> Material and methods
A sequential methodology in four stages was followed for the development of these recommendations.
Systematic review of evidence
Firstly, a systematic review was conducted of the interventions for improving treatment adherence in inflammatory conditions, which was used as the basis for preparing the initial recommendations. The search was started on March, 24th, 2013, without any time limitations, in the following databases: EBM Reviews, Cochrane Database of Systematic Reviews (2005 to March 2013), Embase (1988 to 2013, week 16) and MedLine through the OVID platform (1946 until the current day). The search strategy in order to identify all relevant studies included terms associated with the conditions studied, adherence and persistence, and was limited to studies published in English and Spanish (Table 1). Studies were considered eligible if: (i) they included patients with a confirmed diagnosis of an inflammatory condition (Rheumatoid Arthritis, Polyarticular Juvenile Idiopathic Arthritis, Axial Spondyloarthritis, Ankylosing Spondylitis, Psoriatic Arthritis, Psoriasis, Crohn's Disease, and Ulcerative Colitis) under drug treatment; (ii) there was an assessment of any intervention or method used for improving drug treatment adherence; and (iii) they presented a comparative or non-comparative experimental design, including observational studies or systematic reviews. The following were excluded: non-systematic bibliographic reviews, editorials and opinion articles, as well as presentations to congresses. Two independent reviewers selected the studies according to previously defined criteria, in two structured stages of reading the title and abstract (1) and the complete text (2). Discrepancies were sorted out by consensus.
The relevant data from the studies selected were extracted in evidence tables, where interventions for improving treatment adherence were classified, according to their nature, into informative (targeted to promote higher knowledge and awareness among patients about their condition and treatment), educational/behavioural (developed to influence and modify patient behaviour, through reminders for taking their medication, or rewarding the desired behaviour), or mixed (including both types of interventions previously mentioned)6. Finally, preliminary recommendations were prepared, based on evidence and classified according to the recommendations by the Oxford Centre for Evidence-Based Medicine (Randomized Clinical Trials (RCTs), Cohort Studies (CS) and CT systematic reviews: levels 2a-2c; Case-Control Studies (CCS) and CCS systematic reviews: level 3a-3b; Series of cases: Level 4; Expert opinion: Level 5)7, and these were subsequently presented to the experts involved.
Expert Group Meeting
During June, 2013, the contents of the initial recommendations were discussed in 3 sequential meetings, following a methodology of nominal groups with the different expert groups: hospital pharmacy, nursing, and medical specialties (Rheumatology, Gastroenterology, and Dermatology). Experts were selected according to their professional experience regarding treatment of patients diagnosed with chronic inflammatory conditions, and their previous experience in terms of initiatives for improving treatment adherence. The meetings included 12 Hospital Pharmacists, 7 nurses, and 11 physicians (5 Rheumatologists, 3 Gastroenterologists, and 3 Dermatologists). The evidence outcomes were presented in each meeting, and the main points of interest were discussed, as well as the preliminary recommendations. Based on the comments and suggestions by each expert group, a second version of the recommendations was prepared.
Electronic vote of recommendations
The second version of recommendations was submitted to one single individual vote through an electronic survey with a 5-item Likert Scale (from "Complete agreement" to "Complete disagreement"), in order to assess the level of agreement with each recommendation. Those experts who showed a certain degree of disagreement with any of the recommendations were asked to provide comments for improving and accepting the wording.
Final Meeting for Recommendations
]]> On September, 2013, the final meeting was held in order to establish the recommendations, with the participation of all experts. The outcomes of the electronic survey were presented, and those recommendations without unanimous agreement were discussed and modified, while the rest were considered as final recommendations. Those new recommendations created by discussion underwent a final vote within the same meeting, and a final agreement was reached in terms of their final version.
Results
Systematic Review results
A total of 1,115 single quotes were identified based on the search strategy (Fig. 1), while 84 articles were considered potentially relevant based on the review of title and abstract. The complete text of these articles was then reviewed. After applying the eligibility criteria, 30 articles were selected which, after review, were classified as publications assessing: informative interventions (n = 7), educational or behavioural interventions (n = 2) or mixed interventions (n = 17, belonging to 15 studies). Four (4) systematic reviews of evidence were also included.
