Bisfosfonatos y Patología Oral I. Aspectos generales y preventivos
Bisphosphonates and Oral Pathology I. General and preventive aspects
Nerea Ponte Fernández1, Ruth Estefania Fresco1, José Manuel Aguirre Urizar1
(1) Medicina Bucal. Departamento de Estomatología. Unidad de Patología Oral y Maxilofacial.
Servicio Clínica Odontológica. Universidad del País Vasco EHU
Dirección para correspondencia
]]>
RESUMEN
Los bisfosfonatos constituyen un grupo de fármacos capaces de modular el recambio óseo y disminuir su remodelado cuando existe una reabsorción excesiva. Por ello están indicados en numerosas patologías óseas como la osteoporosis postmenopáusica o la osteolisis asociada al cáncer de mama o al mieloma múltiple. En los últimos años y a raíz de su utilización masiva se han ido publicando numerosos casos de complicaciones asociadas a su uso. Entre los posibles efectos adversos más importantes se encuentran los orales, con la aparición de ulceraciones y sobre todo los casos de osteonecrosis de los maxilares asociados a esta terapéutica. En esta revisión analizamos las características generales de estos medicamentos y su mecanismo de actuación, así como los efectos adversos descritos, especialmente los orales y maxilofaciales, haciendo una referencia especial sobre la prevención de la osteonecrosis de los maxilares, a la luz de los casos descritos en la literatura médica y odontológica. El protocolo preventivo refuerza el papel fundamental del odontólogo en la prevención efectiva de este proceso antes, durante y después del tratamiento.
Palabras clave: Bisfosofonatos, efectos adversos, osteonecrosis, maxilar, prevención.
ABSTRACT
Bisphosphonates constitute a group of drugs capable of modulating bone turnover, and reduce its remodelling when an excessive resorption occurs. This is why they are indicated in a large group of bone diseases like postmenopausal osteoporosis or osteolisis associated with breast cancer or multiple myeloma. Over the last years and due to their extensive use, many cases of complications associated with their use have been published. Among the most important possible adeverse effects are the oral ones, with the appearance of ulcerations and, especially, osteonecrosis of the jaws associated with this therapy. In this paper, we have analyzed the general characteristics of these drugs and their mechanisms of action as well as the described adverse effects, especially oral and maxillofacial has been made special reference regarding the prevention of osteonecrosis of the jaws, hightened by cases described in medical and odontological literature. The preventive protocol backs up the fundamental role of the odontologist in the effective prevention of this process before, during and after the treatment.
Key words: Bisphosphonates, adverse effects, osteonecrosis, jaws, prevention.
Aspectos generales
]]> Los bisfosfonatos (BFF) son fármacos análogos no metabolizados de los pirofostatos endógenos, capaces de fijarse al hueso e inhibir la función de los osteoclastos reduciendo el recambio óseo y disminuyendo el remodelado activo en los lugares donde existe una reabsorción ósea excesiva (1). Se han utilizado en pastas dentales como agentes antiplaca y en pruebas diagnosticas como transporte del Tc99 (2).El primer BFF que se introdujo fue el etidronato con baja potencia y tendencia a producir osteomalacia (3). Buscando compuestos más potentes y sin efectos indeseados, se desarrollaron los de segunda (alendronato, pamidronato, ibandronato, tiludronato) y los de tercera generación (risedronato, ácido zoledrónico, minodronato). Pequeños cambios en la estructura de los BFF modifican sus propiedades fisicoquímicas, biológicas, terapéuticas y toxicológicas (4).
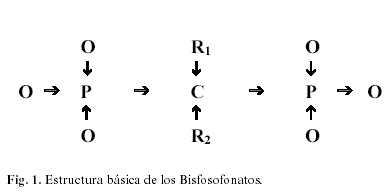
La estructura de los BFF (Figura 1) muestra dos grupos fosfatos unidos a un átomo de carbono (P-C-P), es responsable en buena parte, de su baja biodisponibilidad. Como los pirofosfatos, los BBF se unen fuertemente a la hidroxiapatita, explicando sus acciones farmacológicas en el hueso (5). Los radicales R1 influyen en esta afinidad, así los OH aumentan la fijación, mientras que el Cl la reduce. Ello explicaría por qué los BFF con grupos OH (alendronato, pamidronato, ) se fijan mas que el clodronato (6).
