
Rhabdomyosarcoma of the Oral Tissues two new cases and literature review
Cristiane Miranda França 1, Eliana M. M. Caran 2, Maria Teresa S. Alves 3,
Adriana D. Barreto 4, Nilza N. F. Lopes 5
(1) Oral Pathologist, DDS, PhD, Biodentistry Post-Graduation Program, Dental School, Ibirapuera University
(2) Pediatric Oncologist, MD, PhD, Pediatric Institute GRAACC, Medical School of São Paulo, Federal University of São Paulo
(3) Pathologist, MD, PhD, Pediatric Institute GRAACC, Medical School of São Paulo, Federal University of São Paulo. ]]>
(4) DDS, Pediatric Institute GRAACC, Medical School of São Paulo, Federal University of São Paulo
(5) DDS, Head of Dental Service, Pediatric Institute GRAACC, Medical School of São Paulo, Federal University of São Paulo
ABSTRACT
Rhabdomyosarcoma (RMS) is a malignant soft tissue neoplasm consisting of cells derived from the primitive mesenchyme that exhibit a profound tendency to myogenesis. About 35% of RMS arises in the head and neck, being classified as parameningeal and non-orbital non-parameningeal forms. Parameningeal tumors carry the worst prognosis. The use of contemporary, multi-agent chemotherapy, radiotherapy, and surgery has made treatment of the disseminated disease possible, and has significantly improved overall survival from 25% in 1970 to 70% in 1991. Here, we present the management of two cases of orofacial RMS in adolescents: an 18-year-old, white female that had a 9-month history of a nodule in the left buccal mucosa, and a 19-year-old, white male who had been aware of a nodule in the left, posterior maxillary ridge with progressive growth for 4 months. Before final diagnosis, both cases were previously treated as inflammatory lesions. Their clinicopathological aspects, treatment, and poor survival as a consequence of delays in diagnosis are discussed.
Key words: Rhabdomyosarcoma, oral diseases, diagnosis.
Introduction
]]> Rhabdomyosarcoma (RMS) is a malignant, soft tissue neoplasm consisting of cells derived from the primitive mesenchyme that exhibit a profound tendency to myogenesis. (1) Each year, 4.6 per million U.S. children and adolescents younger than 20 years of age are diagnosed with RMS. (2) In Brazil, the estimated incidence is 7.8 per million children under 15 years of age. (3) The use of contemporary, multi-agent chemotherapy, radiotherapy, and surgery has made treatment of the disseminated disease possible, and has significantly improved overall survival from 25% in 1970 to 70% in 1991. (4)About 35% of RMS arises in the head and neck. According to their anatomical location and propensity for invasion of the central nervous system, these RMSs are divided in orbital, parameningeal and non-orbital non-parameningeal forms. Parameningeal tumors carry the worst prognosis. (4)
The histopathological and molecular spectrum manifested by RMS has led to many classification systems. These differing morphological features were recognized in the mid-1900s by Horn and Enterline who divided rhabdomyosarcomas into embryonal (ERMS), alveolar, botryoid, and pleomorphic subtypes. (1,5) Thus, a proliferation of subtle differences in diagnostic criteria had developed. (1) In 1994, histological and biological studies have resulted in the International Classification of RMS into prognostically significant and diagnostically consistent subgroups based on the level of agreement and power of prognostic prediction. Four broad subtypes of RMS were established: (a) botryoid and spindle cell RMS (both less common variants of ERMS); (b) embryonal RMS, generally having a superior prognosis; (c) alveolar (including the solid-alveolar variant) RMS, generally having a poorer prognosis and (d) undifferentiated sarcoma, also generally having a poorer prognosis. Finally, a category of sarcoma nor otherwise specified was created for tumors that could not be classified into an specific subtype. (1,5) Recently it was added to this classification a subtype of RMS with rhabdoid-like features, whose prognosis is not presently valuable. (6) It is now apparent that rhabdomyosarcoma comprises a group of morphologically similar but biologically diverse lesions. (1)
Here, we present the management of two cases of orofacial RMS in adolescents, and discuss their clinicopathological aspects, treatment, and poor survival of these as a consequence of delays in diagnosis.