Preparation of Recommendations
Literature review resulted in an initial list of 8 recommendations based on evidence, which was discussed and modified at each of the three expert meetings, thus reaching a preliminary list of recommendations with an Expert Level of Evidence in the majority of cases. This preliminary list of recommendations was distributed into 7 blocks that describe the process of assessment for treatment adherence: the importance of adherence, communication with patients, patient stratification according to their risk of non-adherence, estimating/assessing/monitoring adherence, interdisciplinary approach of the intervention, individualization, and strategies in order to improve adherence. The individual voting through electronic questionnaire showed a very high level of agreement among experts. From the 14 recommendations submitted to vote, the experts showed a unanimous level of agreement for 7, and for the remaining 7, only 1 to 3 experts declared to be partially in disagreement. The latter were reviewed, modified, and a consensus was reached in the final meeting, with a unanimous vote (Table 2). Each one of the 14 final recommendations are detailed below, divided into blocks, as well as their justification according to the level of evidence for each one:
Block 1: The importance of Adherence
Recomendation 1.1
To promote and generate lines of research in order to determine the relationship between the level of treatment adherence and health outcomes.
]]> Level of evidence: 5.Block 2: Communication with Patients
Recommendation 2.1
To promote the development of communication abilities in healthcare professionals in order to encourage a fluent and honest communication with patients based on mutual trust.
Level of evidence: 2a
Comments to the recommendation: A systematic review by Pasma et al.8 about the factors associated with treatment adherence in patients with Rheumatoid Arthritis identified that a good communication between healthcare professional and patient is required in order to convince the patient of the importance of treatment.
Recommendation 2.2
In order to achieve higher treatment adherence and, consequently, better health outcomes, the following are necessary:
• Early and continuous information and education for patients about the disease.
]]> • Acceptance of their disease by patients.• Patient commitment with the agreed-upon treatment strategy.
Level of evidence: 1a
Comments to the recommendation: Joosten et al.9 studied the evidence of efficacy in shared decision making in terms of health outcomes. The authors reviewed 11 relevant publications about different conditions, and reached the conclusion that the process of shared decision making can be a good method in order to achieve patient commitment, particularly in chronic conditions. However, they identified the need for further studies in order to assess its efficacy. Likewise, the most recent recommendations by EULAR (European League Against Rheumatism) for management of Rheumatoid Arthritis with Disease Modifying Drugs10 have also included as one of its essential principles the concept of treatment as a shared decision between patient and Rheumatologist. This principle is based on the discussion of the treatment plan, its objectives, and the reasons for its selection.
Recommendation 2.3
Healthcare professionals must advice patients about reliable information sources regarding their disease and treatment, adapted to their information needs (for example, web-pages of patient associations or scientific societies).
Level of evidence: 5.
Block 3: Patient Stratification according to their Risk of Non-Adherence
Recommendation 3.1
For the patient stratification process, the following must be taken into account:
]]>• Aspects associated with the knowledge and acceptance of patients of their disease, their sociocultural and economic setting, their expectations, fear of adverse effects, their comorbidities, physical difficulties for administration, etc.
• Potential barriers for treatment adherence: treatment complexity, treatment tolerability, easy access to health professionals, etc. Level of evidence: 2b.
Comments to the recommendation: Patient stratification according to their risk of non-adherence has been identified as an increasing need in order to personalize the strategies for improving adherence. Even though no specific risk pattern has been identified for non-adherent patients, some tools have been developed11, which have been used in prospective studies with positive results12.
Block 4: Estimating / Assessing / Monitoring Adherence
Recommendation 4.1
To estimate and analyze adherence periodical/y is a key aspect in the management of patients with chronic diseases, and essential for making informed decisions about patient treatment.
Level of evidence: 5.