Los BFF se liberan cuando el hueso donde se depositan es reabsorvido, lo que explicaría su larga vida media (7). Esta liberación es multifásica y en el caso del alendronato, es de 10,5 años. Esta circunstancia explicaría su acción persistente a largo plazo en el hueso (8).
Mecanismo de accion
Los BFF inhiben la reabsorción ósea disminuyendo la actividad reabsortiva de los osteoclastos(9), promoviendo su apoptosis (10), evitando su formación a partir de precursores hematopoyéticos (11) o afectando a los osteoblastos(12).
Una vez en el interior de los osteoclastos, las cadenas R2 determinan la potencia y eficacia del fármaco. En el caso del clodronato (Cl en R2) y el etidronato (CH3 en R2), actuarían a través de su metabolización a análogos tóxicos del ATP, lo que induciría la apoptosis osteoclástica (13).
Diferentes estudios (14,15) han demostrado el papel de la FPP-sintasa como diana molecular de los BFF nitrogenados. Estos fármacos inducen la apoptosis de los osteoclastos mediante la inhibición de la biosíntesis de colesterol y de la vía del mevalonato, al inhibir la síntesis del farnesilpirofosfato (16-19). Estas alteraciones afectan la organización del citoesqueleto, el tráfico de vesículas y la formación del borde en cepillo osteoclástico.
]]> Los BFF ejercen también alguna acción sobre los osteoblastos, todavía no totalmente aclarada, disminuyendo la apoptosis y estimulando la secreción de inhibidores del reclutamiento de los osteoclastos (20).También se ha descrito un efecto antiangiogénico de los BFF mediante la inhibición de las células endoteliales, disminuyendo su proliferación e induciendo su apoptosis (21).
Indicaciones terapeuticas
Las indicaciones de los BFF han sufrido una gran evolución desde que se empezaron a utilizar en el tratamiento de varios desórdenes óseos y del metabolismo del calcio (22, 23).
Actualmente las principales indicaciones de los BFF son:
1.- En la osteoporosis postmenopaúsica y la inducida por corticosteroides. En estos procesos el BFF más utilizado ha sido el alendronato, que evita la aparición de fracturas patológicas (24).
2.- En la enfermedad de Paget, para mejorar la morfología ósea y disminuir el dolor (25).
3.- En la hipercalcemia asociada a malignidad, tratando de corregir la hipercalcemia, reducir el dolor, prevenir el desarrollo de lesiones osteolíticas y fracturas (26).
4.- En metástasis óseas del cáncer de mama y de próstata, para aliviar el dolor, evitar las fracturas y la hipercalcemia. (27, 28).
]]> 5.- En el mieloma múltiple, para reducir la patología ósea asociada como los colapsos vertebrales y las fracturas, y también del dolor óseo (29, 30).Los BFF orales son potentes inhibidores de los osteoclastos, pero menos eficaces en el tratamiento de los procesos óseos asociados a enfermedades malignas, por lo que estarían principalmente indicados en el tratamiento de la osteoporosis. Por el contrario, los BFF sistémicos se indican en pacientes con cáncer de mama metastático, mieloma múltiple, hipercalcemia por malignidad, enfermedad de Paget y en pacientes con metástasis óseas de tumores sólidos (próstata, pulmón ).
Efectos adversos generales
Los BFF se toleran bien en general si se administran correctamente, no obstante, se han descrito diferentes efectos adversos y complicaciones asociadas a su uso.
En el caso de los BFF orales, los efectos secundarios digestivos son los más frecuentemente descritos: erosiones, úlceras gástricas, esofagitis y estenosis esofágica.
Se han descrito casos aislados de uveitis en pacientes tratados con alendronato (31, 32), pamidronato (33, 34), y recientemente con ácido zoledrónico (35).
La administración continuada de etidronato puede producir un cuadro de osteomalacia que desaparece al suspender la terapia (36, 37). No obstante, los BFF más modernos carecen de este efecto adverso.
Algunos modelos experimentales han planteado que la inhibición del remodelado óseo podría llevar a una disminución de la resistencia ósea aunque no hay evidencias clínicas, al menos con la administración de BFF orales. De hecho, incluso con los BFF más potentes no se anula por completo el proceso de remodelado (38).