Case reports
Case 1
The Pediatric Institute GRAACC is a branch of the Medical School of São Paulo of the Federal University of São Paulo. Before beginning any procedures, the patients or their guardians receive a detailed, informed consent form that the case may be used for didatic purposes. Only those who sign the form, of their own free will are included in published reports or study groups.
An 18-year-old, white female had a 9-month history of a nodule in the left buccal mucosa. Initially, she consulted her dentist, who performed a dental extraction. Subsequently, at the same site, she noted a painless slowly-growing swelling, and one month later she was submitted to an incisional biopsy that disclosed a histological diagnosis of "solid alveolar rhabdomyosarcoma". The patient was treated employing VAC chemotherapy (vincristine, actinomycin-D, cyclophosphamide) alternated with ifosfamide/vepeside, and complementary radiotherapy, receiving 3600 cGy. The tumor progressed and the patient was referred to our service.
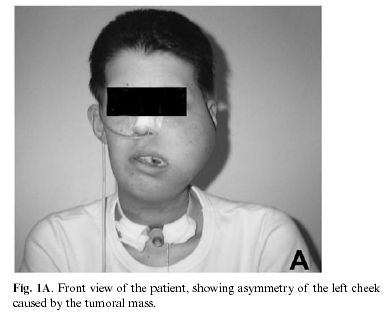
At the time of admission, the patient exhibited an extensive swelling in the oral cavity with imminent airway obstruction. A tracheostomy was performed immediately (Figure 1A). An intra-oral exam was not possible owing to the extent of the tumor. Computerized tomography scans showed an infiltrative soft tissue mass causing maxillary destruction and displacement of the adjacent structures (Figure 1B). Metastasis screenings indicated liver and pulmonary involvement. Revision of the biopsy material confirmed the diagnosis of RMS but the subtype was changed to "undifferentiated sarcoma" due its negativity to immunohistochemical tests for myogenin, desmin, muscle-specific actin, leukocyte common antigen and cytokeratins (Figures 1C and 1D). Chemotherapy with topotecan, navelbin, carboplatin and vepeside was initiated resulting in the partial regression of the tumor. The patient also received complementary radiotherapy, reaching 7300 cGy in the oral cavity and 1200 cGy in the lung. The patient died from respiratory failure caused by metastases in lung 20 months after admission to our service.
]]>
Case 2
A 19-year-old, white male had been aware of a nodule in the left, posterior maxillary ridge with progressive growth for 4 months. Initially, the patient was seen by a physician who prescribed antibiotic/anti-inflammatory therapy. After two months an unsuccessful attempt was made to drain the lesion. A biopsy was performed and a "malignant tumor" indicated. At the time of admission to our service, the patient presented facial asymmetry with an asymptomatic, fetid swelling on the left side (Figure 2A). Intra-oral examination showed a 5 x 6 cm, red, firm mass with grayish areas of central necrosis, covering the left side of the maxillary gingiva, from the central incisor to the second molar (Figures 2B and 2C). The teeth involved in the mass showed mobility although the patient still could talk and feed properly.
]]>Computerized tomography scans revealed an irregular destruction of the maxillary bone (Figures 3A and 3B). Findings by computerized chest tomographies, complete blood cell count, and blood chemistry were all within normal limits. An incisional biopsy was performed, and the specimens disclosed embryonal rhabdomyosarcoma (Figure 3C). The immunohistochemical tests were positive for desmin, vimentin and muscle actin, and negative for cytokeratins and S100 protein (Figure 3D). Myogenin showed a cytoplasmic and nonspecific background staining. The patient began a combined VAC chemotherapy consisting of cyclophosphamide, vincristine and actinomycin-D; however, the tumor continued to spread, and after four cycles reached 6 x 8 cm. The treatment protocol was altered to topotecan, navelbine, ifosfamide, and vepeside, with an associated dose of radiotherapy (5040 cGy). The tumor increased in size despite treatment, and the patient died from lung metastases 8 months after admission to the service.