Recommendation 4.2
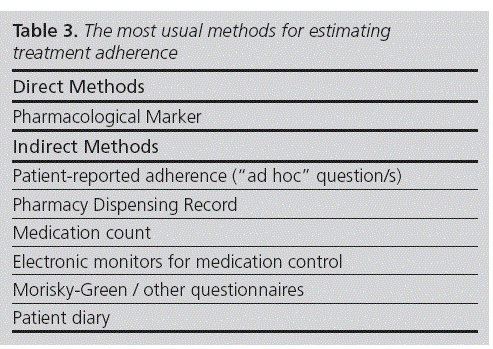
Tools for assessing adherence must be simple, in order to encourage their use in daily c/inical practice, and they should be preferab/y validated in the population of interest (Table 3). It is advisable to use simultaneous/y at least two tools for assessing adherence, adapted to the daily practice of each healthcare professional.
]]>
Level of evidence: 5.
Block 5: Interdisciplinary Approach of the Intervention
Recommendation 5.1
Adherence must be approached by consensus and in an interdiscip/inary manner by al/ agents (healthcare professionals, patients, caregivers and relatives, and managers) involved in comprehensive patient care; it would be advisable to encourage dialogue forums between them. Level of evidence: 5.
Recommendation 5.2
Healthcare professionals involved in patient care should share direct technologies for information and communication in the clinical record setting, to allow a better coordination.
Level of evidence: 5.
]]> Block 6: IndividualizationRecommendation 6.1
For each patient, it is recommended to assess the factors predicting treatment adherence before and after treatment. It is recommended that these interventions should be individualized, because:
• There is no unique intervention that can be recommended to all patients.
• Risk factors differ according to patient characteristics, type of disease, and treatment.
• The needs and beliefs of each patient are varied, regarding their disease, its treatment, and potential side effects.
Level of evidence: 5.
Block 7: Strategies
Recommendation 7.1
]]> Interventions targeted to improving treatment adherence, adapted to the individual needs of patients, and approved by consensus by the interdisciplinary teams, should include standard informative aspects, educational / behavioural aspects, or a combination of both.Level of evidence: 5.
Recommendation 7.2
In paediatric patients, programs for improving treatment adherence should include the active participation of their relatives or carers.
Level of evidence: 1b
Comments to the recommendation: Carers or relatives will usually play an active role in the studies of interventions for treatment adherence in paediatric patients. Those studies identified in the process of systematic review that included paediatric population involved carers or relatives in all cases, as active participants of the strategy13,14.
Recommendation 7.3
For dependent patients, or those who require assistance by a carer or relative, the intervention programs for improving adherence must include the active participation by their carers or relatives.
Level of evidence: 5.
Recommendation 7.4
]]> It is recommended to use comprehensive strategies sustained over time between the healthcare professional and the patient, targeted to encourage treatment adherence. These should include, for example (Table 4):
• Oral, written and/or electronic channels of information.
• Adaptation of the treatment strategy.
• Tools as reminders for administration as well as for collecting medication.
Level of evidence: 1b
Comments to recommendation: In the process of evidence systematic review, different articles were identified that assessed the use of various interventions for improving treatment adherence in patients with systemic inflammatory diseases15,16,17,18. To develop this Recommendation, those interventions that had proven efficacy for improving adherence were included, as well as other potential strategies suggested by experts and agreed upon during the consensus process.
]]>Discussion
The objective of these recommendations is to create awareness among all healthcare professionals involved in managing patients diagnosed with systemic inflammatory conditions about the importance of treatment adherence, how to communicate with patients, how to detect and assess patient adherence, and which strategies could be applied in order to improve adherence. This is the first multidisciplinary document of recommendations prepared in Spain for this area.
Regarding the methodology used in the development of recommendations, standard processes have been taken into account, such as a systematic literature review, as well as more qualitative aspects, such as the dynamics of recommendations conducted in each one of the meetings with healthcare professionals. A structured survey has also been used, as the most adequate tool for the final quantification of the degree of agreement with each one of the recommendations, so that all participants had the same level of representation in the final recommendations. All this leads to a robust process of preparation of recommendations, at a methodological level, due to the combination of different strategies.