Los efectos secundarios generales de los BFF intravenosos son similares a los de los orales, y se han descrito algún caso de flebitis (18%). Además, se ha descrito febrícula transitoria y escalofríos (10-41%) y un síndrome pseudogripal (20%) en los dos primeros días (39).
]]> Se han referido casos de hipocalcemia, habitualmente asintomática, tras altas dosis de BFF (40, 41). No obstante, la mayoría de los pacientes no la sufren gracias al aumento compensatorio de paratohormona (42).Hay que tener cuidado cuando se administran BFF intravenosos en grandes cantidades ya que se ha descrito que su inyección rápida puede provocar una insuficiencia renal, alformar una fase sólida en la sangre, que se retiene en el riñón (43).
Efectos adversos orales
Se han descrito algunos casos de ulceraciones crónicas en la mucosa oral asociados a la toma de alendronato en pacientes con osteoporosis (44). Estos casos correspondian a mujeres con osteoporosis menopáusica que tomaban alendronato y presentaron ulceraciones en la mucosa oral secundarias a una administración inadecuada, que cedieron con corticoide tópicos. Se han descrito otros casos de ulceraciones orales asociados generalmente a una inadecuada toma de BFF orales (45, 46). En la Figura 2 se presenta un caso reciente de una mujer de 55 años con ulceraciones en el suelo de la boca asociada a la toma de alendronato.
Desde hace 4 años se han ido publicando numerosos casos de una forma especial de osteonecrosis de los maxilares (ONM) en pacientes que estaban en tratamiento con BFF de alta efectividad, preferentemente con pamidronato y zolendronato y más raramente con alendronato y afectos de mieloma o cáncer de mama (47-50).
Para explicar la aparición de la ONM se han propuesto dos teorías, una relacionada con la acción de los BFF sobre el recambio óseo y otra por su efecto antiangiogénico (51). La teoría principal señala que la ONM estaría facilitada por el cese de la remodelación ósea por el efecto inhibidor sobre los osteoclastos. Los BFF se concentrarían en gran cantidad en los huesos maxilares, ya que tienen un mayor aporte sanguíneo que otros huesos y un intercambio óseo más rápido, relacionados con su gran actividad y con la presencia de los dientes. Esta circunstancia unida a la aparición frecuente de patologías dentales, tratamientos odontológicos y la fina mucosa que recubre el hueso maxilar, explicarían por qué esta condición osteonecrótica se manifiesta especialmente en estos huesos.
El mantenimiento del remodelado óseo es crítico para mantener la viabilidad ósea. Si se disminuye severamente la función osteoclástica, los osteocitos no son reemplazados y tampoco se mantiene la red capilar ósea, posibilitando la aparición de una necrosis ósea avascular (52). La rotura de la mucosa oral, bien por una ulceración traumática o por un acto quirúrgico, provoca una necrosis ósea local que va a progresar cuando fracasa la cicatrización ósea. El riesgo de ONM aumenta con la manipulación dental y con la mala higiene, ya que el hueso queda expuesto a la microbiota oral y se sobreinfecta apareciendo dolor, tumefacción, supuración y necrosis ósea progresiva, difícil de controlar (53).
La otra hipótesis etiopatogénica se basa en evidencias experimentales, según las cuales los BFF potentes también inhiben la neoangiogénesis capilar, disminuyendo la formación de capilares e inhibiendo los factores de crecimiento endoteliales (21), favoreciendo la necrosis avascular. Sin embargo, esta no debe ser la única causa ya que no se ha asociado a la utilización de otras drogas antiangiogénicas más potentes que los BFF (54, 55).
]]> Se han descrito casos de ONM asociados a BFF tanto en pacientes que tomaban la medicación durante años como durante unas pocas semanas (53). Sin embargo, en un estudio sobre pacientes con mieloma múltiple, se determinó que el riesgo de padecer ONM era tiempo-dependiente y llega a ser significativo a partir de los 12 meses y más después de los 36 meses (56).
Prevencion de la osteonecrosis maxilar asociada a BFF
Como en cualquier otro planteamiento profiláctico las medidas para prevenir la aparición de una osteonecrosis maxilar deberían implantarse antes de iniciar el tratamiento con BFF.
a.- Medidas antes de iniciar el tratamiento con BFF
Tan pronto como se considere necesario administrar BFF por parte del oncólogo u otro especialista, el paciente debe ser referido a un odontólogo para realizar un examen bucal urgente (57), que debe consistir en una exploración clínica y radiográfica completa.