Discussion
Oral rhabdomyosarcomas are classified within the non-parameningeal group of tumors, which present a better prognosis and tend not to invade the central nervous system. (4) Further, with the advent of combined surgical, chemotherapeutic, and radiotherapeutic management of RMS, the five-year survival rate is approximately 85% for this RMS subtype. (7) Even so, we present two new cases of oral rhabdomyosarcoma in adolescents with a fatal outcome.
The young man exhibited embryonal RMS, no metastases on initial treatment, and an 8-month survival period after admission to our service. On the other hand, the woman suffered from an undifferentiated sarcoma with metastases at admission, dying 20 months later. The opposite outcome would be expected. This is a conflicting data with the literature (7-9), and we did not have the possibility to perform molecular pathology investigations, but they may have answered our question about the survival of the patients. Recent studies suggest that the application of molecular pathology to classification of RMS subtypes may help to resolve some of these controversies by the use of more objective criteria based on genetic differences between the subtypes of RMS. For example, it has been described that patients with the variant PAX7-FKHR translocation have a more favorable prognosis than do those with the more common PAX3-FKHR translocation. Even more intriguing is a preliminary report suggesting a strikingly better outcome among patients with metastatic disease and variant-translocation-positive alveolar RMS (estimated 4-year overall survival, 75% vs. 8% for patients with PAX3-FKHR-positive alveolar RMS). (8)
Although myogenin is considered a sensitive and specific marker for RMS, more specific than desmin and muscle-specific actin and more sensitive than myoglobin, (10) in our cases, clinicopathological data, morphology and the immunohistochemical panel were more useful in the RMS diagnosis than myogenin alone.
]]> The incidence of RMS is highest in children 1-4 years of age, falling to a lower rate at 10-14 years, and remaining steady between 15-19 years of age. (11) This tumor is usually fast-growing and infiltrative, and often appears as an enlarging, painless mass. Initial symptoms may be vague, and may mimic other childhood and adolescent, soft-tissue sarcomas such as fibrosarcoma, leiomyosarcoma and neurofibrosarcoma. (12)Bras et al. (1987) found that of 12 patients with RMS, the interval between the first symptoms and biopsy ranged from 0 to 26 weeks, with a median of 7 weeks and an average of 7.4 weeks. (13) In most cases, when initially examined, the patients already exhibit large tumors due to their rapid growth and the frequent delay in medical consultation. Since the late 1970´s it is believed that suspected infection constitutes an important cause of the delay in RMS diagnosis. (13) Our patient cases confirm this finding, although, given the lack of inflammatory symptoms in both cases, there was a misdiagnosis of dental abscess or pulpitis. This misdiagnosis delayed the patients´ diagnoses for 3 months, and the lesions were inappropriately managed, increasing the patients´ morbidity.
In the advanced stages of the disease, because of the infiltrative growth, and depending on the site of the tumor, pain, paresthesia, loosening of the teeth, and trismus occur. (13,14) The first patient showed involvement of the teeth as a consequence of tumor growth, while the second patient had affected teeth since the very beginning of her complaint.
Site predilection in the oral cavity varies according to different authors, some finding that the palate is the commonest site, while others report that the tongue is the most common site. (14,15) The patients described here had lesions of the hard palate and alveolar ridge.
Conclusion
Despite marked improvement in RMS survival over the past decades, it is known that, in addition to a poor prognosis for patients over ten-years old, delays in diagnosis have a profoundly unfavorable impact on prognosis as illustrated by the cases presented here. Finally, when a non-resolving nodule arouses suspicion, before the clinician begins antibiotic/anti-inflammatory therapy, support for the hypothesis of infection or inflammatory disease must be obtained from anamnesis and physical examination. Otherwise, a biopsy should be performed and the patient immediately referred to a recognized diagnostic center. The initial management of any tumor is critical to a satisfactory outcome of the disease.