On the other hand, if we review the different consensus or recommendations published so far in Spain, the majority have focused on HIV infection19 and psychiatric conditions20,21,22. The GESIDA/SEFH/PNS multidisciplinary consensus on adherence in HIV infection shares some of the recommendations collected in our study. Particularly, these show the importance of assessing the factors of risk for non-adherence, the need of a multidisciplinary approach to address adherence, the need to assess it through different methods, the importance of communication between the healthcare professional and the patient, and the need to individualize those interventions targeted to improve treatment adherence19. The GESIDA recommendations have been recently used to assess the perspective of the different healthcare professionals involved (hospital pharmacists, physicians and nurses) regarding the support measures for treatment adherence; differences in their perspectives have been observed, based on lack of time and training23. In the Psychiatry area, consensus has focused on schizophrenia and major depression. In the case of schizophrenia, 2 unidisciplinary consensuses have been developed by Psychiatrists20 and nurses21, with different perspectives, because the former is mostly focused on improving adherence through psycho-educational strategies targeted to increasing patient awareness of their condition, and the latter on improving strategies to be implemented in daily practice. The consensus on major depression also stresses that knowledge about the disease and the importance of receiving drug treatment in order to improve the disease symptoms are the essential aspects in order to improve treatment adherence22.
Also regarding chronic conditions, different regional and national agencies have driven initiatives and policies for the improvement of treatment adherence24,25. In the European setting, a consensus document for improving treatment adherence has been recently published26, developed through the Delphi methodology, with a panel of 50 experts from 14 countries representing organizations of patients, carers, health providers and professionals, politicians, academicians, and industry representatives. Twenty-five (25) potential solutions were prioritized for improving adherence, primarily an improvement in education and information for patients, patient involvement in drug therapy decisions taking into account their preferences, and a higher education and training for healthcare professionals.
Another important aspect to be considered is the impact of non-adherence in health outcomes. The impact of lack of adherence has been widely studied in conditions such as HIV infection27, psychiatric disorders28, or cardiovascular conditions29, demonstrating the association between the lack of adherence in a generic way with a higher mortality rate4. However, this impact has not been so widely explored in systemic inflammatory conditions.
This is a dynamic document that is open to review, in order to incorporate any new recommendations based on the outcomes of new studies assessing strategies targeted to improving therapeutic adherence in these patients, and showing improvements in health outcomes. Among many discussions and debates generated during the meetings of the expert panel, one need stood out: to generate new evidence through pilot studies or other type of observational studies to assess the impact of interventions for improving adherence in patients with inflammatory diseases, and the optimal frequency for this assessment from the point of health outcomes, and always from the multidisciplinary perspective of healthcare professionals involved in the process.
In the conditions studied, there is a variation in the type of population affected, and their course and prognosis; this could represent a limitation for their joint analysis. However, these are chronic clinical entities that share common characteristics, and have a high impact on the quality of life of patients; therefore, an overall document can allow the healthcare professional to apply the recommendations which are more adequate for each case.
On of the main limitations of this document is the lack of involvement by patient associations in these recommendations. As previously discussed, the point of view of patients is essential in order to approach adherence from a comprehensive perspective. However, this group has not been involved in the development of this first document.
]]> New studies are required in the setting of daily clinical practice, to generate evidence regarding the impact of interventions on improving adherence in patients with inflammatory diseases and their association with health outcomes. However, this article collects all aspects to be taken into account in order to detect, monitor and assess therapeutic adherence in a multidisciplinary manner, actively involving the patient in this process, and allowing all healthcare professionals in Spain to consult and use it at any time.
Conflict of Interests
Fernando Bermejo hereby declares that he has received grants for research, and has worked as a lecturer and advisor for MSD and Abbvie.