El tratamiento bucal en estos pacientes está dirigido a la eliminación de todos los focos infecciosos y a la prevención de la necesidad de realizar procedimientos dentales invasivos en un futuro cercano. Por ello la terapia preventiva debe ser agresiva y debe incluir: extracciones dentarias, cirugía periodontal, tratamientos de endodoncia en dientes con viabilidad asegurada, control de caries, restauraciones dentales y colocación de prótesis si fuera preciso. Estos pacientes no son candidatos a la colocación de implantes ya que suponen un elemento de riesgo (51). Se recomienda la eliminación preventiva, un mes antes de iniciar el tratamiento con BFF, de los torus mandibulares grandes o de los torus palatinos recubiertos por una fina mucosa, por el peligro de ulceraciones.
Un paciente sin patología cardiaca no requiere profilaxis antibiótica ante procedimientos no invasivos pero sí ante los invasivos; en este caso el antibiótico de elección es la penicilina y sus derivados. Para pacientes alérgicos a la penicilina, las combinaciones de quinolonas y metronidazol han demostrado ser eficaces. La clindamicina sola no está recomendada por su escasa actividad contra los Actinomyces, Eikenella corrodens y especies similares que frecuentemente colonizan el hueso expuesto en la cavidad oral (51).
Si el paciente sólo requiere cuidado dental no invasivo no es necesario retrasar el tratamiento con BFF. Pero si el paciente requiere la realización de procedimientos invasivos se debe diferir un mes el tratamiento con BFF para permitir que el hueso se recupere y cicatrice perfectamente. Se recomienda plantear un programa continuado de vigilancia cada 4 meses con control de placa y medidas higiénicas estrictas.
b.- Medidas durante el tratamiento con BFF
]]> El odontólogo debe examinar cuidadosamente la cavidad oral buscando la presencia de exposiciones óseas en las áreas más comúnmente afectadas, como la zona posterior y lingual de la mandíbula. Se realizará un estudio radiográfico completo buscando evidencias de osteolisis, osteosclerosis, ensanchamiento del espacio periodontal y afectación periodontal.Se debe considerar la realización de una limpieza dental cuidadosa y el uso preventivo de flúor y clorhexidina, así como la instauración de medidas de higiene bucal estrictas (51).
Deben realizarse precozmente todas las restauraciones precisas para tratar de conservar los dientes y evitar la aparición de focos infecciosos. Si el diente no es restaurable, la endodoncia y la amputación de la corona son preferibles a la extracción. Los tratamientos de endodoncia se deben realizar con procedimientos que impliquen un mínimo trauma sobre el periodonto marginal y apical (58).
En relación con la enfermedad periodontal sólo se extraerán los dientes que presenten un movilidad grado 3 o mayor o aquellos asociados a un absceso periodontal. En estas situaciones, hay que dar un tratamiento antibiótico adecuado, según la pauta indicada anteriormente (51).
Los procedimientos quirúrgicos están contraindicados; no obstante, en los que tienen que someterse a cirugía, puede ser recomendable la retirada del BFF ya que se puede producir una mala recuperación si sigue bajo tratamiento. No obstante, no se sabe con certeza si la retirada del BFF va a prevenir la aparición de la ONM, ya que su vida media es larga (59). Cuando se tiene que realizar un procedimiento quirúrgico oral en un paciente en tratamiento con BFF hay que tomar unas precauciones especiales como: realizar técnicas estrictamente asépticas, realizar una cirugía atraumática y lograr un cierre por primera intención cuando sea posible (60).
Cuando sea necesario el uso de prótesis, deben ser preferiblemente fijas y bien realizadas, para evitar en lo posible la aparición de ulceras traumáticas secundarias.
En los pacientes bajo tratamiento con BFF se recomienda plantear un programa continuado de vigilancia cada 3 o 4 meses con control de placa y medidas higiénicas estrictas.
Agradecimientos: A Mr. David Hallett por su ayuda en la traducción al ingles de este trabajo.