References
1. Parham DM. Pathologic classification of rhabdomyosarcomas and correlations with molecular studies. Mod Pathol 2001;14:506-14. [ Links ]
2. Ries LAG, Smith MA, Gurney JG eds. Cancer incidence and survival among children and adolescents: United States SEER Program 1975-1995. Bethesda, National Cancer Institute; 1999. p. 111-24. [ Links ]
3. Stiller CA, Parkin DM. Geographic and ethnic variations in the incidence of childhood cancer. Br Med Bull 1996;52:682-703. [ Links ]
4. Crist WM, Anderson JR, Meza JL, Fryer C, Raney RB, Ruymann FB, et al. Intergroup rhabdomyosarcoma study-IV: results for patients with nonmetastatic disease. J Clin Oncol 2001;19:3091-102. [ Links ]
5. Asmar L, Gehan EA, Newton WA, Webber BL, Marsden HB, van Unnik AJ, et al. Agreement among and within groups of pathologists in the classification of rhabdomyosarcoma and related childhood sarcomas. Report of an international study of four pathology classifications. Cancer 1994;74:2579-88. [ Links ]
6. Kodet R, Newton WA Jr, Hamoudi AB, Asmar L. Rhabdomyosarcomas with intermediate-filament inclusions and features of rhabdoid tumors. Light microscopic and immunohistochemical study. Am J Surg Pathol 1991;15:257-67. [ Links ]
7. Stiller CA, Stevens MC, Magnani C, Corazziari I. EUROCARE Working Group. Survival of children with soft-tissue sarcoma in Europe since 1978: results from the EUROCARE study. Eur J Cancer 2001;37:767-74. [ Links ]
8. Sorensen PH, Lynch JC, Qualman SJ, Tirabosco R, Lim JF, Maurer HM. PAX3-FKHR and PAX7-FKHR gene fusions are prognostic indicators in alveolar rhabdomyosarcoma: a report from the childrens oncology group. J Clin Oncol 2002;20:2672-9. [ Links ]
9. Meehan S, Davis V, Brahim JS. Embryonal rhabdomyosarcoma of the floor of the mouth. A case report. Oral Surg Oral Med Oral Pathol 1994;78:603-6. [ Links ]
10. Cessna MH, Zhou H, Perkins SL, Tripp SR, Layfield L, Daines C, et al. Are myogenin and myoD1 expression specific for rhabdomyosarcoma? A study of 150 cases, with emphasis on spindle cell mimics. Am J Surg Pathol 2001;25:1150-7. [ Links ]
11. Hicks J, Flaitz C. Rhabdomyosarcoma of the head and neck in children. Oral Oncol 2002;38:450-9. [ Links ]
12. McDowell HP. Update on childhood rhabdomyosarcoma. Arch Dis Child 2003;88:354-7. [ Links ]
13. Bras J, Batsakis JG, Luna MAO. Rhabdomyosarcoma of the oral soft tissues. Oral Surg Oral Med Oral Pathol 1987;64:585-96. [ Links ]
14. Chen SY, Thakur A, Miller AS, Harwick RD. Rhabdomyosarcoma of the oral cavity. Report of four cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1995;80:192-201. [ Links ]
15. Pavithran K, Doval DC, Mukherjee G, Kannan V, Kumaraswamy SV, Bapsy PP. Rhabdomyosarcoma of the oral cavity--report of eight cases. Acta Oncol 1997;36:819-21. [ Links ]
![]() Correspondence
Correspondence
Profa. Dra. Cristiane Miranda França
Av. Conselheiro Rodrigues Alves # 948, apto 93 Vila Mariana
São Paulo SP Brazil 04014-002 ]]>
Email: crispadron@uol.com.br
Received: 14-08-2005
Accepted: 4-01-2006