Bibliography
1. Thorneloe RJ, Bundy C, Griffiths CE, Ashcroft DM, Cordingley L. Adherence to medication in patients with psoriasis: a systematic literature review. Br J Dermatol. 2013;168:20-31. [ Links ]
2. Lopez A, Billioud V, Peyrin-Biroulet C, Peyrin-Biroulet L. Adherence to Anti-TNF Therapy in Inflammatory Bowel Diseases: A Systematic Review. Inflamm Bowel Dis. 2013;19:1528-33. [ Links ]
]]>3. McHorney CA, Victor Spain C, Alexander CM, Simmons J. Validity of the Adherence Estimator in the Prediction of 9-Month Persistence with medications prescribed for chronic diseases: A prospective analysis of Data from pharmacy claims. Clin Ther. 2009;31:2584-607. [ Links ]
4. Hugtenburg JG, Timmers L, Elders PJ, Vervloet M, van Dijk L. Definitions, variants, and causes of nonadherence with medication: a challenge for tailored interventions. Patient Prefer Adherence. 2013 ;7:675-82. [ Links ]
5. Medicines adherence: involving patients in decisions about prescribed medicines and supporting adherence. National Collaborating Centre for Primary Care (UK). Royal College of General Practitioners (UK); 2009 Jan. [ Links ]
6. Kripalani S, Yao X, Haynes RB. Interventions to enhance medication adherence in chronic medical conditions: a systematic review. Arch Intern Med. 2007;167:540-50. [ Links ]
7. Oxford Centre for Evidence-based Medicine - Levels of Evidence. Oxford, UK, Marzo 2009 (consultado 12 de diciembre de 2014). Disponible en: http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/. [ Links ]
]]>8. Pasma A, van't Spijker A, Hazes JM, Busschbach JJ, Luime JJ. Factors associated with adherence to pharmaceutical treatment for rheumatoid arthritis patients: a systematic review. Semin Arthritis Rheum. 2013;43:18-28. [ Links ]
9. Joosten EA, DeFuentes-Merillas L, de Weert GH, Sensky T, van der Staak CP, de Jong CA. Systematic review of the effects of shared decision-making on patient satisfaction, treatment adherence and health status. Psychother Psychosom. 2008;77:219-26. [ Links ]
10. Smolen JS, Landewe R, Breedveld FC, Dougados M, Emery P, Gaujoux-Viala C, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Ann Rheum Dis. 2010;69:964-75. [ Links ]
11. INTERMED Complexity Assessment Grid (IM CAG version 6). INTERMED Foundation. Amsterdam, The Netherlands, 2009 (consultado 10 de octubre de 2013). Disponible en: http://www.intermedfoundation.org/uploads/10/32/103299ff97ac58bcee216085ec23a708/Description-of-IM-CAG-v6-including-interview-and-score-December-2009.pdf. [ Links ]
12. Stockl KM, Shin JS, Lew HC, Zakharyan A, Harada AS, Solow BK, et al. Outcomes of a rheumatoid arthritis disease therapy management program focusing on medication adherence. J Manag Care Pharm. 2010;16:593-604. [ Links ]
]]>13. Hommel KA, Hente E, Herzer M, Ingerski LM, Denson LA. Tele-health behavioral treatment for medication nonadherence: A pilot and feasibility study. Eur J Gastroenterol Hepatol. 2013 ;25:469-73. [ Links ]
14. Hommel KA, Hente EA, Odell S, Herzer M, Ingerski LM, Guilfoyle SM, et al. Evaluation of a group-based behavioral intervention to promote adherence in adolescents with inflammatory bowel disease. Eur J Gastroenterol Hepatol. 2012;24:64-9. [ Links ]
15. Balato N, Megna M, Di Costanzo L, Balato A, Ayala F. Educational and motivational support service: A pilot study for mobile-phone-based interventions in patients with psoriasis. Br J Dermatol. 2013;168:201-5. [ Links ]