Bibliografía
]]>1. Rodan GA, Reszka AA. Bisphosphonate mechanism of action. Curr Mol Med 2002;2:571-7. [ Links ]
2. Mellibovsky L. Osteoporosis, bisfosfonatos y calcitonina. JANO 2001; 61:47-54. [ Links ]
3. Fleish H. Bisphosphonates: Mechanisms of Action. Endocr Rev 1998; 19:80-100. [ Links ]
4. Fleish H. The role of bisphosphonates in breast cancer: Development of bisphosphonates. Breast Cancer Res 2002; 4: 30-4. [ Links ]
5. Jung, A, Bisaz S, Fleisch H. The binding of pyrophosphate and two diphosphonates on hydroxyapatite crystals. Calcif Tissue Res 1973;11: 269-80. [ Links ]
6. Shinoda H, Adamek G, Felix R, Fleisch H, Schenk R, Hagan P. Structure-activity relationships of various bisphosphonates. Calcif Tissue Int 1983;35:8799. [ Links ]
7. Kasting GB, Francis MD. Retention of etidronate in human, dog, and rat. J Bone Miner Res 1992; 7: 513-22. [ Links ]
8. Khan SA, Kanis JA, Vasikaran S, Kline WF, Matuszewski BK, McCloskey EV et al. Elimination and biochemical responses to intravenous alendronate in postmenopausal osteoporosis. J Bone Miner Res 1997; 12:1700-7. [ Links ]
9. Flanagan AM, Chambers TJ. Inhibition of bone resorption by bisphosphonates: Interactions between bisphosphonates, osteoclasts, and bone. Calcif Tissue Int 1991;49:407-15. [ Links ]
10. Hughes DE, Wright KR, Uy HL, Sasaki A, Yoneda T, Toodman GD et al. Bisphosphonates promote apoptosis in murine osteoclasts in vitro and in vivo. J Bone Miner Res 1995;10:1478-87. [ Links ]
11. Boonekamp PM, van der Wee-Pals LJ, van Wijk-van Lennep MM, Thesing CW, Bijvoet OLM. Two modes of action of bisphosphonates on osteoclastic resorption of mineralized matrix. Bone Miner 1986;1:27-39. [ Links ]
12. Sahni M, Guenther HL, Fleisch H, Collin P, Martin TJ. Bisphosphonates act on rat bone resorption through the mediation of osteoblasts. J Clin Invest 1993;91:2004-11. [ Links ]
13. Frith JC, Monkkonen J, Auriola S, Monkkonen H, Rogers MJ. The molecular mechanism of action of the antiresorptive and antiinflammatory drug clodronate: evidence for the formation in vivo of a metabolite that inhibits bone resorption and causes osteoclast and macrophage apoptosis. Arthritis Rheum 2001;44:2201-10. [ Links ]
14. Ciosek CP, Magnin DR, Harrity TW, Logan JV, Dickson JK, Gordon EM et al. Lipophilic 1,1-bisphosphonates are potent squalene synthase inhibitors and orally active cholesterol lowering agents in vivo. J Biol Chem 1993;268:24832-7. [ Links ]
15. Fisher JE, Rodan GA, Reszka AA. In vivo effects of bisphosphonates on the osteoclast mevalonate pathway. Endocrinology 2000;141:4793-6. [ Links ]
16. Luckman SP, Coxon FP, Ebetino FH, Russell RGG, Rogers MG. Heterocycle-containing bisphosphonates cause apoptosis and inhibit bone resorption by preventing protein prenylation: evidence from structure-activity relationships in J774 macrophages. J Bone Miner Res 1998; 13:1668-78. [ Links ]
17. Luckman SP, Hughes DE, Coxon FP, Russell RGG, Rogers MG. Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of gtp-binding proteins, including ras. J Bone Miner Res 1998;13:581-9. [ Links ]
18. van Beek E, Pieterman E, Cohen L, Löwik C, Papapoulos S. Farnesyl pyrophosphate synthase is the molecular target of nitrogen-containing bisphosphonates. Biochem Biophys Res Commun 1999;264:108-11. [ Links ]
19. Bergstrom JD, Bostedor RG, Masarachia PJ, Rewszka AA, Rodan GA. Alendronate is a specific, nanomolar inhibitor of farnesyl diphosphate synthase. Arch Biochem Biophys 2000;373:231-41. [ Links ]
20. Fleish HA. Bisphosphonates: preclinical aspects and use in osteoporosis. Ann Med 1997;29:55-62. [ Links ]
21. Fournier P, Boissier S, Filleur S, Guglielmi J, Cabon F, Colombel M et al. Bisphosphonates inhibit angiogenesis in vitro and testosterona-stimulated vascular regrowth in the ventral prostate in castrated rats. Cancer Res 2002;62:6538-44. [ Links ]
22. Green JR, Rogers MJ. Pharmacologic profile of zoledronic acid; a highly potent inhibitor of bone resorption. Drug Rev Res 2002; 55: 210-44. [ Links ]
23. Lenz JH, Steiner-Krammer B, Schmidt W, Fietkau R, Mueller PC, Gundlach KK. Does avascular necrosis of the jaws in cancer patients only occur following treatment with bisphosphonates?. J Cranio-Maxillofac Surg 2005;33:395-403. [ Links ]
24. Bagger YZ, Tanko LB, Alexandersen P, Ravn P, Christiansen C. Alendronate has a residual effect on bone mass in postmenopausal danish women up to 7 years after treatment withdrawal. Bone 2003;33:301-7. [ Links ]
25. Reid IR, Miller P, Lyles K, Fraser W, Brown JP, Saidi Y et al. Comparison of a single infusion of zoledronic acid with risedronate for Pagets disease. N Engl J Med 2005;353:898-908. [ Links ]
26. Berenson JR, Lichtenstein A, Porter L, Dimopoulos MA, Bordoni R, George S et al. Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. N Engl J Med 1996;334: 48893. [ Links ]
27. Michigami T, Hiraga T, Williams PJ, Niewolna M, Nishimura R, Mundy GR et al. The effect of the bisphosphonate ibandronate on breast cancer metastasis to visceral organs. Breast Cancer Res Treat 2002;75:249-58. [ Links ]
28. Bauss F, Body JJ. Ibandronate in metastatic bone disease: a review of preclinical data. Anticancer Drugs 2005;16:107-8. [ Links ]
29. Berenson JR, Rosen LS, Howell A, Porter L, Coleman RE, Morley W et al. Zoledronic acid reduces skeletal-related events in patients with osteolytic metastases. Cancer 2001;91:1191-200. [ Links ]
30. Roodman GD. Mechanisms of bone metastasis. N Engl J Med 2004; 350: 1655-64. [ Links ]
31. Mbekeani JN, Slamovits TL, Schwartz BH, Sauer HL. Ocular inflammation associated with alendronate therapy. Arch Ophthalmol 1999; 117:8378. [ Links ]
32. Malik AR, Campbell SH, Toma NM. Bilateral acute anterior uveitis after alendronate. Br J Ophthalmol 2002;86:1443. [ Links ]
33. Ghose K, Waterworth R, Trolove P, Highton J. Uveitis associated with pamidronate. Aust N Z J Med 1994;24:320. [ Links ]
34. Fraunfelder FW, Fraunfelder FT, Jensvold B. Scleritis and other ocular side effects associated with pamidronate disodium. Am J Ophthalmol 2003;135:21922. [ Links ]
35. Durnian JM, Olujohungbe A, Kyle G. Bilateral acute uveitis and conjunctivitis after zoledronic acid therapy. Eye 2005;19:2212. [ Links ]
36. Jowsey J, Riggs BL, Kelly PJ, Hoffman DL, Bordier P. The treatment of osteoporosis with disodium ethane-1-hydroxy-1,1-diphosphonate. J Lab Clin Med 1971;78:57484. [ Links ]
37. Boyce BF, Smith L, Fogelman I, Johnston E, Ralston S, Boyle IT. Focal osteomalacia due to low-dose diphosphonate therapy in Pagets disease. Lancet 1984;1:8214. [ Links ]
38. Riancho JA. Bifosfonatos orales en el tratamiento de la osteoporosis. En: Riancho JA, González J. Manual Práctico de osteoporosis y enfermedades del metabolismo mineral. Madrid: Jarpyo Editores; 2004 . p. 184-5. [ Links ]
39. Hernández JL. Bifosfonatos intravenosos y osteoporosis. En: Riancho JA, González J. Manual Práctico de osteoporosis y enfermedades del metabolismo mineral. Madrid: Jarpyo Editores; 2004. p. 190. [ Links ]
40. McIntyre E, Bruera E. Symptomatic hypocalcaemia after intravenous pamidronate. J Palliat Care 1996;12:46-7. [ Links ]