16. Cook PF, Emiliozzi S, El-Hajj D, McCabe MM. Telephone nurse counseling for medication adherence in ulcerative colitis: A preliminary study. Patient Educ Couns. 2010;81:182-6. [ Links ]
17. Elkjaer M, Shuhaibar M, Burisch J, Bailey Y, Scherfig H, Laugesen B, etal. E-health empowers patients with Ulcerative Colitis - a randomised controlled trial of the web-guided Constant-care approach. Gut. 2010;59:1652-61. [ Links ]
]]>18. Cross RK, Cheevers N, Rustgi A, Langenberg P, Finkelstein J. Randomized, controlled trial of home telemanagement in patients with ulcerative colitis (UC HAT) Inflamm Bowel Dis. 2012;18:1018-25. [ Links ]
19. Panel de Expertos de Gesida Recomendaciones GESIDA/SEFH/PNS para mejorar la adherencia al tratamiento antirretroviral (actualizado Junio 2008; consultado 21 octubre 2013). Disponible en: http://www.gesida-seimc.org/pcientifica/fuentes/DcyRc/Gesida_dcyrc2008_adherenciaTAR.pdf. [ Links ]
20. Roca M, Cañas F, Olivares J, Rodríguez A, Giner J. Treatment adherence in schizophrenia. Spanish clinical consensus. Actas Esp Psiquiatr. 2007;35(1 Suppl):1-6. [ Links ]
21. García I, Granada JM, Leal MI, Sales R. LLuch MT, Fornés J, et al. Adherencia al tratamiento en la esquizofrenia: Consenso de enfermería en salud mental. Actas Esp Psiquiatr. 2010;38(Suppl 1):1-45. [ Links ]
22. Montejo AL, Menchón JM, Carrasco JL, Franco M, Martín Carrasco M, Moriñigo A. Guía de evaluación y mejora del cumplimiento en el tratamiento a largo plazo del Trastorno Depresivo Mayor. Actas Esp Psiquiatr 2010;38(Suppl. 2):1-27. [ Links ]
]]>23. Morillo Verdugo R, Jiménez Galán R, Almeida González C. Perspectiva multidisplicinaria del apoyo a la adherencia antirretroviral en Andalucía. Estudio Andhalusida. Farm Hosp. 2012;36:410-23. [ Links ]
24. Plan de Salud de Cataluña 2011-2015. Generalitat de Catalunya. Departamento de Salud, 2012. Consultado 21 octubre 2013. Disponible en: http://www20.gencat.cat/docs/salut/Home/El%20Departament/Pla_de_Salut_2011_2015/documents/arxius/plan%20de%20salud%20Catalunya_es.pdf. [ Links ]
25. Navarra. ORDEN FORAL 146/2013, de 4 de octubre, de la Consejera de Salud, por la que se crea el Comité de Expertos para la conciliación y la mejora de la adherencia al tratamiento de los pacientes crónicos. Boletín Oficial de Navarra, 29 de octubre de 2013, núm 209, pp. 11356-7. [ Links ]
26. Clyne W, White S, McLachlan S. Developing consensus-based policy solutions for medicines adherence for Europe: a Delphi study. BMC Health Serv Res. 2012;12:425. [ Links ]
27. García de Olalla P, Knobel H, Carmona A, Guelar A, López-Colomés JL, Caylà JA. Impact of adherence and highly active antiretroviral therapy on survival in HIV-infected patients. J Acquir Immune Defic Syndr. 2002;30:105-10. [ Links ]
]]>28. Ascher-Svanum H, Faries DE, Zhu B, Ernst FR, Swartz MS, Swanson JW. Medication adherence and long-term functional outcomes in the treatment of schizophrenia in usual care. J Clin Psychiatry. 2006;67:453-60. [ Links ]
29. Corrao G, Parodi A, Nicotra F, Zambon A, Merlino L, Cesana G, et al. Better adherence to antihypertensive medications reduces cardiovascular risk. J Hypertens.2011; 29:610-18. [ Links ]
![]() Dirección para correspondencia:
Dirección para correspondencia:
Correo electrónico: miquel.ribera@uab.cat
(Miquel Ribera-Pibernat).
Recibido el 3 de noviembre de 2015; ]]> aceptado el 23 de junio de 2016.
]]>