41. Mishra A, Wong L, Jonklaas J. Prolonged symptomatic hypocalcaemia with pamidronate administration and subclinical hypoparathyroidism. Endocrine 2001;14:159-64. [ Links ]
42. Fraser WD, Logue FC, Gallacher SJ, OReilly DS, Beastall GH, Ralston SH et al. Direct and indirect assessment of the parathyroid hormone response to pamidronate therapy in Pagets disease of bone and hypercalcemia of malignancy. Bone Miner 1991;12:113-21. [ Links ]
43. Bounameaux HM, Schifferli J, Montani JP, Jung A, Chatelanat F. Renal failure associated with intravenous diphosphonates. Lancet 1983;1:471. [ Links ]
44. González-Moles MA, Bagán-Sebastián JV. Alendronate-related oral mucosa ulcerations. J Oral Pathol Med 2000;29:514-8. [ Links ]
45. Mundy GR, Guise TA. Hypercalcemia of malignancy. Am J Med 1997;103:134-45. [ Links ]
46. Schmutz JL, Barbaud A, Trechot P. Alendronate and mouth ulcers: careful interrogation of the patient to start with. Ann Dermatol Venereol 2005;132:930. [ Links ]
47. Marx RE. Pamidronate (Aredia) and zolendronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 2003;61:1115-7. [ Links ]
48. Jiménez Y, Bagán JV. Bisphosphonates, as a new cause of drug-induced jaw osteonecrosis: an update. Med Oral Patol Oral Cir Bucal 2005; 10:88-91. [ Links ]
49. Bagán JV, Murillo J, Jiménez Y, Poveda R, Milian MA, Sanchos JM et al. Avascular jaw osteonecrosis in association with cancer chemoterapy: series of 10 cases. J Oral Pathol Med 2005;34:120-3. [ Links ]
50. Pastor-Zuazaga D, Garatea-Crelgo J, Martino-Gorbea R, Etayo-Pérez A, Sebatián-Lopez C. Osteonecrosis maxilar y bisfosfonatos. Presentación de tres nuevos casos. Med Oral Patol Oral Cir Bucal 2006;11:57-60 [ Links ]
51. Marx RE, Sawatari Y, Fortin M, Broumand V. Bisphosphonate-induced exposed bone (osteonecrosis/osteopetrosis) of the jaws: Risk factors, recognition, prevention, and treatment. J Oral Maxillofac Surg 2005;63:1567-75. [ Links ]
52. Carter G, Goss AN, Doecke C. Bisphosphonates and avascular necrosis of the jaw: a possible association. Med J Aust 2005;182:413-5. [ Links ]
53. Migliorati CA. Bisphosphanates and oral cavity avascular bone necrosis. J Clin Oncol 2003;21:4253-4. [ Links ]
54. Migliorati CA. Bisphosphonate-associated oral osteonecrosis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2005;99:135. [ Links ]
55. Stinchcombe TE, Mitchell BS, Depcets-Smith N, Orlowski RZ, Shea TC, Baldwin AS et al. PS-341(Bortazomib) is active in multiple myeloma. Preliminary report of a phase I trial of the proteasome inhibitor PS-341 in patients with hematologic malignancies. Blood 2000;96:516. [ Links ]
56. Brian GM, Durie MD. Zometa and osteonecrosis of the jaws a new update. Jul 2004. htpp://myeloma.org/main.jsp.aredia/ [ Links ]
57. Greenberg MS. Intravenous bisphosphonates and osteonecrosis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2004;98:259-60. [ Links ]
58. Katz H. Endodontic implications of bisphosphonate-associated osteonecrosis of the jaws: a report of three cases. J Endod 2005;31:831-4. [ Links ]
59. Berenson JR. Recommendations for zoledronic acid treatment of patients with bone metastases. Oncologist 2005;10:52-62. [ Links ]
60. Robinson NA, Yeo JF. Bisphosphonates a word of caution. Ann Acad Med Singapore 2004;33:48-9. [ Links ]
![]() Dirección para correspondencia:
Dirección para correspondencia:
Prof. José Manuel Aguirre Urízar
Medicina Bucal. Departamento de Estomatología.
Universidad del País Vasco EHU
Barrio Sarriena s/n
Leioa 48940. Vizcaya
E-mail: otpagurj@lg.ehu.es
Recibido: 29-03-2006 ]]> Aceptado: 26-06-2006
]]>